RP-HPLC Method Development and Validation for Simultaneous Estimation of Glabridin, Glycyrrhizinic Acid and Total Flavonoids in Glycyrrhiza Glabra Extract: Stability Studies of Constituents
Shrikant Kulkarni1, Shital Palghadmal2, Atul Sawant1, Shrinivas Bhope2 and Sriram Padmanabhan3*
1Analytical Development Laboratory, Sava Healthcare Limited, India
2Herbal Division, Sava Healthcare Limited, India
3Head R & D, Sava Healthcare Limited, India
Submission: April 09, 2021; Published: May 10, 2021
*Corresponding author: Sriram Padmanabhan, Head R & D, Sava Healthcare Limited, Research Center, MIDC, Chinchwad, India
How to cite this article:Shrikant K, Shital P, Atul S, Shrinivas B, Sriram P. RP-HPLC Method Development and Validation for Simultaneous Estimation of Glabridin, Glycyrrhizinic Acid and Total Flavonoids in Glycyrrhiza Glabra Extract: Stability Studies of Constituents. Glob J Pharmaceu Sci. 2020; 8(3): 555738. DOI: 10.19080/GJPPS.2021.08.555738.
Abstract
Objectives: The major bioactive phytoconstituents of Glycyrrihiza glabra include Glabridin (GB), glycyrrihizic acid (GA) and total flavonoids (TF). This article describes the development and validation of HPLC method for simultaneous estimation of GB, GA, and TF in liquorice as per of ICH guidelines. Materials and methods: The chromatographic separation was achieved by gradient program using a Inertsil ODS 3V column (250mm×4.6mm ID, 5μm particle size) at 40°C temperature with KH2PO4 buffer as mobile phase A and pure acetonitrile as mobile phase B at a flow rate of 1 mL/min. Quantification of all the three constituents was performed at two different wavelengths, 280 nm for GB, TF and 254 nm for GA. Results and conclusions: The developed method showed the limit of quantitation (LOQ) of 0.2 μg/mL for both GB and GA, over the concentration range of 0.10 – 5.025 μg/mL (r2=1.000) for GB and concentration range of 0.19–4.747 μg/mL (r2=0.999) for GA with 95.2-103.4% recovery for all the three constituents. The present method was successfully validated with excellent selectivity, linearity, sensitivity, precision and accuracy for GB, GA, and TF in liquor ice. Also, the present work illustrates data on the stability of GB, GA, and TF in liquorice extract by six different experimental factors such as temperature, illumination, humidity, acid hydrolysis, base hydrolysis and hydrogen peroxide oxidation.
Keywords: Glycyrrhizin acid; Glabridin; Total flavonoids; HPLC; Development; Validation; Quantification; Stability
Abbreviations: ARDS: Acute Respiratory Distress Syndrome; PDA: Photo Diode Array; ICH: International Conference on Harmonization; LOD: Limit of Detection; LOQ: Limit of Quantification; RSD: Relative Standard Deviation
Introduction
Liquorice, (glycyrrhiza glabra linne), an herbal medicine and a natural sweetener, is used worldwide. Biologically active components isolated from liquorice include triterpenes, saponins, flavonoids, isoflavonoids, and chalcones each of the component having various medicinal uses for treatment of ulcer, inflammation, blood pressure, seizures, viral infections, allergies along with its well-known antioxidant pharmacological activity [1,2] with different extraction procedures employing the use of water, ethanol, acetone, and ethyl acetate. Used as a treatment for hepatitis C in Japan, Glycyrrihizin also acts as an emulsifier and gel-forming agent in foodstuff and cosmetics with anti-viral activities on HIV-1, hepatitis viruses A, B and C [3-5].
Many of the active constituents of liquorice such as glycyrrhizic acid and liquiritin are also known to prevent replication of viruses such as COVID-2 causing the Corona infection, a pandemic declared by WHO recently that more than 10,778, 206 patients affected by this virus with a fatality rate of 3-4% [6]. GA can be useful to treat respiratory infections and acute respiratory distress syndrome (ARDS) [3]. Hence, this article describing simultaneous estimation methods for three constituents of liquorice would be immensely useful.
In this article, we report a validated HPLC method to estimate glabridin, glycyrrihizic acid and total flavonoid simultaneously in a single HPLC run. Various analytical methods for separate quantification of glabridin have been previously published, such as HPLC [7-9], HPTLC [10], capillary-zone electrophoresis [11,12], and liquid chromatography–tandem mass spectrometry [13] with every method having some limitations with respect to sensitivity, linearity, resolution efficiency etc. The HPLC method reported by Shanker et al. [7] has a narrow linearity range, inefficient separation of glabridin from other extract constituents, and low analyte recovery while the HPLC method by Kamal et al. has limitations in efficient separation of constituents [14].
Materials and Methods
The purified glycyrrhizin ammonical hydrate (Lot no. T20F068, Purity – 93.0%, validity- June 2022) and glabridin (Lot no. T18F128, Purity – 99.3%, validity- June 2022) was procured from Natural Remedies Pvt. Ltd. Bangalore India was used for qualification and quantitation. Two commercial samples of Glycyrrhiza glabra (water and acetone extract), manufactured by Sava Healthcare Ltd. India, was used.
Methanol (HPLC grade) was purchased from Finer Chemicals while HPLC grade acetonitrile, ortho-phosphoric acid, monobasic potassium phosphate (KH2PO4), hydrochloric acid and sodium hydroxide were purchased from Rankem Chemicals. Hydrogen peroxide (batch no. CL86680696, analytical reagent) was purchased from Merck, USA. Elix Milli Q system water used for all preparations. 0.45μm nylon syringe filters (MDI Membrane Technologies, Ambala Cantt, India) were used for sample filtration. HPLC grade, Elix Milli Q system water used for all preparations. 0.45μm nylon syringe filters, (MDI Membrane Technologies, Ambala Cantt, India) used for sample filtration.
Methods
The LC system used for method development and validation of the three constituents GA, GB and TF, consisted of Waters (Alliance e2695 separation module, equipped with Photo Diode Array (PDA) detector (Waters 2998). The HPLC instrument consisted of a Quaternary gradient pump, an online degasser, an auto-sampler, a thermostatically controlled column compartment. For reproducibility Shimadzu (LC 2010C HT) system (Shimadzu Corporation, Kyoto, Japan) was used.
Preparation of mobile phase and diluent
Mobile phase A contained monobasic potassium phosphate buffer (1.36mg/ml). Mobile phase B contained acetonitrile (100%). Methanol (100%) was used as diluent. Preparation of stock standard solutions and mixed standard solutions of glycyrrhizin acid and glabridin 2.6mg of glycyrrhizin ammoniacal hydrate reference standard (equivalent to 2.5mg of glycyrrhizic acid) was dissolved in 25mL of diluent to yield GA solution of 0.1 mg/mL while glabridin standard was prepared by dissolving 2.5mg of glabridin reference standard in 25mL diluent. To make a mixed standard solution, 2mL of each of glycyrrhizic acid standard stock solution and glabridin standard stock solution was diluted to 20mL with methanol to yield a concentration of GA and GB as 0.01mg/mL.
Preparation of test sample solution
50mg of powdered extract sample was dissolved in to a 50mL volumetric flask with diluent, sonicated for a few minutes and used for analysis.
Selection of wavelength
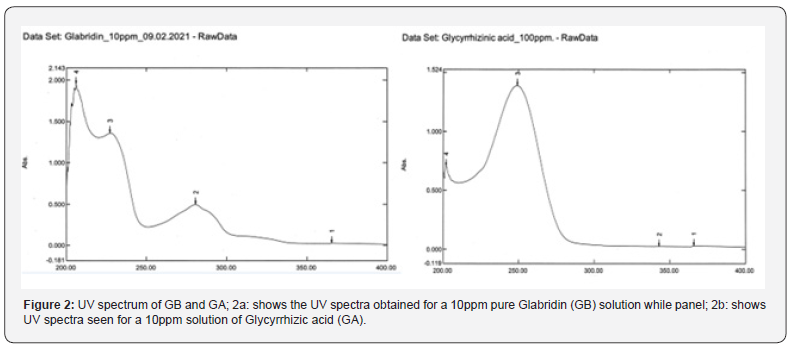
Determination of detection wavelength was based on the scanned UV spectrum of the GB and GA solution between 200 to 400nm. Based on UV spectrum for GB and TF, the chosen wavelength was 280nm while it was 254nm for GA.
Chromatographic conditions
The chromatographic separation, using HPLC, was performed with gradient elution of mobile phase A (KH2PO4 buffer) & mobile phase B (pure acetonitrile). The gradient program was set as (time/% B) 0/5, 25/40, 40/42, 45/60, 50/60, 55/80, 60/80, 61/5, 65/5 which pumped the phases at a rate of 1mL/min. The injection volume of the samples was kept at 20μL and the elutents such as GA was examined at 254nm and GB and TF were observed at 280 nm at a fixed wavelength as well as scanned over range of 190-400nm for characterization of GA, GB, TF and impurities or degradants by using the diode array detector.
Method validation
The developed HPLC method was validated in terms of the following parameters: System suitability, specificity (selectivity and forced degradation), sensitivity (LOD & LOQ), linearity, precision, accuracy, filter study, stability of analytical solutions and robustness studies. The validation was done as per the guidelines of International Conference on Harmonization (ICH) for validation of all the developed analytical procedures. Throughout the validation study, the system suitability parameters such as resolution, tailing factor and theoretical plates were monitored.
Specificity and selectivity
The specificity and selectivity of method was investigated by injecting blank samples (methanol), individual selectivity solutions and sample solutions to demonstrate the absence of interference with elution of GB, GA and TF peaks and to prove the selective nature of peaks. The forced degradation study enabled us to evaluate the resolution of the analyte from other interfering or degradation products.
Sensitivity (LOD & LOQ).
The sensitivity of the analytical technique, described in this paper, was estimated in terms of limit of detection (LOD) and limit of quantification (LOQ) that were defined based on signal to noise ratio of 3:1 and 10:1, respectively and calculation from the calibration line at low concentration levels (0.095-5μg/mL). LOQ was taken as lowest concentration of GB and GA that could be quantitatively determined with acceptable accuracy and precision.
Linearity
To study the linearity of analytical procedure developed, six working standard solutions of GB and GA at different concentration levels (0.5-15μg/mL) were injected into HPLC system in three individually replicates. To determine that the instrumental response was directly proportional to analyte concentration, calibration curve was constructed by plotting concentration of GB and GA on X-axis and average peak area on Y-axis. Regression equation and value of co-relation coefficient was calculated using linear regression analysis.
Precision and accuracy
For precision studies, six extract samples at similar concentration levels, were analyzed on two consecutive days (inter-day precision) and six times during the same day (intra-day precision). The precision of proposed method was obtained by calculating the relative standard deviation (RSD) values observed for intra-day and inter-day analysis with acceptance criteria of not more than 5%. The accuracy of measurement method was assessed via the methodological recovery by injecting extract samples at three different concentration levels (50%, 100% and 150%) and analyzed. The percentage deviation of the determined concentration of extract samples and theoretical concentration expressed the recovery of the developed method.
Filter study
To evaluate the suitability of filter for sample preparation method performed the filter interference study on 0.45μm Nylon (mdi) filter and it was compared with the sample centrifuged at 5,000rpm for 10min.
Stability of analytical solutions
To evaluate stability of samples during the analysis, stability of GB and GA samples was studied at room temperature (at 25°C for 3 days) and at cold (at 10°C for 3 days) and after going through three freeze-and-three room temperature cycles for every 24 h, GB and GA samples were reanalyzed and compared with freshly prepared standard solution.
Robustness
Robustness study was performed by introducing small variations in flow rate (1.0±0.1mL min), column oven temperature (40±5°C), gradient composition (±5% of mobile phase B) and detection wavelength (For Glycyrrhizic acid 254nm±3nm and for Glabridin 280nm±3nm) to evaluate the methods’ capacity to remain unaffected.
Stability and degradation studies
The stability and degradation study of GB, GA and TF was carried out to demonstrate stability indicating method stability studies of constituents, extract samples insight into degradation pathways. During this study, the sample extract was exposed to various stress conditions like heat (60°C) for 24h and 168h; 25°C with 60% relative humidity (RH) and 40°C with 75% RH, for 24h and 168h; UV visible light (1.2million lux hours) for 27h; acid and alkali hydrolysis with 0.1N, 1N and 2N HCl and NaOH solutions for 1h at room temperature and oxidative degradation with 0.3%, 3% and 30% H2O2 for 1 h at room temperature.
Results
Selection of wavelength
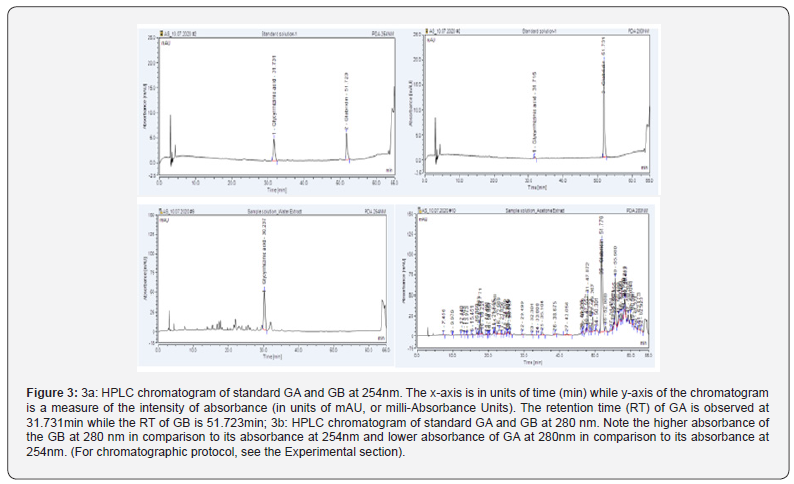
Figure 1 gives the structures of GB and GA. Based on UV spectra of GB and GA solutions and HPLC chromatographic purity, 280nm was selected for GB and 254nm for GA (Figure 2). The HPLC assay chromatograms for GB and TF at 280nm and for GA at 254nm are presented in (Figure 3). The wavelength of 280nm was selected as a suitable detection wavelength for GB and TF while 254nm was chosen for detecting GA since at this wavelength, the baseline was clear without interference from diluent (methanol) and mobile phase buffer.
HPLC method development and method optimization
The present work was aimed at developing simple, rapid, and economical assay method for GB, GA, and TF in extract samples. The RP-HPLC separation was developed on Inertsil C18 column under gradient elution with acceptable resolution, use of cost-effective solvents and ease of preparation. Quantitative analysis was achieved with high chromatographic response peak and optimum resolution. The GB, GA, and TF along with their degradation products were difficult to separate to be analyzed on HPLC by published methods (data not shown). To enable better resolution and detection, the HPLC conditions were optimized. A series of mobile phases, with varying different concentration of KH2PO4 buffer, with different organic modifiers were tested.
Acetonitrile was chosen as the organic portion of the mobile phase due to its lower background noise and strong elution effect on targeted compounds and degraded products. Also, with acetonitrile, the analysis time is shortened in comparison to methanol. We believe that this could be due to the strong hydrophobic properties of the octadecyl silane (C18) column than C8 column, whose bonding reactions with the impurities (long chain alkane) were stronger than the octyl silane phase for column packing. Different columns with (C18) and (C8) packing materials were tested. However, a complete resolution of all the interested compounds was achieved with Inertsil ODS 3V column (250x4.6mm, 5mm). This column gave a better resolution with steady baseline for the peaks of interest.
Different diluents like methanol, acetonitrile and water were tested for the efficient extraction of targeted compounds and degradation products of GA, GB and TF. Methanol was found to be the most suitable diluent for this purpose. The chromatogram for a mixed standard solution of GA and GB indicates that GA shows good absorbance at 254nm (Figure 3a) while GB shows lesser absorbance at 254 in comparison to 280nm (Figure 3b). Hence, liquorice samples prepared using water (GA is isolated specifically with water) was examined at 254nm (Figure 3c) while the acetone extract sample of liquorice (where GB is isolated specifically), was examined at 280nm (Figure 3d).
System suitability
The system suitability test was determined by injecting five replicates of the mixed standard preparation. The relative standard deviation (RSD) of each compound’s peak area and retention time was found less than 2.0%, tailing factors was found less than 1.5 and theoretical plates were at least 5,000 in overall all validation study. The results indicated that the system was suitable for all the peaks.
Specificity
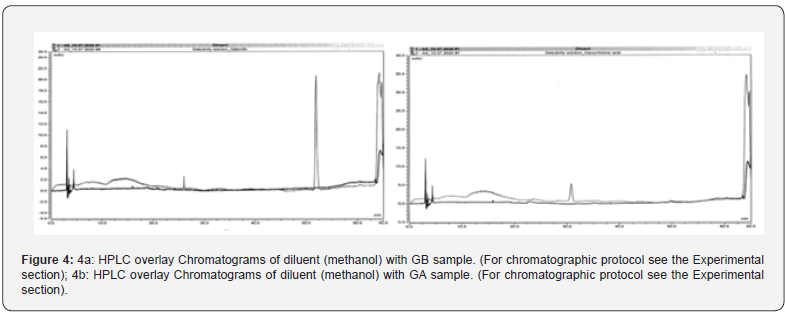
Figure 4a & 4b shows the overlay of chromatograms for GA and GB standards respectively and assay samples with that of blank sample (methanol). No interference with GA, GB peaks was observed as blank did not show any interfering peaks at the retention time of GA (31min), GB (51min) peaks. Specificity of the method can thus be concluded based on the chromatographic peak purity observed in the chromatograms.
Linearity
The calibration curve of both GA and GB was constructed by plotting a graph of peak mean area versus concentration. The linearity of calibration curves was evaluated using linear regression analysis. For GA regression equation was: Y=13,581.1115x+5,311.3742 with 0.999 as co-relation coefficient and for GB regression equation was: Y=49,938.8954x+28,480.6544 with 0.9974 as co-relation coefficient, which meets the analytical method validation acceptance criteria. Hence linearity of method was proved over the concentration of 0.5-15μg/mL for both GA and GB under the experimental conditions described (Figures 5a & 5b).
Sensitivity
Calibration line at low concentration levels (0.095-5μg/mL) for GB and GA was plotted and obtained LOD for GB was 0.13 and LOQ was 0.41μg/mL similarly for GA, LOD was 0.16 and LOQ was 0.49μg/mL. Although the method has capable to calculate the lowest amount 0.2μg/mL of both GB and GA with acceptable accuracy and precision. Based on recovery of quantification at LOQ concentration level (0.2μg/mL) the average % recovery for GB was 105.5% with 2.62% precision RSD and for GA was 106.0% with 0.84% precision RSD, which meets the acceptance criteria for method recovery and precision. Also signal to noise ratio at 0.2μg/mL concentration level for GB was 25.7 and GA was 18.7. Therefore, lowest concentration 0.2μg/mL was experimentally considered as LOQ of the assay method for both GB and GA.
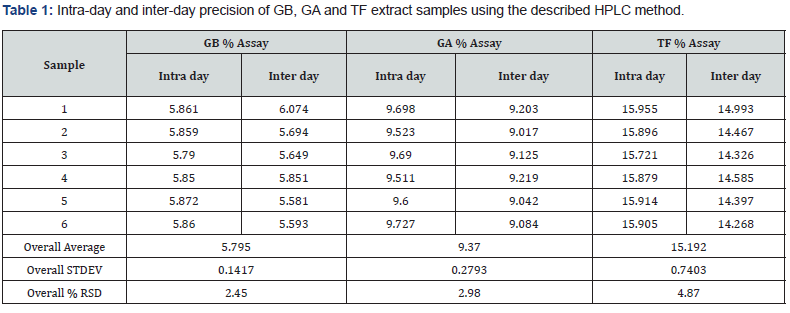
The intra-day and inter-day precision of method was expressed as RSD value. The RSD values for intra- and inter-day assay of extract samples, of both water and acetone extract of liquor ice, did not exceed 5%, indicating that the developed method has a good precision. The results are presented in (Table 1).
Accuracy
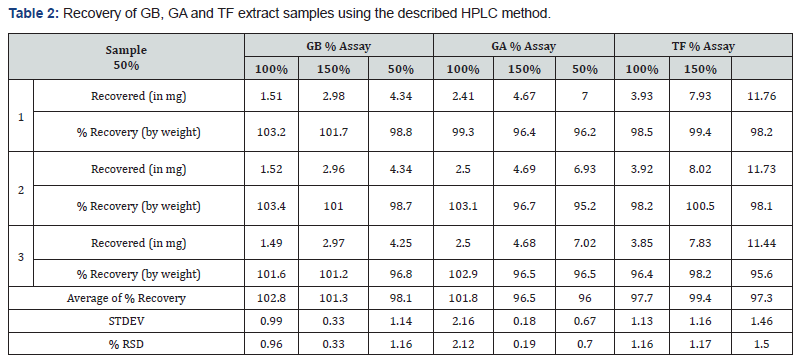
The accuracy of method was assessed by recovery studies on extract samples. The percent recovery by the assay of GB samples ranged from 96.8 to 103.4%, for GA samples ranged from 95.2 to 103.1% and for TF samples ranged from 96.4 to 100.5%. The good recovery values for accuracy study ascertain that the developed method is accurate (Table 2).
Stability of analytical solution
Stability results were obtained from the stability studies of standard and samples stored at room temperature (at 25°C for 3 days) and at cold (at 10°C for 3 days). Based on the results, GB and GA standard and sample solutions was found to be stable up to 3 days, both at room temperature (25°C) and at cold (10°C) and TF sample solutions was found to be stable up to 2 day at room temperature (25°C) but stable for 3 days at cold (10°C).
Stability and degradation studies
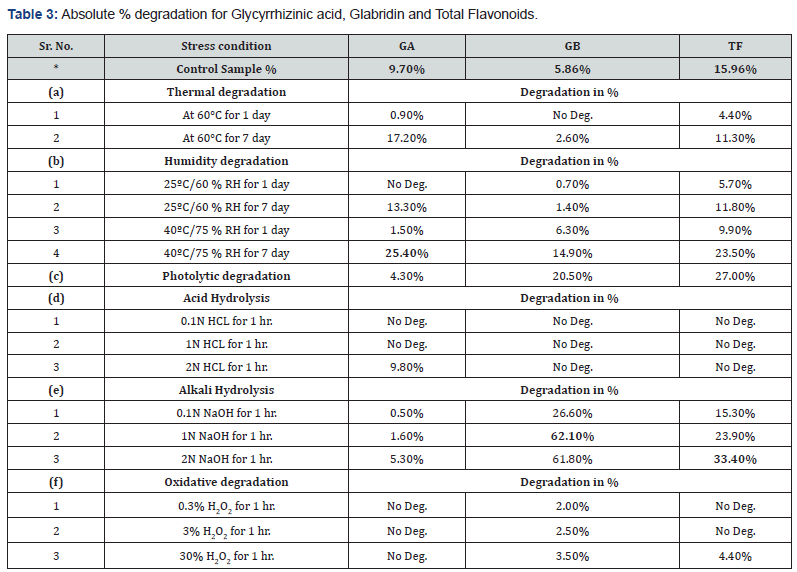
During this study, the sample extract was exposed to various stress conditions like acid hydrolysis, oxidation, temperature, temperature and humidity, illumination, and alkali hydrolysis. The results indicate that GA shows maximum degradation when exposed to temperature (60oC) in comparison to GB and TF. Similarly, GA is more prone to degradation when exposed to high humidity (40oC/75% for 7 days) in comparison to GB and TF. As far as photolytic degradation is concerned, GA showed minimum degradation (4%) in comparison to almost 25% degradation seen in GB and TF. All the three constituents like GA, GB and TF were found to be affected minimally by oxidation but by alkali treatment, GB and TF were more susceptible in comparison to GA. It is tempting to speculate that one could adopt acid hydrolysis of the liquorice extract to achieve higher yields of purer GB and TF content selectively since only GA showed 9.8% degradation by acid treatment (Table 3).
Comparison of sensitivity of the HPLC methods
Table 4 summarizes the comparisons of all the existing methods wrt LOD, LOQ for GA, GB, and TF. It is clear from this table that there is no method in the literature that has estimated all the three constituents by a single HPLC run. It is also evident from this table that the LOD and LOQ values for the three constituents are highly variable. While Kamal et al. [14] reported LOD for glabridin as 0.35ppm and LOQ as 1ppm, Shanker et al. [5] obtained for the analysis of glabridin, with LOD and LOQ of 19.5ppm and 65ppm.
LI et al. [15] have shown the LOD for glabridin estimation as 0.49ppm by ultra-performance convergence chromatography (UPC) method and the LOQ as 1.61ppm. The LOD and LOQ for glycyrrihizin was found to be 3.08ppm and 10.27ppm respectively by De et al. [16] with no attempts to quantify glabridin. The LOD for glycyrrhizic acid by HPLC was 300ppm [17] while the established LOD for GA was found to be 1060ppm [18]. Ao et al. have reported the LOD and LOQ for glabridin in liquorice root extracts as 34 and 114 ppm respectively [19]. while Tian et al. [8] showed the LOD for glabridin and Glycyrrihizic acid to be as 0.229 and 0.464ppm respectively. Nocca et al. [20] have also reported LOD of 11.4 ppm and LOQ of 13.7ppm for glabridin of liquorice extract with no such values for GA. Viswanathan & Mukne [10] report the LOD and LOQ for glabridin in their HPLC method as 0.5ppm and 2ppm respectively
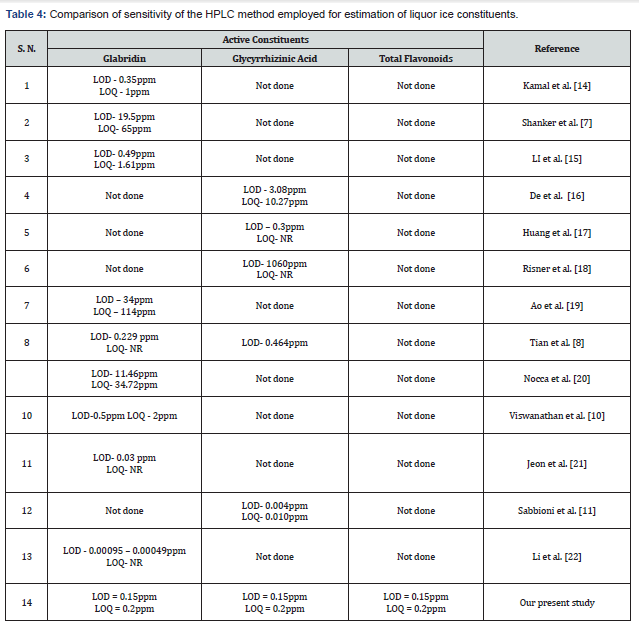
There are certain papers that have reported appreciable LOD and LOQ values as compared to our present reported values of 0.15 and 0.2ppm, both for GB and GA. These include a report of the LOD of glabridin in a cosmetic cream by HPLC with photodiode array detection (PAD) was reported 0.03ppm [21] and the LOD of 0.00095 and LOQ of 0.00049ppm respectively for glabridin [22] and Sabbioni et al. [11] reporting a value of 0.004ppm for LOD of GA and 0.10ppm for LOQ for GA. We believe that such low values of LOD and LOQ is due to the use of an ultrahigh-performance liquid chromatography tandem mass spectrometry (UHPLC-MS/MS) with the use of a highly sensitive detector assay for glycyrrihizin and glabridin, since availability of such high-end instruments is scarce in all labs, our method using regular RP-HPLC method with appreciable LOD and LOQ appears an interesting alternative.
Discussion
About 30 species of Glycyrrhiza are reported to date and liquorice has benefits of treating coughs, colds, and chills [23]. Known for its anti-inflammatory, anti-viral, antioxidant, antiulcerative and antitussive activity [24], liquorice is also used in tackling allergies. Other promising uses includes its anticarcinogenic [25] and anti-hepatotoxic [26,27] properties.
Glabridin and Glycyrrhizic acid are the two main active principals of liquorice. Several literatures exists for determination of glabridin and glycyrrhizic acid from liquorice roots. The content of glabridin in liquorice extract was found to be 3.90% by HPLC and 3.79% by HPTLC [8] while we see the concentration as 5% in our regular extraction process with acetone as the extraction solvent (data not shown). To the best of our knowledge, a HPLC method for simultaneous estimations of GA, GB and TF is not available while methods for separate estimations of these constituents are available.
Glabridin detection was done at 230nm for HPLC by Parveen et al. which is one of the absorption maxima for Glabridin solution [28]. However, we noticed that at 230nm, the peaks of total flavonoids were negligible while at 254nm and 280nm, the peaks of TF were consistent and significantly higher. Hence our method of using 254 nm for glabridin estimation appears the right strategy. Also, the concentrations of flavonoid in the liquorice samples have been estimated as quercetin equivalents (QE). While Husain et al. [29] report a value of 2.25μg/g, Velvizhi & Annapurani [30] report the concentration of total flavonoids as 64μg/mg and Tohma & Gulçin [31] report 4.2μg QE/mg to name a few. The existing reports of TF amount in liquorice clearly indicates that the TF values are variable and cannot be attributed to the method of extraction since the extracts are prepared using solvents such as ethanol. Our present observations of nearly 150mg TF/g of liquorice extract are the highest estimation of TF to date and this could be attributed to the use of sensitive and a robust RP-HPLC method described here. We have compared the observed TF content by the method described here with reported spectroscopic method [32] and found the latter method to underestimate the TF content (data not shown).
The RP-HPLC method described here was employed to measure the GA content in liquorice root samples and the concentrations found were in the range 100-150g GA/kg of dry matter (i.e., 10–15 %). This is consistent with the GA values reported in the literature using HPLC ranging from 7.1 [33] to 8.7% [34]. Jirawattanapong et al. synthesized a derivative of glabridin with an effort to improve its skin penetration properties and enhanced stability in formulation [9] and we believe the current sensitive HPLC estimation method for glabridin would prove useful for scientists engaged in such research.
A few of the liquorice flavonoids such as licuraside, isoliquiritin, and licochalcone A have found use as effective antibrowning and depigmenting agents [35]. The importance of flavonoids is due to their antioxidant activity in vitro and in vivo. The flavonoids of liquorice have 100 times more antioxidant activity than the antioxidant activity of vitamin E. Since such flavonoids are effective scavengers of most oxidizing molecules, and free radicals are causative agents for human health hazards such as cancer, aging, heart diseases, and gastric problems [36], the accurate estimation of TF will have clinical significance.
Licorice is known for its anti-viral, anti-inflammatory, antitumor, anti-microbial, and antioxidant properties [37] and used to treat cough owing to its demulcent and expectorant properties. Having more than 20 triterpenoids and approximately 300 flavonoids, liquorice finds its use also in the treatment of acidity, jaundice, hiccough, hoarseness, bronchitis, and diarrhea [38]. Among licorice’s constituents, Glycyrrihizic acid or Glycyrrihizin has been shown to exhibit anti-viral and anti-microbial activity against HIV, HCV, influenza HSV, rotavirus, coxsackievirus, HRSV, and HBV [39] and hence our present paper highlighting accurate estimation of total flavonoids, glycyrrihizic acid and glabridin using RP-HPLC in a single run assumes critical importance.
Conclusion
A systematic quantitative RP-HPLC method with UV detection for GB, GA and TF was successfully developed and validated. The described method is simple, sensitive, accurate and validated as per regulatory guidelines and subsequently successfully applied to stability studies of GB, GA and TF. Factors that influence the stability of GB, GA and TF have also been studied and presented in this article. The present study provides data for the quality control of commercial water and acetone extract samples of liquorice using a precise HPLC method for simultaneous estimation of GA, GB and TF. The chromatographic conditions and sample preparation method is ideal for carrying out stability studies for GB, GA and TF prodrugs.
Acknowledgement
The authors thank the production team of Sava Herbals at Malur, Bangalore, India for providing liquorice plant extract for the experimental work described in this paper. Thanks, are also due to Mr. Vinod Jadhav, Chairman Sava Healthcare Limited, India and Mr. Avinaash Mandale, MD, Sava Healthcare Limited, India for their support and encouragement.
References
- Visavadiya NP, Narasimhacharya AV (2006) Hypocholesterolaemic and antioxidant effects of Glycyrrhiza glabra (Linn) in rats. Mol Nutr Food Res 50(11): 1080-1086.
- Quintana SE, Cueva C, Villanueva Bermejo D, Moreno Arribas MV, Fornari T, et al (2019) Antioxidant and antimicrobial assessment of liquorice supercritical extracts. Ind Crops Prod 139(1): 111496.
- Bailly C, Vergoten G (2020) Glycyrrhizin: An alternative drug for the treatment of COVID-19 infection and the associated respiratory syndrome? Pharmacol & Therapeut 214: 107618.
- Cinatl J, Morgenstern B, Bauer G, Chandra P, Rabenau H, et al. (2003) Glycyrrhizin, an active component of liquorice roots, and replication of SARS-associated coronavirus. Lancet 361: 2045-2046.
- Patel SKS, Lee J K, Kalia VC (2020) Deploying biomolecules as anti-COVID-19 agents. Indian J Microbiol 60(3): 263-268.
- Orhan IE, Deniz FSS (2020) Natural products as potential Leads against coronaviruses: Could they be encouraging structural models against SARS‑CoV‑2? Nat Prod Bioprospect 10: 171-186.
- Shanker K, Fatima A, Negi AS, Gupta VK, Darokar MP, et al. (2007) RP-HPLC method for the quantitation of Glabridin in Yashtimadhu (Glycyrrhiza glabra). Chromatographia 65: 771-774.
- Tian M, Yan H, Row KH (2008) Extraction of glycyrrhizic acid and glabridin from liquorice. Int J Mol Sci 9: 571-577.
- Jirawattanapong W, Saifah E, Patarapanich C (2009) A validated stability indicating HPLC method for analysis of glabridin prodrugs in hydrolysis studies. Drug Discov Ther 3(3): 97-103.
- Viswanathan V, Mukne AP (2016) Development and validation of HPLC and HPTLC methods for estimation of glabridin in extracts of Glycyrrhiza glabra. J AOAC Int 99(2): 374-379.
- Sabbioni C, Mandrioli R, Ferranti A, Bugamelli F, Saracino MA, et al. (2005) Separation and analysis of glycyrrhizin, 18beta-glycyrrhetic acid and 18alpha‑glycyrrhetic acid in liquorice roots by means of capillary zone electrophoresis. J Chromatogr A 1081: 65-71.
- Rauchensteiner F, Matsumura Y, Yamamoto Y, Yamaji S, Tani T (2005) Analysis and comparison of radix Glycyrrhizae (liquorice) from Europe and China by capillary-zone electrophoresis (CZE). J Pharm Biomed Anal 15: 594-600.
- Aoki F, Nakagawa K, Tanaka A, Matsuzaki K, Arai N, et al. (2005) Determination of glabridin in human plasma by solid-phase extraction and LC-MS/MS. J Chromatogr B 15: 70-74.
- Kamal YT, Singh M, Tamboli ET, Praveen R, Zaidi SMA, et al. (2012) Rapid RP-HPLC method for the quantification of glabridin in crude drug and in polyherbal formulation. J Chrom Sci 50(9): 779-784.
- LI L, Zhu W, Yang J, Liu X, Dong Y (2018) Rapid quantitative analysis of six flavonoids in liquorice by ultra-performance convergence chromatography. Food Sci Technol 1: 1-6.
- De AK, Datta S, Mukherjee A (2012) Quantitative analysis of Glycyrrhizic acid from a polyherbal preparation using liquid chromatographic technique. J Adv Pharm Technol Res (2012) 3(4): 210-215.
- Huang T, Chen N, Wang D, Lai Y (2014) Infrared-assisted extraction coupled with high performance liquid chromatography (HPLC) for determination of liquiritin and glycyrrhizic acid in liquorice root. Anal Methods 6: 5986-5991.
- Risner CH (2008) Evaluation of HPLC mobile phases and extracts for the determination of glycyrrhizic acid in Liquorice and Tobacco. J Liq Chromatogr R T 31(9): 1337-1345.
- Ao M, Shi Y, Cui Y, Guo W, Wang J, et al. (2010) Factors influencing Glabridin stability. Nat Prod Commun 5(12): 1907-1912.
- Nocca G, Callà C, Santini SA, Amalfitano A, Marigo L, et al. (2018) Quantitative determination of 18-β-glycyrrhetinic acid in HepG2 cell Line by high performance liquid chromatography method. Intl J Anal Chem 2018: 1-5.
- Jong Sup J, Han Taek K, Myeong Gil K, Moon Seog O, Se Ra H, et al. (2016) Simultaneous detection of Glabridin, (−)-α-Bisabolol, and Ascorbyl Tetraisopalmitate in whitening cosmetic creams using HPLC-PAD. Chromatographia 79(14): 851-860.
- Li G, Nikolic D, van Breemen RB (2016) Identification and chemical standardization of Liquorice raw materials and dietary supplements using UHPLC-MS/MS. J Agric Food Chem 64(42): 8062-8070.
- Mamedov NA, Egamberdieva D (2019) Phytochemical constituents and pharmacological effects of Liquorice: A review. Plan Hum Heal 3: 1-21.
- Pastorino G, Cornara L, Soares S, Rodrigues F, Oliveira MBPP (2018) Liquorice (Glycyrrhiza glabra): A phytochemical and pharmacological review Phytother Res 32(12): 2323-2339.
- Satomi Y, Nishino H, Shibata S (2005) Glycyrrhetinic acid and related compounds induce G1 arrest and apoptosis in human hepatocellular carcinoma HepG2. Anticancer Res 25(6): 4043-4047.
- Wan X Y, Luo M, Li X D, He P (2009) Hepatoprotective and anti-hepatocarcinogenic effects of glycyrrhizin and matrine. Chem Biol Interact 181: 15-19.
- Wu YT, Shen C, Yin J, Yu JP, Meng Q (2006) Azathioprine hepatotoxicity and the protective effect of liquorice and glycyrrhizic acid. Phytother Res 20(8): 640-645.
- Parveen R, Kamal YT, Singh M, Tamboli ET, Rahman S, et al. (2011) Development of analytical methods (RP-HPLC and HPTLC) for the fast analysis of Glabridin in crude drug and Unani formulations. Planta Med 77: 90.
- Husain A, Ahmad A, Mujeeb M, Khan SA, Alghamdi AG, et al. (2015) Quantitative analysis of total phenolic, flavonoid contents and HPTLC fingerprinting for standardization of Glycyrrhiza glabra roots. Herbal Medicine 1(1): 1-9.
- Velvizhi S, Annapurani S (2018) Estimation of total flavonoid, phenolic content, and free radical scavenging potential of Glycyrrhiza glabra. Asian J Pharm Clin Res 11(4): 231-235.
- Tohma HS, Gulcin I (2010) Antioxidant and radical scavenging activity of aerial parts and roots of Turkish liquorice (Glycyrrhiza glabra). Int J Food Prop 13: 657-671.
- Handique P, Deka AK, Deka DC (2020) Antioxidant properties and phenolic contents of traditional rice-based alcoholic beverages of Assam, India. Natl Acad Sci Lett43: 501-503.
- Lauren DR, Jensen DJ, Douglas JA, Follett JM (2001) Efficient method for determining the glycyrrhizin content of fresh and dried roots, and root extracts, of Glycyrrhiza species. Phyochem Anal 12(5): 332-335.
- Fenwick GR, Lutomski J, Nieman C (1990) Liquorice, Glycyrrhiza glabra L-composition, uses and analysis. Food Chem 38: 119-143.
- Fu B, Li H, Wang X, Lee FSC, Cu S (2005) Isolation and identification of flavonoids in Liquorice and a study of their inhibitory effects on tyrosinase. J Agric Food Chem 53(19): 7408-7414.
- Rajpurohit A, Nayak DS, Patil S, Mahadevan KM (2017) In vitro antioxidant, antimicrobial and admet study of novel furan/benzofuran c-2 coupled quinoline hybrids. Int J Pharm Pharm Sci 9(10): 144-153.
- Kim HS, Suh KS, Ko A, Sul D, Choi D, et al. (2013) The flavonoid glabridin attenuates 2-deoxy-D-ribose-induced oxidative damage and cellular dysfunction in MC3T3-E1 osteoblastic cells. Int J Mol Med 31: 243-251.
- Damle M (2014) Glycyrrhiza glabra (Liquorice) - a potent medicinal herb. Int J Herb Med 2: 132- 136.
- Wang L, Yang R, Yuan B, Liu Y, Liu C (2015) The antiviral and antimicrobial activities of licorice, a widely used Chinese herb. Acta Pharm Sin B 5(4): 310-315.






























