Simultaneous and Direct Estimation of Glymes in Pharmaceuticals and Biopharmaceuticals by Headspace Gas Chromatograph Coupled with Mass Spectrometer
Balaji Nagarajan* and Sayeeda Sultana
St. Peter’s Institute of Higher Education and Research, India
Submission: May 04, 2021; Published: May 19, 2021
*Corresponding author: Balaji Nagarajan, St. Peter’s Institute of Higher Education and Research, Avadi, Chennai, Tamil Nadu, India
How to cite this article:Balaji N, Sayeeda S. Simultaneous and Direct Estimation of Glymes in Pharmaceuticals and Biopharmaceuticals by Headspace 0099 Gas Chromatograph Coupled with Mass Spectrometer. Glob J Pharmaceu Sci. 2021; 8(3): 555740. DOI: 10.19080/GJPPS.2021.08.555740.
Abstract
The analytical approach used for the effective trace-level determination of monoglyme and diglyme in pharmaceuticals and biopharmaceuticals employs a simple sample preparation technique. This method involves headspace gas chromatograph coupled with mass spectrometer and is based on an oven scheduling approach which uses helium gas as the mobile phase; a complete sample analysis is conducted in less than 1h which does not include the injection time of standard replicates. The detection and quantitation limits of this advanced technique are 0.01 and 0.03μg/mL for both monoglyme and diglyme, respectively. The successful separation of monoglyme and diglyme was confirmed by determining their corresponding specific molecular masses and its fragments. This method is precise, sensitive, accurate, linear, and robust as determined by method validation performed according to the International Council on Harmonization validation guidelines Q2 (R1) and United States Pharmacopoeia. In this study, monoglyme and diglyme were not detected in the samples of pharmaceuticals and biopharmaceuticals when using this technique. By headspace chromatograph coupled with mass spectrometer, monoglyme and diglyme can be easily estimated at trace levels during the commercial preparation of biologically and chemically active substances in both the biopharmaceutical and pharmaceutical industries. The main benefit of this research work is to get the GC-FID and GCMS data simultaneously to determine monoglyme and diglyme at micro-level in Pharmaceuticals and Biopharmaceuticals.
Keywords: GC-MS; Headspace gas chromatography; Glymes; Class-2 solvent; Biopharmaceuticals; Pharmaceuticals
Abbreviations: GC: Gas Chromatography; GC-HS: Headspace Gas Chromatography; FID: Flame Ionization Detector; ICH: International Conference on Harmonization, GC-HS-MS: Gas chromatograph equipped with Headspace Sampler coupled through Mass Spectrometer; SVHC: Substance of Very High Concern; USA: United States Pharmacopoeia
Introduction
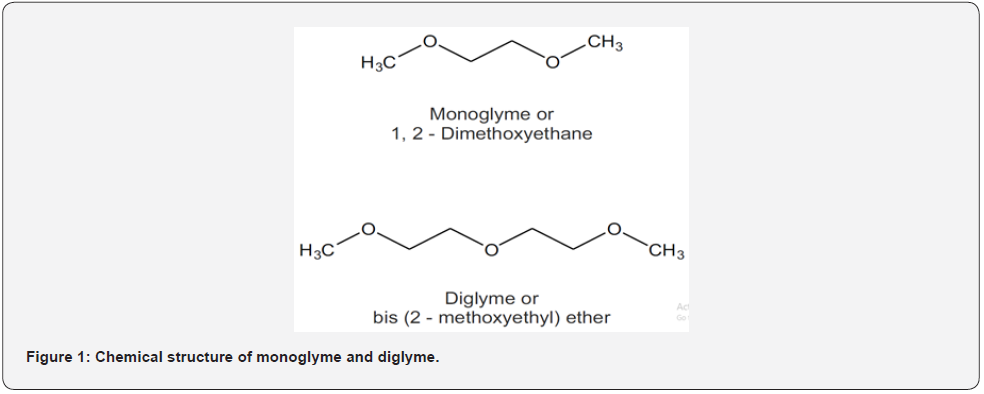
Monoglyme is of importance in the lithium battery industry. It has been commercially synthesized by reacting dimethyl ether and ethylene oxide. Monoglyme is used as a low-viscosity solvent of electrolytes in lithium batteries and as a coordinating solvent in laboratories. It is often used as an alternative to diethyl ether and tetrahydrofuran owing to its relatively higher boiling point; it acts as a bidentate ligand for some metal cations [1-3]. Monoglyme (chemical name 1,2-dimethoxyethane) is listed as a Class 2 solvent in the International Conference on Harmonization (ICH) guideline on impurities: a guideline for residual solvents Q3 (R6) and United States Pharmacopoeia (USP), with a permissible daily exposure of 1.0mg/day and concentration limit of 100μg/mL in pharmaceutical products. Diglyme (chemical name bis(2-methoxyethyl) ether) is a solvent with a high boiling point and is not listed in the ICH Q3 (R6) and USP. Diglyme is miscible with water and organic solvents; it smells sweet. It is used as a solvent in organometallic reactions such as Grignard reaction and metal hydride reduction, and diborane reaction for hydroboration; it also serves as a chelate for alkali metal cations [4,5]. The European Chemicals Agency lists diglyme as a substance of very-high concern (SVHC) owing to its reproductive toxicity [6]. The structures of monoglyme and diglyme are presented in figure 1.
Several analytical techniques for the determination of monoglyme and diglyme using chromatographic techniques have been reported [7-10]. However, these methodologies have limited potential for routine use or widespread deployment. For instance, Cao et al. determined glymes in a fuel exhaust using graphitized carbon black by solvent extraction followed by gas chromatography (GC) coupled with MS (GC-MS) [7]. In their study, seven glymes were determined with detection limits ranging from 1.5 to 13.2μg/m3 using a large sample volume of approximately 3.4L. Owing to the need for high sample quantity and the difficulty in extracting the drug substances/drug products, this method is not suitable for use in pharmaceutical and biopharmaceutical industries. Bensoam et al. evaluated monoglyme, diglyme, and triglyme by solid phase microextraction using gas chromatography with a flame ionization detector [8]. This technique is highly complicated for determining glymes in drug substances/drug products.
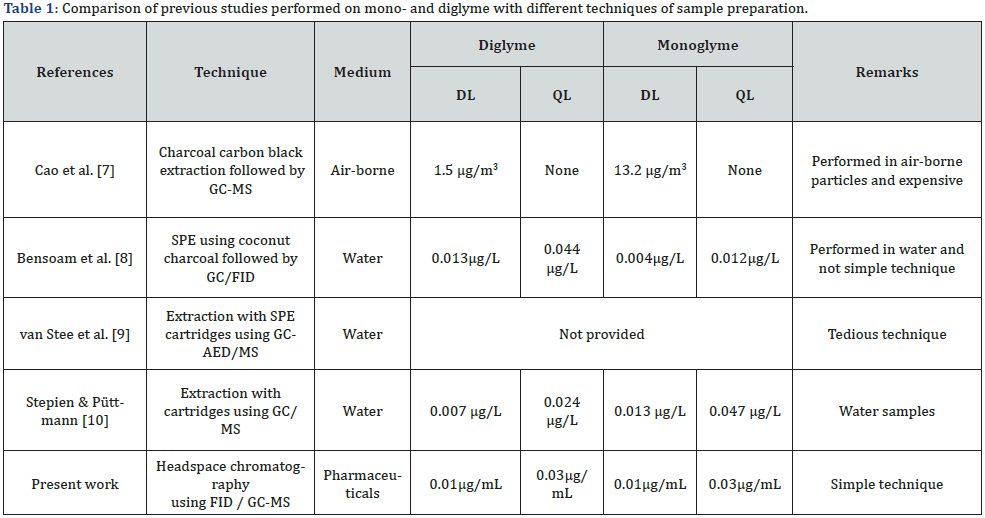
Diglyme, triglyme, and tetraglyme were detected in a screening study of a wide range of contaminants in surface water employing XAD-4 (macroporous polystyrene-divinylbenzene copolymer) and XAD-8 (macroporous polymethylmethacrylate) together with detection using a gas chromatograph equipped with an atomic emission detector and mass spectrometer [9]. As solid-phase extraction was used for the analysis, samples cannot be analyzed without treating the specimen. Stepien and Püttmann validated the method for identifying four glyme compounds in water; it was applied to analyze numerous surface water samples by GC-MS [10]. The sample quantity was approximately 0.5-1.0L that is a relatively high quantity for extraction in pharmaceutical industries. A comparison of different techniques of sample preparation is provided in table 1.
To date, simple sample preparation approaches for the detection and quantification of monoglyme or diglyme have not been developed; in particular, to our knowledge, there is no report on the use of headspace chromatograph coupled with mass spectrometer and flame ionization detector to determine monoglyme and diglyme at the micro-level in the presence of other residual solvents in both pharmaceutical drugs (for example, in Saxagliptin, Memantine, and Etravirine) and biopharmaceutical drugs (for example, in Oncaspar, Pegaspargas, Peginterferon alpha 2b, and Pegasta, which is generically known as Pegfilgrastim).
A literature survey revealed that there is no report of sample preparation methodology with the simultaneous estimation of monoglyme and diglyme at the micro-level in both the above pharmaceutical and biopharmaceutical drugs in the presence of other residual solvents. Food and Drug Administration (FDA) reviewer guidelines and the USP require the resolution between peaks to be not less than 1.5; this is the recommended system suitability criterion to ensure the separation of peaks in any kind of chromatographic techniques. A significant separation is achieved between monoglyme and diglyme in the presence of other residual solvents with a resolution factor of 1.5. The present study aimed to establish a simple quality control method for determining trace levels of monoglyme and diglyme in the presence of other residual solvents used in the manufacture of both pharmaceutical and biopharmaceutical drugs. We used headspace gas chromatograph with mass spectrometer and flame ionization detector as the analytical tools.
Materials and Methods
Materials
Saxagliptin, Memantine, and Etravirine drug substance samples were procured from Sigma Aldrich (Bengaluru, India). Saxagliptin tablets Onglyza® (5mg), memantine tablets Menata® (5mg), and etravirine tablets Intelence® (100 mg) drug-product samples were bought from Indiamart supplier (Chennai, India). Pegaspargase Injection 3750IU/5mL Hamsyl®, Peginterferon Alpha-2b for Injection PegiHep120® (120μg/0.5mL), and Pegfilgrastim Pegasta® 6mg Injections were purchased from Indiamart supplier (Bengaluru, India). Monoglyme, diglyme, methanol, ethanol, acetone, isopropanol, acetonitrile, dichloromethane, n-hexane, ethyl acetate, tetrahydrofuran, toluene, n-heptane, 2-butanone, cyclohexane, diethyl ether, benzene, triethylamine, methyl tertiary butyl ether, methyl isobutyl ketone, diisopropyl ether, dimethylformamide, dimethylacetamide, dimethyl sulfoxide, and benzyl alcohol were bought from Thermo Fisher Scientific (Mumbai, India). The VF-624 GC capillary column was obtained from LCGC Bioanalytic Solutions LLP (Hyderabad, India). Helium gas cylinder was procured from Indo Gas Agencies (Chennai, India). Development and validation studies were carried out using a 7890A gas chromatograph equipped with a G1888 headspace sampler (Agilent, Santa Clara, CA, USA) and coupled with 5973C GC-MS (Agilent, Singapore) and Flame Ionization Detector was utilized to identify and quantify the molecular mass of each monoglyme and diglyme chromatographic peak.
Preparation of diluent
Seven hundred mL of dimethyl sulfoxide and 300mL of water were added into a 1000mL glass bottle that was then covered with a lid and placed at 25°C for 30min mixed well.
Preparation of standard stock solution
Monoglyme and diglyme (25mg each) reference standards were added into a 50mL volumetric flask and then the volume was made up to 50mL with the diluent.
Preparation of standard solution
To prepare the standard solution, 0.1mL of the stock solution was transferred into a 100mL volumetric flask and the volume was made up to 100mL with the diluent. The concentration of the standard solution of both monoglyme and diglyme was approximately 1μg/mL. with respect to the analyte concentration.
Preparation of sample solution
One thousand mg each of Drug substances saxagliptin, memantine, etravirine, powdered drug products of saxagliptin tablets of (Onglyza®), memantine tablets (Menata®), etravirine tablets (Intelence®) and biopharmaceuticals of Pegaspargase Injection (Hamsyl®), PEGinterferon Injection (PegiHep120®), and Pegfilgastim Injection (Pegasta®) were added into individual headspace vials, and 2mL of diluent were added into each headspace vial to prepare sample solutions.
Method
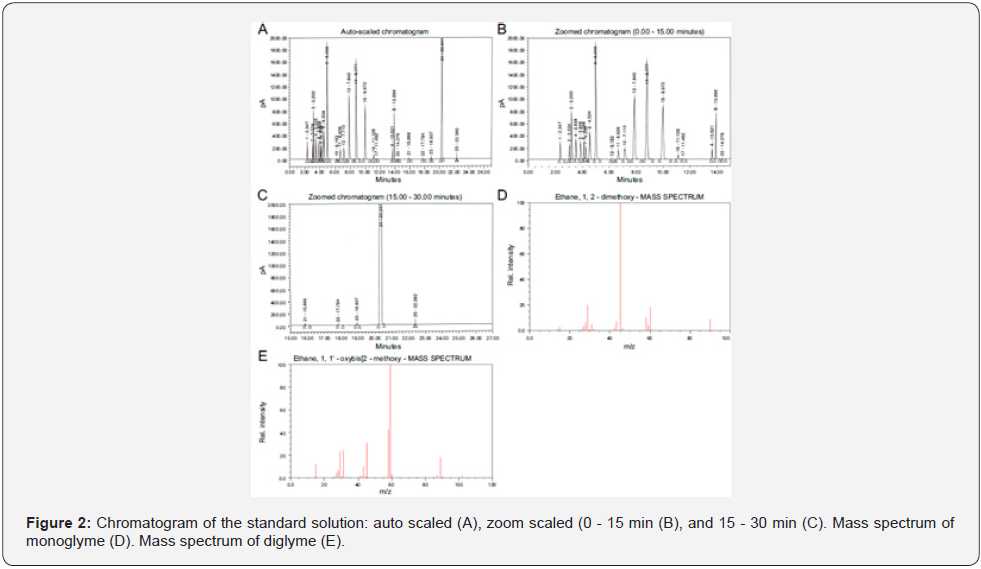
The gas chromatographic FID and mass spectrometric conditions were as follows; the column: VF-624 (30m × 0.25mm × 1.4μm); the oven temperature program: preliminary temperature of approximately 45°C with a holding time of 10 min; the stepup ramp 15°C/min; the final temperature 220°C after holding for approximately 5.33min. The split injection mode with a ratio of 10:1 was used. The column flowrate was 2.0mL/min. The injection port and FID temperatures were 220°C and 250°C, respectively. The oxidizer flow as 400 mL/min for Air gas and the fuel flow rate as 40mL/min for Hydrogen gas with the flow rate of 30mL/min for helium as make-up gas. The conditions of headspace (HS) were as follows: the oven, loop, and transfer line temperatures are 80°C, 175°C, and 175°C, respectively, and the GC cycle time, vial equilibration time, vial pressure time, loop filling time, loop equilibration time, and injection time are 40min, 10min, 30s (0.5min), 9s (0.15min), 6s (0.1min), and 60s (1.0min), respectively. The vial pressure was maintained at 10psi. The MS source and MS quadrupole temperature as 230°C and 150°C with the auxiliary temperature of 280°C. The mobile phase was helium for GCMS. The selected ion monitoring of 45 and 60 with the dwell of 400 milliseconds for monoglyme and diglyme, respectively. The one end of the VF-624 column was attached to Front In-let GC and other end primarily associated into MS splitter which consists of one input and two outputs. One output was connected into MS detector and other output was linked to Flame Ionization Detector of GC. Using this technique, data can be generated simultaneously in Signal plot of GC-FID and Total ion chromatogram of GC-MS. Samples (2.0mL each) of diluent as the blank, standard solution as the standard, sample solution as the test sample, and tolerance sample solution as the spiked sample were pipetted into individual sub headspace vials, sealed using automatic vial crimpers, and placed in the respective positions of the automated headspace sampler. The specificity study was performed by adding ~20mg each of methanol, ethanol, acetone, isopropanol, acetonitrile, dichloromethane, n-hexane, ethyl acetate, tetrahydrofuran, toluene, n-heptane, 2-butanone, cyclohexane, diethyl ether, benzene, triethylamine, methyl tertiary butyl ether, methyl isobutyl ketone, diisopropyl ether, dimethylformamide, dimethylacetamide, dimethyl sulfoxide, and benzyl alcohol into individual headspace vials, 2.0mL of diluent were added to each, and the vials were sealed tightly. The samples were injected into the chromatograph for analysis. The chromatograms of the standard solutions are shown in figure 2.
Data analysis
The gas chromatographic and mass spectrometric data were recorded using ChemStation and MSD Chemstation Software (Agilent, Santa Clara, CA, USA). This headspace chromatography (GC-HS-MS) method was developed and validated using a 7890A gas chromatograph equipped with a G1888 headspace sampler and coupled with 5973C GC-MS (Agilent, Gurugram, India). The linearity analysis and plot creation were performed using Analyseit for Microsoft Excel 5.68 (Microsoft Corporation, Redmond, WA, USA and Analyse-it software limited, Leeds, UK). Analyse-it software was used for the statistical analyses.
Results and Discussion
Method development
The separation of monoglyme and diglyme in the presence of other residual solvents such as methanol, acetone, isopropyl alcohol, acetonitrile, dichloromethane, n-hexane, ethyl acetate, tetrahydrofuran, toluene, n-heptane, 2-butanone, cyclohexane, methyl tertiary butyl ether, methyl isobutyl ketone, diisopropyl ether, dimethylformamide, dimethylacetamide, and dimethyl sulfoxide was challenging in this study. To achieve separation, the VF-624 capillary column rather than others, such as DB-1, DB-5, DB-Wax, DB-1701, and DB-FFAP bonded phases was used. There are studies on the detection of either monoglyme or diglyme [6- 15]. The VF-624 GC low bleed capillary column was chosen based on the mid-polar nature of the bonded phase, less noise level, inertness, and improved detection level.
The selection of pharmaceutical drug substances, drug products, and biopharmaceuticals was based on the solvents used in their pharmaceutical production and availability/cost of biopharmaceuticals in the market, and their utility in evaluating the method specificity. However, a resolution of less than 1.0 between monoglyme and diglyme was obtained with 15 and 20m capillary GC columns; this resolution is not recommended by the pharmacopeia. To improve the resolution, the column length was increased to 30m with an internal diameter of 0.25mm and a packing particle size of 1.4μm.
Analytical method validation
The developed headspace chromatograph FID and mass spectrometric method was validated following the ICH guidelines Q2 (R1) and USP [16,17]. Precision, detection limit, quantitation limit, linearity and range, recovery, specificity, robustness, and solution stability were selected as the method validation parameters [16,17] and are discussed below.
System suitability and precision
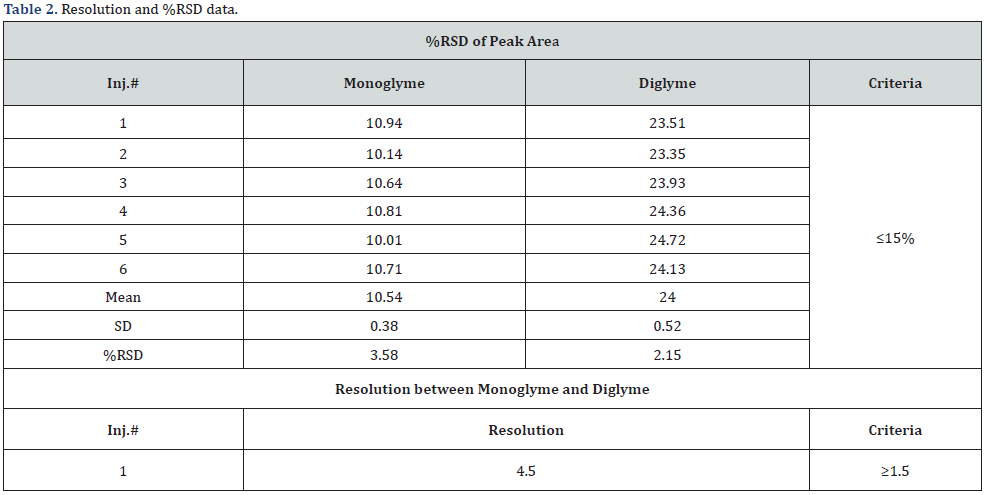
The standard solution, prepared as mentioned in section 2, was analyzed six times by the headspace chromatograph to prove that the method was precise and suitable for the intended purpose. The resolution was approximately 4.5 between monoglyme (Peak A in Figure 2a) and diglyme (Peak B in Figure 2a) determined from the first injection of the standard solution; the %RSD was less than 4% obtained from six injections of standard solutions. The system suitability and precision should meet the acceptance criteria of resolution not less than 1.5 and %RSD of not more than 15.0% [16,17]. Based on the above results, the system was deemed to be suitable for determining the monoglyme and diglyme content. The results are tabulated in table 2.
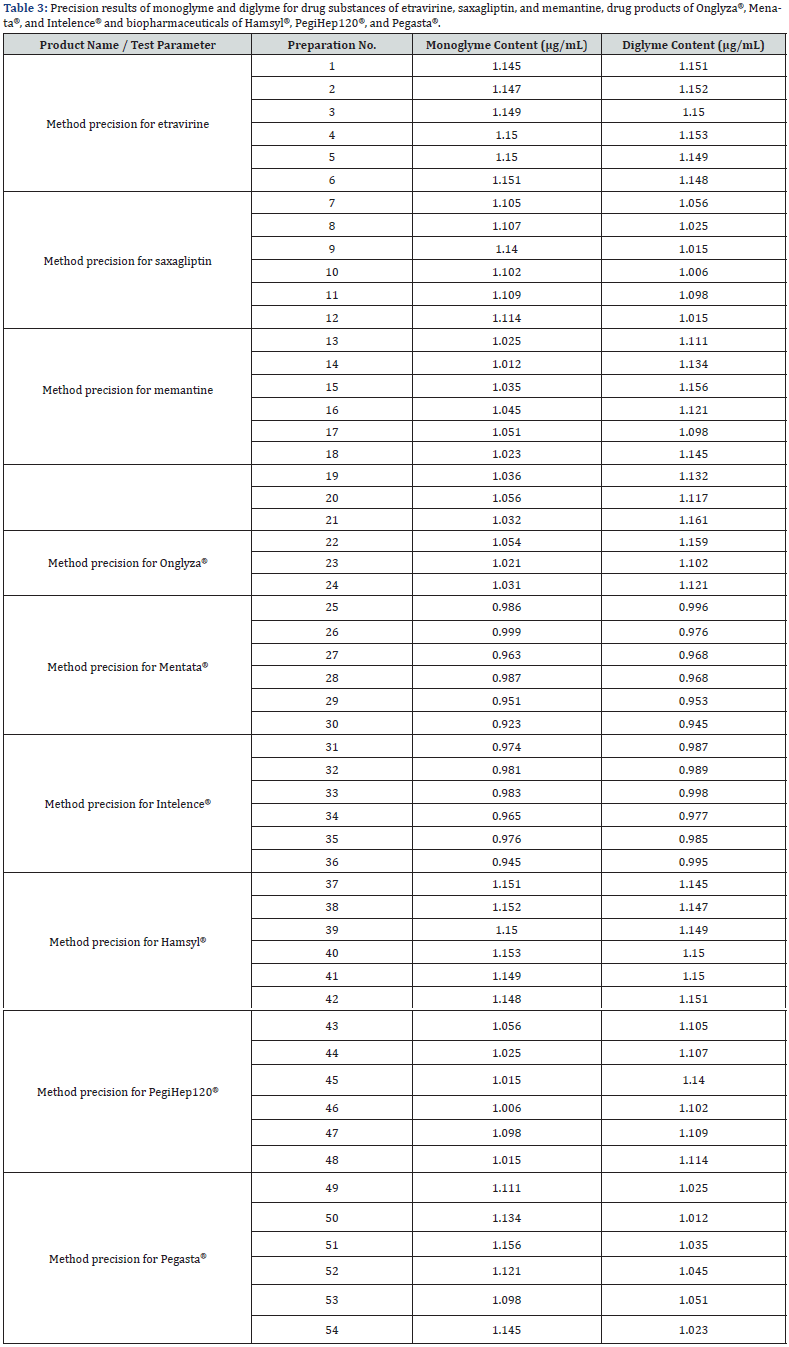
Method precision
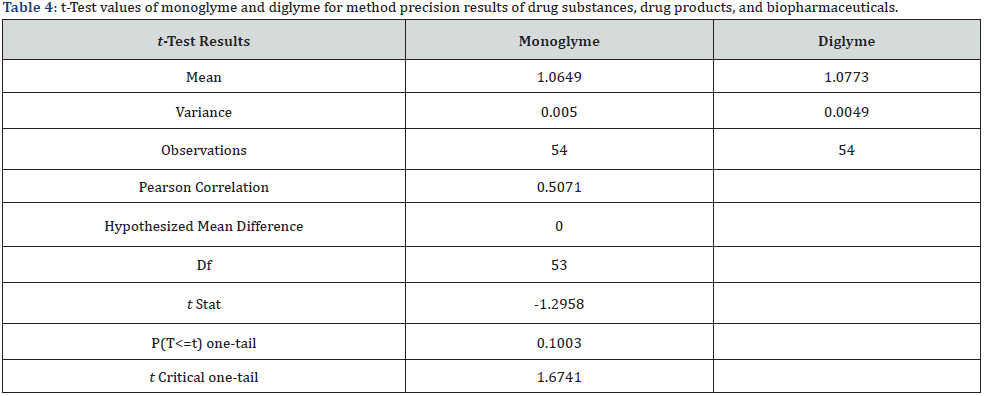
Method precision was determined using the tolerance sample solution. The %RSD was determined for monoglyme and diglyme content by injecting the spiked tolerance sample six times. The %RSD for monoglyme and diglyme was less than 0.5% that is well within the acceptance criterion of 5.0% [16,17]. The intermediate precision was analyzed by a different analyst using columns of a different lot number, different instrument, and on a different day. The %RSD of monoglyme and diglyme content was less than 4% and met the acceptance criterion of not more than 5.0% [16,17]. The data obtained are tabulated in (Table 3). These findings show that the method is precise for the instrument concerned. A t-test was performed to analyze the method precision and intermediate precision results of drug substances of saxagliptin, memantine, and etravirine, drug products of saxagliptin tablets (Onglyza®), memantine tablets (Menata®), etravirine tablets (Intelence®), and biopharmaceuticals Pegaspargase Injection (Hamsyl®), PEGinterferon Injection (PegiHep120®), and Pegfilgastim Injection (Pegasta®). The t-stat value was either greater than +2.0 or less than -2.0 is acceptable, indicating that the values (Table 4) are statistically significant with sufficiently strong evidence.
LOD and LOQ
The LOD and LOQ were established using the signal-to-noise approach recommended by the ICH guidelines Q2 (R1) and USP and its limits were 3 and 10 of LOD and LOQ, respectively. The solutions were prepared and serially diluted to obtain the required signal-to-noise ratio using the standard solutions and diluent. The detection and quantification limits for monoglyme and diglyme were 0.01 and 0.03μg/mL, respectively, demonstrating that the technique was sufficiently sensitive for determining the monoglyme and diglyme content in all pharmaceutical and biopharmaceutical samples.
Linearity and range
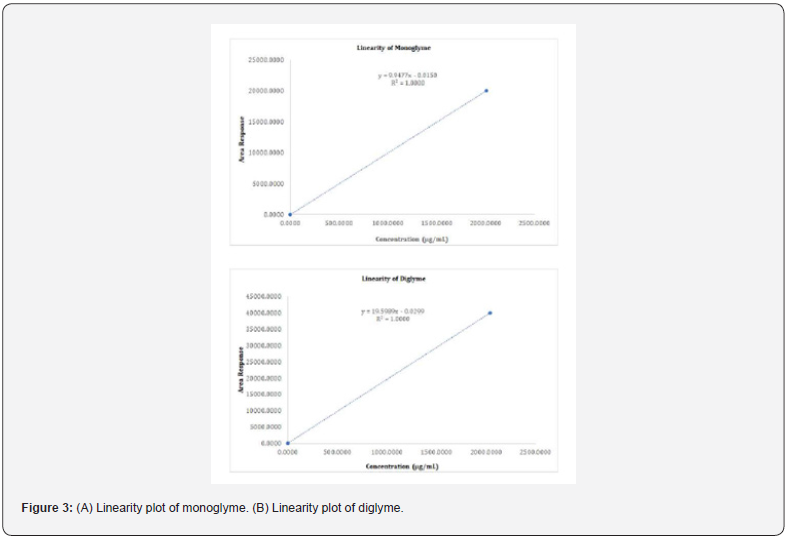
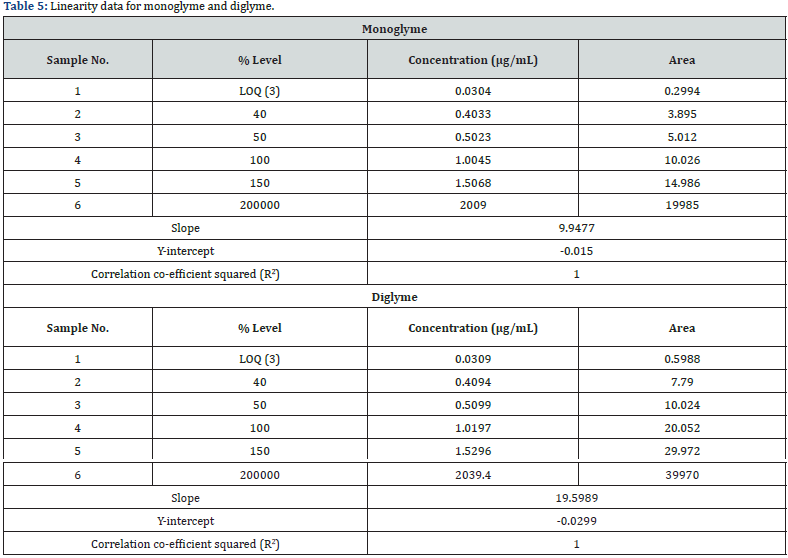
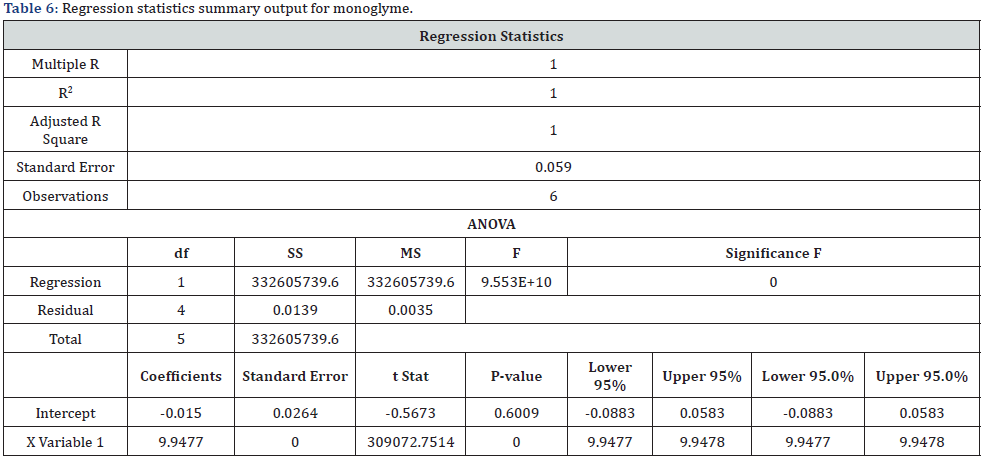
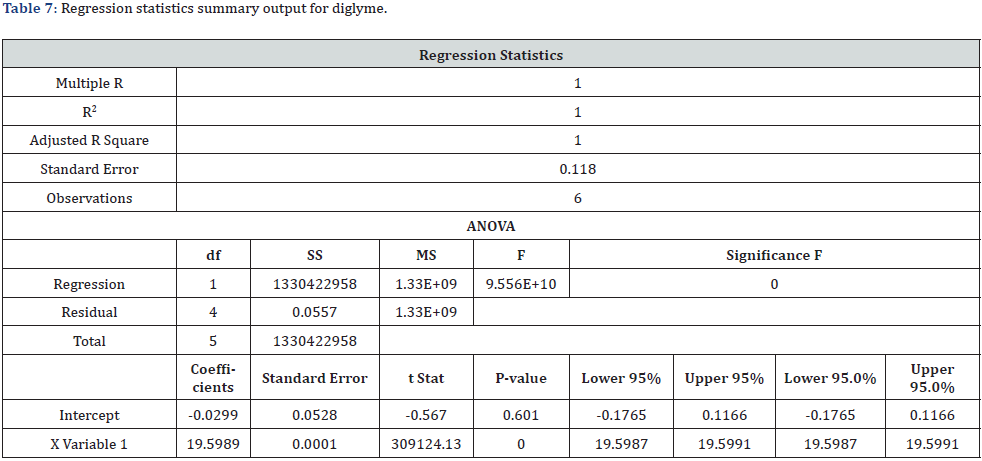
The linearity of the method was established by analyzing serially diluting solutions containing both monoglyme and diglyme at the starting level of LOQ and several times the strength of the standard solution (i.e., 200000%) [16,17]. The linearity solutions were prepared at the LOQ level of 40%, 50%, 100%, 150%, and 200000% of the sample strength. The LOQ and 200000% solutions were injected six times and the other linearity solutions were injected twice into the chromatograph. A correlation coefficient of 1.0000 was obtained for both monoglyme and diglyme, higher than the 0.98 limits for the correlation coefficient [16,17]. The range was calculated from the linearity data of the LOQ and 200000% solutions that represented the lower and upper levels of sample strength. The specified concentration range (0.03 - 2000μg/mL) for both monoglyme and diglyme is presented in Table 5. The technique was found to be linear, and the range was consistent with the ICH guidelines Q2 (R1) and USP [16,17]. The regression statistics summary output showed that the regression model cannot be rejected. The obtained real values Table 6&7 passed close to but not exactly through the origin. The linearity graph for both monoglyme and diglyme is shown in figure 3.
Accuracy
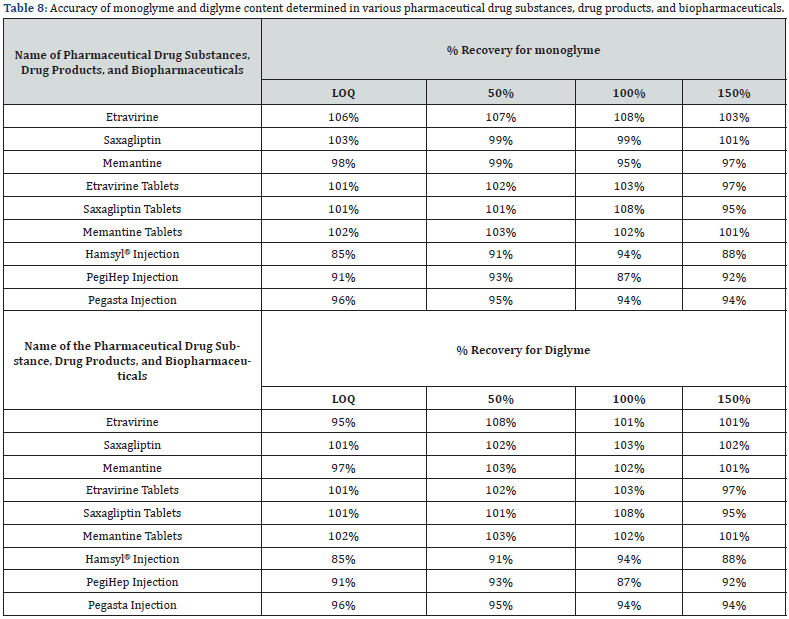
The accuracy of the technique was established using the tolerance sample solutions. The accuracy of the method was evaluated at the lowest, specification, and highest levels of LOQ, 100%, and 150%, respectively. The required recovery value limit is between 80% and 120%. Each LOQ, 100%, and 150% levels tolerance sample solutions were prepared in triplicate to prove the accuracy of the method. These solutions were injected once into the chromatograph, and the recovery value of monoglyme and diglyme in drug substances of saxagliptin, memantine, etravirine, drug products of saxagliptin tablets (Onglyza®), memantine tablets (Menata®), etravirine tablets (Intelence®), and biopharmaceuticals of Pegaspargase Injection (Hamsyl®), PEGinterferon Injection (PegiHep120®), and Pegfilgastim Injection (Pegasta®) were calculated. The obtained values 85% - 108% were within the acceptable limits. The results are shown in table 8.
Specificity
The specificity of the method was established by injecting each solvent into the chromatograph. There should not be any interference at the retention time of monoglyme and diglyme peaks in blank and other solvent solutions. Failure in specificity tests leads to the misinterpretation of results in the samples. Furthermore, the results of spiking of solvents with the sample indicated that monoglyme and diglyme were not co-eluted with the other solvents. The data showed that the technique was specific for the determination of monoglyme and diglyme.
Robustness
The robustness of the technique was established by varying the oven temperature, column flow rate, and headspace oven temperature. The variations were evaluated at oven temperatures of 40°C and 50°C, column flow rates of 1.8 and 2.2mL/min, and headspace oven temperatures of 100°C and 120°C. The system suitability criteria such as the resolution and %RSD of monoglyme and diglyme remained unchanged. Thus, the developed technique was found to be robust and suitable for the intended analysis.
Solution stability
The solution stability was determined using the standard and sample solutions. The standard and sample solutions were prepared and injected immediately at time 0 h. These standard and sample solutions were then kept at the laboratory temperature for 24h and injected at the 24h time. The %RSD of the standard solution was 15.0, and no difference was observed in the content of monoglyme and diglyme compared with the sample solution.
Limitations
A limitation of this method is the 30m length of the capillary column. When the column length was reduced to 15 or 20m, the total flow rate (column, split flow, and septum purge flow) was also modified to maintain the total flow rate at approximately 20mL/ min as recommended by Agilent experts for the reproducibility.
However, further adjustments would be needed to obtain a robust headspace chromatographic with mass spectrometric method.
Conclusion
Herein, we report a versatile headspace gas chromatograph coupled with mass spectrometer method for the simultaneous separation and quantification of monoglyme and diglyme in pharmaceutical drug substances, drug products, and biopharmaceuticals at the micro-level. The developed method is precise, sensitive, linear, accurate, specific, and robust for the detection and quantification of monoglyme and diglyme in both chemically and biologically active substances. Molecular masses were confirmed for monoglyme and diglyme by GC-MS. Using headspace chromatograph with FID and mass spectrometer, monoglyme and diglyme can easily be estimated at trace levels during the commercial preparation in both chemically and biologically active substances in both industries. The detection and quantitation of glymes may be further simplified using headspace chromatography with the required analyte concentration or improved sampling technique at the ng/mL level upon further research.
References
- D Berndt, D Spahrbier (2005) Batteries in Ullmann’s Encyclopedia of Industrial Chemistry. Wiley Weinheim, p. 23-65.
- S Rebsdat, D Mayer (2000) Ethylene Glycol in Ullmann’s Encyclopedia of Industrial Chemistry. Wiley Weinheim, pp. 531-544.
- J A Riddick, W B Bunger, T K Sakano (1985) Techniques of Chemistry in Organic solvents: physical properties and methods of purification. Fourth edition Wiley, pp. 802-1130.
- M W Rathke (1978) Boranes in functionalization of olefins to amines: 3-pinanamine. Org Synth 58: 32-36.
- E Negishi, H C Brown (1983) Perhydro-9b-boraphenalene and perhydro-9b-phenalenol. Org Synth 61: 103-111.
- European Chemicals Agency (2011) Inclusion of Substances of Very High Concern in the Candidate List.
- X L Cao, J Zhu (2011) Monitoring method for airborne glymes and its application in fuel exhaust emission measurement. Chemosphere 45: 911-917.
- J Bensoam, A Cicolella, R Dujardin (1999) Improved extraction of glycol ethers from water by solid-phase micro extraction by carboxen polydimethylsiloxane-coated fiber. Chromatographia 50: 155-159.
- L L P van Stee, P E G Leonards, W M G M van Loon, A J Hendricks, J L Maas, et al. (2002) Use of semi-permeable membrane devices and solid-phase extraction for the wide-range screening of microcontaminants in surface water by GC-AED/MS. Water Res 36(18): 4455-4470.
- D K Stepien, W Püttmann (2012) Simultaneous determination of six hydrophilic ethers at trace levels using coconut charcoal adsorbent and gas chromatography/mass spectrometry. Anal Bioanal Chem 405(5): 1743-1751.
- M Kucharska, W Wesołowski (2017) 1,2-Dimethoxyethane determination in working air with gas chromatography-mass spectrometer. Podstawy i Metody Oceny Środowiska Pracy 33: 133-147.
- K P Shah, S Kumar, M Kurmi, D Gohil, S Singh (2018) Successful development by design of experiments of a gas chromatography method for simultaneous analysis of residual solvents of classes 1 and 2. J Chromatograph Sci 56: 473-479.
- A Deshpande, B Sirisha, G R Deshpande, J M Babu, R M Krishna, et al. (2016) Identification and quantification of an unknown peak in residual solvent analysis by GC using relative response factor. Int J Pharm Sci Res 7: 1343-1352.
- F Al Taher, B Nemzer (2018) Determination and quantitation of residual solvents in natural food ingredients using a static headspace gas chromatography with flame ionization detection and mass spectrometric detection method. J Food Prot 81(10): 1573-1581.
- K U Schwenke, S Meini ,S X Wu, H A Gasteiger, M Piana (2013) Stability of superoxide radicals in glyme solvents for non-aqueous Li-O2 battery electrolytes. Phys Chem Chem Phys 38: 11830-11839.
- International Conference on Harmonisation (ICH) of Technical Requirements for the Registration of Pharmaceuticals for Human Use, Q2 (R1): impurities: Guidelines for Residual Solvents.
- United States Pharmacopeia (USP): USP-NF 43 <467> Residual Solvents.






























