Heralding the Drug Related Problems by Using Hepler-Strand Classification in a Tertiary Care Teaching Hospital
Divya Gopineni1, Manohar Reddy YV2*, Reddy Mahesh N2, Rajesh A2 and Karun Kumar J2
1 Department of Pharmacy Practice, Sri Padmavathi School of Pharmacy, India
2 Department of Pharmacy Practice, Sri Padmavathi School of Pharmacy, India
Submission: February 05, 2020; Published: February 19, 2020
*Corresponding author: Manohar Reddy YV, Department of Pharmacy Practice, Sri Padmavathi School of Pharmacy, Tiruchanoor, Tirupati, Andhra Pradesh, India
How to cite this article:Divya G, Manohar R YV, Reddy M N, Rajesh A, Karun K J. Heralding the Drug Related Problems by Using Hepler-Strand Classification in a Tertiary Care Teaching Hospital. Glob J Pharmaceu Sci. 2020; 7(5): 555725. DOI: 10.19080/GJPPS.2020.07.555725.
Abstract
Drug related problems (DRP) are generally referred as “an event (or) circumstance involving in drug treatment at all stages from prescribing to dispensing, which interferes actually or potentially with the patient achieving an outcome of pharmaceutical care. Various classifications were involved in drug related problems among which Hepler–Strand classification is widely followed. This study aimed to identify and report the drug related problems by using Hepler-strand classification and to educate the patients regarding safe use of medications. This is a Prospective observational study which was conducted over a period of 6 months in inpatient wards of General Medicine, Pediatrics and General Surgery at Sri Venkateswara Ramnarayan Ruia Government General hospital. A total of 150 patients were analysed during the study period. Among them76 (50.7%) were male and 74 (49.3%) were female patients. Among 150 prescriptions majority were found with DRP’s 89 (59.4%) followed by prescriptions without DRP’s was 61 (40.6%). Within 89 prescriptions a total of 277 DRP’s were observed. It includes 107 DRP’s in surgery; followed by 131 DRP’s in general medicine and 39 DRP’s in paediatrics. In a total of 277 DRP’s drug interactions was found to be in 264 cases, ADR’s in 4 cases, others in 9 cases. In those 264 drug interactions, drug-drug interactions (205), followed by drug-food interactions (59) were observed. The involvement of the clinical pharmacist may also improve the better usage of drugs in hospital and to decrease the economic status of patient.
Keywords:Drug related problems; Pharmaceutical care; Hepler–Strand classification; Drug interactions; Adverse drug reactionsn
Abbreviations: DRP: Drug related problems; PCNE: Pharmaceutical Care Network Europe
Introduction
Definition
Drug related problems (DRP) is defined as “an event (or) circumstance involving in drug treatment at all stages from prescribing to dispensing, which interferes actually or potentially with the patient achieving an outcome of pharmaceutical care” [1,2].
These are common in the patients, which are caused by polypharmacy, physiological changes related to age, the pharmacokinetics, and the Pharmacodynamics. So, very generally guidelines should be avoided, and individualized strategies should be used, which should be the best for every patient, and every situation, where pharmacist-physician-patient communication and individualized pharmacotherapy monitoring are the most important.
Various classifications were involved in drug related problems such as, ABC, American Society of Hospital pharmacist (ASHP), Cipolle/Strand/Morley, Granada Consensus, Hanlon approach, Hepler – strand, Krska et al system, Mackie, NCC – MERP, Pharmaceutical Care Network Europe (PCNE), PI – Doc, Westerlund. Among which Hepler–Strand classification is widely followed.
Hepler–Strand classification
Hepler and Strand also introduced several categories of DRPs. In this approach, problems and causes were not separated.
Definition: An event or circumstance involving a patient’s drug treatment that actually or potentially interferes with the achievement of an optimal outcome [3,4].
In this classification, the DRPs were classified as follows:
a) Untreated indication: Does the patient have an untreated medical condition or indication which may benefit from drug therapy? When reviewing the indication for drug therapy, it’s most significant to consider whether the indication may be an unrecognized ADR. For example: a patient who complains about diarrhoea may be taking antibiotics or other drugs which causes to this problem.
b) Improper drug selection: Does the patient have a medical condition for which the incorrect drug is being taken? To make sure that the most suitable drug has been chosen to treat the patient’s medical condition. For example: a short course of a non-steroidal anti-inflammatory agent is the firstline treatment for acute gout.
c) Sub therapeutic dosage: Does the patient have medical condition for which too small of the correct drug is being taken? The dose and dosing regimen should be individualized based on the patient’s medical condition.
d) Over dosage: Does the patient have medical problem for which too much amount of the correct drug is being taken? It may also take place if a patient takes a drug for a longer period than needed.
e) Adverse drug reaction: Does the patient have a medical condition which is the result of an ADR? The detection of ADR is crucial in the management of any patient since failure to recognize an ADR mat result in continuing patient morbidity
f) Failure to receive drugs: Does the patient have a medical condition that is the result of him/her not receiving a drug? It may due to many factors including non-adherence, poor administration technique, missed doses due to medication errors, sub-standard drugs, non-availability of prescribed drugs, and patient’s incapability to pay for the medication.
g) Drug interactions: Does the patient have a medical condition that is the result of a drug-drug or drug-food interaction? It vary in their clinical significance, and the pharmacist desires to make professional judgment of change in drug therapy is necessary or not [5,6].
h) Drug use without an indication: Is the patient taking a drug for which there is no valid indication? Care is needed here, as the indication for which a drug is used may not be immediately obvious [7,8].
Drug related problems are very common manifesting 54.8% are due to Therapeutic failure, 32.9% are due to adverse reactions, 12.3% are due to Overdose, 49.3% are due to Avoidable [9].
This study aimed to identify and report the drug related problems by using Hepler-strand classification and to educate the patients regarding safe use of medications.
Methodology
A Prospective observational study was conducted over a period of 6 months in inpatient wards of General Medicine, Pediatrics and General Surgery at Sri Venkateswara Ramnarayan Ruia. Apart from those who are not willing to sign in informed consent form, Poison cases, Special population includes pregnancy, Inpatients of burns ward & psychiatry, Tuberculosis, HIV & Opportunistic infection patients, Emergency department rest all inpatients of three departments were included in the study.
Method of Data Collection
A specially designed proforma was used for collecting data which includes patient demographics, past medical history, family and surgical history, co-morbidities, diagnosis and present medications prescribed for each patient. The data was obtained by direct patient interview and from patient case profiles. All inpatient cases in general medicine, paediatrics and general surgery wards were screened for DRP’s. The collected data were analyzed for screening of different categories of DRP’s.
Results
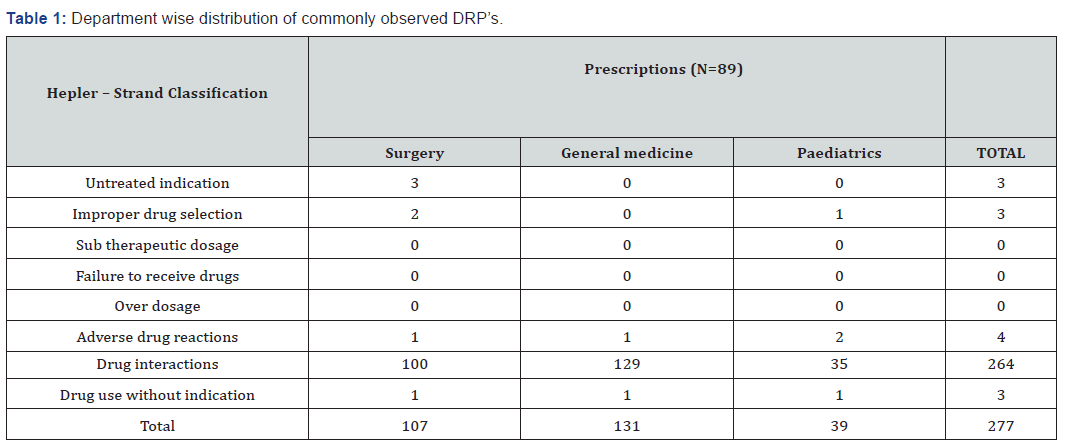
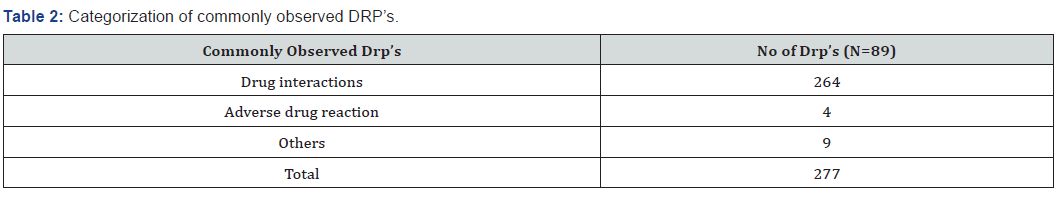
A total of 150 patients were analysed during the study period. Among them76 (50.7%) were male and 74 (49.3%) were female patients. The maximum number of prescriptions was found to be in > 61 years age group (25%) followed by < 30 years age group (24%), 41-50 years age group (19%), 31-40 years age group (16%), and 51-60 years age group (16%). Among 150 prescriptions majority were found with DRP’s 89 (59.4%) followed by prescriptions without DRP’s was 61 (40.6%). Within 89 prescriptions a total of 277 DRP’s were observed. It includes 107 DRP’s in surgery; followed by 131 DRP’s in general medicine and 39 DRP’s in paediatrics (Table 1 & 2).
In a total of 150 patients the observed DRP’s were drugdrug interactions (205), drug-food interactions (59). 264 drug interactions majority of drug interactions in Moderate 178 (67.42%), followed by Minor 65 (24.62%) and Major 21 (7.96%).
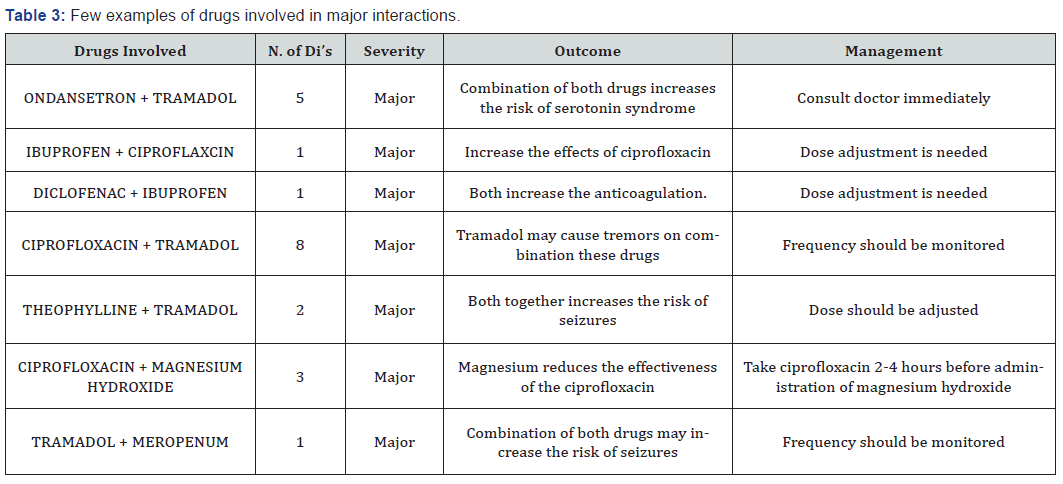
Common drug’s involved in drug interactions were Pantoprazole + Iron Folic Acid, Ondansetron + Tramadol, Ferrous Sulfate + Pantoprazole, Furosemide + Pantoprazole, Ciprofloxacin + Tramadol, Ceftriaxone + Amikacin, Propranolol + Furosemide, Tinidazole + Metronidazole, Calcium Carbonate + Ferrous Sulfate, Theophylline + Pantoprazole, Ferrous Sulfate + Food, Amlodipine + Food, Ciprofloxacin + Food, Acetaminophen + Food, Theophylline + Food (Table 3 & 4).
Other drug related problems
Among 9 other DRP’S, untreated indication (3), improper drug selection (3), and Drug use without an indication (3) were observed.
Discussion
A prospective observational study is one of the best trails to test this particular DRP classification in the practice setting; to assess and evaluate the DRP’s involved in practice of hospital settings. A total of 150 prescriptions were collected from 3 departments each contains 50 prescriptions in tertiary care teaching hospital. This study supports with Costa S, et al. [10] The Indian reports states, DRP’s are more common now a day due to lack of knowledge. Out of 150 prescriptions 89 prescriptions were identified with DRP’s during our study we found that the male patients (50.7%) were found to be higher than the female patients (49.3%). Considering the age group in general medicine and surgery, the maximum number of prescriptions was found to be in > 61 years age group (25%) followed by < 30 years age group (24%), 41-50 years age group (19%), 31-40 years age group (16%), and 51-60 years age group (16%).
In pediatrics, we found the maximum number of prescriptions was found to be in 5-12 years age group (40%) followed by 1-5 years age group (32%) and < 1 year age group (28%).
Among 150 prescriptions majority were found with DRP’s 89 (59.4%) followed by prescriptions without DRP’s 61 (40.6%).
Among 89 prescriptions a total no. of 277 DRP’s were observed in 3 departments. Maximum of 131 DRP’s in general medicine, followed by 107 DRP’s in surgery and 39 DRP’s in pediatrics. And majority of DRP’s with drug interactions (264) followed by adverse drug reaction [4] and other DRP’s [9] which includes UNTREATED INDICATION, DRUG USE WITHOUT AN INDICATION and IMPROPER DRUG SELECTION. This is similar to Peter C et al. [11] study11.
In those 264 drug interactions, drug-drug interactions (205), followed by drug-food interactions (59) were observed.
In our study, we observed severity of 264 drug interactions. Majority of drug interactions in MODERATE 178 (67.42%), followed by MINOR 65 (24.62%) and MAJOR 21 (7.96%). This is similar to Peter C, et al. [11] study [11].
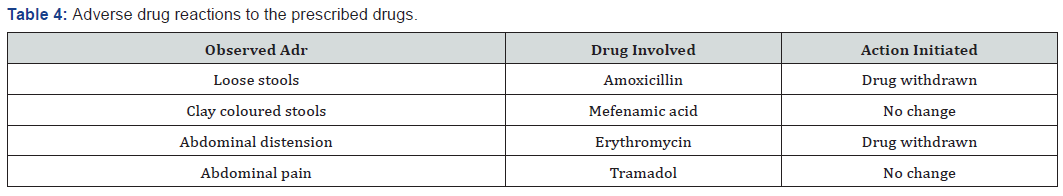
In this study, we found 4 ADR’s among the 264 DRPs. Loose stools, which is caused by amoxicillin and action initiated was drug withdrawn as a positive reaction, clay colored stools, caused by Mefanamic acid and no change in prescription as negative reaction, abdominal distension, caused by erythromycin and action initiated was drug withdrawn as a positive reaction and abdominal pain which is caused by Tramadol and action was not taken as negative reaction.
Clinical pharmacists can play an important role in identifying and resolving DRPs through cooperation with patients and other health-care providers. Potential and actual DRPs can be identified through medication profile reviews, and these problems can be prevented by monitoring therapeutic plans [12]. Clinical pharmacists are the upcoming breed of pharmacists in our country. Clinical pharmacists can contribute improved patient outcomes by monitoring the drug therapy and can also promote rational use of drugs. Clinical pharmacists can provide reactive intervention involving in the patient care and also can offer services like drug information to the other members of the health care team so that effective therapeutic decision can be made [13].
Our study has a number of limitations. The study was prospective observational and seasonal variations were not considered. The data presented here will be useful in future, and development of drug use in hospitals. Finally, this classification permits a useful survey of the type of DRPs faced in practice setting and coordinates to the pharmacist’s intervention and patient outcomes.
Conclusion
From this study it is concluded that majority of prescriptions were found with DRPs, and majorly DRPs were observed in general medicine department. In that, commonly observed DRPs were drug interactions. All these interactions were potential drug interactions and no one interaction is observed among the patients clinically. The involvement of the clinical pharmacist may also improve the better usage of drugs in hospital and to decrease the economic status of patient.
References
- Sudheer Kumar D, Krishnaveni J, Manjula P (2010) “Fundamentals of clinical pharmacy practice”. Pharma Med Press
- Revi Kumar KG, Miglani BD (2012) “A textbook of Pharmacy practice”. Career Publications: 233-257.
- Parthasarathi G, Karin nyfort-Hansen, Milap C Nahata (2012) “A textbook of Clinical Pharmacy Practice Essential concepts and skills”. Universities Press, Second edition: 190-210.
- Adusumilli PK, Adepu R (2014) “Drug related problems: An overview of various classification systems”. Asian Journal of Pharmaceutical and Clinical Research 7(4): 1-10.
- Van Mil JF, Westerlund LT, Hersberger KE, Schaefer MA (2004) “Drug related problem classification systems”. Ann Pharmacother 38(5): 859-867.
- Ruths S, Viktil KK, Blix HS (2007) “Classification of drug related problems”. Translation of topical issues articles published in The Journal of the Norwegian Medical Association 127(23): 3073-3076.
- Linda M, Peter C, Robert J Cipolle, Ruthanne R (1990) “Drug related problems: their Structure and Function”. DICP, the Annals of Pharmacotherapy 24(11): 1093-1097.
- Karen Baxter (2008) “Stockley’s drug interactions”. Pharmaceutical press, Eighth edition.
- Sireesha A (2016) Int J of Res. in Pharmacology and Pharmacotherapeutics 5(4): 311-317.
- Costa N, Santos C, Madeira A, Santos MR, Santos R (2004) “Using two different classifications of drug related problems in community pharmacies”. The Portuguese experience Farmacometer 2(4): 267-285.
- Andrew C, Peter C, Gregory M, Shane L (2007) A comparison of drug related problems identified in RMMRs and HMRs. Australian pharmacist 26(5): 414-420.
- Movva R, Jampani A, Nathani J, Pinnamaneni SH, Challa SR (2015) A prospective study of incidence of medication-related problems in general medicine ward of a tertiary care hospital. J Adv Pharm Technol Res 6(4): 190-194.
- Parveen S (2018) “Clinical pharmacists’ role in identification of drug related problems in a tertiary care teaching hospital in Kadapa.”.” IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) 17(12): 32-37.






























