Method Development and Validation of in Vitro Release Test for Fluorometholone Ophthalmic Suspension using Franz Diffusion Cell System
Hicran Gür Dereli*, Ecem Kuzpınar Şengün, Ceren Okuyucu Genç, Banu Özkırım Arslan and Mine Uz Gökalp
Department of Analytical Research & Development, Abdi İbrahim Research & Development Center (R&D), Turkey
Submission: November 02, 2021; Published: November 18, 2021
*Corresponding author: Hicran Gür Dereli, Department of Analytical Research & Development, Abdi İbrahim Research & Development Center, Turkey
How to cite this article:Hicran G D, Ecem K Ş, Ceren O G, Banu Ö A, Mine U G. Method Development and Validation of in Vitro Release Test for Fluorometholone Ophthalmic Suspension using Franz Diffusion Cell System. Glob J Pharmaceu Sci. 2021; 9(2): 555758. DOI: 10.19080/GJPPS.2021.09.555758.
Abstract
The in Vitro release method to be used must have dissolution conditions capable of distinguishing even small changes in the formulation or production process required to develop the generic product. It should measure the amount of active substance released during the in Vitro release test in an accurate, robust and repeatable manner. The aim of the study is to demonstrate the similarity by comparing the test and reference product for Ophthalmic Suspensions containing Fluorometholone (FLU) with the Bootstrap F2 method. In the experiment, pH 7.4 ATF / STF (Artificial / Simulated Tear Fluid) medium was used. Since the FLU molecule is not soluble in water, 2% Sodium dodecyl sulphate was applied as a surfactant to increase the solubility of FLU. By Franz Diffusion Cell method, using Nylon Membrane Disc Filter an in Vitro release profile of 48 hours at 37°C at 750 rpm rotation speed was performed. Last but not the least, a selective and validated HPLC method was used for the analysis of released drug molecules .
Keywords: In vitro release test; Suspension; Franz diffusion cell; Fluorometholone; Method validation
Introduction
In vitro release testing (IVRT) is used to observe the release and diffusion of drug products and it is considered an essential tool in formulation development [1,2]. IVRT has also been used to detect formulation changes, and its use to waive bioequivalence studies following post-approval modifications to a product has been accepted significantly [3]. The IVRT application has many additional benefits, such as its use in product development, determination of critical production variables, simulation of in Vitro performance, and product performance evaluation, including uniformity of each batch number [4,5]. The IVRT was created by the USP as a complementary method and complementary devices and procedures for these tests are described in USP General Section <1724> [6]. In vivo refers to the tests, experiments and procedures that researchers perform on a living organism such as a human, laboratory animal or plant. In vitro describes medical procedures, tests and experiments performed outside of a living organism. The In vitro study is performed in controlled environments such as a test tube, petri dish etc. and is a simpler research methodology. Researchers can perform more detailed analyzes and study biological effects on more in vitro subjects than in animal or human experiments. For this reason, it is more preferred than in Vitro studies [7]. There are many dissolution test devices developed over the years. The Vertical Diffusion Cell (VDC) proposed by the USP is one of the more widely accepted devices [8]. In order to in Vitro diffusion studies and is by far the most widely used in Vitro model for the study of drug release. These cells can be used with numerous modifications in basic design. For this reason, a release test for FLU agent has been developed easily in ophthalmic product using Franz diffusion cells too [9]. During the release in the Franz Diffusion Cell, the diffusion of molecules from a region of high concentration (donor room) to a region of low concentration (receptor chamber) occurs. The membrane acts as a rate limiting step to maintain a diffusional collapse. As the drug concentration in the donor compartment decreases, the drug concentration in the receptor environment increases [4]. FLU is a widely used anti-inflammatory ophthalmic formulation that elicits a lower ocular hypertensive response than other glucocorticoid drugs. This serves to mitigate the risk of steroid induced glaucoma. FLU is a corticosteroid drug commonly used for inflammatory diseases and dry eye syndrome. FLU solution is difficult to prepare due to its low water solubility. The commercially available FLU formulation is a suspension [10].
The fundamental purpose of this article is to present the process of IVRT method development with Franz Diffusion Cell device in FLU containing Ophthalmic Eye Suspensions, the methodology used with details about the application techniques, equipment, and the published results. The in Vitro release method to be developed must have dissolution conditions capable of distinguishing even minor changes in the formulation or production process required for generic product development. It should measure the number of active substances released during the dissolution test in an accurate, robust, selective and repeatable manner. In this study, a method has been developed that can detect even minor changes that may occur in the formulation and production process and the accuracy, repeatability and selectivity of the method have been proven by validation studies.
Materials and Methods
Materials
FLU WS, FLU 21-Hydroxy Analog Impurity Standard, FLU 21-Mesiloxy Analog Impurity Standard, FLU 9Br Analog Impurity Standard, FLU Delta 9.11 Impurity Standard and FLU 9β, 11β Epoxy Analog Impurity Standard were purchased from API supplier. FLU Impurity A was obtained from USP®. Sodium Dodecyl Sulfate (SDS), acetonitrile and sodium chloride were purchased from J.T.Baker®. Methanol, sodium perchlorate monohydrate, perchloric acid and calcium chloride were received from Merck®. Also, hydrochloric acid and sodium bicarbonate were purchased from Sigma Aldrich® and STB®, respectively. All materials detailed above were used in analytical grade.
Sterile eye drop containing fluorometholone
Sterile Eye Drop, Suspension product was developed by Abdi İbrahim R&D Center, Turkey. Efemoline Sterile Eye Drop, Suspension was used as reference product was obtained from Thea Pharma Pharmaceutical Trade Limited Company, Turkey.
HPLC analysis of fluorometholone
FLU assay analyzes were completed using a Waters HPLC system with UV detector conditioned at 210nm.
The mobile phase was a mixture of acetonitrile and buffer solution (55/45, v/v). The buffer solution prepared by weighing 5.0 g of sodium perchlorate monohydrate reagent (NaHClO4.H2O) into a 1000mL graduated flask. After dissolving in 900 mL of water, the pH of the solution was adjusted to 2.9±0.1 with 20% (v / v) perchloric acid solution and made up to its volume with water. Hichrom Lichrospher, CN (250 x 4.6mm, 5μm; Merck®) column was used at a flow rate of 1.5mL/min, the column temperature was set to 30°C, while the sample temperature was set to 25°C. The injection volume was chosen as 20μL. Chromatographs were processed using Empower® 3Build 3471 software.
In vitro release testing of suspensions
The Vertical Franz diffusion Cells with a volume of 7mL (Model A, Hanson ®) were used to determine the in vitro drug release of the FLU suspensions. The release test was carried out in pH 7.4 artificial/simulated tear fluid (ATF/STF) (0.0067% NaCl, 0.002% NaHCO3 and 0.0000604% CaCl2) which was containing 2.0 % SDS. Freshly prepared medium was placed in the receptor chamber and the bath temperature was allowed to reach 37±0.5°C. Pre- Sterilized Nylon-66 Membrane Disc Filter (MDI®, 0.45 μm pore size) was used as an artificial membrane and the membrane was saturated in STF medium for about 30 minutes before analysis was initiated. Then, a saturated membrane, white silicon ring, screwed glass cell, metal cell ring to was used sequentially were placed on each Franz diffusion cell and the parts were fixed together using the clamps. The stirring speed of the Franz diffusion cells was set at 750 rpm. With the help of an automatic collector, 1.5mL samples were taken at the 1st, 2nd, 3rd, 4th, 5th, 6th, 7th, 8th, 12th, 18th, 24th, 36th and 48th hours. Sample quantities are automatically completed with media on volume by device. The collected samples were analyzed on the HPLC instrument by a method validated at 210nm wavelength.
In Vitro Release Test Method Development
HPLC method
Even if a validated method of analysis is available for the drug active substance and its related compounds, it may not be suitable for the analysis of these compounds in the selected in vitro release medium. To ensure the quality of the results of in vitro release studies, the method developed requires a full validation. The results of the completed Validation parameters belong to developed method are shared in the section of Method Validation Study.
Selection of membrane
The following properties of the membrane preferred for in vitro release tests should have:
a) Provide an inert retention surface for formulations and should not be a barrier to release.
b) Since the drug active molecule is “released” from the dosage form of the ingredient, it should allow it to spread easily into the receiving environment.
c) There should be no physical or chemical interaction between the membrane and the formulation.
d) The membrane must not contain any “leachable substance” that could affect the assay results of the agent.
Membranes should show the minimum resistance of the diffusion mixture and it should not be effective in determining the release rate. Frequently used synthetic membrane varieties are Silicone, Cellulose, Polysulfone, Nylon membrane, Dialysis bag, GFD / D-Filter, etc. When choosing a synthetic membrane, the following features should be considered:
Pore size and product viscosity: Typically, 0.45μm pore size is sufficient. However, materials with high viscosity may require a larger pore size to ensure diffusion is not restricted.
Hydrophobic and hydrophilic materials: Typically, hydrophobic membrane is used for hydrophilic API and hydrophilic membrane is used for hydrophobic API. This reduces the likelihood that the API with the membrane will interfere.
Easy accessibility as a commercial: Membranes that are difficult to obtain may cause delays in testing. For this reason, priority should be given to using easily accessible membranes [11]. In the light of the information given above, 3 different commonly used synthetic membranes were selected and 48-hours in vitro release studies were completed keeping all other parameters of the in vitro release medium constant to be used in this study. Dialysis Membrane Filter, MDI 0.45um Filter and PALL 0.45um Filter were made to work with. The analysis results given in Table 1 were compared, it is seen that the filter with the highest permeability was the MDI 0.45um Filter.
Selection of receiving medium
While determining the optimum solution environment,the medium that simulates the application site of the drug and dissolves the active substance best is determined. In IVRT, the primary criterion is that it simulates the in vitro conditions of the working medium as much as possible, rather than the solubility of the active substance reaching 100%. In the literature, simulated tear fluid is often the first choice for in vitro release studies into the eye product [12]. However, when using this medium, optimum dissolution for the product may not always be achieved. The most important factor for the selection of the receiving medium is the solubility of the active ingredient in the medium. While it is desirable to have a receptor medium similar to the physiological state of the eye, it is also necessary to ensure that the receptor environment can be measured without suppressing the release of the drug. In order to prevent the suppressing the release of the drug, surfactants and organic solvents can be used to increase solubility [13,14]. The receiving medium must provide a dissolution medium by creating a profile that increases over time for the active substance [3]. In addition to all these, the pH of the medium is another important factor to be considered. The choice of the pH of the aqueous component of the medium should be based on the pH of the formulation, the pH solubility profile of the active ingredient, and the pH of the target membrane [15]. While a IVRT method development phase, the following specific equipment related parameters should be considered.
a) Temperature: It has been observed in the literature that 37°C is frequently used for eye products. [16,17].
b) Stirring : 200 to 900 rpm [18].
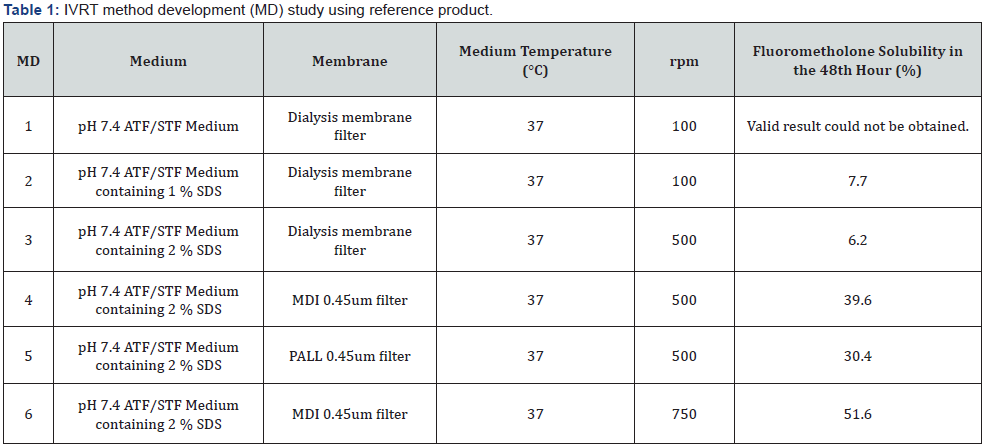
As a result of the method development studies summarized in Table 1, it was decided to the final IVRT method by evaluating all parameters and results. Method conditions are given in below.
IVRT Conditions
Media : pH 7.4 SLS/ATF
Membrane : MDI 0.45um filter
Media Temperature : 37 °C
FDC Rotation : 750 rpm
Sampling Volume : 1.5 mL
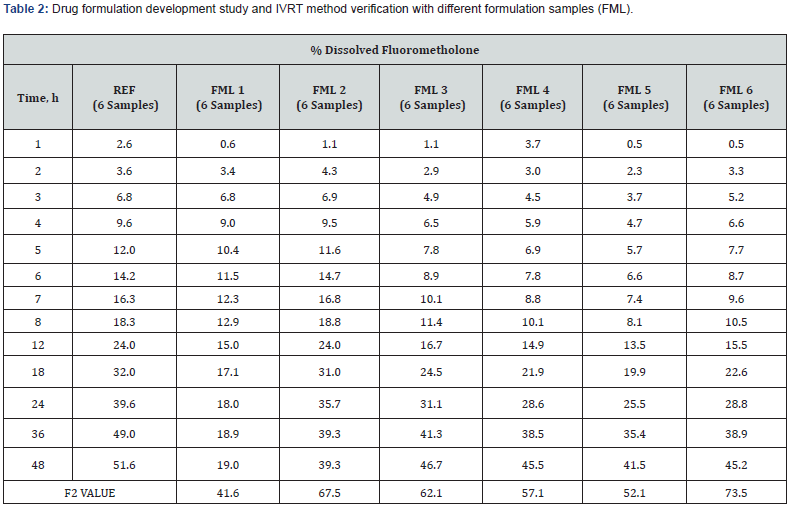
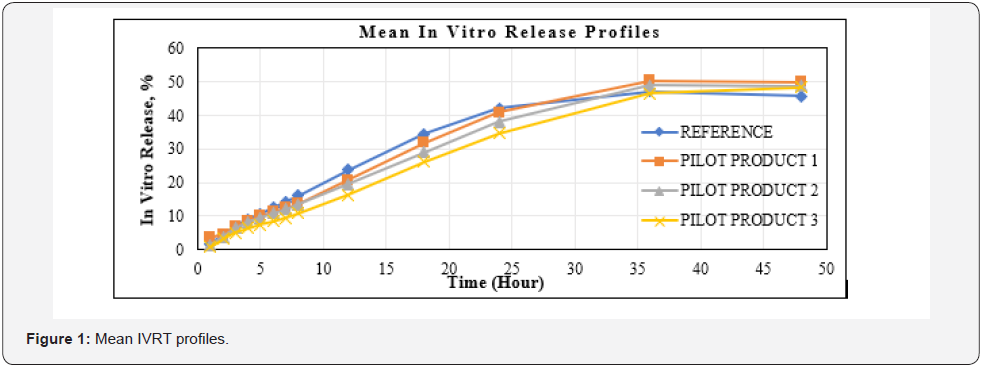
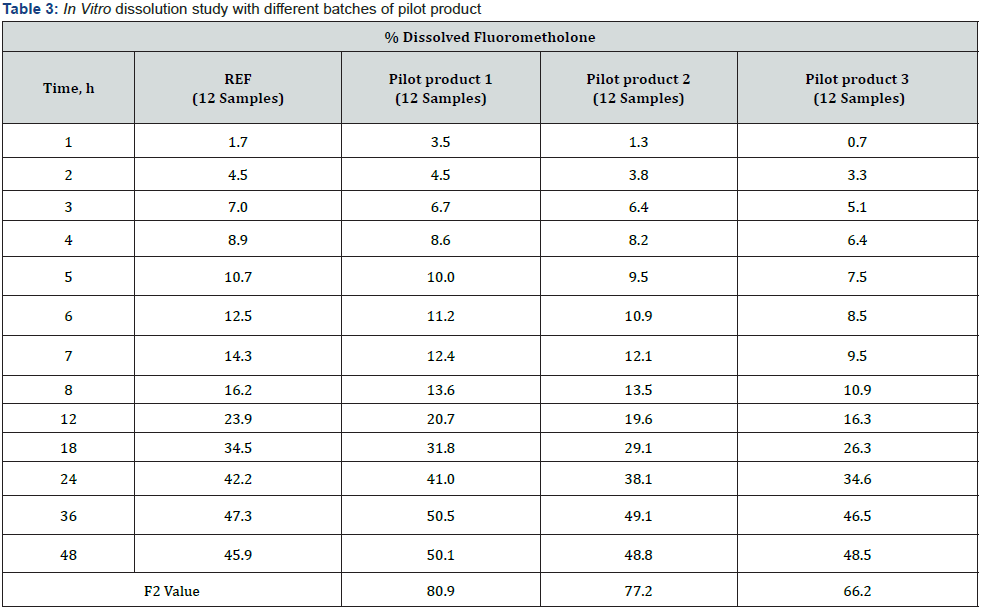
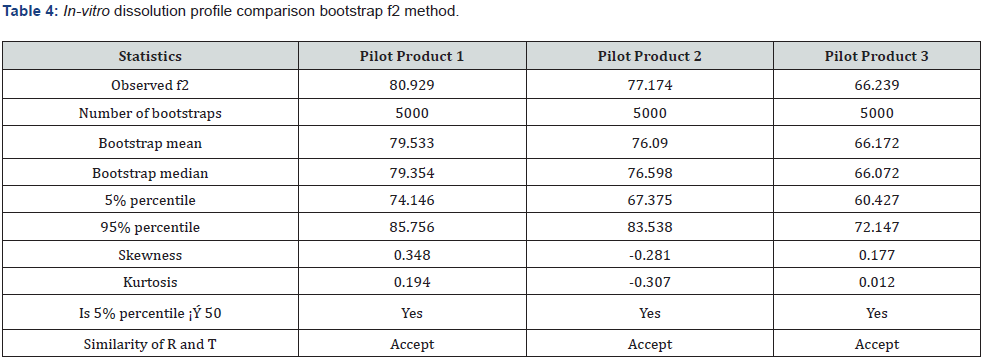
IVRT is a critical test for evaluating the safety, efficacy, and quality of drug delivery systems, but there are no complementary or regulatory standards. For method development, it is important to use more complex environments that simulate the composition of biological fluids to which the drug product will be administered and / or absorbed. The surfactant was added to the STF medium used in this study. Thus, qualitative and quantitative evaluable results were obtained. At the end of the 48th hour, over 45% in vitro release results were obtained [19]. In Table 2, release profiles and F2 results are given to compare the similarity between trial productions (FML) made during the formulation development phase and the reference product. IVRT studies were carried out with an optimized method. Using the developed IVRT method, test products with different unit formulas were analyzed and it was observed that the method was distinctive in each formulation change. It can be said that the in vitro release profiles of formulations with an F2 result greater than 50 are similar between the reference and test samples.
Calculations
In the release test performed using the Franz diffusion cell device, samples obtained from the 48-hours profile study were analyzed by the assay method using the HPLC device. The cumulative calculation of the obtained data was made as follows.
An0 (in the nth period, uncorrected% dissolution rate result)
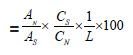
AN : Fluorometholone peak area obtained from sample solution
AS : Fluorometholone peak area average on chromatograms from standard solution
CS : Fluorometholone concentration in standard solution, mg/mL
CN : Fluorometholone concentration in sample solution, mg/mL
L : Label (1.0 mg/mL)
An (in the nth period, corrected% dissolution rate result) =
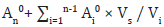
An0 : in the nth period, uncorrected% dissolution rate result (%)
Ai0 : in the ith period, uncorrected% dissolution rate result (%)
Vs : Automatic withdrawal volume reduced in each period (1.5 mL)
Vi : in the ith period, total media volume (7 mL)
n : Number of periods
Results and Discussion
Materials
As a result of the formulation development studies conducted to obtain a similar release profile with the reference product, the final unit formula was decided by considering the F2 values, and then pilot production was carried out. The IVRT method validation studies have been completed by using the pilot product and reference product. Release study for three different batches test products and a reference product having the same manufacturing method were performed using the Franz Cell in vitro release testing method which was validated. Quantitation of drug substance release values (%) in medium were calculated at defined periods. The reference and generic product were compared using Bootstrap methodology which is applied to derive confidence intervals for F2 (Similarity Factor) based on quantities of in vitro release similarity, and this approach could be considered the preferred method over F2 [20]. In order to compare the individual release profiles, Figure 1 are illustrated in Table 3 & 4.
Method Validation Study
Materials
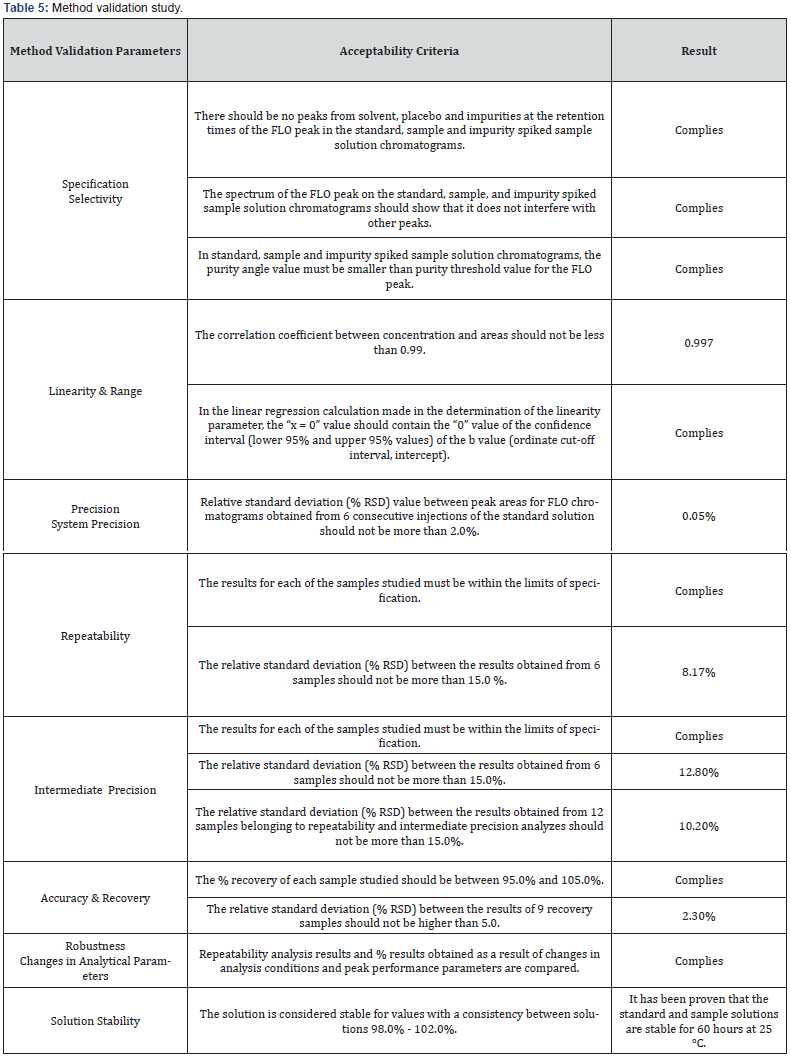
Specificity, linearity and range, repeatability, precision, accuracy and recovery, robustness method validation parameters were applied to the IVRT method using the Franz Diffusion Cell device for Fluorometholone in the Sterile Eye Drops, Suspension product, and the method validation was successfully completed. According to validation studies results: In Vitro Release Test method is specific for Fluotometholone and robust in view of changes to flow rate, wavelength, column temperature. The linearity, accuracy, and recovery of the method in the range of 0.001428 mg/mL to 0.214230 mg/mL has been proved. The correlation coefficient between the concentration and areas in the concentration ranges of 0.001428 mg/mL to 0.214230 mg/mL (r2 = 1.00) is not less than 0.99 as (Table 5) it should be.
Conclusion
Materials
In conclusion, IVRT method was developed and applied for three different batches test product and reference product. Most importantly, the IVRT study applied is a sensitive and reproducible method used to evaluate product similarities for ophthalmic suspensions. Method reliability was proved by method validation study.
Acknowledgement
Materials
The authors thank Dr. Mustafa Uygun in the Department of Research & Development in Abdi İbrahim Pharmaceutical Company for their support and useful discussion. This work was supported by Abdi İbrahim Pharmaceutical Company, Turkey.
References
- Ayres JW, Laskar PA (1974) Diffusion of Benzocaine from Ointment Bases. J Pharm Sci 63(9): 1402-1406.
- Zatz JL (1995) Drug Release from Semisolids: Effect of Membrane Permeability on Sensitivity to Product Parameters. Pharm Res 12(5): 787-789.
- SUPAC-SS (1997) Nonsterile Semisolid Oral Dosage Forms, Scale-Up and Post approval Changes: Chemistry, Manufacturing, and Controls; In Vitro Dissolution Testing and In Vivo Bioequivalence Documentation; Guidance for Industry. U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), U.S. Government Printing Office, Washington, DC, USA.
- Olejnik A, Goscianska J, Nowak I (2012) Active Compounds Release from Semisolid Dosage Forms. J Food Drug Anal 101(11): 4032-4045.
- Chang R, Raw A, Lionberger R, Yu L (2013) Generic Development of Topical Dermatologic Products: Formulation Development, Process Development, and Testing of Topical Dermatologic Products. AAPS J 15(1): 41-52.
- Semisolid Drug Products (2016) Performance Tests. In: The United States Pharmacopoeia and National Formulary USP 37-NF 32. The United States Pharmacopoeial Convention Inc Rockville MD, pp. 1273-1284.
- https://mpkb.org/home/patients/assessing_literature/in_vitro_studies?do=admin.
- Salamanca CH, Ocampo AB, Lasso JC, Camacho N, Yarce CJ (2018) Franz Diffusion Cell Approach for Pre-Formulation Characterisation of Ketoprofen Semi-Solid Dosage Forms. Colombia 10(3).
- Semisolid Drug Products (2016) Performance Tests. In: The United States Pharmacopoeia and National Formulary USP 39-NF 34. The United States Pharmacopeial Convention, Inc Rockville, MD 38, pp. 1869-1881.
- K Baba, Hashida N, Tujikawa M, Quantock AJ, Nishida K (2020) The generation of fluorometholone nanocrystal eye drops, their metabolization to dihydrofluorometholone and penetration into rabbit eyes.
- KD Thakker, WH Chern (2003) Development and Validation of In Vitro Release Tests for Semisolid Dosage Forms-Case Study.
- MG Hewitt, PWJ Morrison, HM Boostrom (2020) In Vitro Topical Delivery of Chlorhexidine to the Cornea: Enhancement Using Drug-Loaded Contact Lenses and beta-Cyclodextrin Complexation, and the Importance of Simulating Tear Irrigation. Mol Pharmaceutics 17(4): 1428-1441.
- (1092) The Dissolution Procedure: Development and Validation.
- DT Birnbaum, JD Kosmala (1999) Controlled release of b-estradiol from PLAGA microparticles: The effect of organic phase solvent on encapsulation and release, USA.
- I Kanfer, S Rath, P Purazi, NA Mudyahoto (2017) In Vitro Release Testing of Semi-Solid Dosage Forms.
- SC Chattaraj, I Kanfer (1995) Release of acyclovir from semi-solid dosage forms: A semi-automated procedure using a simple plexiglass flow-through cell. Int J Pharm 125(2): 215-222.
- RB Saudagar, PS Deore (2016) Formulation and Characterization of Fluorometholone Nanosuspension for Ophthalmic Drug Delivery by Precipitation Method, India.
- P Naik, SM Shah (2016) Influence of Test Parameters on Release Rate of Hydrocortisone from Cream: Study Using Vertical Diffusion Cell.
- European Medicines Agency (2010) Guideline on the Investigation of
- European Medicines Agency (2018) Question and answer on the adequacy of the Mahalanobis distance to assess the comparability of drug dissolution profiles.






























