Development and Evaluation of Microballons Based Extended Release Drug Delivery System for Hypertension Therapy
Ganesh S Bangale1*, GV Shinde2and Rajesh KS3
1Government College of Pharmacy, Amravati (M.S.), India
2Mill Laboratories Pvt Ltd, Gujarat, India
3Zydus Healthcare Ltd, India
Submission:March 09, 2019; Published:August 05, 2019
*Corresponding author:Ganesh S Bangale, Government College of Pharmacy, Amravati-444601, Maharashtra, India
How to cite this article: Ganesh S Bangale, Government College of Pharmacy, Amravati-444601, Maharashtra, India Glob J Pharmaceu Sci. 2019; 7(3): 555718. DOI: 10.19080/GJPPS.2019.06.555718.
Abstract
The objective of the present work was to formulate and evaluate micro balloons of Captopril to for prolongation of gastric residence time. The micro balloons were prepared by the solvent diffusion technique using different ratio of Ethyl cellulose, HPMCK4M & acrylic polymers as Eudragit RL100. Micro balloons characterized for micrometrics properties found in passable to good range, the yield of micro balloons was up to 71.20 ±4.68 to 92.25 ±2.34 % and % entrapment efficiency was found to be 71.40±1.64 to 87.34±2.54% respectively. The size of micro balloons formulations was in the range of 159.1±4.09 to 203.42±5.61. The shape and surface morphology of prepared micro balloons were characterized by scanning electron microscopy.
The micro balloons formed were additionally found to be floating over gastric juice for > 12 hours. A broad range of drug release pattern could be achieved by varying the drug and polymer amount, the best results were obtained in F-4 formulation at the ratio of drug: polymer (1:1) containing HPMC K4M and Eudragit RL100 as a polymer exhibited prolong drug release over 12 hrs. Higher values of correlation coefficients were obtained with Higuchi’s square root of time kinetic. The mechanism of the release of Captopril from formulation was found to be Non-fickian transport. Drug–Polymers interaction has been determined based on FTIR studies. Optimized formulation (F-4) was stable up to 90 days prior storage at 40±20C & 5-8±20C in terms of % yield, %E.E. & % drug released. Non-significant difference was observed in above analyzed parameters, supported by t crit > t stat by t test with p =0.05. The developed Floating micro balloons of Captopril might be clinically used for prolonged drug release in GIT, for better drug utilization and improved patient compliance.
Keywords: Captopril, Micro balloons, Ethyl cellulose, HPMC K4M And Eudragit RL100, Solvent evaporation
Introduction
Multi-particulate drug delivery systems are mainly oral dosage forms consisting of a multiplicity of small discrete units, each exhibiting some desired characteristics. In this system, the dosage of the drug substance is divided on a plurality of subunits, typically consisting of thousands of spherical particles with diameter of 0.05- 2.00mm1. Thus, multi particulate dosage forms are pharmaceutical formulations in which the active substance is present as several small independent subunits, deliver the recommended total dose. Recently much emphasis is being laid on the development of multi particulate dosage forms in preference to single unit system because of their potential benefits such as increased bioavailability, reduced risk of systemic toxicity, reduced risk of local irritation and predictable gastric emptying. Multi particulate systems show better reproducible pharmacokinetic behavior than conventional (monolithic) formulations.
Drug safety may also be increased by using multi particulate dosage forms, particularly for modified release systems. conventional GI transiting dosage forms do not have enough residence time in intestinal absorption region to afford a true once-daily oral delivery system; gastro-retentive micro balloons may be better alternative to slowly release the drug in stomach and upper intestine. This, in turn, would ensure around the clock-controlled delivery of the antihypertensive agent for better management of Hypertension and related indications [1]. Captopril is an anti-hypertensive agent used in treatment of hypertensive disorder. It is highly water-soluble drug with absolute bioavailability of 70-75%.
Captopril is competitive inhibitor of angiotensin-converting enzyme (ACE), the enzyme responsible for the conversion of angiotensin I (ATI) to angiotensin II (ATII). The half-life of Captopril is < 2hr. respectively [2,3]. Present attempts have been to achieve a sufficiently prolonged drug release as per the pharmacokinetic need while providing a controlled release of captopril for 24hrs. Microencapsulation technique, which is a process of applying a thin coating on the small drug particle, shows the following advantages over matrix technique:
a) Provides protection from gastric irritation
b) Microcapsules contain small amount of drug, which is released slowly at the absorption site
c) Microparticles prepared by microencapsulation technique release small drug particle, which float on the GIT fluid and in turn release the drug for extended duration achieving uniform distribution [4,5].
Material and Methods
Captopril was obtained from Cadila healthcare pvt ltd Ahmadabad, Span 80, Liquid paraffin purchased from Chemdyes Corporation, Rajkot, India., HPMC K4M, Ethyl cellulose from Molychem, Mumbai, India, Eudragi RL-100 was obtained from Sigma Aldrich, Mumbai, India All other solvent and reagents were of analytical grade.
Determination of UV Absorption Maxima
10μg/ml solution of captopril was prepared in methanol. The absorbance was measured at entire range of UV (200-400nm) for determination of λ max (wavelength maxima).
Calibration Curve of Captopril in 0.1N HCl Solution (pH 1.2)
Accurately weighed and transferred 100mg of captopril into a 100ml volumetric flask, dissolved and adjusted the volume up to 100ml with 0.1N HCl to get stock solution A (1000μg/ ml). From the stock solution A, 5ml was pipetted out into a 50ml volumetric flask and volume was made with 0.1N HCl to get stock solution B (100μg/ml). From the stock solution B, 2, 4, 6, 8 and 10ml were pipetted out and made up to 50ml with 0.1 N HCl in 50ml volumetric flask to get 4, 8, 12, 16 and 20μg/ ml concentration solutions respectively and absorbance was recorded at 212 nm by UV visible spectrophotometer. Each study was conducted in triplicate [6,7].
Drug-Excipients Compatibility study
FTIR was carried out for solid samples to detect if any interaction were present between the drug and polymers. The samples were prepared by potassium bromide disk method. Powders (Captopril, HPMC K4M, Ethylcellulose, Eudragit RL100) were triturated in agate mortar and pestle to produce fine and uniform mixture. The pellets were prepared by compressing the powders at 20 psi. Pure KBr powder was used as background for baseline correction. Prepared sample disk was placed in sample holder and transferred to sample compartment. Samples were scanned in the region of 4000- 400cm-1 in an FTIR spectrophotometer (Shimadzu 8400S, Japan) [8,9].
Formulation of Captopril Loaded Microballoons
Formulation of Microballoons has been carried out using different polymers, total nine batches were formulated, and composition was shown in (Table 1).
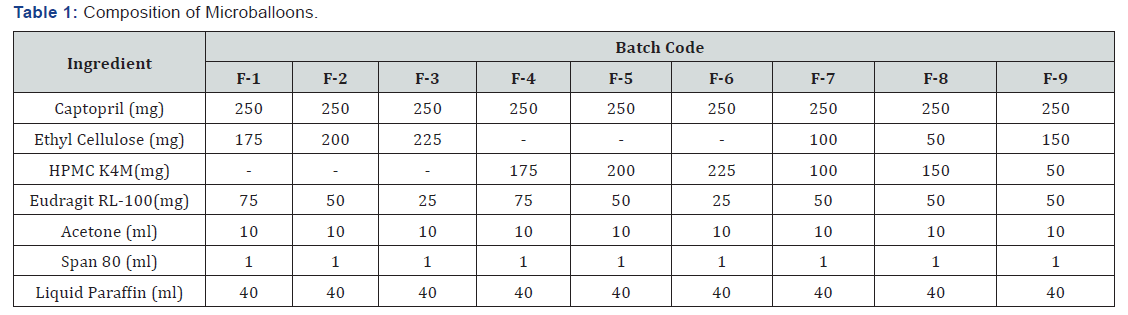
Non-Aqueous Solvent Evaporation Method
a) Dissolved accurately weighed quantity of Drug and Polymer mixtures using 10 ml of acetone in 100 ml beaker.
b) In a separate beaker measured and transferred 40ml of liquid paraffin and added 1ml of span 80.
c) Polymeric solution was slowly introduced into liquid paraffin by stirring at 700rpm using a mechanical stirrer equipped with a three bladed propeller at room temperature resulting formation of w/o emulsion. The emulsion was stirred for 3hr to allow the solvent (Acetone) evaporation and diffusion completely.
d) After 3hr the Microballoons were collected by filtration.
e) The micro balloons were washed repeatedly with petroleum ether (40-60oC) until free from liquid paraffin.
f) The collected microballoons were dried for 1 hr at room temperature and subsequently stored in a dedicator over fused CaCl2 until further study [10-13] (Figure 1).
Evaluation of Microballoons
Micromeretics Study
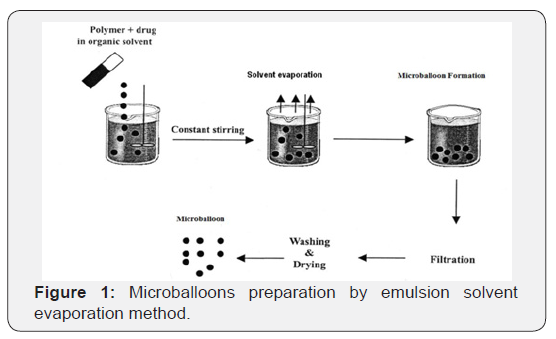
Determination of Bulk Density
Bulk density/fluff density It is the ratio between a given mass of a powder and its bulk volume. Bulk density of the formulated micro balloons was determined by taking a known quantity about 2 g of formulated micro balloons in a clean measuring cylinder, and initial volume was measured [14,15]. The bulk density was calculated by the following equation:
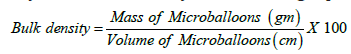
Determination of Tapped density
Tapped density of micro balloons was done by the tapping method [15]. Formulated micro balloons (2g) were transferred into 10mL measuring cylinder. After observing the initial volume of micro balloons, the tapping was continued on a hard surface until no further change in volume was calculated using tapped density apparatus. The tapped density was calculated according to the following formula
% Compressibility Index
The percentage compressibility of powder is a direct measure of the potential powder arch or bridge strength and stability and is computed according to the following equation

Angle of repose
The flow property of floating micro balloons is usually assessed by determining the angle of repose. The angle of repose of micro balloons was determined by fixed funnel method. The micro balloons could fall freely through a funnel until apex of conical pile just touched the tip of the funnel. The angle of repose was determined according to the following formula
Ө = tan-1 (h/r)
h=height of the pile
r = radius of the pile
Yield of micro balloons
The prepared Micro balloons were collected and weight. The measured weight was divided by the total amount of all the non-volatile components used for the preparation of the Micro balloons [16].
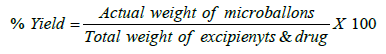
Particle size analysis
The particle size analysis of micro balloons was determined with an optical microscopic method. The particle size of the prepared micro balloons was dispersed in glycerin, and a drop of the above dispersion was transferred on a glass slide and observed under an optical microscope under regular polarized light, and the mean particle size was calculated by measuring 100 micro balloons (n=3) with the help of a calibrated eyepiece micrometer and stage micrometer. The average diameter was calculated using the following formula:
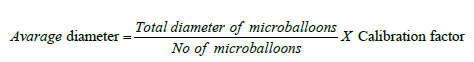
Drug content of the micro balloons
100 mg of micro balloons was crushed in a glass mortar and vortex mixed for 2 hours with methanol. The extract was transferred in to a 50ml volumetric flask and the volume was made up to the mark using methanol and filtered. Drug content was analyzed after suitable dilution by UV spectrophotometer at a wavelength of 212nm against suitable blank. All the studies were carried out in triplicate [17].
Drug Entrapment Efficiency (DEE)
To determine the incorporation efficiency, 50mg of micro balloons were taken and dissolved in 25mL of methanol. Then, the solution was filtered to separate shell fragments. The estimation of drug was carried out using a UV double-beam spectrophotometer (Shimadzu UV-1700 series) at the λ max of 212nm. Drug entrapment efficiency of the micro balloons was calculated using the following formula [18,19].
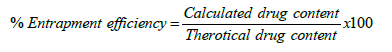
In-vitro Buoyancy)
In-vitro floating study was carried out in a USP dissolution test apparatus 2 containing 250ml of 0.1N HCl as dispersion medium and 0.1% Span 80 as a surfactant. Microballoons were spread over the surface of dispersing medium, at 37 ± 0.5oC and paddle rotated at 100 rpm. Each fraction of micro balloons floating on the surface and those settled down was collected at a predetermined time point. The collected samples were weighed after drying [20].
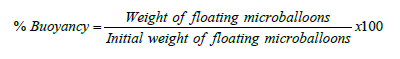
Surface topography (SEM)
The external morphology (Intact micro balloons) and internal morphology (Sliced micro balloons) of micro balloons was analyzed by scanning electron microscopy (JSM-6510LV Jeol-Japan). The morphology of microballoons both before and after dissolution was examined by scanning electron microscopy at angle of incidence 900 with accelerating voltage 5KV [21,22].
In-vitro Drug Release Study
In vitro drug release studies were carried out in USP type II dissolution test apparatus. Dissolution studies were performed in 500ml dissolution medium (0.1N HCl, pH 1.2) at 50rpm maintained at 37 ± 0.5oC. Microballoons equivalent to 50mg of the pure drug were used for dissolution study. At predetermine time intervals an aliquot of 2ml was withdrawn and replenished with fresh medium. Amount of drug in each aliquot was determined on a UV-spectrophotometer at 212nm using a suitable blank. All trials were conducted in triplicate and the average (±SD) reading was noted. Similarly, in-vitro release studies were carried out for the marketed products [23,24].
Release Kinetics
Zero Order Equation
To study the release kinetics, data obtained from In vitro drug release studies were plotted as cumulative amount of drug released versus time [21,25,26].
Qt = K0.t
Where, Qt = percentage of drug released at time t
K0 = release rate constant.
First order equation
The data obtained are plotted as log cumulative percentage of drug remaining versus time which would yield a straight line with a slope of -K/2.303
ln (100 - Qt) = ln 100 – K1.t
here, K1 = first order release rate constant.
Higuchi’s equation
The data obtained were plotted as cumulative percentage drug release versus square root of time.
Qt = KH.t 1/2
Where, KH = Higuchi release rate constant.
Hixson-crowell equation
To study the release kinetics, data obtained from In vitro drug release studies were plotted as cube root of drug percentage remaining in matrix versus time
W0 1/3 – W11/3 = KHC.t
Where, KHC= Hixson-Crowell rate constant.
Korsmeyer-peppas equation
Korsmeyer et al. (1983) derived a simple relationship which described drug release from a polymeric system equation
Qt/Q∞ = KKp.tn
Where, Qt/Q∞ = fraction of drug released at time t,
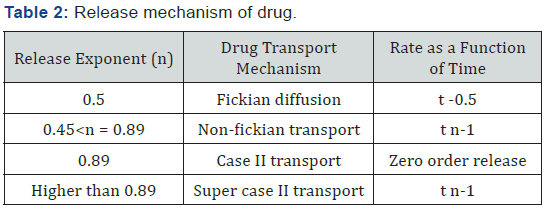
KKP = korsmeyer-peppas rate constant compromising the structural and geometric characteristics of the device, n = release exponent, which is indicative of the mechanism of drug release. To study the release kinetics, data obtained from In vitro drug release studies were plotted as log cumulative percentage drug release versus log time (Table 2).
Stability study
The formulation (F-4) were subjected to accelerated stability studies as per ICH (The International Conference of Harmonization) guidelines. The microballoons were packed in well closed container and subjected to elevated temperature and humidity condition of 40± 2°C & 5-8 ± 2°C respectively. Sample were withdrawn at the end of everyone month and evaluated for active drug content, drug entrapment efficiency, and In-vitro drug release study [27].
Result & Discussion
UV spectrum of captopril
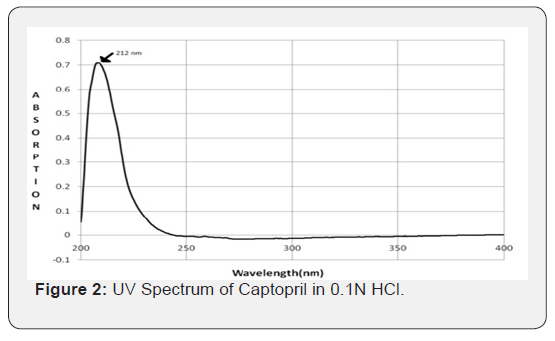
Wavelength of maximum absorption (λ max) of captopril was determined in 0.1N HCl and λ max was found to be 212 nm by UV-Visible spectrophotometer (Figure 2).
Standard calibration curve of captopril
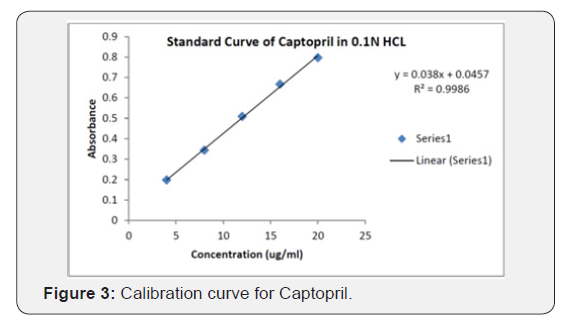
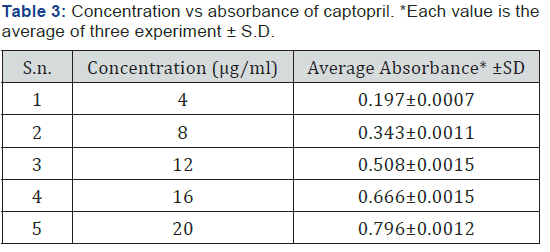
Standard curve was prepared to obey Beer’s Law in the concentration range of 4-20 μg/ml. Estimation of captopril was carried out by UV spectrophotometer at λ max 212 nm in 0.1N HCl respectively. The linear co-efficient was found to be respectively 0.9986 in 0.1N HCl which is closer to 1 at concentration range between 4-20μg/ml. The regression equation generated was generated and using this regression co-efficient equation the assay and % CDR were calculated (Table 3) (Figure 3).
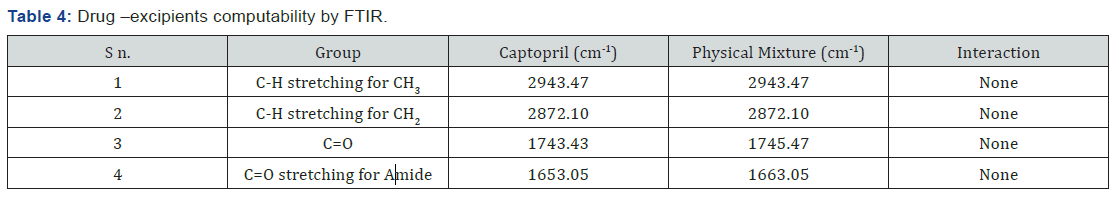
Drug-excipients compatibility study
FTIR spectra of captopril
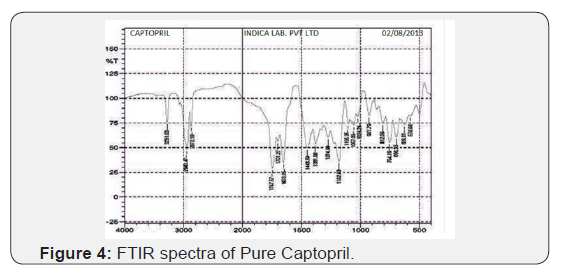
Characteristics peak of C-H stretching for CH3, CH2 and C=O appeared at 2943.47, 2872.10 and 1743.43 cm-1 respectively (Figure 4).
FTIR spectra of physical mixture
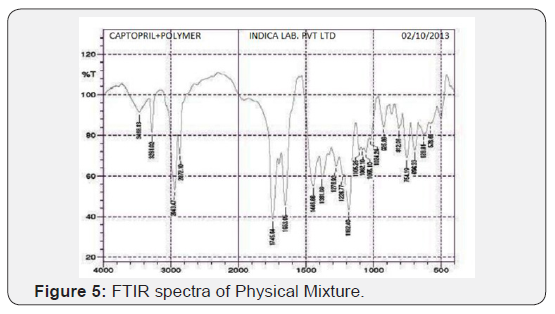
The IR spectra of drug polymer physical mixture (1:1) indicated no, potential interaction between drug and polymer and hence EC, HPMC K4M and Eudragit RL100 were chosen as polymers for further investigations. Chances of interaction between the –C=O groups of Captopril and hydroxyl group of ethyl cellulose exist but no other potential interaction or shift in the position of peak occurs was observed (Table 4) (Figure 5).
Micrometrics study of microballoons
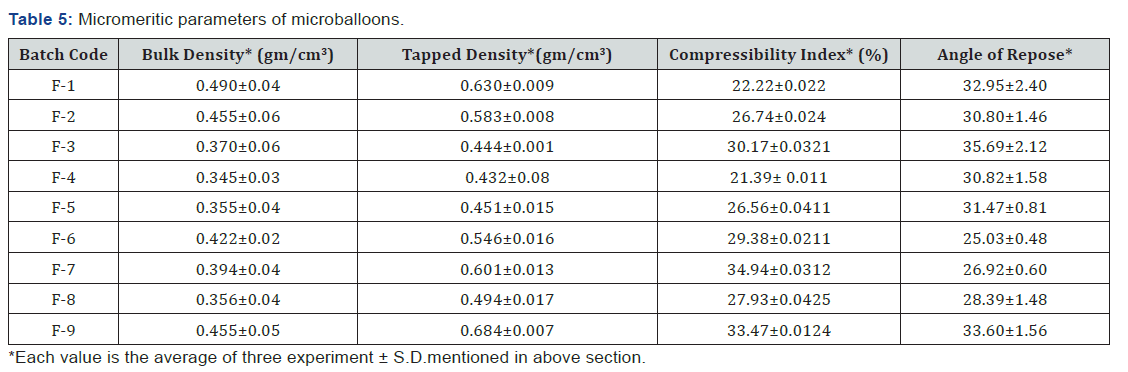
Interparticulate interactions that influence the bulking properties of a powder flow. A comparison of the bulk density and tapped density can give a measure of the relative importance of this interaction in a given powder; such a comparison is often used as an index of the ability of the powder to flow. The bulk density and tapped density for microballoons was found to be 0.490±0.1 to 0.594±0.04 gm/cm3 respectively. The value for compressibility index was found to be 21.39 + 1.011 to 34.94±0.0312 that reflects the acceptable flow property. All above properties mentioned above and value obtained for microballoons was near to boundary of standard limit (Table 5).
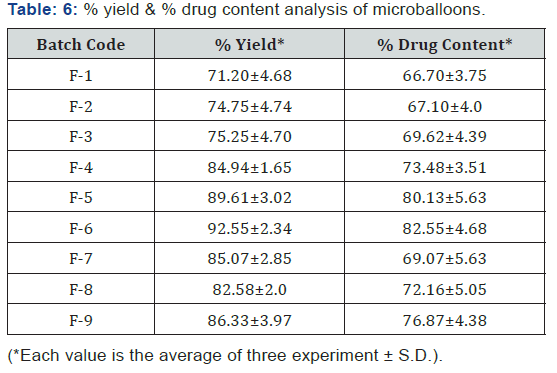
Determination of % yield & drug content of microballoons
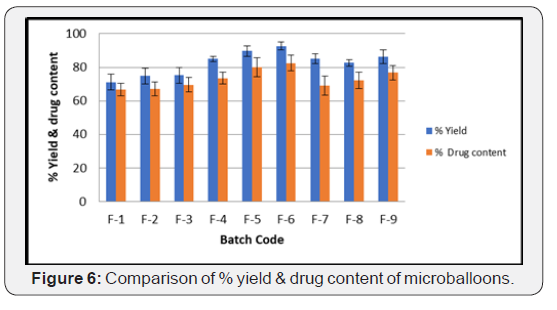
Microballoons were weighed after drying, and the percentage yield was calculated. The percentage yield of microballoons containing EC is less as (71.20±4.68 to 75.25±4.70%) when compared with HPMC-containing microballoons (84.94±1.65 to 92.55±2.34%). Microballoons with Eudragit, EC & HPMC show percentage yield ranging from (85.07±2.85 to 86.33±3.97%) respectively, concludes that no significant difference in entrapment efficiency by incorporation of Eudragit. It clearly indicates that % yield of microballoons were increased when HPMC K4M polymer is incorporated in the formulations. Drug content also strongly depends on % yield of microballoons i.e. HPMC K4M based formulation such as F-4, F-5 & F-6 shows highest drug content as (73.48±3.51, 80.13±5.63 & 82.55±4.68) supported by % yield values (Table 6) (Figure 6).
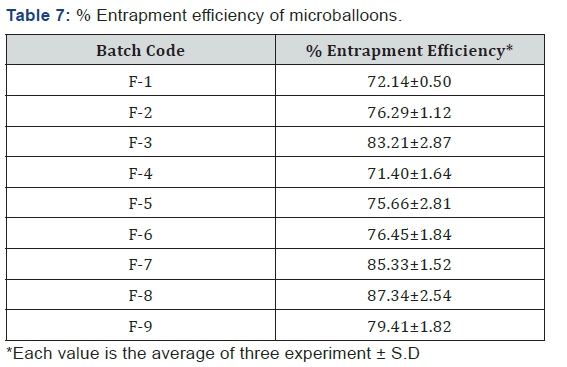
Determination of % entrapment efficiency
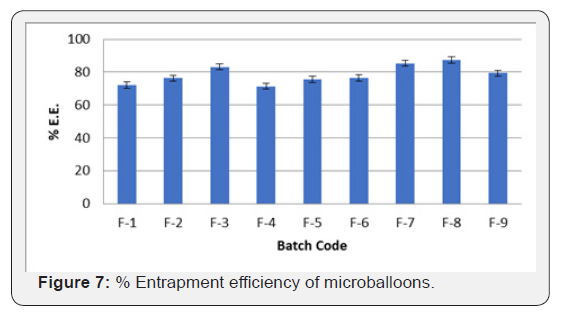
The percent drug entrapment efficiency (% DEE) of all formulation batches were found to be in the range of 71.40±1.64 % to 87.34±2.54 % as shown in (Figure 7). The % drug entrapment efficiency was more when the concentration of polymer (HPMC K4M) was increased in the drug: polymer ratio. Drug encapsulation efficiency was found to be increased due to increase in the concentration of gelling polymer leading to increase in cross linking structure and viscosity of internal phase which leads to reduce migration of drug in aqueous phase (Table 7).
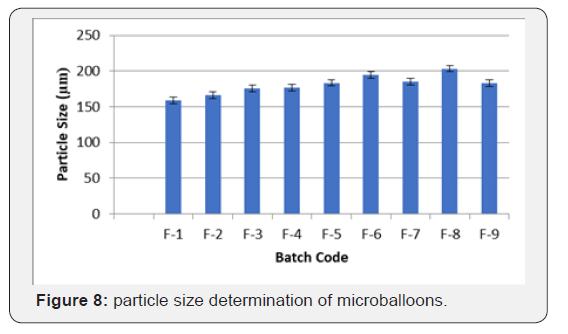
Determination of mean particle size
The mean particle size of all formulations was ranged from 159.1±4.09μm to 203.42±5.61μm. Formulation F-1 to F-3 showed smallest size due to ethyl cellulose which was low viscous in nature. Higher particle size was obtained when the proportion of HPMC K4M was increased in formulations. This was due to a significant increase in viscosity in a fixed volume of solvent, thus causing an increase in emulsion drop size and finally increases in size of particles. Increased speed of mixing (stirring) resulted in the formation of small-sized microballoons (Table 8) (Figure 8).
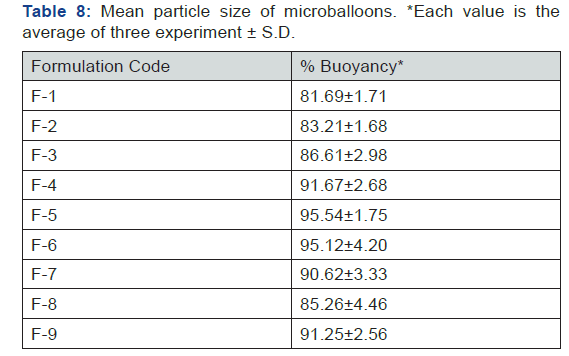
In-vitro buoyancy (%)
F-1 to F-9 was varied from 81.69 ±1.71% to 95.54 ±1.75%. It was observed that all the formulations were able to remain float in a continuous manner on the dissolution medium over a period of 12h. The In vitro buoyancy of microballoons of captopril was due to the presence of pores, a hollow structure and low bulk density. It may also deduce that these microballoons can float in gastric fluid retarding the passage of the microballoons into the intestinal region and increasing their residing time in the stomach. The In vitro % buoyancy was found to increase with increasing concentration of polymer (HPMC K4M) based formulations like F-4, F-5 & F-6 due to swelling property & hollow core formation which posses’ density less than gastric fluid (Table 9) (Figure 9).

Scanning electron microscopy of formulation F-4
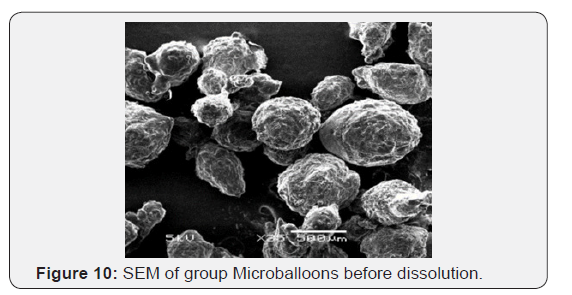
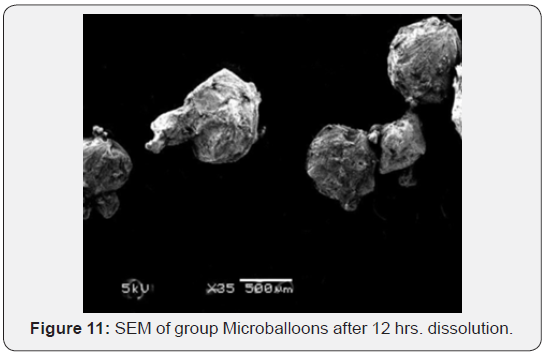

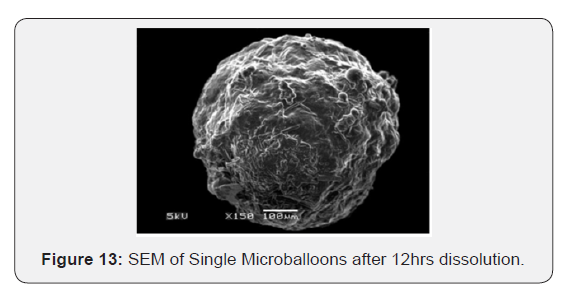
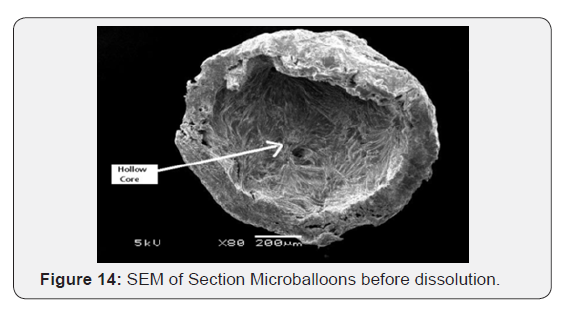
The surface morphology of captopril loaded floating microballoons was investigated by SEM. It was seen that microballoons were spherical in appearance and exhibit a range of sizes within each batch. The surface was observed to be smooth, dense and less porous, whereas the internal core was highly porous. The less porous outer surface and highly porous internal surface supported controlled release of drug from the microballoons and good buoyancy. The porous nature and cavity formed in the microballoons would indicate the floating behavior of microballoons (Figure 10-14).
In-vitro drug release study
Release study of captopril from formulations of microballoons was carried out in 0.1 N HCL for 12 hrs. & % drugs released reported in (Table 10) (Figure 15). Different amount of EC from were used for preparing the polymer solution for formulation F-1 to F-3, as the concentration of EC increased the drug release was greatly retarded. The drug release ranged from 74.5% at high EC concentration (F-3) to 82.86% at low-EC concentration. (F-1). The increased concentration of polymer increased the density of polymer matrix, which results in increased diffusional path length; this decreased the overall release of drug from polymer matrix (Figure 16).
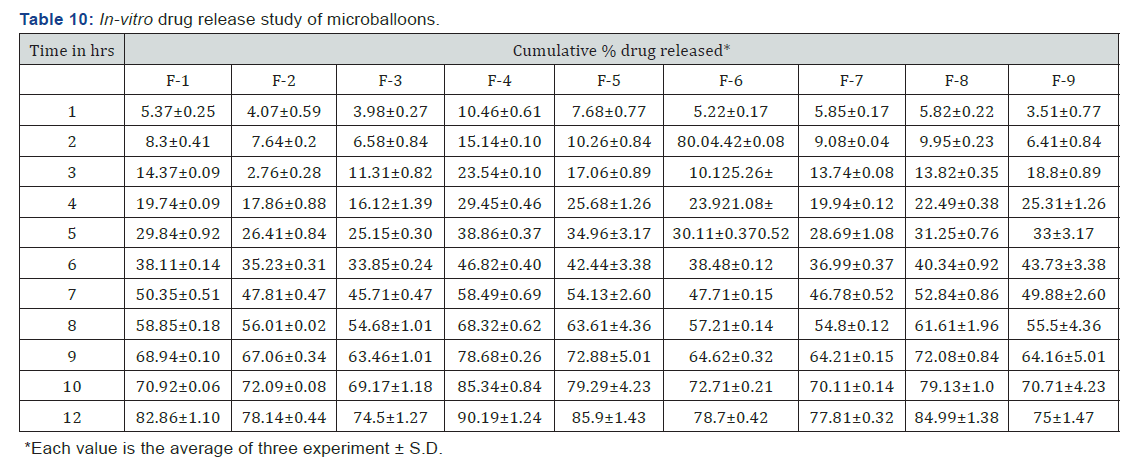
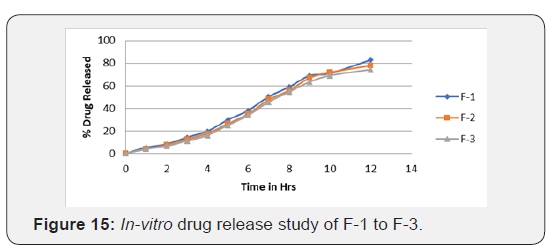
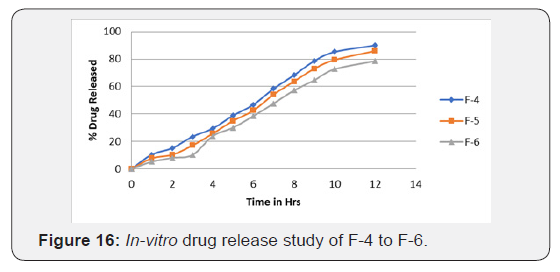
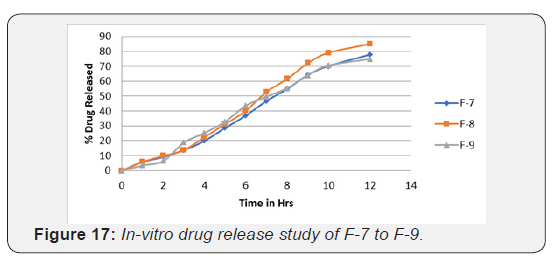
Another formulation F-4 to F-6 prepared by using HPMC K4M polymer gave good release as compared to EC. HPMC K4M was found to be more satisfactory; since it showed prolonged release at the end of 12 h (Figure 17). Microballoons prepared with Combination of EC, HPMC K4M and Eudragit RL100 were fine in size but release profile of formulations F-7, F-8, F-9 show slow drug release profile compared to other batches. Formulation containing polymer as Eudragit RL 100 is acrylic polymer insoluble in acidic medium hence release rate of drug was slower than formulation containing HPMC, Ethyl cellulose polymers.
Release rate kinetics
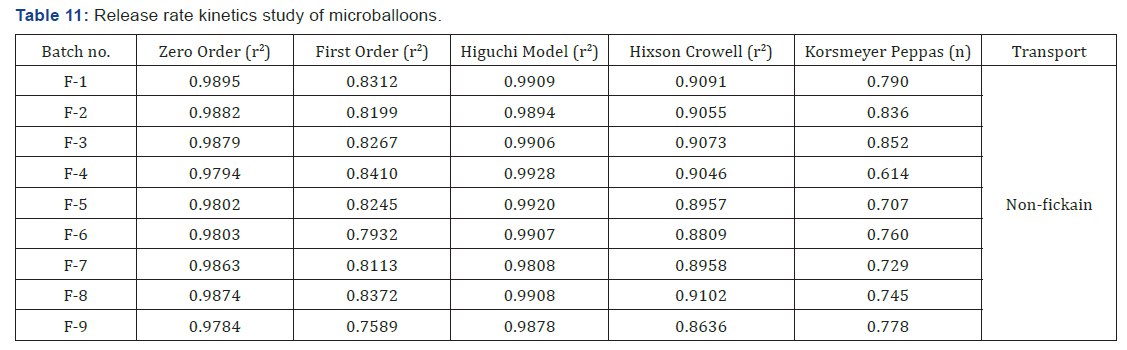
The kinetic investigations of the release profile gave us useful insight into the mechanism of drug release from the microballoons. The release from optimized formulation did not show any burst effect or lag time, which is indicative of a homogeneous drug distribution, in the microballoons. The dissolution data was subjected to regression analysis and were fitted to five kinetic models, viz., Zero order, First order, Hixson- Crowell and Higuchi. Most formulation followed Higuchi square root kinetics characteristic of matrix system. Drug release mechanisms evaluated using the Korsmeyer -Peppas model. In this model, the value of n identified the release mechanism of drug. The n value for most of the batches was found between 0.5 and 1, which confirmed that mechanism of drug release follows an anomalous or Non-fickian transport since their ‘n’ exponent was 0.45 < n = 0.89 (Table 11) (Figure 18-22).
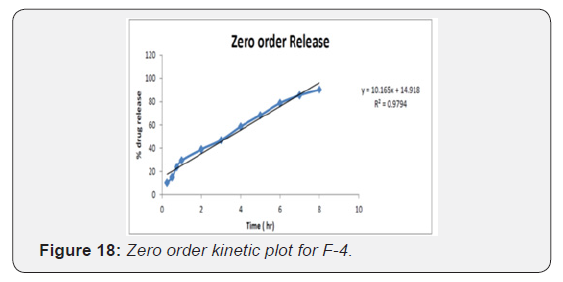
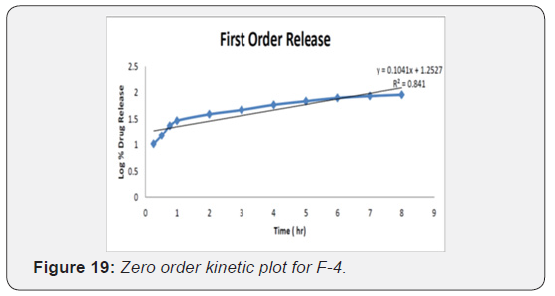
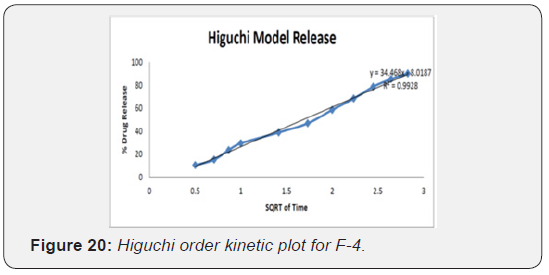
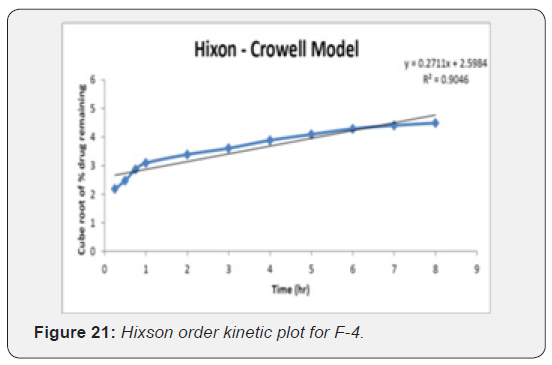
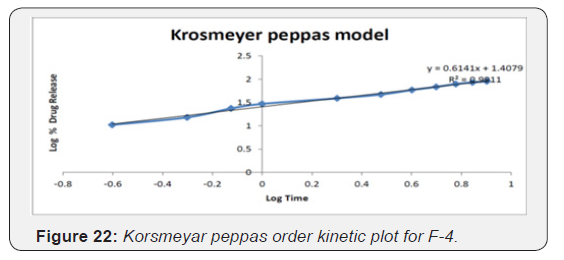
Stability study for F-4 formulation
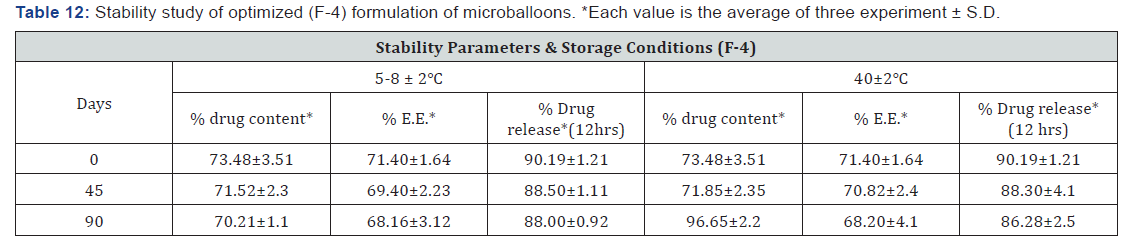
Optimized batch (F-4) was tested for stability study at 40±2℃ / 75% ± 5% RH, 5-8 ±2℃ for 90 days. Stability studies indicated that, no significant changes were observed with respect to % Drug content, % DEE and In vitro drug release after three months. It showed that optimized batch F-4 was stable up to 3 months (Table 12).
Student t test
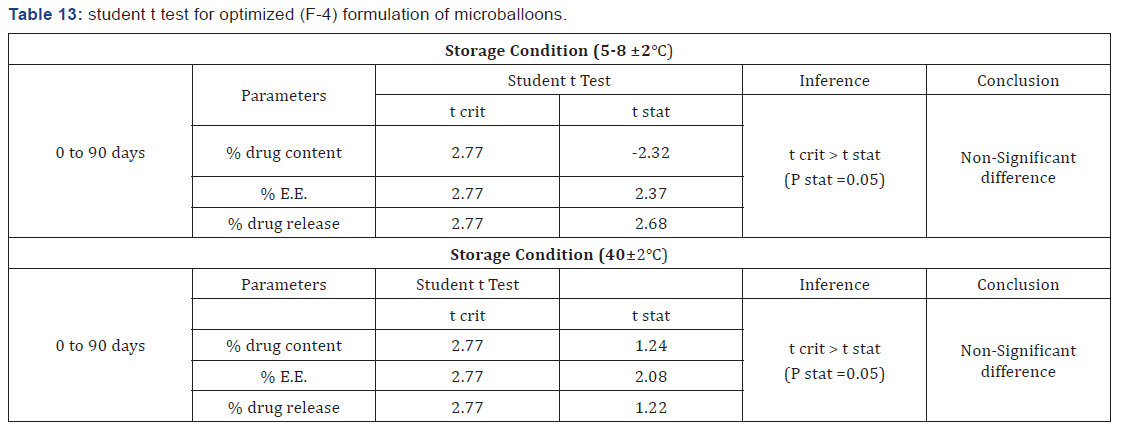
Effect of storage conditions on different characteristics properties of F-4 was statistically analyzed by student t test. Non-significant difference was observed in above analyzed parameters, supported by t crit > t stat by t test with p =0.05 (Table 13).
Conclusion
Hypertension, being a worldwide menace to the aging population and active work force alike, has attracted very wide attention of the medical and paramedical fraternity. Captopril is a front-line drug used in initiation and maintenance therapy of hypertension and myocardial infarction. It is a safe and efficacious candidate of choice to start chronic treatment. However, the dosing frequency is more than once and hence has less patient compliance. Patient compliance is expected to improve by formulating Captopril floating microballoons for prolonged use. Keeping in view these lacunae in achieving optimum therapy with Captopril, a sustained release floating drug delivery system in the form of floating Microballoons were developed using widely accepted and physiologically safe excipients and using technically simple, quick and reproducible methodologies. The FTIR studies did not detect any drug – excipients physical interaction and hence were compatible for formulation development. The optimized formulations F-4 was found to be comparable in drug release profile to target area in time. Therefore, the optimized multi-unit floating Captopril delivery system is expected to provide the clinician with a new choice of an economical, safe and efficacious arsenal in the management of hypertension.
References
- Ganderton D, Johnson P, Lloyd-Jones JG (1987) Drug Delivery Systems. (3rd edn), Chichester, Ellis Horwood, pp. 188-191.
- Jawahar N, Eagappanath T, Venkatesh N, Jubie S, Samanta MK (2009) Preparation and Characterisation of PLGA-Nanoparticles Containing an Anti-Hypertensive Agent. Int J Pharm Tech Res 1(2): 390-397.
- (2010) Daily med Drug list.
- Lachman L, Liberman HA, Kanig LJ (1987) The Theory and Practice Industrial Pharmacy; (3rd edn), Bombay, Varghese publishing house, pp. 412-29.
- Robinson TR, Lee HLV (1987) Controlled Drug Delivery Fundamentals and Applications; (2nd edn), NY, Marcel Dekker, Inc. pp, 4-61.
- Harrizul Rivai (2018) Development and Validation of Analysis Methods of Captopril in Tablets with Methods of Area Under Curves and Absorbance by Ultraviolet-Visible Spectrophotometry. Journal of Pharmaceutical Sciences and Medicine 3(4): 1-10
- Nief Rahman Ahmed (2013) Indirect Spectrophotometric Determination of Captopril in Pharmaceutical Tablets and Spiked Environmental Samples. Iraqi National Journal of Chemistry 49: 1-118.
- Mohan Kamila M, Mondal N, Kanta Ghosh L, Kumar Gupta B (2009) Multiunit Floating Drug Delivery System of Rosiglitazone Maleate: Development, Characterization, Statistical Optimization of Drug Release and In Vivo AAPS Pharm Sci Tech 10(3): 887-899.
- Perumandla PK, Priya S (2014) Formulation and in vitro Evaluation of Floating Microspheres of Dextromethorphan Hydrobromide. Int J Pharm Pharm Sci 6(4): 206-210.
- Struebel A, Siepmann J, Bodmeier R (2003) Multiple Units Gastroretentive Drug Delivery Systems: A New Preparation Method for Low Density Microballoons. J Microencapsul 20(3): 329-347.
- Aksh Yadav (2011) Gastroretentive Microballoons of Metformin: Formulation Development and Characterization. Journal of Advanced Pharmaceutical Technology & Research 2(1): 51-55.
- Rajeev Garg, GD Gupta (2010) Gastroretentive Floating Microspheres of Silymarin: Preparation and in- vitro Trop J Pharm Res 9(1): 59-66.
- Charulatha R, Damodharan N, Sundaramoorthy R, Abhilash G (2012) Design and Evaluation of Acebrophylline Sustained Release Matrix Tablets. Der Pharm Lett 4(2): 530-535.
- Ganesh S Bangale, Rajesh KS (2011) Natural Mucoadhesive Material Based Buccal Tablets of Nitrendipine-Formulation and in-vitro Journal of Pharmacy Research 4(1): 33-38.
- B stephen Rathinaraj, S Vijaya Kumar, Ganesh Sheshrao Bangale (2010) Preformulation Study of Buccoadhesive Monolayered Tablets Carvedilol. International Journal of Pharma and Bio Sciences 1(2): 1 -10.
- Nutan MTH, Soliman MS, Taha EI (2005) Optimization and Characterization of Controlled release Multiparticulate Beads Coated with Starch Acetate. Int J Pharm 294(1-2): 89-101.
- Dinarvand R, Mirfattahi S, Atyabi F (2002) Preparation Characterization and in vitro Drug Release of Isosorbide Dinitrate Microballoons. J Microencapsul 19(1): 73-81.
- Peeyush Bhardwaj, Himanshu Chaurasia (2010) Formulation and In-vitro Evaluation of Floating Microballoons of Indomethacin. Acta Poloniae Pharmaceutica - Drug Research 67(3): 291-298.
- Joydeep Dutta, G Ganesh Kumar (2011) Formulation and Evaluation of Gastroretentive Floating Microballoons of Norfloxacin. Indo American Journal of Pharmaceutical Research 1(4): 332-342.
- Sato Y, Kawashima Y, Takeuchi H, Yamamoto H (2004) In-vitro Evaluation of Floating and Drug Releasing Behaviors of Hollow Microspheres (microballoons) Prepared by the Emulsion Solvent Diffusion Method. European Journal of Pharmaceutics and Biopharmaceutics 57: 235-243.
- Bangale GS, Rajesh KS, Shinde GV (2018) Development and optimization of liposomal drug delivery system by 32 factorial design for cancer therapy. Indian Drugs 55(5): 14-24.
- Judith K (2010) Temoporfin loaded liposomes: Physicochemical characterization, Euro J Pharm Sci 40(4): 305-15.
- Prakash K, Raju PN, Shanta KK, Lakshmi MN (2007) Preparation and Characterization of Lamivudine Microcapsules Using Various Cellulose Polymers. Trop J Pharm Res 6(4): 841-847.
- Yogesh S Gattani, Durgacharan A Bhagwat, Akhil P Maske (2008) Formulation and Evaluation of Intragastric Floating Drug Delivery System of Diltiazem Hydrochloride. Asian J Pharm 2(4): 228-231.
- Dash S, Murthy PN, Nath L, Chowdhury P (2010) Kinetic Modeling on Drug Release from Controlled Drug Delivery Systems. Acta Pol Pharm 67(3): 217-223.
- Srivastava AK, Ridhurkar DN, Wadhwa S (2005) Floating Microspheres of Cimetidine: Formulation, Characterization and in vitro Acta Pharm 55(3): 277-285.
- Bajaj S, Singla D, Sakhuja N (2012) Stability Testing of Pharmaceutical Products. J Appl Pharm Sci 2(3): 129-138.






























