Rotundic Acid Promotes Angiogenesis Potentially by Activating the STAT3 -JAK Signaling Pathway
Zhiqiang Gong1,2, Yu Huo2, Xianfu Liu2, Wenjun Tang2, Tianxiu Meng2, Yanhong Huang2, Lifang Yang1* and Chenglin Li2*
1Department of Guangxi Key Laboratory of Chemistry and Engineering of Forest Products, Guangxi University for Nationalities, China
2Department of Faculty of Chinese Medical Science, Guangxi University of Chinese Medicine, China
Submission: November 02, 2021; Published: November 18, 2021
*Corresponding author: Lifang Yang, Department of Guangxi Key Laboratory of Chemistry and Engineering of Forest Products, Guangxi University for Nationalities, China
*Chenglin Li, Department of Faculty of Chinese Medical Science, Guangxi University of Chinese Medicine, China
How to cite this article:Zhiqiang G, Yu H, Xianfu L, Wenjun T, Tianxiu M, et al. Rotundic Acid Promotes Angiogenesis Potentially by Activating the STAT3- JAK Signaling Pathway. Glob J Pharmaceu Sci. 2021; 9(2): 555759. DOI: 10.19080/GJPPS.2021.09.555759.
Abstract
Context: Conventional revascularization strategies for ischemic heart disease (IHD) are designed to prompt reperfusion of the coronary artery to salvage cardiomyocytes, but they may lead to myocardial reperfusion injuries.
Objective: This work investigated the pro-angiogenic effect of Rotundic acid (RA) to provide the experimental basis for angiogenesis-mediated drug therapy of IHD.
Materials and methods: Human umbilical vein endothelial cells (HUVEC) were treated with vehicle or RA at 4, 8, 12, or 16μM for 24 h. A model of hindlimb ischemia was established using C57BL/6 mice. In sham-operated animals, only the femoral artery was isolated. Mice in the supplementation group were injected with RA (50 mg/kg body weight/day) for 7 days.
Results: RA promoted cell proliferation, adhesion, migration and tube formation in the HUVECs in a dose-dependent manner. The ED50 of RA to improve cell adhesion is 8μM. In mice, RA promoted angiogenesis after ligation of the femoral artery and ameliorated the ischemic conditions. Intriguingly, more blood flow recovery was observed in the RA supplemented mice compared to the Vehicle-treated mice (0.85±0.05 vs. 0.71±0.10 on day 3; 0.94±0.10 vs. 0.75±0.08 on day 7). In HUVEC, RA increased the phosphorylation of STAT3 and JAK, which could be the mechanism by which RA mitigated IHD.
Conclusion: This is the first study that shows RA promotes angiogenesis under both normal and ischemic conditions. RA mitigates IHD potentially by activating the JAK-STAT3 pathway. Further clinical trials are warranted to verify the clinical implication.
Keywords: Rotundic acid; Angiogenesis; Ischemic heart disease; STAT3; JAK
Abbreviations: IHD: Ischemic Heart Disease; RA: Rotundic Acid; HUVEC: Human Umbilical Vein Endothelial Cells; WHO: World Health Organization; IHD: Ischemic Heart Disease; NF: Nuclear Factors; JAK: Janus Kinase; IL: Interleukin; IFNS: Interferons; NIH: National Institutes of Health; HUVEC: Human Umbilical Vein Endothelial Cells; PBS: Phosphate Buffered Saline; IHC: Immunohistochemistry; WCL: Whole Cell Lysates; RIPA: Radioimmunoprecipitation Assay
Introduction
Based on the World Health Organization (WHO) report, ischemic heart disease (IHD) is the leading cause of death in developed and developing countries Finegold et al. [1]. Traditional revascularization strategies, such as primary coronary intervention (PCI), mainly focus on promoting coronary reperfusion to replenish cardiomyocytes Ong & Foussas [2,3]. However, these strategies may lead to microcirculatory dysfunction Stone & Ergul et al. [4,5]. Therefore, methods need to be taken during the angiogenesis-mediated microvascular recovery to improve reperfusion and reduce adverse sequences. Angiogenesis is a physiological or pathological process characterized by the sprouting of new blood vessels from existing vasculature Adair & Cheng et al. [6,7]. In addition to IHD, angiogenesis plays a critical role in various physiological process such as wound healing Poustchi et al. [8], where angiogenic capillary sprouts enter the fibrin/fibronectin-rich wound clot and generated a microvascular network in the granulation tissue. Signal transducer and activator of transcription (STAT) is a group of nuclear factors (NF) that modulate various cellular physiological and biological activities Loh et al. [9]. Among the STAT family members, STAT3 is the most extensively studied NF due to its critical role in cancer formation and cardiac pathogenesis for angiogenesis Chen, Johnson & Cheng et al. [10-12]. Janus kinase (JAK) is a family of non-receptor tyrosine kinases Yamaoka et al. [13]. In response to cytokine ligands, such as Interleukin (IL)-6 and Interferons (IFNs), cytokine receptors are activated, which may recruit JAK proteins by facilitating the binding of JAKs to the cytokine receptor subunit β (gp130) and the phosphorylation of STAT3 at tyrosine 705 residue Huang & Johnson et al. [11,14] Since the JAK-STAT3 signalling pathway plays a vital role in angiogenesis, it is receiving increasing attention in the cardiovascular field as a potential molecular target for angiogenesis-mediated therapy. Ilex rotunda (Aquifoliaceae), Holly Family, is commonly used in Chinese herbal medicine in southern China. The medicine is made from the bark or leaves and shows effects of clearing away heat and toxic substances, removing dampness, and relieving swelling and pain. It is mostly used to treat various infectious diseases Wang et al. [15]. It has been reported that its bark extract is capable of increasing coronary flow, improving hypoxia tolerance, lowering blood pressure and protecting myocardial ischemia in rats Liu et al. [16]. Rotundic acid (RA) belongs to the pentacyclic triterpenoid family and is mainly found in water fly thrips such as Ilex rotundus, which are widely distributed in China Haraguchiet & Xie et al. [17,18]. Previous studies reported the anticancer efficacy of RA both in vitro and in vivo Nan & Roy et al. [19,20]. However, the effects of RA in treating IHD remain enigmatic. This study explores the potential efficacy of RA in IHD treatment, using both in vitro and in vivo methods. We hypothesized that RA is able to enhance blood flow recovery after IHC treatment by promoting angiogenesis. We also investigated the potential mechanism by which RA promoted angiogenesis.
Materials and Methods
Animals
Six-week-old male C57BL/6 mice were obtained from the Department of Laboratory Animals, Chinese Academy of Sciences (Shanghai, China). All animals received humane care, and all experimental procedures of the animals were in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (NIH) and approved by the Experimental Research Ethics Committee of Chinese Medical Science Guangxi University of Chinese medicine (GXUTCM0933162).
Cell culture
Primary human umbilical vein endothelial cells (HUVEC) were purchased from ScienCell (Carlsbad, CA, USA) and cultured in MCDB131 (PAA, Pasching, Austria) containing 5% fetal bovine serum (Hyclone, Logan, UT), 1% endothelial cell growth supplement (ScienCell, Carlsbad, CA, USA), and 1% penicillin streptomycin (Gibco, Grand Island, NY, USA). Only cells at passages 4-6 were used for the experiments. Transformed HUVEC were obtained from American Type Culture Collection (Rockville, MD, USA) and cultured in DMEM (Gibco) containing 10% FBS and 1% penicillin streptomycin. Four biological replicates were used in the assays.
Cell viability assay
RA was purchased from Push Biotechnology (Sichuan, China). Primary HUVECs were plated in 96-well plates and treated with 4,8,12 and 16μM RA (Push Biotechnology) for 24h. Then the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2Htetrazolium bromide assay was conducted using a commercialized kit (Thermosphere Scientific, Waltham, MA, USA) as per the manufacturer’s instructions. The absorbance at 570 nm in DMSO was recorded as a reflection of cell viability.
Cell proliferation assay
Primary HUVECs were seeded in 96-well plates. After RA treatment for 24 h, proliferating cell nuclei were stained with an EdU assay kit (RiboBio, Guangzhou, China) and detected by fluorescence microscopy. DAPI (4’,6-diamidino-2-phenylindole) was used for cell nuclear counterstain.
Cell adhesion assay
Primary HUVECs were plated in 48-well plates precoated with 50μg/mL collagen I (BD, Franklin Lakes, NJ, USA) and treated with RA at 4,8,12, and 16μM for 10 min. Then, cells were washed with phosphate buffered saline (PBS) and fixed with 4% formaldehyde. Afterwards, cells were stained with a crystal violet staining solution. Cells in three random fields in each well were counted and the average was reported.
Wound healing test
Primary HUVECs were plated in 48-well plates and treated with hydroxyurea (Sigma, St. Louis, MO, USA) to inhibit cell proliferation. Once a confluent cell monolayer was formed, it was scratched with a yellow pipette tip to create a scratch wound. Afterwards, cells were treated with RA at 16μM for 24h. Photographs were taken with a Zeiss digital camera at baseline (0h) and 24h. The wound area was calculated to determine the migration distance.
Trans well migration assay
Primary HUVECs were seeded in the upper chamber precoated with 50μg/mL collagen I, and RA at 16μM was added to the lower chamber. Cells were allowed to migrate for 8h and then fixed with 4% formaldehyde. Non-migrating cells in the upper chamber were removed with a cotton swab, whereas migrating cells were stained with crystal violet. Cells in three random fields in each well were counted and the average was reported.
Tube formation test
Primary HUVECs were seeded in 48-well plates pre-coated with matrigel (BD Biosciences, CA, USA). Cells were treated with RA for 6h, and the tubule structure was subsequently photographed and quantified by measuring the length of each tube using the software Image-Pro Plus 6. Tube length in three random fields per well was calculated, and the average was reported.
Hindlimb ischemia model
Mice were intraperitoneally administered pentobarbital sodium at a dose of 20 mg/100 g body weight. A solution containing 16.7 mg/mL sodium pentobarbital was prepared fresh daily. C57BL/6 mice were anesthetized with sodium pentobarbital and placed on a heating pad (37°C) under an Olympus stereotactic microscope. A longitudinal 5 mm incision was made along the left femoral vessel starting from the groin crease. The femoral artery was carefully isolated without injuring veins or nerves, and the ligation was performed using triple surgical knots 7.0 silk sutures. Successful ligation was verified by laser Doppler perfusion imaging, and color-coded perfusion images were recorded as a reflection of the perfusion defect in the left foot after hindlimb ischemia. In sham-operated animals, only the femoral artery was isolated without ligation. Mice in the supplementation group were intraperitoneally injected with RA (50 mg/kg body weight/day) for 7 days, while mice in the vehicle group were intraperitoneally injected with saline.
Laser doppler perfusion imaging
Hindlimb perfusion was assessed using a Pericam PSI system-hemoperfusion imager (Perimed, Stockholm, Sweden) by scanning the animals on a heating plate maintained at 37°C. Perfusion was evaluated at the indicated times (before, immediately after, and 7 days after femoral artery ligation), and the flow ratio was determined by dividing the perfusion value of the ischemic hindlimb by that of the non-ischemic hindlimb from the same animal.
Immunohistochemistry (IHC)
At day 10 after femoral artery ligation, mice were sacrificed, and their gastrocnemius muscles were excised for fixation and embedded in paraffin. The endothelial cells in paraffin sections were detected by using a polyclonal CD31 antibody (Santa Cruz, CA, USA) and then stained with HRP-conjugated secondary antibody and diaminobenzidine substrate (BlueGene Biotech, Shanghai, China). Three random fields per section were captured using a Zeiss digital camera.
Micro angiogram
For the hindlimb ischemia model, mice were anesthetized by sodium pentobarbital on day 10 after femoral artery ligation, and catheters (PE-10, BD) were implanted into 0.5cm of the abdominal aorta to reach the iliac bifurcation. Hindlimb vessels were then perfused by sequential manual injections of heparinized saline (10U/mL), nitroglycerin (100μg/mL), and barium sulfate (size, 1μm, 0.1g/mL). Subsequently, mice were placed in an X-ray chamber and angiographic images were acquired using an in vivo FX PRO system (Carestream, Rochester, NY, USA). Vessel density was determined by pixel analysis using Image J software.
Protein extraction and western blot
Whole cell lysates (WCL) were extracted by using the methods described in the previous studies (20). Briefly, cell media was aspirated, followed by washing with chilled PBS and centrifugation at 2,500 rpm for 10 min. Afterwards, WCL was extracted by using radioimmunoprecipitation assay (RIPA) buffer containing 1% protease inhibitors. Western blot was performed as described previously Cheng & Kan et al. [21,22]. In brief, protein extracts were heated with 5×loading buffer (Fermentas, Vilnius, Lithuania) and then loaded onto SDS-PAGE gels (Thermo Fisher Scientific, MA, USA). Separated proteins were transferred onto polyvinylidene difluoride membranes (Millipore, MA, USA) and then blocked with 5% nonfat dry milk in TBS (Amresco) containing 0.1% Tween-20. Subsequently, the membranes were incubated with primary (p- STAT3, STAT3, p-JAK, JAK, and GAPDH) and secondary antibodies (Cell Signaling Technology, MA, USA). HRP-bound proteins were detected by Immobilon Western Chemiluminescent HRP Substrate (Millipore, MA, USA).
Statistical analysis
Quantitative data were expressed as mean±SEM. Statistical significance of differences was evaluated using unpaired Student’s t-test or one-way ANOVA. A p-value less than 0.05 was considered statistically significant.
Results
Rotundic acid promotes proliferation, adhesion and migration
As a result, RA treatment at different dosages (4,8,12 and 16μM) significantly increased cell viability and cell proliferation in primary HUVECs (Figure 1A & 1B). Furthermore, we found that RA treatment significantly promoted cell adhesion in a dose-dependent manner (Figure 1C). The ED50 of RA to improve cell adhesion is 8μM. Interestingly, in the adhesion assay, RA treatment for only 10 min was able to induce cell adhesion, suggesting that RA may induce an acute response in the HUVEC cells.
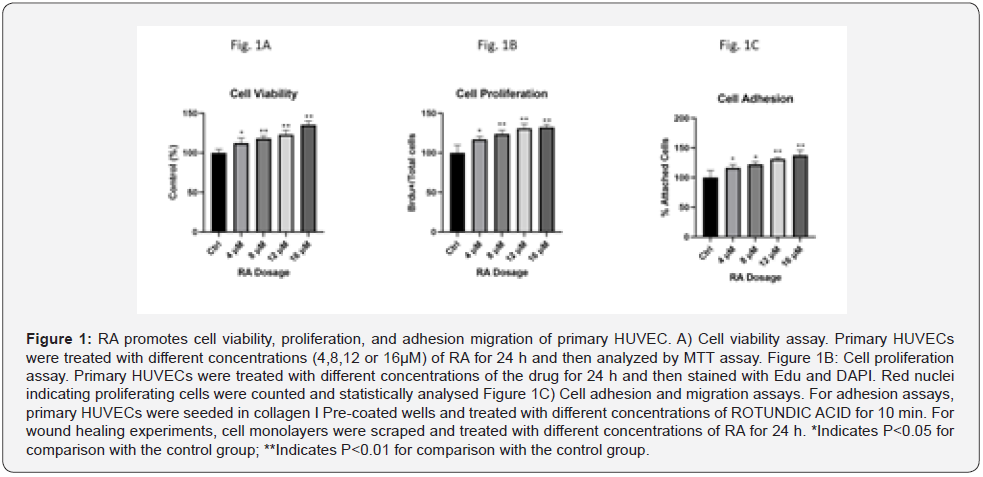
Rotundic acid promotes tube formation
In the current study, both wound-healing assay and transwell chamber assay showed increased HUVEC cell migration in response to RA treatment (Figure 2). In the 2D Matrigel experiments, tube length and morphology significantly improved with RA treatment, suggesting that RA supplementation may enhance tube network formation in the endothelial cells (Figure 3).
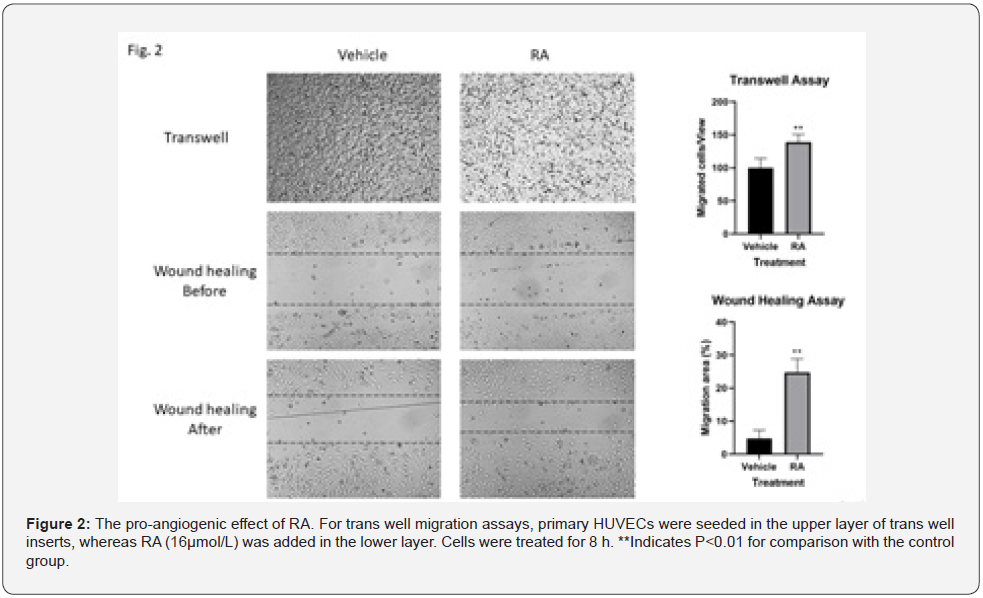
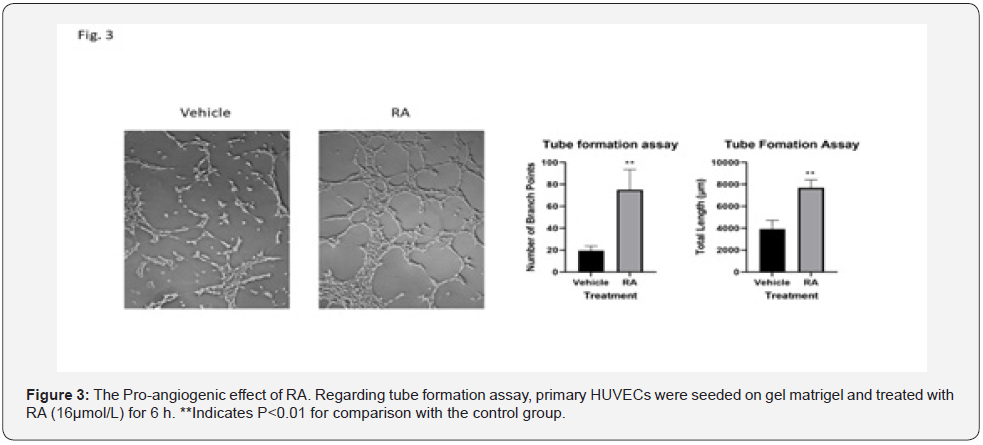
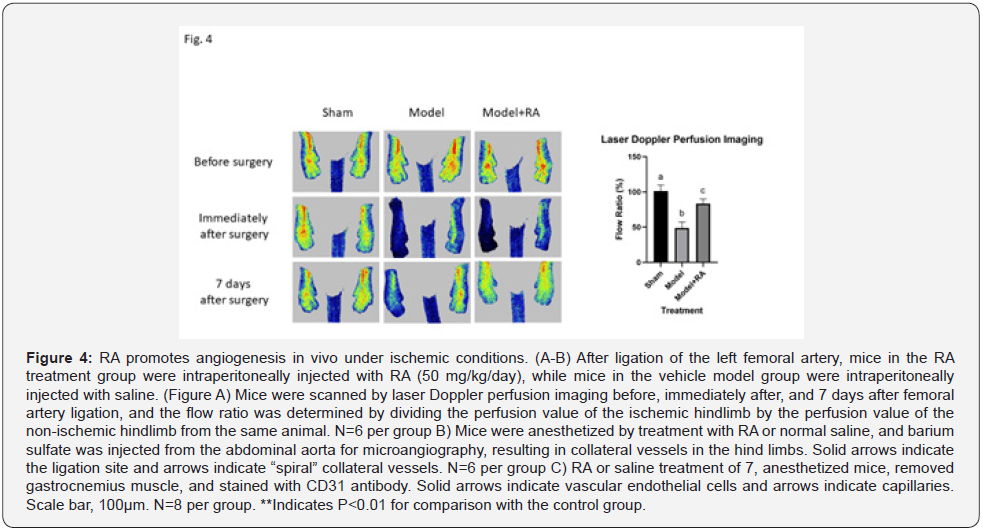
Rotundic acid promotes angiogenesis in vivo under both normal and ischemic conditions
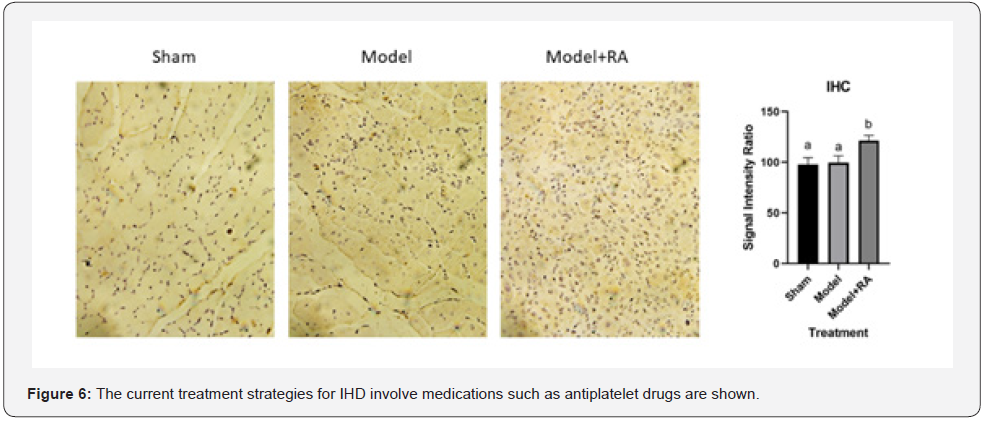
To investigate the possible therapeutic potential of RA in ischemic diseases, we utilized a mouse hindlimb ischemia model. After the femoral artery ligation, blood flow was significantly reduced in the ischemic hindlimb (Figure 4A). Serial analysis of hindlimb perfusion by laser Doppler imaging revealed significantly more blood flow recovery in the RA supplemented mice compared to the Vehicle-treated mice (0.85±0.05 vs. 0.71±0.10 on day 3, 0.94±0.10 vs. 0.75±0.08 on day 7). Our microangiography analysis showed that ligation resulted in collateral artery development in ischemic hindlimbs, which is an adaptive pathway for tissue salvage during arterial occlusion. Intriguingly, RA supplementation led to a significant increase in angiographically visible collateral vessels with a typical “spiral” appearance (Figure 4B). After further exploring the underlying mechanism, we found that CD31, a specific endothelial cell marker, significantly increased in the gastrocnemius muscle of the mice with RA supplementation, compared to the mice in the vehicle group (Figure 4C). Such results suggest that RA may enhance blood supply by increasing the micro vessel density of the hindlimb to mitigate ischemic conditions. Therefore, our in vivo results indicate that RA may have therapeutic potential for ischemic diseases by promoting angiogenesis or astrogenesis.
P-JAK and p-STAT3 were enhanced after rotundic acid treatment
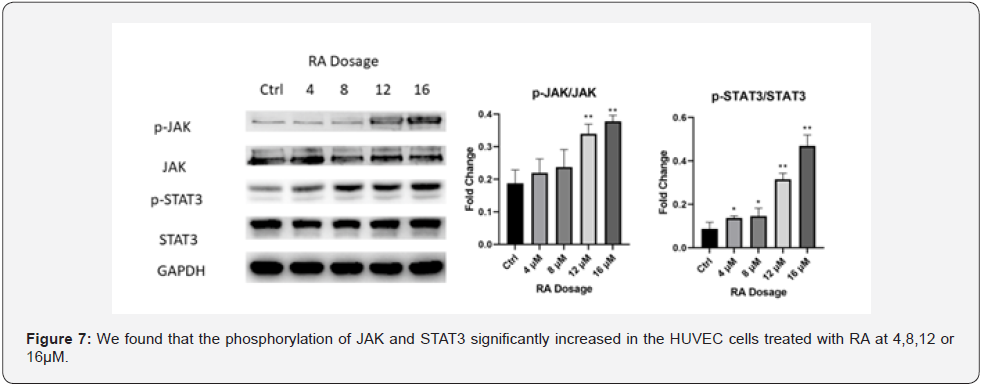
After further exploring the underlying molecular mechanism by which RA enhanced angiogenesis, we found that the phosphorylation of JAK and STAT3 significantly increased in the HUVEC cells treated with RA at 4,8,12 or 16μM (Figure 5). This suggests that RA may improve angiogenesis by modulating the JAK-STAT3 signalling pathway.
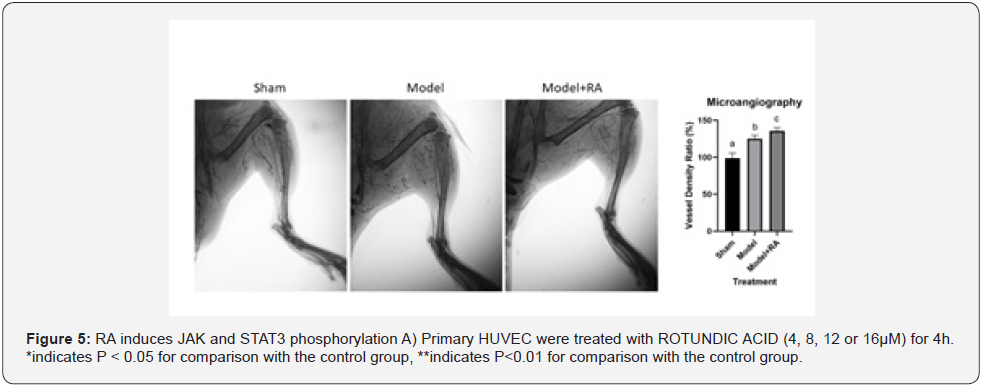
Discussion
The current treatment strategies for IHD involve medications such as antiplatelet drugs (aspirin), antilipemic drugs (statins), or vasodilators, or revascularization strategies, such as PCI or coronary artery bypass grafting (CABG), prompting coronary reperfusion to save viable cardiomyocytes Yellon & Hausenloy [23]. However, during the treatment, myocardial reperfusion injuries may occasionally occur, mainly due to microcirculatory dysfunction caused by oxidative stress, calcium overload, and inflammation Park & Yellon et al. [23,24]. In addition, considering the population who are vulnerable to PCI or coronary artery bypass grafting procedures, such as elderly patients Peterson et al. [25] novel strategies need to be explored to reduce the adverse effects and procedural risks. In the former study, FGF or VEGF was injected as a plasmid into the ischemic myocardium of IHD patients, thereby reducing symptoms and improving myocardial perfusion by improving angiogenesis Ng & D’Amore [26]. Such results highlighted the potential efficacy of angiogenesismediated therapy in promoting the outcome of the IHD treatment. Phytochemicals have shown various beneficial effects against multiple diseases Cheng & Balbuena et al. [27,28]. In the past three decades, the cardiovascular protective effects of Ilex pubescens have been studied de Oliveira et al. [29]. It has been shown that the aqueous extract of Ilex pubescens (100 mg/kg) could increase the coronary flow and significantly improve the hypoxia tolerance of the rats Fang et al. [30]. Additionally, researchers showed that Ilex pubescens treatment at 100 and 200 mg/kg substantially mitigated focal cerebral ischemia/reperfusion injuries and reduced proinflammatory cytokine levels Fang et al. [31] Interestingly, the extracted flavonoids from Ilex pubescens also displayed beneficial effects in enhancing blood circulation and antioxidative capacities in the rats with cerebral ischemia reperfusion injuries Miao et al. [32]. Since RA is one of the main active ingredients of Ilex pubescens, we believe that it is capable of improving the outcomes of IHD by reducing myocardial reperfusion injury. Interestingly, our results were inconsistent with the prior study showing that RA inhibited HUVEC proliferation and migration in vitro. Two reasons that may explain the discrepancy from the studies: first, they did not provide the source of RA. We purchased the RA from Push Biotechnology, which was a pure compound. If they extracted RA in-house, there would be a mixture of other phytochemicals, which may impose inhibitory effects to the cell viability. Secondly, the dosage used in the prior report was high (10-60μM), but the dosage of RA in our current project was 4 - 16μM, which was one third of the dosage used in the prior study. However, one of the (Figures 6 & Figure 7) limitations in this study is that we were not able to determine whether the efficacy of RA in preventing cerebral ischemia reperfusion injuries was comparable with that of Ilex pubescens. Since the multiple phytochemicals in a whole food may generate synergistic effects and display more potent beneficial effects than single compound Li & Kan et al. [33,34], it would be interesting to explore if the angiogenesis-promoting effects of Ilex pubescens stemmed from itself or from RA. Another limitation is that there was a lack of rationale of selecting day-10 to observe microangiography. Therefore, multiple time-points will be adapted to observe microangiography in our next steps. In this study, we demonstrated that RA promoted proliferation, adhesion, migration, and tube formation of primary HUVEC under both normal and ischemic conditions. Our data also showed that RA promoted angiogenesis in vivo. In particular, using a hindlimb ischemia model, we revealed the therapeutic potential of RA against IHD. JAK-STAT3 also plays an important role in cell survival. This pathway was first elucidated as a key transcription factor in tumour angiogenesis and was subsequently shown to be required after ischemic injury Kishore & Verma [35], Harhous et al. [36]. Previous studies reported that STAT3 knockout in cardiomyocytes resulted in reduced left ventricular capillarization Hilfiker- Kleiner et al. [37]. In contrast, cardiac-specific activation of STAT3 was demonstrated to promote cardiac vascularization Osugi et al. [38]. Given the important role of STAT3 in angiogenesis, we first examined whether RA had any effect on STAT3 activity and found that RA induced JAK and STAT3 phosphorylation. Therefore, our results suggested that JAK-STAT3 might be a key signalling pathway by which RA induced angiogenesis. However, our current study only showed an association between JAK-STAT3 and angiogenesis, not causality. Thus, in the following work, we will investigate the causality of JAK-STAT3 activation in RA-induced angiogenesis by using a STAT3-knockout mouse model. In addition, JAK/STAT3 has been suggested to play a primary role in inflammationassociated pathogenic angiogenesis, such as retinopathy, and this might yield side effects of RA. Since the activation of JAK/STAT3 is a [39,40] two-sided sword that could either mediate rescuing ischemic tissue or induce pathogenic angiogenesis, it is critical to monitor the degree of JAK/STAT3 activation, which would be an interesting research topic that warrants further investigation. Nevertheless, in our current study, we did not observe such side effects in the animals.
Conclusion
This is the first study that shows RA promotes angiogenesis under both normal and ischemic conditions. In addition, this is also the first instance a study has elucidated that the molecular mechanism of RA improves IHD treatment outcomes by highlighting JAK_STAT3-mediated angiogenesis. Further clinical trials are warranted to verify the clinical implication.
References
- Finegold JA, Asaria P, Francis DP (2013) Mortality from ischaemic heart disease by country, region, and age: statistics from World Health Organisation and United Nations. Int J Cardiol 168(2): 934-945.
- Ong AT, Serruys PW (2006) Complete revascularization: coronary artery bypass graft surgery versus percutaneous coronary intervention. Circulation 114(3): 249-255.
- Foussas SG, Tsiaousis GZ (2008) Revascularization treatment in patients with coronary artery disease. Hippokratia 12(1): 3-10.
- Stone GW, Webb J, Cox DA, Brodie BR, Qureshi M, et al. (2005) Distal microcirculatory protection during percutaneous coronary intervention in acute ST-segment elevation myocardial infarction: a randomized controlled trial. JAMA 293(9): 1063-1072.
- Ergul A, Alhusban A, Fagan SC (2012) Angiogenesis: a harmonized target for recovery after stroke. Stroke 43(8): 2270-2274.
- Adair TH, Montani JP (2010) Overview of angiogenesis. In: Angiogenesis. California, USA: Morgan & Claypool Life Sciences pp. 1-12.
- Cheng J, Miao B, Hu KQ, Fu X, Wang XD (2018) Apo-10′’-lycopenoic acid inhibits cancer cell migration and angiogenesis and induces peroxisome proliferator-activated receptor γ. J Nutr Biochem 56: 26-34.
- Poustchi F, Amani H, Ahmadian Z, Niknezhad SV, Mehrabi S, et al. (2021) Combination Therapy of Killing Diseases by Injectable Hydrogels: From Concept to Medical Applications. Adv Healthcare Mater 10(3): 200-210.
- Loh CY, Arya A, Naema AF, Wong WF, Sethi G, et al. (2019) Signal transducer and activator of transcription (STATs) proteins in cancer and inflammation: functions and therapeutic implication. Front Oncol 9: 48.
- Chen Z, Han ZC (2008) STAT3: a critical transcription activator in angiogenesis. Med Res Rev 28(2): 185-200.
- Johnson DE, O Keefe RA, Grandis JR (2018) Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat Rev Clin Oncol 15(4): 234-248.
- Cheng J, Eroglu A (2021) The Promising promising effects of astaxanthin on lung diseases. Adv Nutr 12(3): 850-864.
- Yamaoka K, Saharinen P, Pesu M (2004) Holt VET, 3rd, Silvennoinen O, O Shea JJ. The Janus kinases (Jaks). Genome Biol 5(12): 253.
- Huang G, Yan H, Ye S, Tong C, Ying QL (2014) STAT3 phosphorylation at tyrosine 705 and serine 727 differentially regulates mouse ESC fates. Stem Cells 32(5): 1149-1160.
- Wang C, Chao Z, Sun W, Wu X, Ito Y (2014) Enrichment and purification of pedunculoside and syringin from the barks of Ilex rotunda with macroporous resins. J Liq Chromatogr Relat Technol 37(4): 572-587.
- Liu C, Shen YJ, Tu QB, Zhao YR, Guo H, et al. (2018) Pedunculoside, a novel triterpene saponin extracted from Ilex rotunda, ameliorates high-fat diet induced hyperlipidemia in rats. Biomed Pharmacother 101: 608-616.
- Haraguchi H, Kataoka S, Okamoto S, Hanafi M, Shibata K (1999) Antimicrobial triterpenes from Ilex integra and the mechanism of antifungal action. Phytother Res 13(2):151-156.
- Xie JB, Bi ZM, Li P (2003) HPLC-ELSD determination of triterpenoids and triterpenoid saponins in Ilex pupurea leaves. Yao Xue Xue Bao 38(7): 534-536.
- Nan ML, Wang X, Li HJ, Yu DH, Sun WY, et al. (2019) Rotundic acid induces Cas3-MCF-7 cell apoptosis through the p53 pathway. Oncol Lett 17(1): 630-637.
- Roy G, Guan S, Liu H, Zhang L (2019) Rotundic acid induces DNA damage and cell death in hepatocellular carcinoma through AKT/mTOR and MAPK pathways. Front Oncol 9: 545.
- Cheng J, Liu C, Hu K, Greenberg A, Wu D, et al. (2017) Ablation of systemic SIRT1 activity promotes nonalcoholic fatty liver disease by affecting liver-mesenteric adipose tissue fatty acid mobilization. Biochim Biophys Acta Mol Basis Dis 1863(11): 2783-2790.
- Kan J, Cheng J, Guo J, Chen L, Zhang X, et al. (2020a) A novel botanical combination attenuates light- induced retinal damage through Antioxidant antioxidant and prosurvival mechanisms. Oxid Med Cell Longev 7676818.
- Yellon DM, Hausenloy DJ (2007) Myocardial reperfusion injury. N Engl J Med 357(11): 1121-1135.
- Park JL, Lucchesi BR (1999) Mechanisms of myocardial reperfusion injury. The Annals of thoracic surgery 68(5): 1905-1912.
- Peterson ED, Alexander KP, Malenka DJ, Hannan EL, O Conner GT, et al. (2004) Multicenter experience in revascularization of very elderly patients. Am Heart J 148(3): 486-492.
- Ng YS, D Amore PA (2001) Therapeutic angiogenesis for cardiovascular disease. Curr Control Trials Cardiovasc Med 2(6): 278-285.
- Cheng J, Miller B, Eroglu A (2020b) The efficacy of carotenoids in DNA repair in lung cancer. Curr Dev Nutr 4(2): 99.
- Balbuena E, Cheng J, Eroglu A (2021) The role of lycopene in chronic lung diseases. Antioxidants. In: Antioxidants. London, UK: IntechOpen.
- De Fang X, Li Y, Qiao J, Guo Y, Miao M (2017) Neuroprotective effect of total flavonoids from Ilex pubescens against focal cerebral ischemia/reperfusion injury in rats. Molecular medicine reports 16(5): 7439-7449.
- JR, Camargo SEA, De Oliveira LD (2019) Rosmarinus officinalis L. (rosemary) as therapeutic and prophylactic agent. J Biomed Sci 26(1): 5.
- Fang X, Li Y, Zheng Y, Wang Y, Feng S, et al. (2020) Ethanol extracts from Ilex pubescens promotes cerebral ischemic tolerance via modulation of TLR4-MyD88/TRIF signaling pathway in rats. J Ethnopharmacol 256: 112680.
- Miao M, Cao L, Xu K, Xin W, Zheng Y (2017) Intervention action of total flavonoids from root of Ilex pubescens in cerebral ischemic tolerance with blood stasis. Saudi J Biol Sci 24(3): 729-736.
- Li CC, Liu C, Fu M, Hu KQ Aizawa K, et al. (2018) Tomato powder inhibits hepatic steatosis and inflammation potentially through restoring SIRT1 activity and adiponectin function independent of carotenoid cleavage enzymes in mice. Mol Nutr Food Res 62(8): 1700738.
- Kan J, Cheng J, Xu L, Hood M, Zhong D, et al. (2020b) The combination of wheat peptides and fucoidan protects against chronic superficial gastritis and alters gut microbiota: a double-blinded, placebo-controlled study. Eur J Nutr 59(4): 1655-1666.
- Kishore R, Verma SK (2012) Roles of STATs signaling in cardiovascular diseases. JAKSTAT 1(2): 118-124.
- Harhous Z, Booz GW, Ovize M, Bidaux G, Kurdi M (2019) An update on the multifaceted roles of STAT3 in the heart. Front Cardiovasc Med 6: 150.
- Hilfiker KD, Hilfiker A, Drexler H (2005) Many good reasons to have STAT3 in the heart. Pharmacol Ther 107(1): 131-137.
- Osugi T, Oshima Y, Fujio Y, Funamoto M, Yamashita A, et al. (2002) Cardiac-specific activation of signal transducer and activator of transcription 3 promotes vascular formation in the heart. J Biol Chem 277(8): 6676-6681.
- Cheng J, Balbuena E, Miller B, Eroglu A (2021) Bridge the gap - the efficacy of -carotene in immune sensing of LPS and intestinal barrier integrity in colon epithelial cells. Curr Dev Nutr 5(2): 71.
- Cheng J, Miller B, Balbuena E, Eroglu A (2020a) Lycopene protects against smoking-induced lung cancer by inducing base excision repair. Antioxidants (Basel) 9(7): 643-657.






























