Assessment of the Antisickling Activities of Extracts and Isolated Compounds from Dried Leaves of Rhaphiostylis beninensis [Planch. Ex Benth]
Ajayi TO1, Akintayo CO2, Olaniran DM1, Iyobhebhe M3, Ojo OA3 and Owolabi OV4
1Department of Pharmacognosy, University of Ibadan, Nigeria
2Department of Physiology, Afe Babalola University, Nigeria
3Department of Biochemistry, Landmark University, Omu Aran
4Department of Biochemistry, Afe Babalola University, Nigeria
Submission: February 01, 2021; Published: April 20, 2021
*Corresponding author: Akintayo CO, Department of Physiology, College of Medicine and Health Sciences, Afe Babalola University, Nigeria
How to cite this article:Leaves of Rhaphiostylis beninensis [Planch. Ex Benth]. Glob J Pharmaceu Sci. 2020; 8(2): 555734. DOI: 10.19080/GJPPS.2021.08.555734.
Abstract
Introduction: Sickle cell disease is one of the most severe monogenic disorders of the blood in the world, with the haemoglobin undergoing polymerization in absence of oxygen, resulting in erythrocyte rigidity and vaso-occlusion, which is central to the pathophysiology of this disease. Rhaphiostylis beninensis [Planch. ex Benth] (Icacinaceae) has been known for its folkloric use as it possesses some of the imperative medicinal activities including anti-inflammatory, analgesic, aphrodisiac and antimicrobial activities. This assessment is aimed at determining the antisickling activity of dried leaves of Rhaphiostylis beninensis [Planch. ex Benth] (Icacinaceae) Materials and Methods: This was based on the principle of haemoglobin gelation on deprivation of oxygen with sodium metabisulphite-induced sickling action resulting in haemolysate formation which is then mixed with the various concentrations of the crude extract, and isolated compounds of Rhaphiostylis beninensis [Planch. ex Benth] (Icacinaceae) and then analyzed with ultraviolet spectrophotometer to determine the change in absorbance (optical density) against time. Results and Discussion: The rate of polymerization, relative percentage polymerization and relative percentage inhibition suggests that the antisickling activity of Rhaphiostylis beninensis [Planch. ex Benth] (Icacinaceae) may be concentration-dependent and thus warrants for further and elaborate investigation to prove vividly if in-fact the crude extracts, fractions or isolated compounds from the plant possess significant antisickling property. Conclusion: Considerable level of antisickling activity is required to certify the use in the management of sickle cell disease, where positive results may lead to revolutionary development of new lead candidate for the management or treatment of sickle cell disease.
Keywords:Antisickling; Sickle cell disease; Rhaphiostylis beninensis; Isolated compounds; Polymerization
Introduction
Sickle cell disease (SCD) is one of the most prevalent type of hemoglobinopathy which is also known as drepanocytosis; a life-long genetic blood cell disorder characterized by erythrocytes or red blood cells that presents a rigid, sickle shape and anomaly in structure of the blood cells. It is a genetically inherited disease in which there is a single base substitution in the gene encoding the human β-globin subunit resulting in replacement of β6 glutamic acid by valine, leading to the devastating clinical manifestations of SCD [1,2]. Sickle cell syndrome is inherited as a double-homozygous existence or occasionally, the acquisition of the heterozygous state of the HbSS or HbAS gene from both parents. The prevalence is commonest with the syndromes are sickle cell disease (HbSS), hemoglobin SC disease (HbSC) and hemoglobin Sβ thalassemia (HbSβthal) minor and major. The first-line clinical management of sickle cell disease involves medullar transplantation, continuous and repeated blood transfusion to stabilize the patient’s hemoglobin level, and the use of chemical agents which interfere with the mechanism and/or kinetics of the sickling process. Unfortunately, all current proposed therapies are quite expensive and have attendant risk factors in terms of clinical use [3]. Therefore, there is a need for more definite and effective treatments for the disease. Herbal extracts have been used in African folk medicine for decades in the management of various ailments especially sickle cell disease. Phytochemicals in the plant extracts have the restorative movement and is used as a part of accustomed practice by conventional healers [4]. Various plant parts and extracts have been employed to affect the inversion of the sickled erythrocytes in-vitro. Rhaphiostylis beninensis [Planch. ex Benth] (Icacinaceae) is a woody, sprawling or scrambling glabrous, evergreen shrub or liane native to Tropical Africa, belonging to the family Icacinaceae, and one of three [3] accepted species in the genus Rhaphiostylis. Other species include Rhaphiostylis madagascareinsis Capuron and Rhaphiostylis parvifolia (S. Moore) Exell (“Rhaphiostylis — The Plant List”, 2020). It is traditionally used as an anti-inflammatory by the Bantu people of Africa. Traditionally, it is used considerably in the management of rheumatic disorders, skin illness, mental illness, convulsion and problems of the eye [5]. It is also believed to be useful as an insect repellent, as well as in the management of abnormal growth in some various part of the body [6]. Rhaphiostylis beninensis [Planck ex Benth] (Icacinaceae) is found in the South and Eastern parts of Nigeria and other West African countries [7]. It is called by different local names in Nigeria such as “Kpolokoto” by Igbos, “Usuende” by Benin and “Umeni” by the Urhobos [8]. The plant is used in ethnomedicine in the treatment of several illness and symptoms which includes fever, rheumatism, constipation, mental disorder, pain management and in ophthalmology [5,9]. It is broadly known in ethnomedicine in which the parts of a plant or the whole plant would possess or be able to dispense recuperating properties. It is recognized worldwide that conventional medications can be investigated and abused to be utilized for manufactured pharmaceutical products for improved wellbeing administration. Because of high death rate of patients with sickle cell disease, particularly in young ones, and since chemotherapy has its undesirable effects, there is necessities for reasonable medication advancement that must grasp manufactured medications as well as normal items (phytomedicines/home grown medications), normally happening hostile to sickling operators which can be gotten from out immense timberland assists and can be utilized to viably deal with the sickle cell patient and treat frail condition going with this issue. The present correspondence audits the accessible literature revealing the anti-sickling properties if numerous plants as revealed in the blood tests.
Materials and Methods
Materials and chemicals
The materials used in the assay include Parahydroxybenzoic acid, sodium metabisulphite, methanol, ethyl acetate, distilled water, Pasteur pipette, 4ml of sickle-cell blood in EDTA bottle, test tubes, test-tube rack, conical flasks, centrifuge machine, 5 smallsized glass tubes with cover, weighing balance, measuring cylinder, syringe, spectrophotometer, clock, aluminum foil, separating funnel, beakers, Glass cylinder, micropipette and vortex mixer
Plant material
Rhaphiostylis beninensis [Planch. ex Benth] (Icacinaceae) leaves were collected from Sagamu environs, Ogun State, Nigeria, including the aerial parts of the plant for the purpose of identification. The plant was authenticated at Forest Herbarium Ibadan located at Jericho, Ibadan as voucher specimen FHI – 108407.
Extraction of plant material
The dried leaves of Rhaphiostylis beninensis [Planch. ex Benth] (Icacinaceae) were hand-picked from the stem and blended with grinding machine. The ground leaves (50g) were subjected to cold extraction using ethyl acetate as solvent over a period of 72 hours. The extract was then filtered and then the filtrate was concentrated using rotary evaporator to derive a paste of crude of extract.
Blood sample collection
Upon submission of application for ethical approval to UI/ UCH bioethics committee and the participants consented to the purpose of sample collection, fresh blood samples from confirmed sickle cell patient in non-crisis state or that has not been recently transfused with HbAA blood and attending clinic regularly, were collected (2ml each) via venipuncture into anticoagulant bottles (Ethylene diamine tetra-acetic acid, EDTA) by the method described by [10] from the Laboratory Department, Oni Memorial Hospital, Ring Road, Ibadan, Oyo State.
Preparation of red blood cell suspension
Using the haemoglobin gelation experiment, the method used by [11], was employed in this evaluation with the principle that haemoglobin-S (HbSS) undergoes gelation on deprivation of oxygen. The blood samples were centrifuged at about 4000 revolutions per minutes (rpm) for 10 minutes. The blood plasma is then removed with Pasteur pipette and the residual erythrocytes are washed thrice with equal volume of isotonic saline (0.98%w/v NaCl) by repeating the centrifugation. The washed erythrocytes were then suspended in a volume of isotonic saline equivalent to the volume of the displaced plasma and frozen at 0oC. The samples were subsequently thawed to produce the hemolysates for the hemoglobin gelation test.
Anti-sickling assay
The hemoglobin gelation (polymerization) experiment used was based on the method described by [12]. The underlying principle is that hemoglobin-S (HbS) undergoes gelation and polymerization when deprived of oxygen; sodium metabisulphite was used as a reductant or deoxygenating agent, which is responsible for inducing the sickling action of the HbSS. Exactly 4.8 ml of 2% sodium metabisulphite, 0.1 ml HbS hemolysate and 0.1 ml of isotonic saline (0.98% NaCl) were rapidly mixed in a cuvette and the optical density (absorbance) readings taken at 700 nm at two-minute intervals for 10 minutes using a spectrophotometer (Spectrumlab 752s); this served as the negative control for this experiment.
Statistical analysis
Statistical analyses were done using Excel spreadsheet software (v16.0, Microsoft, Redmond, WA, USA). All experiments were conducted in quadruplicate, and the data were expressed as mean ± standard deviation. A p-value (calculated by t-test) of less than 0.05 was considered statistically significant (Table 1).
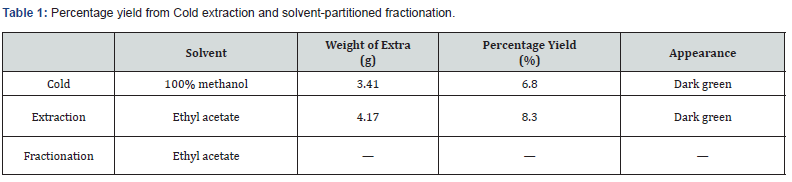
Results
Percentage yield from Cold extraction and solventpartitioned fractionation
The cold extraction of Rhaphiostylis beninensis at 3.41 g using 100 % methanol solvent yielded 6.8 % with a dark green appearance, while extraction with ethyl acetate yielded at 4.17 g yielded 8.3 % with dark green appearance. However, there were zero with no indication of appearance of fractionation using ethyl acetate as a solvent for Rhaphiostylis beninensis respectively
Optical Density (absorbance) of compounds isolated from Rhaphiostylis beninensis
The results from table 2 below indicated the optical density in terms of absorbance of isolated compounds of Rhaphiostylis beninensis from both the positive control, Parahydroxybenzoic acid (PHBA) and negative control: 0.98% NaCl, However, no significant differences (p<0.05) were observed in the isolated compounds of Friedelinol at dosage of 5 mg/ml; 10 mg/ml and Friedelin at dosage of 5 mg/ml and 10 mg/ml when compared with both the negative and positive control groups.

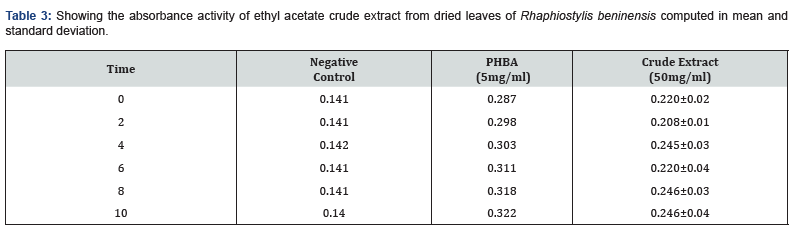
The absorbance activity of ethyl acetate crude extract from dried leaves of Rhaphiostylis beninensis
The results obtained in table 3 also showed no significant change (p<0.05) in the absorbance activity of ethyl acetate crude extract of Rhaphiostylis beninensis dried leaves at 50 mg/ml when compared with the positive control, Parahydroxybenzoic acid (PHBA) at the dosage of 5 mg/ml and negative control: 0.98% NaCl at 2 minutes interval respectively.
Rate of Polymerization, Relative Percentage Polymerization and Relative Percentage Inhibition of sickling of Erythrocytes
The table 4 showed the rate of polymerization of the negative control group at -0.0001 yielded 100% relative percentage of polymerization and 0.0% relative percentage of inhibition. However, the positive control at dose of 5 mg/ml Friedelinol isolation/active compound from Rhaphiostylis beninensis and 10 mg/ml Friedelinol, 5 mg/ml and 10 mg/ml Fredelin yielded 0.0 % relative polymerization and inhibition respectively.
a) Value is expressed as Mean±S.D; Positive control: PHBA - Parahydroxybenzoic acid, Negative control: 0.98% NaCl.
b) Value is expressed as Mean±S.D; Positive control: PHBA - Parahydroxybenzoic acid, Negative control: 0.98% NaCl.
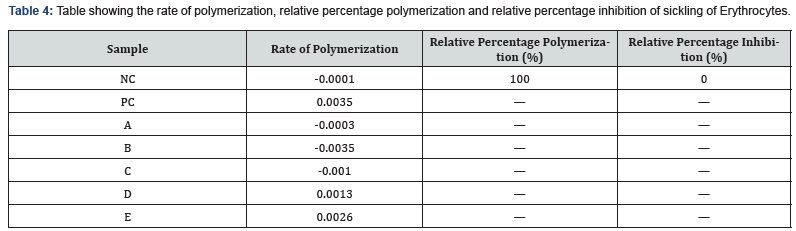
KEYS: NC
Negative Control. PC: Positive Control, A:5mg/ml Friedelinol (Test Compound isolated from Rhaphiostylis beninensis), B:10mg/ ml Friedelinol (Test Compound isolated from Rhaphiostylis beninensis), C: 5mg/ml Friedelin (Test Compound isolated from Rhaphiostylis beninensis), D:10mg/ml Friedelin (Test Compound isolated from Rhaphiostylis beninensis), E: 50mg/ml Crude extract (ethyl acetate extract from dried leaves of R. beninensis)
Discussion
The in-vitro technique employed in this antisickling analysis was based on the simulation of the established in-vivo sicklingprecipitating factor (which is decreased oxygen tension) by using sodium metabisulphite (Na2S2O3) as a physiologically acceptable reductant. Complete sickling of haemolysate was affected by the presence of 2% sodium metabisulphite which deoxygenated the red blood cell thereby creating a state of hypoxia which mimics the events that occurs during sickle cell crisis. This event is responsible for the characteristic sickle cell shapes of the red blood cells.
The blood sample collection from confirmed HbSS patient was done via veno-puncture after establishing that the participants are in non-crisis state and have not been recently transfused with HbAA blood and transferred into anticoagulant bottles (EDTA bottles) to prevent coagulation of the whole blood during the period of transportation. The necessity for the use of erythrocyte suspension instead of the whole blood composition is due to the presence of plasma components and certain metabolic co-factors that can instigate several immunological reactions and eventuality of interactions with the red blood cells such as the presence of thrombin that is implicated in clot formation, which converts fibrinogen to fibrin. The removal of plasma by centrifugation and washing with normal saline is required to preserve the suspended red blood cell and to prevent coagulation upon storage with may inadvertently produce false negative or false positive results. The cold extraction using ethyl acetate and 100% methanol was done to prevent extensive leaching and denaturation of essential secondary metabolites and compounds that may be thermolabile when subjected to heat. Some of the secondary metabolites found in Rhaphiostylis beninensis includes: Alkaloids, Flavonoids, Cardiac glycosides, Tannins, Phlobatannins, Phenols and Terpenoids [13]. The use of methanolic extra is justified due to its moderate polarity and the relative affinity for these secondary metabolites based on the polarity. Solvent-partitioned fractionation is very useful for compounds with varying polarity such that these compounds are partitioned and concentrated in suitable solvent of relative polarities. Some of the solvents used for fractionation include n-butanol, hexane, chloroform, ethyl acetate, dichloromethane and aqueous solvent. Studies have showed different isolated active compounds of promising herbal plants in the management of sickle cell disease [14-17]. The fractionation in this experiment was not sufficient due to the relatively low yield from the cold extraction and any attempt to fractionate crude extract will lead to loss of the extract. The percentage yield of the cold methanolic extraction was calculated to be 6.8% and that from ethyl acetate crude extract was 8.3% while the attempt to fractionate had no yield. Compound isolated from R. beninensis are β-amyrin, Friedelinol, Friedelin and Stigmasterol. The higher the rate of polymerization, the greater the propensity the HbSS sickling activity and thus, the lower the antisickling activity of the test isolated compounds.
In table 4, the values form the rate of polymerization for the negative control, compound Friedelinol at 5mg/ml and 10mg/ ml concentrations and Friedelin at 5mg/ml concentration all displayed gross negative values which is not expected for this anti-sickling assay method [18]. Inadvertently, the negative value for the negative control serving as reference sample along with positive control, impaired the proper analysis of the anti-sickling activity of the test samples and computation of the relative percentage polymerization and inhibition could not be carried out.
However, with the values for rate of polymerization in Table 3, inference can be made that negative control (normal saline) should have 100% rate of polymerization and 0% inhibition of polymerization while positive control (Parahydroxybenzoic acid) while has considerable antisickling activity should be the basis for comparing the anti-sickling activity of test samples at various concentrations. In this assay, the rate of polymerization can be used to analyze the anti-sickling activity of the test samples as lower rate of polymerization indicates increasing anti-sickling activities. Using the negative control as reference with regards to the negative integer value, Friedelinol at 5mg/ml concentration has considerably lower rate of polymerization (-0.0003) and subsequently relative anti-sickling activity compared to negative control (-0.0001) while Friedelinol at 10mg/ml has even lower rate of polymerization (-0.0035) and thus higher anti-sickling activity. Friedelin at 5mg/ml showed lower rate of polymerization compared to negative control in the negative integer, and therefore maybe have considerable anti-sickling activity. Friedelin at 10mg/ml show marked increase in rate of polymerization when compared with the negative control and this suggest reduced anti-sickling activity [14]. The rate of polymerization for the Ethyl acetate extract is relatively higher also when compared to the negative control and this may suggest reduced or absent antisickling activity, this may be as a result of inadequate extraction or concentrating of compounds with anti-sickling activity into ethyl acetate solvent based on relative polarities and affinities.
Conclusion
The results of this study suggest that Rhaphiostylis beninensis may possess varying anti-sickling activities when the isolated compounds are used at different concentrations, in this assay, for instance, Friedelinol showed increasing anti-sickling activity as the concentration increases, while Friedelin showed decreasing activity with increasing concentration. Therefore, further and elaborate assays should be done on methanolic extract and various solvent-partitioned fractions of different plant parts of Rhaphiostylis beninensis. Also, other isolated compounds (Stigmasterol and β-amyrin) from Rhaphiostylis beninensisshould be concisely evaluated for anti-sickling activities.
References
- Mpiana, P T, Tshibangu, D S, Shetonde, O. M Ngbolwa, K N (2007) In vitro antidrepanocytary activity (anti-sickle cell anemia) of some Congolese plants. Phytomedicine 14(2-3): 192-195.
- Nelson D L, Cox MM (2008) Lehninger Principle of Biochemistry. Freeman Press, pp. 75-189.
- Mpiana P T, Mudogo V, Kabangu YF (2009) Antisickling activity and thermostability of anthocyanins extract from Congolese plant, Hymenocardia acida (Hymenocardiaceae). International Journal of Phytochemistry 5: 65-70.
- Belmain S R, Golo P, Andantes H F, Atarigiya H, Chare (2000) Toxicity and repellency of ethnobotanicals used in Ghana as post-harvest protectants. In Abstract of presentations on selected topics at the 14th International Plant Congress (IPPC). Phytoparasitica 28(1): 87-90.
- Odugbemi JE (2008) A Textbook of Medicinal Plants from Nigeria. Lagos University Press Lagos, pp. 597.
- Adjanohoun E J, Ake Assi L (1979) Contribution to the census of medicinal plants from Côte d’ Center National Floristique, University d’Abidjan, Abidjan, Côte d’Ivoire, pp. 351-358.
- Keay RW (1989) Trees of Nigeria. Oxford University Press, UK, pp. 310.
- Lasisi AA, OM Folarin, EO Dare, OA Akinloye, MO Fisuyi (2011) Phytochemical, antibacterial and cytotoxic evaluation of raphiostylis beninensis [Hook F. Ex Planch] stem bark extracts. Int J Pharma Bio Sci 2(3): 489-495.
- Bouquet A, Debray M (1974) Medicinal Plants of Ivory Coast. Trav Doc Orstom 32: 1-4.
- Oyedapo O O, Akinpelu B A, Akinyinka M O, Akinwunmi K F, Sipeolu FO (2010) Red blood cell membrane stabilizing potentials of extracts of Lantana camara and its fractions. International Journal of Plant Physiology and Biochemistry 2(4): 46-51.
- Uwakwe AA, Nwaoguikpe RN (2008) In-vitro antisickling effects of Xylopia aethiopica and Monodora myristica. Journal of Medicinal plant Research 2(6): 119-124.
- Noguchi CT, Schechter AN (1985) Sickle hemoglobin polymerization in solution and in cells. Annual Review of Biophysical Chemistry 14: 239-263.
- Ofeimun J O, Mbionwu M (2014) Cytotoxic growth and inhibitory activity of aqueous extracts of root and leaf of Rhaphiostylis beninensis [Planch ex Benth] and Pyrenacantha staudtii (Icacinaceae). Journal of Pharmacy Bioresources 11(1): 8-14.
- Iwu M N, Igboko O O, Onwubiko H, Ndu U P (1988) Effect of Cajanus cajan on gelation and oxygen affinity of sickle cell hemoglobin. Journal of Ethnopharmacology 23(1): 99-104.
- Said O, Khalil K, Fulder S, Azaizeh H, (2002) Ethnopharmacological survey of medicinal herbs in Israel, the Golan Heights and the West Bank region. J Ethnopharmacol 83(3): 251-265.
- Adejumo O E, Owa-Agbanah I S, Kolapo A L, Ayoola M D (2011) Phytochemical and antisickling activities of Entandrophragma utile, Chenopodium ambrosioides and Petiveria alliacea. Journal of Medicinal Plants Research 5(9): 1531-1535.
- Adejumo O E, Kolapo A L, Roleola O P, Kasim L S (2010) In vitro antisickling activities and phytochemical evaluation of Plumbago zeylanica and Uvaria chamae. African Journal of Biotechnology 9(53): 9032-9036.
- Adejumo O E, Kolapo A L, Folarin A O (2012) Moringa oleifera (Moringaceae) grown in Nigeria: In vitro antisickling activity on deoxygenated erythrocyte cells. J Pharm Bioallied Sci 4(2): 118-122.






























