- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Formulation Design for Poorly Water-Soluble Drug by Using Solid Dispersion of Telmisartan for Solubility and Dissolution Rate Enhancement
A Deevan Paul*, Jada Vinay, K G RajyalakshmiP V Prasad
Academic Consultant, SV University, India
Submission: June 03, 2019; Published:July 12, 2019
*Corresponding author: A Deevan Paul, Academic Consultant, SVU College of Pharmaceutical Sciences, SV University, Tirupati, India
How to cite this article:A Deevan Paul, Jada Vinay, K G Rajyalakshmi, P V Prasad. Formulation Design for Poorly Water-Soluble Drug by Using Solid Dispersion of Telmisartan for Solubility and Dissolution Rate Enhancement. Glob J Pharmaceu Sci. 2019; 7(4): 555716. DOI: 10.19080/GJPPS.2019.06.555716.
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Abstract
Formulating the drug into solid dispersion (SD) by fusion method and solvent evaporation method using different grades of PEG in comparison to plain telmisartan drug with optimised solid dispersion tablets. The Preformulation studies like FTIR and DSC studies for drug excipient compatibility stated that the drug and carrier selected for the study and are compatible for further studies. SD was prepared by using the drug Telmisartan by two methods, fusion method and solvent evaporation method, eighteen formulations were prepared and characterized in terms of various parameters. The in vitro drug for all the formulations were in the range of 82.38%-95.73% for fusion method and 91.45% to 96.81% for solvent evaporation method and tablets it ranges from 95.70% to 99.40%. The in-vitro release studies have shown that the cumulative drug release values were within the range of 14.23%-94.54% for fusion method, 18.57%- 95.89% for solvent evaporation method and 14.35% - 99.53% for tablets. The fast drug release about 99.53% was found in the F22 formulation by solvent evaporation method in solid dispersion tablets in the ratio of 1:2 and drug content was found to be 99.53% and disintegration time was 1.09 seconds and all parameters were found to be greater than all other formulations.
Keywords: Poor solubility; Solid dispersion; Solvent evaporation method; Fusion method
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Introduction
The enhancements of oral bioavailability of poorly water-soluble drugs often show poor bioavailability because of low and erratic levels of absorption. Drugs that undergo dissolution rate limited, in gastrointestinal absorption and it shows improved dissolution and bio availability by reduction in particle size. However, drugs often lead to aggregation and agglomeration of particles, which results in poor wettability [1]. Solid dispersions of poorly water-soluble drugs with water-soluble carriers have been reduced the incidence of these problems and enhanced dissolution.
The development of solid dispersions as a practically viable method to enhance bioavailability of poorly water-soluble drugs overcame the limitations of previous approaches such as salt formation, solubilisation by co solvents, and particle size reduction. Studies revealed that drugs in solid dispersion need not necessarily exist in the micronized state [2-4]. A fraction of the drug might molecularly disperse in the matrix, thereby forming a solid dispersion. When the solid dispersion is exposed to aqueous media, the carrier dissolves and the drug releases as fine colloidal particles. Solid dispersions in water-soluble carriers have attracted considerable interest as a means of improving the dissolution rate, and hence possibly bioavailability, of a range of hydrophobic drugs.
The formulation of poorly soluble drug compound for oral delivery now presents one of the greatest challenges to formulation scientist in the pharmaceutical industry [5-6]. They can be used to increase the dissolution rate of a drug with low aqueous solubility, thereby improving its oral bio availability. Poorly water‐soluble drugs present many difficulties in the development of pharmaceutical dosage forms due to their limited water solubility; slow dissolution rate and low bioavailability [4]. Solid dispersions have been widely reported as an effective method for enhancing the dissolution rate and bioavailability of poorly water-soluble drugs [7]. The dissolution rate is directly proportional to solubility of drug. The therapeutic effectiveness of any drug depends upon the bioavailability; i.e. enough drug must reach the site of action to elicit the desired pharmacological response. The bioavailability affected majorly by two factors i.e. solubility and permeability, other factors are chemical stability,poor dissolution rate, purity. Currently only 8% of new drug candidates have both high solubility and permeability [8-10].
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Material and Methods
Telmisartan from Vasudha Pharma, Polythylene glycol from J&K chemicals, Mumbai, Sodium hydroxide from Merck chemicals, Mumbai, Microcrystalline cellulose form MYL Chem Mumbai, Magnesium stearate from S.D Fine chem. LTD Mumbai, Meglumine from Qualigens Mumbai, Crospovidone XL-10 form Merck Limited and Povidone from MYL Chem Mumbai.
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Preformulation Studies
Preformulation studies
An investigation of physical and chemical properties of a drug substance alone is defined as “Pre-formulation.”
Objective
It generates useful information to the formulator that is useful in developing stable and bio available dosage forms. These are
a) Organoleptic properties.
b) Solubility studies.
Organoleptic properties
Colour, Odour, taste, appearance of the drug play an important role in the identification of sample of above all properties should be recorded in descriptive terminology [11].
Solubility studies
100ml of dissolution medium with various concentration of carrier is taken in stopper flask an excess of drug was suspended in medium and equilibrated by intermittent shaking maintained at 37+0.5c, filter the solution by using what Mann filter paper, filtrate is suitably diluted. It is analyzed by UV spectroscopy [12,13].
Determination of melting point
Melting point of the pure drug was determined by melting point apparatus. Take a little quantity of sample in the capillary tube and placed in the apparatus and switch on the button. Observe through the viewpoint. Temperature was slowly raised and note that the temperature where the sample melts [14].
FTIR (Fourier Transform Infra-Red Spectroscopy) studies
Infrared (IR) spectroscopy studies of Telmisartan, PEG, Croscarmellose sodium, Meglumine and Microcrystalline cellulose were recorded in a FTIR spectrophotometer (Thermo- IR 200) Potassium bromide pellet method was employed, and background spectrum was collected under identical conditions. The spectrum for each sample showed the wavelength of absorbed light which is a characteristic of the chemical bonds in the sample [15-17]. Each spectrum was derived from 16 single average scans collected in the region of 4000 - 400cm-1 at a spectral resolution of 2cm-1.
Differential Scanning Calorimetry (DSC) studies
Thermal analysis of Telmisartan, PEG and physical mixture were recorded. Netzsch DSC 200PC (Netzsche, Selb, Germany), the instrument was calibrated with indium (calibration standard, >99.999%) for melting point and heat of fusion. A heating rate of 10C/min was employed in the range of 25-200C. Analysis was performed under nitrogen purge (20mL/min). The samples were weighted into standard aluminum pans and an empty pan was used as reference. The obtained DSC graphs were interpreted and compared for any presence of interactions [18-20].
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Preparation of telmisartan solid dispersions by fusion method
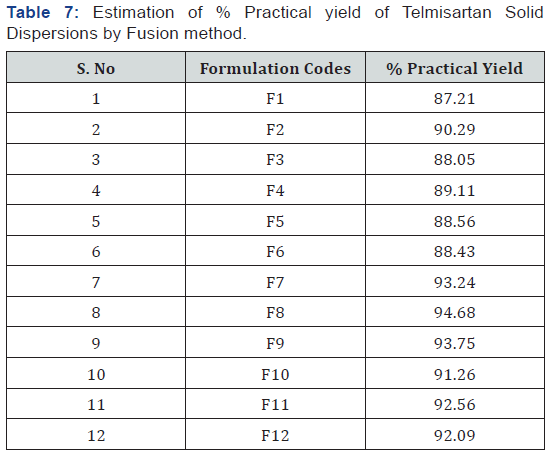
>In present work the drug and carrier were used in different ratios [1:1, 1:2 and 1:3]. The respective amount of polymer (PEG 4000, PEG 6000, PEG8000 and PEG 10000) was placed in a china dish and allowed to melt by heating up to its melting point. To the molten mass, an appropriate amount of drug module was added and stirred constantly until homogenous dispersion was obtained. The mixture was cooled rapidly by placing the dish in an ice bath for 5min to solidify. The solid mass was pulverized, sifted through sieve no. 60 and stored in desiccator for further studies [21-24]. The formulations were coded as F1, F2, F3, F4, F5, F6, F7, F8, F9, F10, F11 and F12 for drug- polymer ratios 1:1, 1:2 and 1:3 respectively (Table 1).
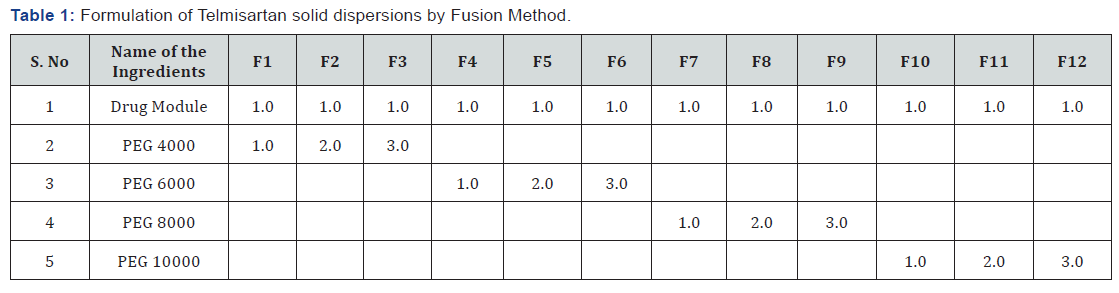
Preparation of Drug module solid dispersions by solvent evaporation method
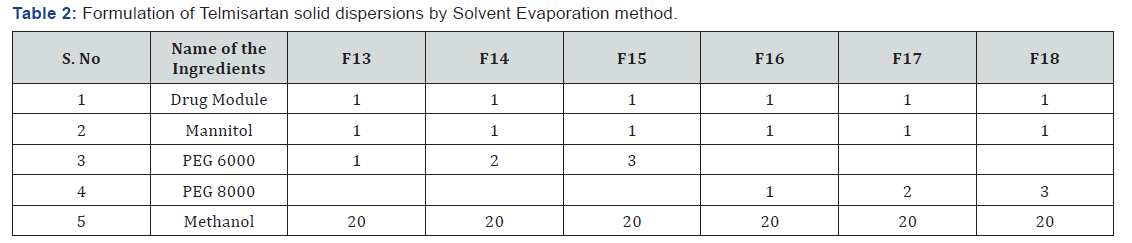
Solid dispersions of drug module with a hydrophilic carrier (PEG 6000, PEG8000) were prepared in different ratios of drug– carrier. The quantity of carriers for optimization was selected based on preliminary trial formulations. The solvent evaporation method was used for the preparation of SD in the present study. In this method, 1.0g of drug module was accurately weighed and dissolved in a minimum amount of methanol in which hydrophilic carrier was suspended [25].
The solvent was evaporated using a water bath at 450C.The obtained solid was pulverized, sieved through a sieve no. 60 and store in airtight containers. The formulations were coded as F13, F14, F15, F16, F17 and F18 for drug- polymer ratios 1:1, 1:2 and 1:3 respectively [26] (Table 2 & 3).
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Evaluation of Solid Dispersion
Prepared solid dispersions were evaluated for the following parameters:
a) Percentage yield
b) Drug content
c) In vitro dissolution studies
Percentage yield
Percentage yield was calculated to know about efficiency of any method and thus its help in selection of appropriate method of production. The final weights of the prepared solid dispersions were taken, and percentage yield was calculated by using the given formula [26,27].
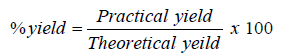
Drug content
Equivalent weight of prepared solid dispersions containing 100mg drug were taken and transferred into 100ml Standard flask Then take 1ml from above solution and diluted up to 100ml simulated salivary fluid pH 6.8 and repeat the same again by take 1ml from above solution and diluted up to 100ml simulated salivary fluid pH 6.8. The resulting solutions were filtered through a 0.45μ membrane filter and diluted accordingly. The absorbance of the solutions was measured at 296 nm. Percentage of drug content was calculated by using the given formula [28].
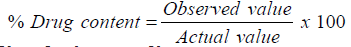
In vitro dissolution studies
In vitro dissolution studies of pure telmisartan and solid dispersions were conducted with the USP type II apparatus (paddle type). The dissolution studies were performed using 900ml simulated salivary fluid of pH 6.8 as dissolution medium at 37±0.5C with 50rpm speed. Samples of each preparation equivalent to 10 mg of drug were added into the dissolution medium. The sample of 5ml aliquots were withdrawn periodically (15, 30, 45 and 60min) and filtered through 0.45μ membrane filter. The withdrawn sample was replaced every time with same quantity of fresh dissolution medium. The filtered solutions were diluted suitably, and the samples were analyzed for their drug content by using UV spectrophotometer at wavelength of 269nm. Percentage of drug dissolved at various time intervals was calculated by plotting time on X- axis against percent cumulative drug release on Y-axis [29-31].
Pre compression characterization of blend
The capecitabine and metoclopramide blend were evaluated for
a) Angle of repose
b) Bulk density
c) Tapped density
d) Carr’s index
e) Hausner’s ratio
Angle of repose
The flow property of blend was determined by the angle of repose values. The maximum angle that can be attained between the surface of the pile of the powder and the horizontal plain is defined as “angle of repose” [32].
Angle of repose was determined by fixed funnel method.
Angle of repose =Tan-1(h /r)
h =Height of pile
r =Radius of pile
7.6. Bulk density
It is defined as the ratio of given mass of powder and its bulk volume. Bulk density values having less than 1.2g/cm3 indicates good packing and greater than 1.5g/cm3 indicates poor packing [33].
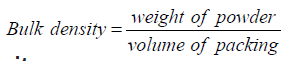
Tapped density
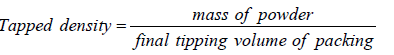
Hauser’s ration
It indicates the flow property of blend; it is defined as the ratio of tapped density and bulk density. It was related to antiparticle friction. Values less than 1.25 indicates good flow property [34].
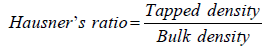
Compressibility
It was obtained from bulk and tapped densities. Less compressibility indicates more flowing property of powder. The CI value less than 10 indicates excellent flow property.
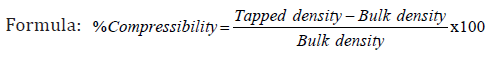
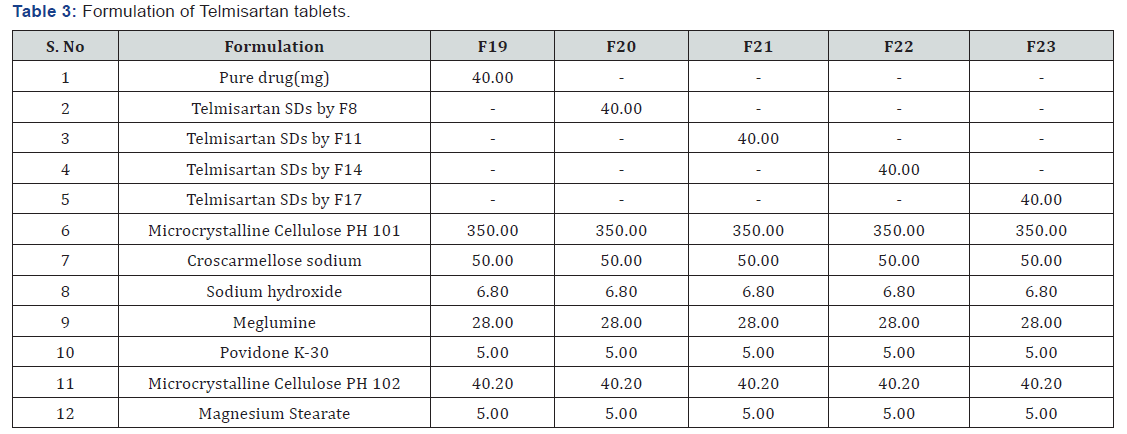
Preparation of tablets
The tablets were prepared by direct compression technique. Powder was compressed by using 12 stationary rotary compression machines with below mentioned tooling [35].
Tooling
Upper Punch: 12.70mm, Circular shaped, standard concave with plain surface.
Lower punch: 12.70mm, Circular shaped, standard concave with plain surface.
Die: 12.70mm
Evaluation of tablets
After the preparation of tablets were evaluated for post compression parameters.
a) Weight variation
b) Thickness
c) Hardness
d) Friability
e) Drug content
f) Disintegration time
g) In-vitro drug release
Evaluation studies are important in the design of tablets and to monitor product quality. There are various standards have been set regarding the quality of pharmaceutical tablets [36,37].
Weight variation test
These tests are based on the comparison of the individual tablets with upper and lower percentage limits of observed sample average(x-mean). USP provides limits for average weight of tablets. When tablets contain more than 150mg these limits are applicable.
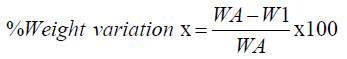
Where WA= Average weight of tablet in mg.
WI= Individual weight of tablet in mg.
Thickness
Thickness was determined using Vernier callipers. It is expressed in millimeter. It was mostly related to the tablet hardness. Can be uses as initial control parameter [38].
Hardness
It is defined as the force required to breaking a tablet diametrically. It was determined using the Monsanto hardness tester. It is measured in kg/cm2. 4kg/cm2 is usually considered as minimum satisfactory value to tablets. Tablet requires a certain amount of hardness to withstanding of mechanical shocks during manufacturing, packing and shipping [39].
Friability
Friability was measured by using Roche friabilator. It is closely related to tablet hardness. It is used in determination of the ability of the tablet to with stand abrasion in packing, hand lining and shipping. Tablets that loss less than 10% of its weight was generally considered as accepted formulation.

Drug content
The drug content was calculated by using following formula.

Disintegration time
It was determined by using disintegration test apparatus using water as immersion fluid. One tablet was placed in each tube of all 6 tubes. Then it was subjected to disintegration at 28- 32 cycles/minute. The time taken for complete disintegration was measured in seconds [40].
In-vitro dissolution studies
Drug release studies were carried out by using USP dissolution test apparatus type II. 900ml dissolution medium (pH 6.8 phosphate buffer) is taken in each bucket. Maintain temperature 37+0.5C. Paddle was rotated at 75rpm for 30 minutes. 1ml of samples was withdrawn at predetermined time intervals of 5, 10, 15, 20, 25 and 30 minutes respectively. Replacing the same amount of dissolution medium by replacing with equal quantity of drug free pH 6.8 phosphate buffer. It was diluted and filtered through membrane filter. Absorbance of sample was analyzed by UV spectrophotometer at 296nm [41-42].
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Results and Discussion
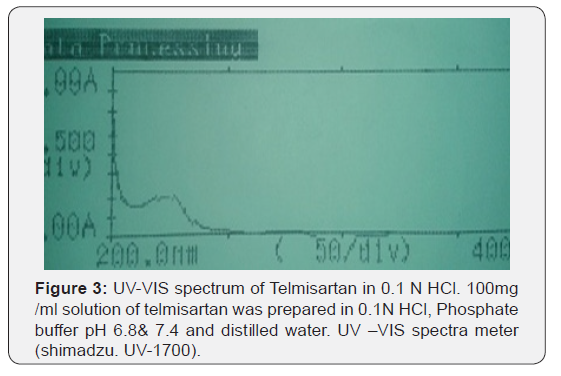
UV-VIS spectrum
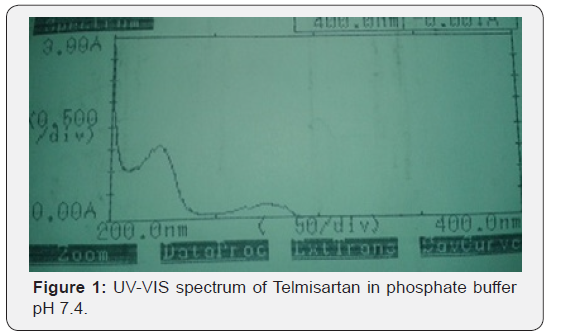
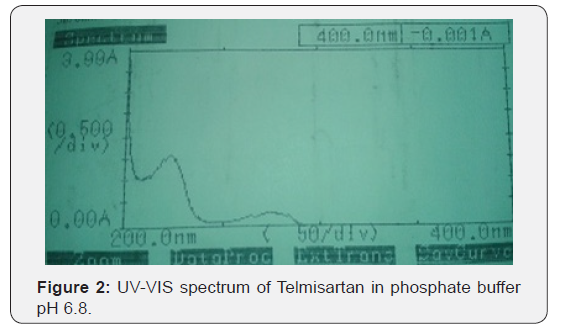
10μg/ml solution of Telmisartan was prepared in 0.1N HCl, Phosphate buffer pH 6.8, Phosphate buffer pH 7.4 and Distilled water. UV-VIS scan was taken between the wavelengths 200- 400nm using UV-VIS spectrophotometer (Shimadzu, UV-1700) (Table 4 & 5).


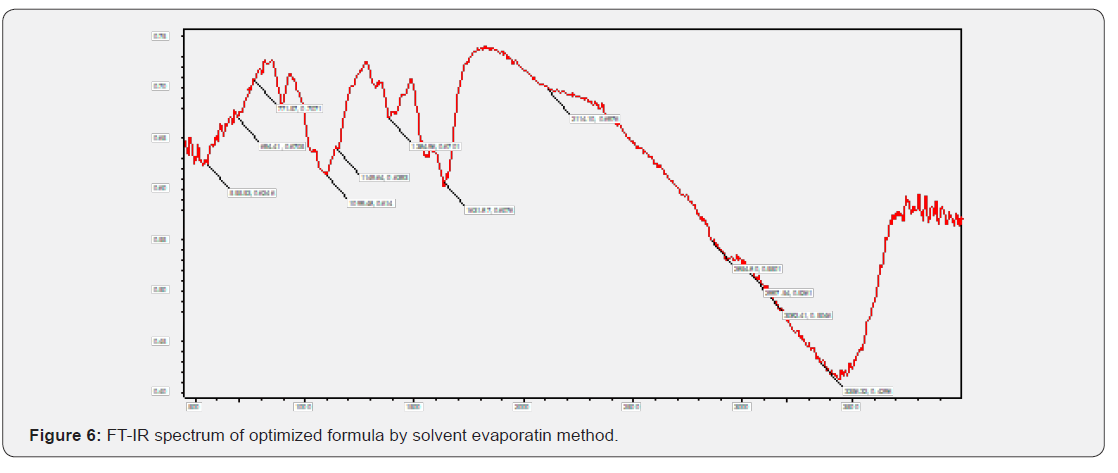
Infrared (IR) spectro scopy studies of telmisartan PEG, Croscarmellose sodium Meglumine and micro crystalline cellulose were recorded in a FTIR spectro photometer (themo- IR200) potassium bromide pellet method was employed and background spectrum was collected under identical conditions the spectrum for each sample showed the wavelength of absorbed light which is a characteristic of the chemical bonds the sample each spectrum was derived from 16 single average collected in the region of 4000-400cm-1 at a spectral resolution of 2cm-1 (Figure 1-5).
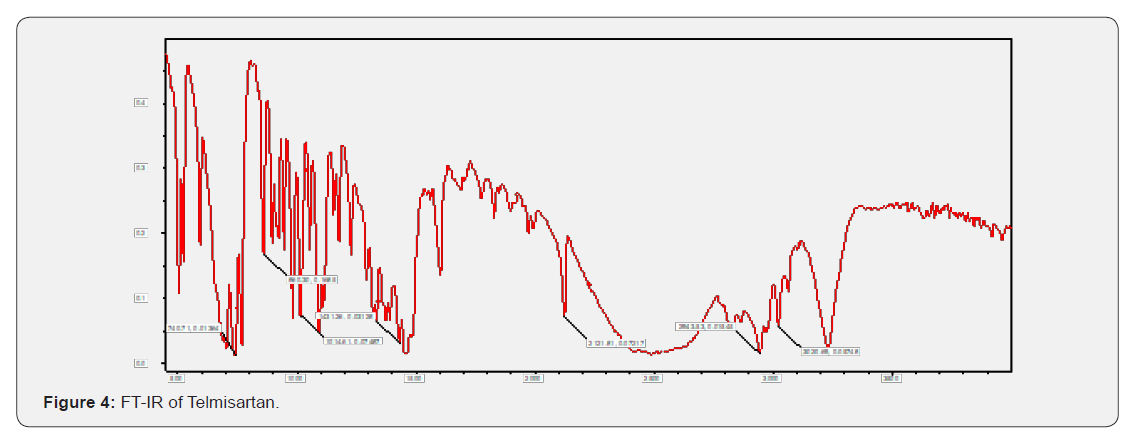
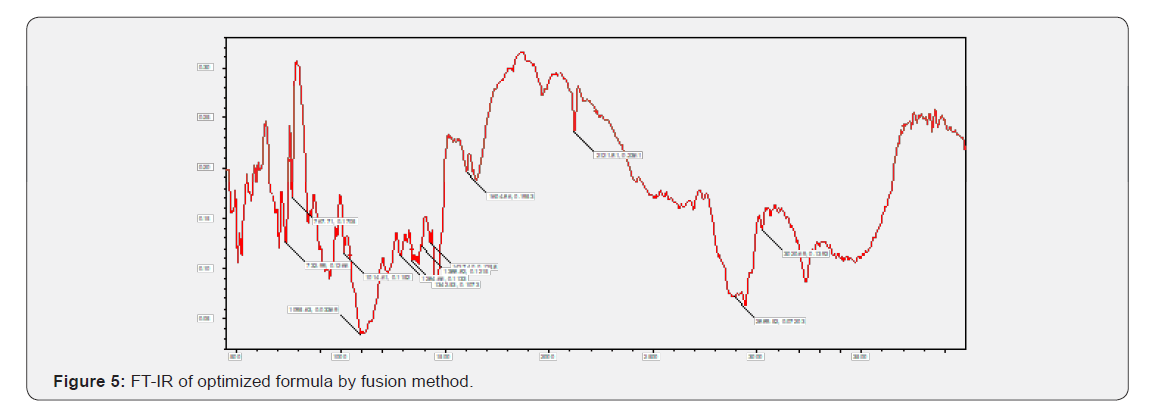
FT-IR Studies did not show any significant interactions between the drugs and their respective excipients. From the compatibility studies PEG, Croscarmellose sodium, Meglumine, Povidone and microcrystalline cellulose are compatible with Telmisartan and its optimized formula for solid dispersion and for tablets states there is no significant interactions between the excipients (Figure 6 & 7).
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Interpretation of Drug –Excipient Compatibility Studies
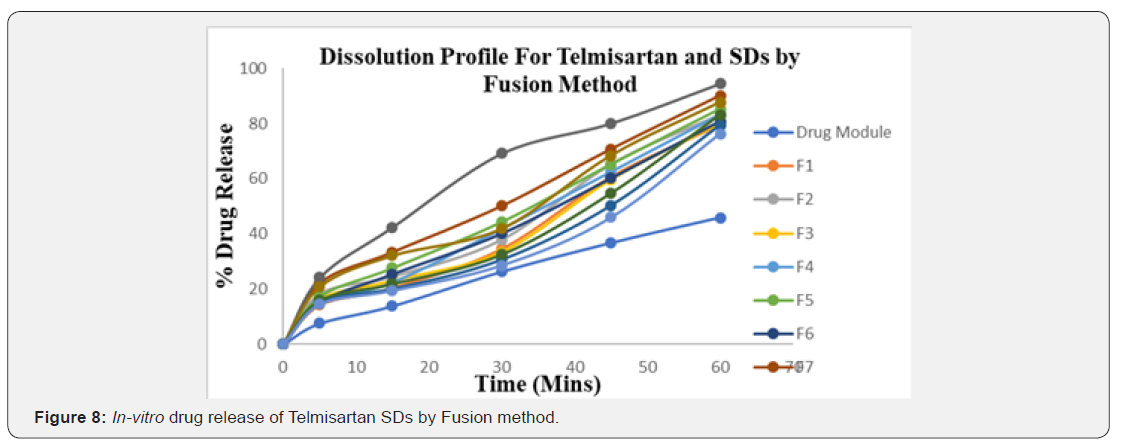
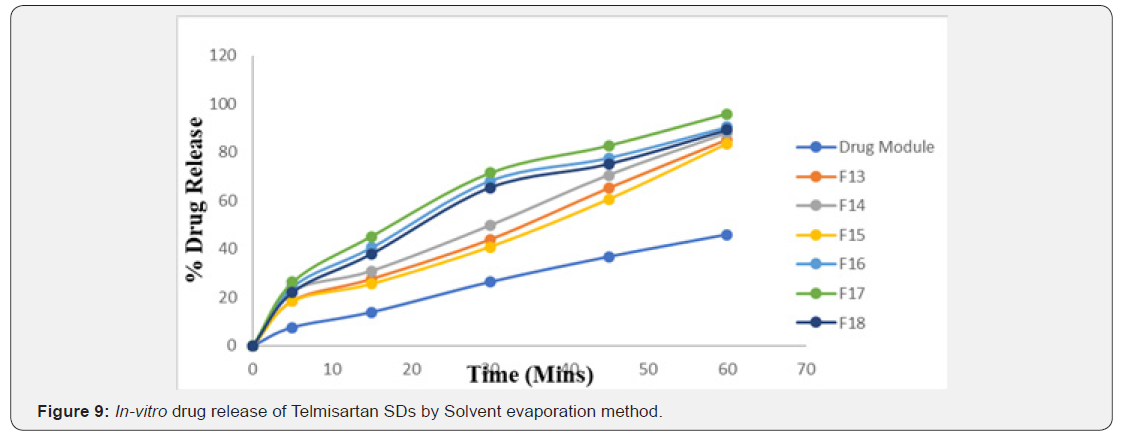
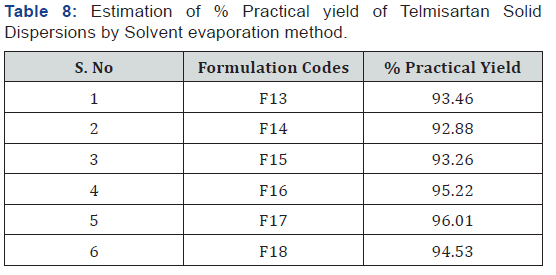
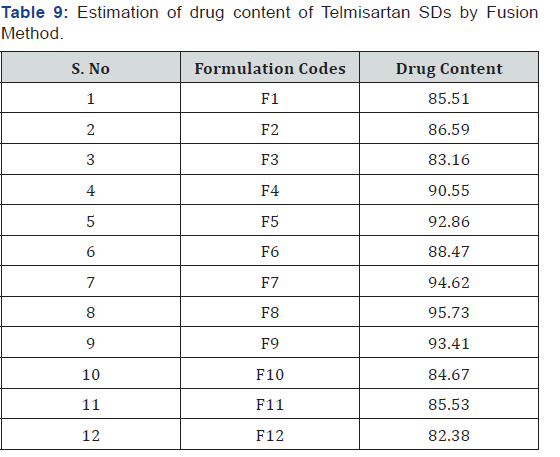
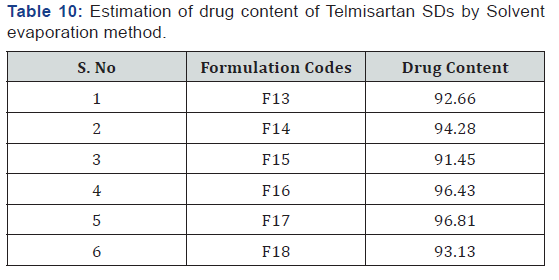
The Preformulation studies for compatibility by FTIR concluded the drug and carrier selected for the study were compatible and can be used for the further studies. The invitro drug content for all the formulations were in the range of 82.38% - 95.73% for fusion method and 91.45% - 96.81% for solvent evaporation method and for tablets it ranges from 95.70%- 99.40% (Table 6-12) (Figure 8 & 9).

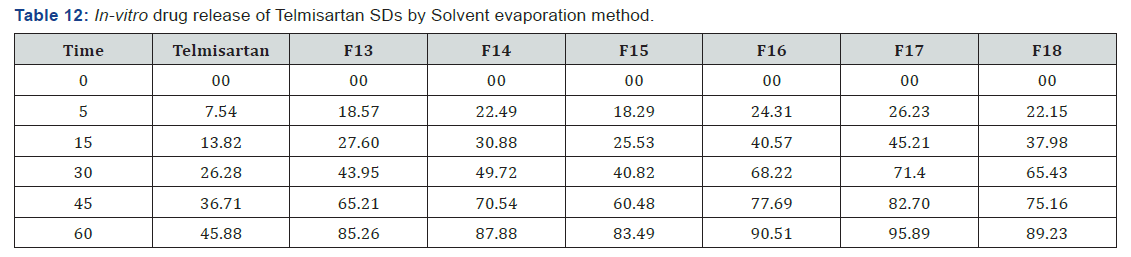
Pre-Compression Parameters
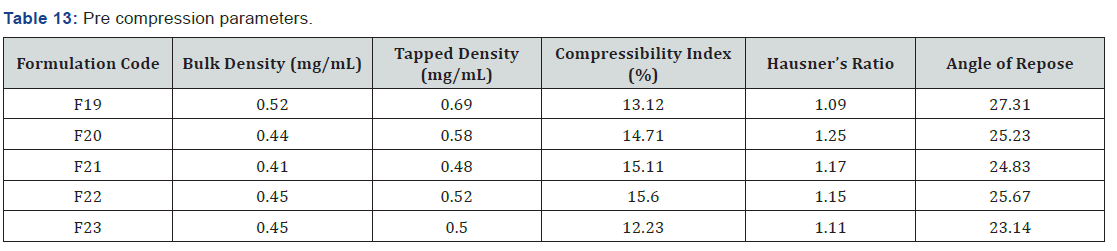
All formulation blends were evaluated to Pre compression parameters such as angle of repose, bulk density tapped density, compressibility index, Hausner’s ratio. Angle of repose values of all formulations blend was found in the range of 23.14 to 25.67 it indicates free flowing of powder blend. The Carr s index values were found in between 12.23 to 15.60 indicates them having good compressibility. Hausner’s ratio was present in the range of 1.11 to 1.25 that indicates good flow of powder blend. All Pre compression parameters were present within the limits whereas for F19 all the parameters were not found to be satisfactory (Table 13).
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Post Compression Parameters
The post compression parameters were measured for tablet. Weight variation was in the range of 522.8 to 528.4mg for 525.00mg. Hardness of all formulation was found to be in the range of 4.3 to 4.9kg/cm2. Friability values were found to be 0.32 to 0.61. Drug content were found to in the range of 95.70 to 99.50. Thickness of IR layer was found between 5.52 to 5.57mm. Disintegration time for all formulation was found to be below 2 minutes except F19 (Table 14 & 15) (Figure 10).
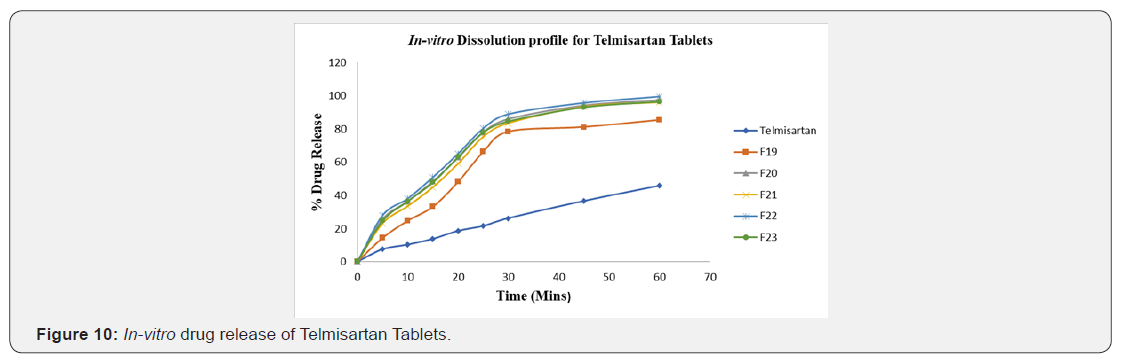
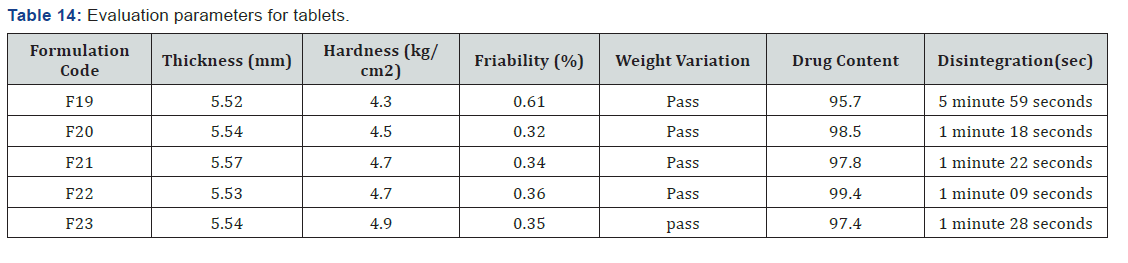
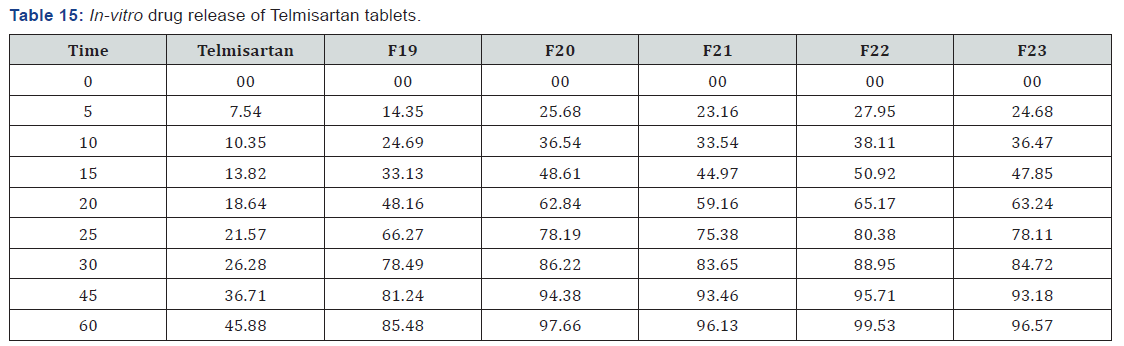
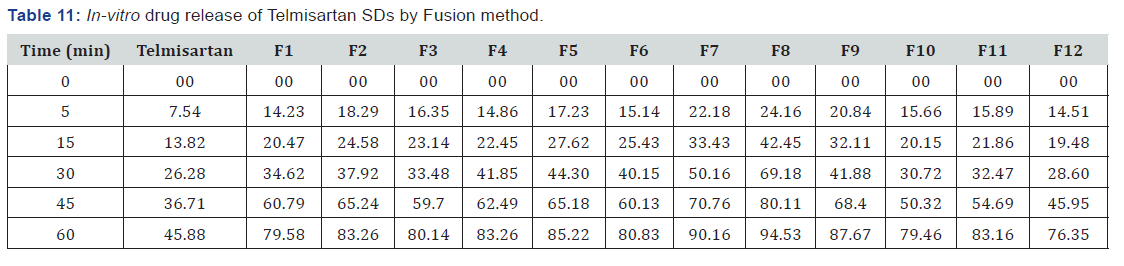
The in-vitro release studies have shown that the % drug release values were within the range of 14.23% - 94.53% for fusion method, 18.57% - 95.89% for solvent evaporation method and 14.35% - 99.53% for tablets.
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Conclusion
The present study was carried out to develop telmisartan immediate release tablets by direct compression method Telmisartan SDs in two methods namely fusion followed by solvent evaporation method respectively which tends to improve the solubility of telmisartan. Formulation characteristics were found to be satisfactory in all formulations shows acceptable internal specification for weight variation, thickness, hardness, friability, drug content, disintegration time and in vitro drug release. The fast drug release about 99.53% was found in the F22 formulation by solvent evaporation method SDs tablets in the ration of 1:2, drug content was found to be 99.53% and disintegration time was 1 minute 09 seconds and all parameters were found to be greater than all other formulations.
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
Acknowledgement
The authors wish to express their heartful gratitude to Prof. K. Thyagaraju, Principal of SVU College of Pharmaceutical Sciences, SV University-517502 for providing their unrestricted support for carrying out this research.
- Research Article
- Abstract
- Introduction
- Material and Methods
- Preformulation Studies
- Preparation of telmisartan solid dispersions by fusion method
- Evaluation of Solid Dispersion
- Results and Discussion
- Interpretation of Drug –Excipient Compatibility Studies
- Post Compression Parameters
- Conclusion
- Acknowledgement
- References
References
- Leuner C, Dressman J (2000) Improving drug solubility for oral delivery using solid dispersions. Eur J Pharm Biopharm 50: 47-60.
- Sekiguchi K, Obi N (1961) Studies on absorption of eutectic mixtures, I. A comparison of the behaviour of eutectic mixtures of sulphathiozole and that of ordinary sulphathiozole in man. Chem Pharm Bull 9: 866-872.
- Goldberg AH, Gibaldi M, Kanig JL (1996) Increasing dissolution rates and gastrointestinal absorption of drug via solid solutions and eutectic mixture II-experimental evaluation of a eutectic mixture; urea acetaminophen system. J Pharm Sci 55: 482-487.
- Kaur R, Grant DJW, Eaves T (1980) Comparison of poly (ethylene glycol) and polyoxy ethylene stearate as excipients for solid dispersions system of griseofulvin and tolbutamide II: Dissolution and solubility studies. J Pharm Sci 69: 1321-1326.
- Yoshioka MB, Hancock C, Zogra G (1995) Inhibition of indomethacin crystallization in poly (vinylpyrrolidone) co precipitates. J Pharm Sci 84: 983-986.
- Vishweshwar P1, McMahon JA, Bis JA, Zaworotko MJ (2006) Pharmaceutical co-crystals. J Pharm Sci 95: 499-514.
- Vadnere MK (1990) Co-precipitates and Melts. In: Swarbrick J, Boylan JC, editors. Encyclopaedia of pharmaceutical technology. Marcel Dekker Inc. New York.
- Chiou WL, Riegelman S (1971) Pharmaceutical application of solid dispersion. J Pharm Sci 60: 1281-1302.
- Kanig JL (1964) Properties of fused mannitol in compressed tablets. J Pharm Sci 53: 188-192.
- Walker SE, Gangley JA, Bedford K, Eaves T (1980) The filling of molten and thyrotrophic formulations into hard gelatin capsule. J Pharm Pharmacol 32: 389-393.
- Dressman J, Leunne C (2000) Improving drug solubility for oral delivery using solid dispersions. Review article. Eur J Pharm Biopharm 50: 47-60.
- Sharma DK, Joshi SB (2007) Solubility enhancement strategies for poorly water-soluble drug in solid dispersion: A Review. Asian Journal of Pharmaceutics 1: 9-19.
- Ambike AA, Mahadik KR, Paradkar A (2005) Spray dried amorphous solid dispersions of simvastatin, a low Tg drug: In vitro and in vivo evaluations. Pharm Res 22: 990-998.
- Takayama K, Nambu N, Nakai T (1982) Factor affecting the dissolution of ketoprofen from solid dispersion in various water-soluble polymers. Chem Pharm Bull 30: 673- 677.
- Subramanian B, Rajewski RA, Snavely K (1997) Pharmaceutical processing with supercritical carbon dioxide. J Pharm Sci 86(8): 885-890.
- Palakodaty S, York P (1999) Phase behavioural effects on particle formation process using supercritical fluids. Pharm Res 16(7): 976-985.
- Sethia SE, Squillante (2002) Physicochemical characterization of solid dispersions of carbamazepine formulated by supercritical carbon dioxide and conventional solvent evaporation method. J Pharm Sci 91(9): 1948-1957.
- Vemavarapu C, Mollan MJ, Needham TE (2002) Crystal doping aided by rapid expansion of supercritical solutions. AAPS Pharm Sci Tec 3(4): E29.
- Muhrer GU, Meier F, Fusaro S, Mazzotti M (2006) Use of compressed gas precipitation to enhance the dissolution behavior of a poorly water-soluble drug: Generation of drug microparticles and drug-polymer solid dispersion. Int J Pham 308(1-2):69-83.
- Jarmer DJ, Lengsfeld CS, Anseth KS, Randolph TW (2005) Supercritical fluid crystallization of griseofulvin: Crystal habit modification with a selective growth inhibitor. Pharm Sci 94(12): 2688-2702.
- Edwards AD, Shekunov BY, Kordikowski A, Forbes RT, York P (2001) Crystallization of pure anhydrous polymorphs of carbamezapine by solution enhanced dispersion with supercritical fluids (SEDS). J Pharm Sci 90(8): 1115-1124.
- Morita M, Hisrota S (1985) Correlation studiesn between thermal and dissolution rate constant of cimitidine drug and tablet. Chem Pharm Bull 33: 2091.
- Shine SC, Oh IJ, Lee YB, Choi HK, Choi JS (1998) Enhancement dissolution of furosemide by co-precipitating or co-grinding with corsprovidone. Int J Pharm 175: 17-24.
- Breitenbach J (2002) Melt extrusion: Fromprocess to drug delivery technology. Pharm Biopharm 54(2): 107-117.
- Choksi R, Zia H (2004) Hot-melt extrusion technique: A review. J Pharm Res 3: 107-117.
- Jorg B (1999) Confocal Raman spectroscopy: Analytical approach to solid dispersion and mapping of drug. Pharm Res 16(7): 1109-1113.
- Verreck G, Six K, Van G, Mooter L, Baert J, et al. (2003) Characterization of solid dispersions of itraconazole and hydroxypropylmethylcellulose prepared by melt extrusion. Int J Pharm 251(1-2): 165-174.
- Breitenbach J, Soliq A (2002) Melt extrusion: from process to drug delivery technology. Eur JbPharm 54(2): 107-117.
- Sushama R, Desai MS, Loyd V, Robert B, Greenwood ML (1989) Effervescent solid dispersions of prednisone, griseofulvin and primidone. Drug Dev Ind Pharm 15: 671-677.
- Ho HO, Shu HL, Tsai T, Sheu MT (1996) The preparation and characterization of solid dispersions on pellets using a fluidized bed system. Int J Pharm 139(1-2): 223-229.
- Yamamoto K, Nakamo M, Arita T, Nakai Y (1974) Preparation and thermal characterization of poly (ethyl oxide)/ griseofulvin solid dispersions for biomedical application. J Pharmaco Biopharm 2: 487-495.
- Hecq JD, Evrard JM, Gillet P, Briquet C (1996) Stability of injectable drugs stored in the freezer & thawed by microwave: actualization in 1996. Pharmakon 106: 83-92.
- Moon YS, Chung KC, Chin A, Gill MA (1995) Stability of Piperacillin sodium-tazobactam sodium in polypropylene syringes and polyvinyl chloride minibags. Am J Health Syst Pharm 52(9): 999-1001.
- Sewell GJ, Palmer AG (1991) Chemical and physical stability of three intravenous infusions subjected to frozen storage and microwave thawing. Int J Pharm 72: 5763.
- Thomas PH, Tredree RL, Barnett MI (1998) Preliminary studies on the freezing and thawing of intravenous solutions. Br J Intravenous Ther 4: 14-21.
- Gopinath C, Vintima Bindu M, Nischala (2013) An overview on bilayered tablet technology. JGTPS 4(2): 1077-1085.
- Panchal Hiten Ashk, Tiwary Ajay Kumar (2012) A novel approaches of bilayer tablet technology a Review. Irjp 3(5): 44-49.
- Morarwars S, Jadhav SB, Kadam VS, Muttepawar SS, Bharkad VB, et al. (2013) Review on bilayer tablet. WJPPS 3(2): 2693-2709.
- Patel, Mehul, Ganesh Nanjam, Sockaa, Kavitha Tamizhmani (2012) Challenges in formulation of bilayered tablets. A Review IJPRD 2(10): 30-42.
- Singh KP, Kumar S (2011) Bilayer and floating bioadhesive tablets: Innovation approach to gastro retention. Journal of drug delivery and therapeutics 1(1): 32-35.
- Balaji G, Gnana Prakash K, Suresh Karudumpala, Venkatesh B (2013) bilayertablet: A Review, International journal of Research and Reviews in pharmacy and applied science 3(4).
- Priyal S, Nilawar, Wankhade VP, Bad Nag DB (2013) An emergency trend on bilayer tablet. IJPRS 3(1).






























