- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Formulation and Optimization of Nanoparticale by 32 Factorial Design for Colon Targeting
Ganesh S Bangale*1, GV Shinde2 and Rajesh KS3
1Government College of Pharmacy, Amravati (M.S.), India
2Parul Institute of Pharmacy, Baroda, India
3I.S.F. College of Pharmacy, Moga, India
Submission: November 06, 2018; Published: January 24, 2019
*Corresponding author: Ganesh S Bangale, Government College of Pharmacy, Amravati (M.S.), India
How to cite this article: N Anjaneyulu, R Naga K, M Ravi K, G Sneha. Skipping Breakfast Everyday Keeps Well-Being Away. Glob J Pharmaceu Sci. 2018; 6(5): 555702. DOI: 10.19080/GJPPS.2019.06.555702.
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Abstract
The aim of the present study was to develop stable nanoparticulate formulation for sustained release of Prednisolone. Chitosan nanoparticles were prepared by ionic gelation method using tripolyphosphate as cross-linking agent. Different nanoparticulate formulations were prepared by using 32 factorial design in which varying the concentration of chitosan (0.1% to 0.3%), concentration of tripolyphosphate (0.02% to 0.03%) as two factors. The effect of these factors on the particle size, % entrapment efficiency and in vitro drug release was evaluated to develop an optimized formulation.
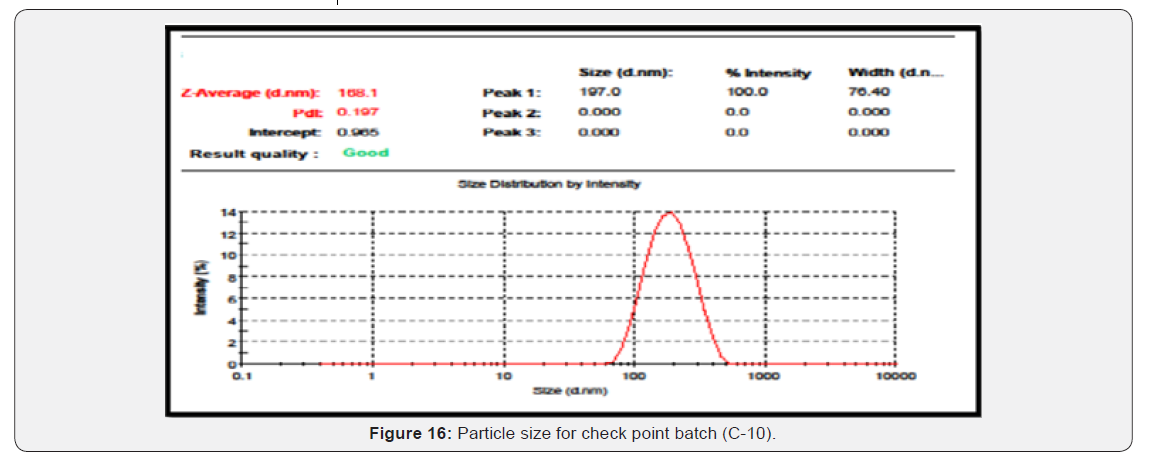
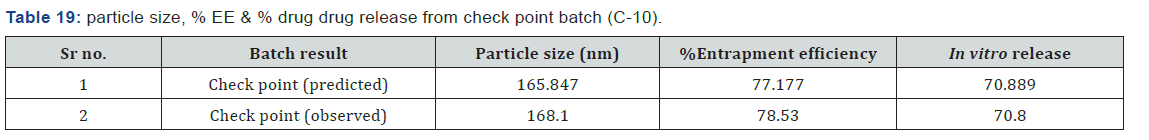
Particle size, % entrapment efficiency and in vitro release of optimized formulation were found to be 168.1nm, 78.53% and 70.80% respectively. ANOVA study applied with p < 0.01 suggests that model is significant & Contour, Surface response & overlay plot was contract to optimize the formulation. Optimized formulation (C-10) showed sustained drug release at the end of 11th hour compared to other formulations. Based on release kinetic model, the drug release data fit well to higuchi model (r2 = 0.9935) indicating the diffusion limited drug release from nanoparticles. Drug release mechanism according to Korsmeyer-Peppas model was found anomalous transport (n = 0.5847). Scanning electron microscopy (SEM) revealed that the nanoparticles were spherical in shape and there was no crystallization of drug and other excipients. Drug-excipients compatibility confirmed by FTIR study.
Keywords: Prednisolone; Chitosan; Nanoparticles; Ionic gelation method; SEM
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Introduction
Inflammatory bowel disease (IBD) represents a group of idiopathic chronic inflammatory intestinal conditions that covers a group of disorders in which the intestines become inflamed (red and swollen), major type of IBD as crohns & ulcerative colitis [1-3]. Oral drug delivery system play promising role to treat above disease but having some limitation such as presystemic elimination, absorption drug through stomach unable to target the intestine as site of action. Nanoparticles are defined as particulate dispersions or solid particles with a size in the range of 10-1000nm. Prednisolone is anti-inflammatory actions of glucocorticoids are thought to involve phospholipase A2 inhibitory proteins, lipocortins, which control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes [4-6]. Present research has been focused on the preparation of nanoparticles using biodegradable hydrophilic polymers such as chitosan by ionic gelation method.
The method involves a mixture of two aqueous phases, of which one is the polymer chitosan, a di-block co-polymer ethylene oxide or propylene oxide (PEO-PPO) and the other is a polyanion sodium tripolyphosphate. In this method, positively charged amino group of chitosan interacts with negative charged tripolyphosphate to form coacervates with a size in the range of nanometer. Coacervates are formed as a result of electrostatic interaction between two aqueous phases, whereas, ionic gelation involves the material undergoing transition from liquid to gel due to ionic interaction conditions at room temperature [7-8]. Resulting approach used for prolonged and/or controlled drug delivery, Improvement of oral bioavailability, Targeted drug delivery to the specific sites, Minimize fluctuation within a therapeutic range, Decreasing dosing frequency, Patient compliance is also improved [9-12]..
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Materials & Methods
Prednisolone was obtained from Cadila healthcare pvt ltd Ahmadabad, Chitoson, Schiff’s reagent purchased from Chemdyes Corporation, Rajkot, India. Sodium tripolyphosphate purchased from Molychem, Bombay, India, Pluronic F-127, Mucin was obtained from Sigma Aldrich, Mumbai, India All other solvent and reagents were of analytical grade.
Analytical Method Development: UV-Visible Spectrophotometric Method

In the present work, Prednisolone was estimated by UVVisible Spectrophotometric method using dissolution media phosphate buffer saline (PBS) pH 7.4. Preparation of phosphate buffer saline (pH 7.4) [13] All ingredients were dissolved in 1 liter of distilled water and pH was adjusted to 7.4 with 1M NaOH (Sodium hydroxide) (Table 1).
Determination Of UV Absorption Maxima
10 μg/ml solution of Prednisolone was prepared in phosphate buffer saline pH 7.4. The absorbance of these was measured at entire range of UV (200-400 nm) for determination of λmax (wavelength maxima).
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Spectrophotometric Analysis of Prednisolone
Preparation of Stock Solution
100 mg Prednisolone was weighed accurately using digital analytical balance and transferred to 100 ml volumetric flask dissolved in phosphate buffer saline pH 7.4 and the final volume was made up to 100 ml with phosphate buffer saline pH 7.4 to get a stock solution A (1000 μg/ml). From the stock solution A, 10 ml was pipette out into 100 ml volumetric flask and the final volume was made up to 100 ml with phosphate buffer saline pH 7.4, to get stock solution B (100 μg/ml) [13].
Preparation of Standard Curve
From the stock solution B, further serial dilutions were made with phosphate buffer saline pH 7.4 to get the solutions in the range of 4-20 μg/ml concentration. The absorbance of the samples was recorded at 248 nm using UV-Visible spectrophotometer against phosphate buffer saline pH 7.4 solution as blank.
Drug-Excipients Compatibility Study
Fourier transform infrared spectroscopy was carried out for solid samples to detect if any interactions were present between the drug and polymers [14]. The samples were prepared by the potassium bromide disc method. Powders were triturated in a small size glass mortar and pestle until the powder mixture was fine and uniform. The pellets were prepared by compressing the powders at 20 psi for 10 min using potassium bromide - press. Pure KBr powder was used as background, and for baseline correction. Prepared sample disc was placed in a sample holder. Afterwards, the sample was transferred to sample compartment. Samples were scanned in the region of 4000-400 cm-1 using a brucker FTIR spectrometer.
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Formulation of Chitosan Nanoparticles (Preliminary Screening)

Chitosan Nanoparticles were prepared by ionic gelation method. First of all measured quantity of chitosan polymer was dissolved in 1%v/v acetic acid solution in one beaker. Solublize Pluronic F-127 in above solution. In another beaker, prepare solution of tripolyphosphate containing drug in distilled water. Add chitosan solution drop wise to the solution of tripolyphosphate under gentle magnetic stirring at room temperature for 1 hr. In all cases, the volume ratio of Chi: TPP solution was 2:1. Nanoparticles formed spontaneously in suspension form and freeze dried it [14] (Table 2).
Evaluation of Nanoparticle (Preliminary Batches)
Preliminary batches further evaluated for to study the influence of polymer, stabilizer, cryoprotectanat on performance of Nanoparticle. The particle size & PDI, % Entrapment efficiency and % drug release study were performed details procedure mentioned in section of characterization.
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
To study all the possible combinations of all factors at all levels, a two-factor, three-level full factorial design was constructed and conducted in a fully randomized order [15]. The dependent variables measured were particle size (Y1), % entrapment efficiency (Y2) and in vitro drug release (Y3) in phosphate buffer saline (pH 7.4). Two independent variables, the concentration of chitosan (X1) and the concentration of tripolyphosphate (X2) were set at three different levels.
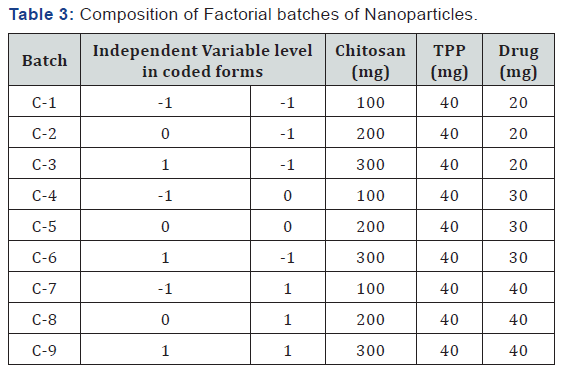
High and low levels of each variable were coded as +1 and -1, respectively and the mean value as zero. 0.1%, 0.2% and 0.3% are low, medium and high level respectively for concentration of chitosan and 0.02%, 0.03% and 0.04% are low, medium and high level respectively for TPP concentration this design was selected as it provides sufficient degree of freedom to resolve the main effects as well as the factor interactions. The conc. Of Stepwise regression analysis was used to find out the control factors that significantly affect response variables (Table 3).
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Characterization of Nanoparticles
Determination of Particle Size and Poly Disparity Index (PDI)
The mean vesicle size and vesicle size distribution was obtained by Zeta sizer. 1 ml suspension was diluted to 100 times with the deionized water. The sample was analyzed using Zeta sizer (Nano ZS, Malvern) [16].
Determination of Zeta Potential
Zeta potential of nanoparticle was measured by dynamic light scattering using Malvern Zetasizer [16].
Determination of % Entrapment Efficiency
% Entrapment efficiency of nanoparticle was determined by ultra filtration method. 2 ml of Nanoparticles suspension was placed into a centrifugal tube which was centrifuged at 10,000 rpm for 15 min at 25°C. The amount of free drug in supernatant was detected by Shimadzu UV1800. The amount entrapped drug was calculated as a result of initial drug minus free drug [17]. % *100 Entrapment efficiency = weight of initial drug weight of free drug Weight of initial drug
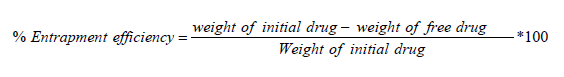
Mucoadhesion Study
Mucoadhesion studies of nanoparticles were performed by mucus glycoprotein assay. Schiff colometric method was used for determining the amount of free mucin to find out amount of adsorbed mucin on the nanoparticles. Calibration curve of mucin was prepared. For that standard solutions of mucin (125, 250, 325, 500 μg/ml) were prepared in distilled water. These samples were incubated at 37°C in a water bath for 1 hour. Then, at room temperature, 0.20 ml Schiff reagent was added to the samples. After 30 minutes the absorbance of the solution was recorded at 556 nm in an ultraviolet spectrophotometer [18]
A standard calibration curve was plotted to calculate the mucin content adsorbed to nanoparticles. Secondly determine the mucoadhesion of the nanoparticles, for that 10 mg of the nanoparticles were dispersed in 20 ml of the mucin solution (0.5 mg/ml). The suspensions were incubated for 1 hour at 37°C with shaking. 0.2 ml Schiff reagent was added to the above solution and kept it at room temperature for 30 min. In order to analyze unadsorbed free mucin, the suspensions were then centrifuged at 12,000 rpm for 5 minutes, and the supernatants were analyzed by spectrophotometer at the visible wavelength of 556 nm.
In vitro Release Study of Nanoparticles
Phosphate buffer saline (PBS) pH 7.4 was selected for the release medium. The lyophilized Prednisolone loaded nanoparticles were suspended in 5 ml phosphate buffer saline (PBS) at pH 7.4 to form the suspension and transferred into a pre- swelled dialysis bag (MW cut-off: 12,000-14,000 Da). The dialysis bag was immersed in 100 ml PBS (pH 7.4). The release study was performed at 37°C and 100 rpm in a constant temperature shaker. After selected time intervals, 5 ml dialysis solution outside the dialysis bag was withdrawn for UV-Vis analysis and replaced with 5 ml fresh buffer solution. Then their absorbance was determined at 248nm by UV-Visible spectrophotometer [19].
SEM Study
The shape and surface morphology of the nanoparticles were studied using scanning electron microscopy (SEM). Nanoparticles were fixed with carbon tape, mounted on metal stubs and then coated with platinum, keeping the acceleration voltage at 10 kV [20].
Release Rate Kinetics
To study the release kinetics of Prednisolone from nanoparticles, the release data were fitted to the following equations
Zero Order Equation
To study the release kinetics, data obtained from in vitro drug release studies were plotted as cumulative amount of drug released versus time
Qt = K0.t
Where, Qt = percentage of drug released at time t K0 = release rate constant [21].
First Order Equation
The data obtained are plotted as log cumulative percentage of drug remaining versus time which would yield a straight line with a slope of -K/2.303
ln (100 - Qt) = ln 100 – K1.t
here, K1 = first order release rate constant [22].
Higuchi’s Equation
The data obtained were plotted as cumulative percentage drug release versus square root of time.
Qt= KH.t 1/2
Where, KH = Higuchi release rate constant [23].
Hixson-Crowell Equation
To study the release kinetics, data obtained from in vitro drug release studies were plotted as cube root of drug percentage remaining in matrix versus time
W0 1/3 – W11/3 = K HC.t
Where, K HC= Hixson-Crowell rate constant [23].
Korsmeyer-Peppas equation
Korsmeyer et al. (1983) derived a simple relationship which described drug release from a polymeric system equation
Qt/Q∞ = KKp.tn
Where, Qt/Q∞ = fraction of drug released at time t, KKP = korsmeyer-peppas rate constant compromising the structural and geometric characteristics of the device,
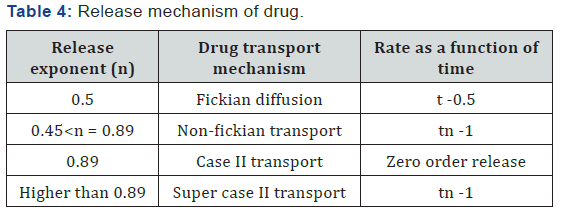
n = release exponent, which is indicative of the mechanism of drug release [24,25]. To study the release kinetics, data obtained from in vitro drug release studies were plotted as log cumulative percentage drug release versus log time (Table 4).
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Results and Discussion
Identification of UV Absorption Maxima (Λmax)
Calibration Curve of Prednisolone in PBS buffer (pH 7.4):
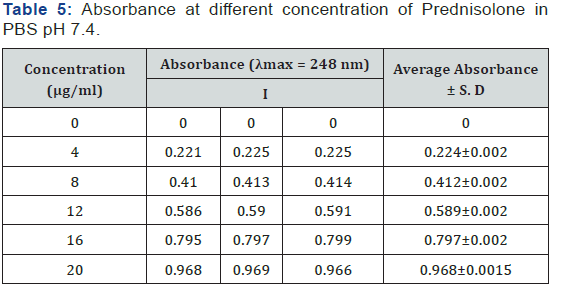
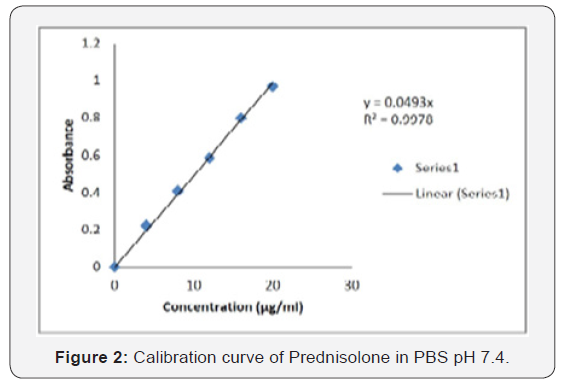
Calibration curve of Prednisolone was prepared in phosphate buffer pH 7.4 at λmax 248 nm. Slope and regression value (r2) was found to be 0.0493 and 0.9978 respectively (Table 5) (Figures 1 & 2).
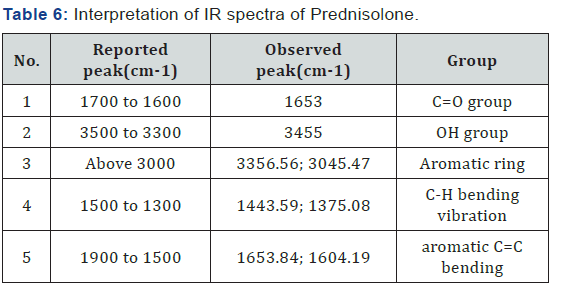
FTIR Spectra of Prednisolone: FTIR spectra of pure drug Prednisolone is shown in fig. 3 and its interpretation is given in (Table 6) (Figure 3).
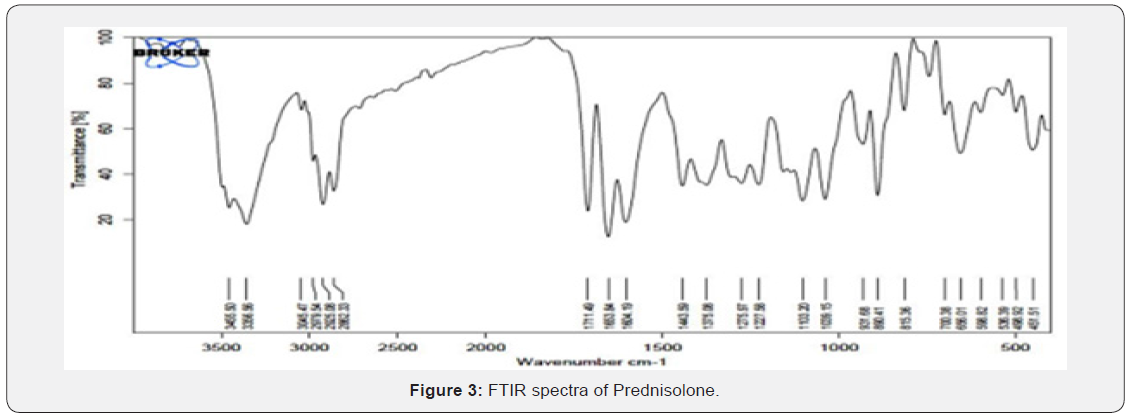
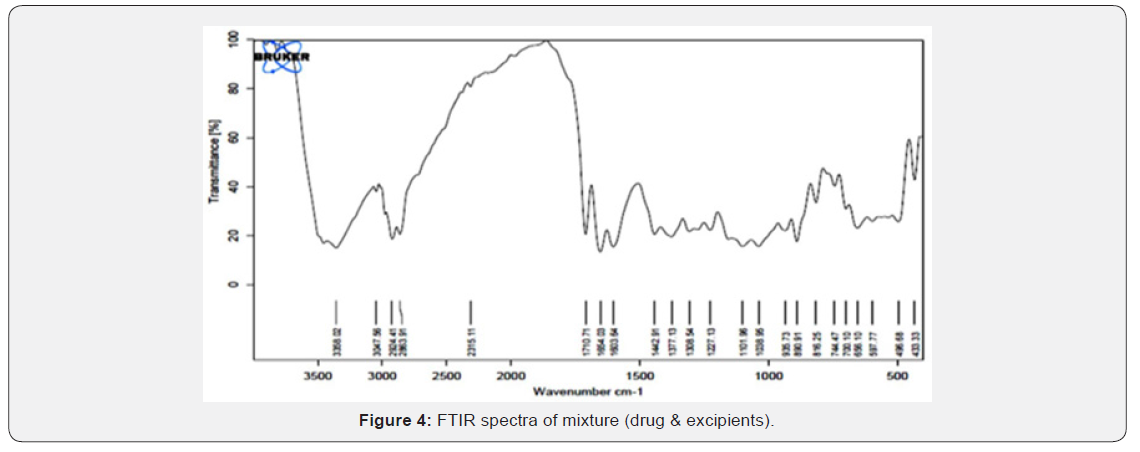
All characteristic peak of drug and polymer were present in FTIR of drug and excipients (Figure 4). In the FTIR Spectra, Prednisolone shows characteristic peak at 1653 cm-1 (carbonyl group), 3455 cm-1 (hydroxyl group), 3045.17, 3356.56 cm- 1(aromatic ring), 1357.08, 1443.59 cm-1(C-H bending vibration). The peaks were also appearing in mixture in Prednisolone and chitosan polymer. So FTIR gave conformation about their purity and showed no interaction between drug and polymer [26].
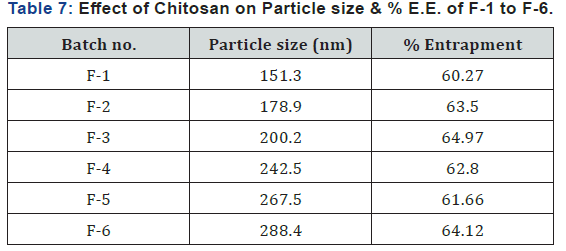
Evaluation of Prednisolone Nanoparticles (Preliminary Batches) Determination of Particle Size & % Entrapment Efficiency (Effect of chitosan concentration)
Here, concentration of chitosan was increased from batch F-1 to F-6 respectively and at that time other components were kept constant. (Table 7) shows the results for particle size and % entrapment. From the result it was seen that particle size of chitosan nanoparticles increases as concentration of chitosan was increased. As concentration of chitosan increased, viscosity of solution increased which prevents effective ionic interaction between tripolyphosphate and chitosan solution that increased the size of nanoparticles and percentage of entrapped drug was found to be above 60. From above batches F-1, F-2, F-3 were selected for factorial design which contains 0.1%, 0.2% and 0.3% concentration of chitosan respectively.
Determination of Particle Size & % Entrapment Efficiency (Effect of Triphosphate concentration)
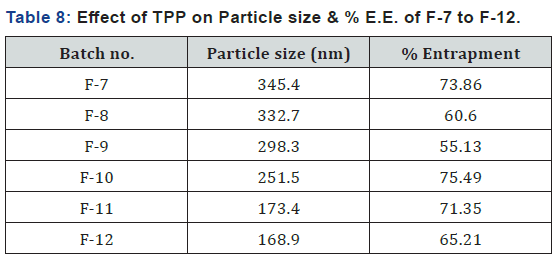
Here, concentration of TPP was decreased gradually from batch F-7 to F-12 and at that time other components were kept constant (Tables 4 & 5) shows the results for particle size and % entrapment (Table 8). From the result it was seen that particle size of chitosan nanoparticles decreases as concentration of TPP was decreased. This could be due to the decrease in the amount of anionic groups in the preparation medium, which causes less electrostatic interaction with positive amino sites on chitosan and drug entrapped in the nanoparticles was above 60%. From above batches we show that better results were obtained when concentration of TPP was between 0.02% to 0.04% so we selected 0.02%, 0.03% and 0.04% as low, medium and high level in factorial design respectively.
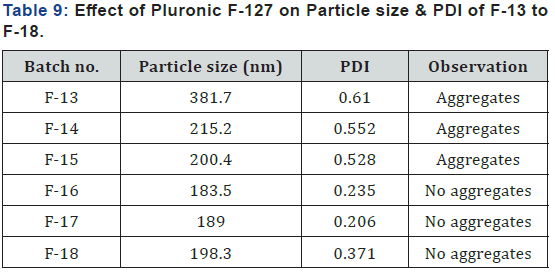
Determination of Particle Size (Effect of Pluronic F-127 concentration)
Screening batches F-13 to F-18 were prepared for screening of pluronic F-127 as stabilizing agent. Here, concentration of pluronic F-127 was increased from batch F-13 to F-18 respectively and results are shown in (Table 9). From the result it was shown that particle size decreased as concentration of pluronic F-127 increased and polydispersity index (PDI) also decreased. Stabilizer was used to stabilize the formulation. When concentration of stabilizer was low, aggregates were formed due to less stability of particles, so size of particle was increased and PDI was also reduced as concentration of stabilizer was increased which indicate presence of monodisperse particle in formulation. From above batches, F-16 batch contains 3% pluronic F-127 was selected to use in factorial batches.
Determination of Particle Size & PDI (Effect of Cryoprotectant concentration)
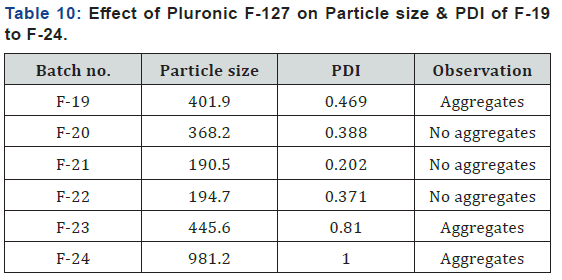

Cryoprotectant was added to the formulation before freeze drying process to prevent damage of internal structure of formulation and prevent formation of aggregates and stabilize the nanoparticles. Here different cryoprotectants with different concentrations were selected and results were showed in (Table 10). From result one showed that lactose gave better results than PDI value and no aggregates were formed after freeze drying process (Figure 5).
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Evaluation of Factorial Batches of Prednisolone Nanoparticles
Determination Particle Size & PDI
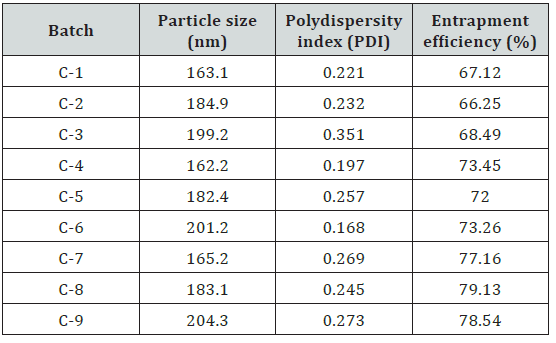
The particle size and size distribution of the Prednisolone loaded nanoparticles in aqueous solution were determined by dynamic light scattering (DLS) and the results were displayed in (Table 11). Particle size is very useful in understanding various properties of the nanoparticles for example dispersion, aggregation and it also affects the biological uptake of the particles. The nanoparticles should be small enough to improve drug delivery, lower the toxicity and for longer duration of time at the site of delivery; they are around 200 nm with particle size distribution (PDI below 0.500). The polydispersity index (PDI) suggested that the obtained Prednisolone loaded nanoparticles were monodisperse and did not aggregate in water. Such ranged nanoparticles may accumulate more readily at the inflammatory site
Determination Entrapment Efficiency (%)
At least 80% of Prednisolone was entrapped in the nanoparticles it was observed that there was increase in %E.E with increase in amount of polymer and cross-linking agent. Hence more time may require by drug molecules for diffusing out of polymer matrix as polymer concentration and crosslinking concentration increases because it form more crosslinked structure of particle.
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Mucoadhesion Study
Calibration Curve of Mucin
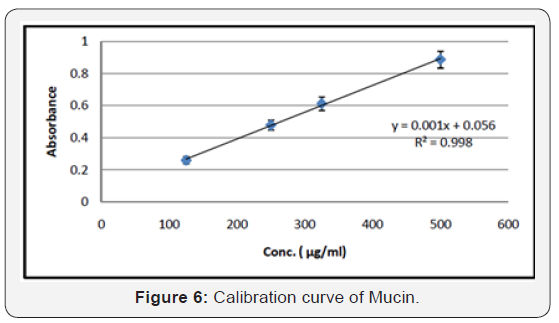
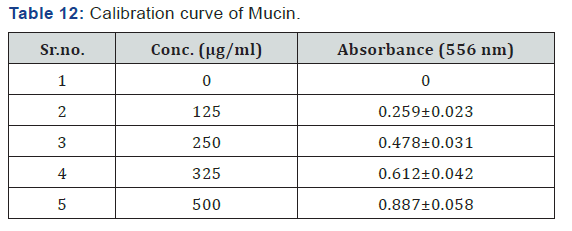
Determination of Mucoadhesion Strength of Mucin
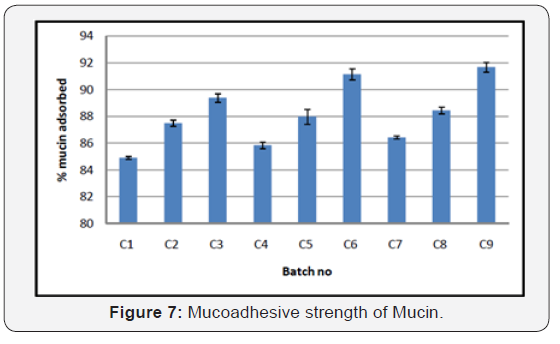

As polymer concentration increase, mucoadhesion property of nanoparticles was also increased. High mucoadhesivity of the nanoparticles is attributed to the hydrogen bond and ionic interaction of the positive charge of chitosan amino groups with mucin chains. Smaller particles show higher mucoadhesion than for larger paticles because small particles provide large surface area and increase in mucin adsorption, which lead to a high mucoadhesive property for the nanoparticles as shown in (Figure 7) (Table 13).
In Vitro Release Study
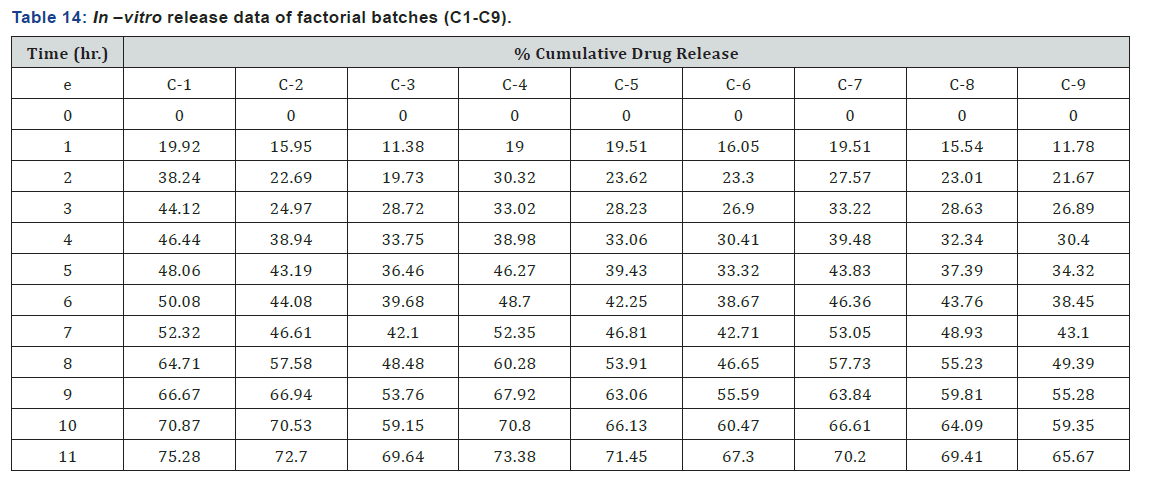

The release profiles of different Prednisolone loaded nanoparticles were investigated in phosphate buffer saline pH 7.4 solution at 37°C. All nanoparticles exhibited a fast release of Prednisolone at the initial stage and a sustained release in the following time. As concentration of polymer and cross-linking agent was increased more cross-linked structure was formed which took more time to diffuse out drug from polymer matrix so it sustained the release of drug. Drug release from higher polymer concentration and TPP concentration were slower than lower polymer concentration and TPP concentration (Table 14) (Figure 8). are show that nanoparticles of Prednisolone give biphasic release behavior. After the initial burst release for about 3 hr., the release rate of Prednisolone slow down and follow Higuchi model. The burst release of nanoparticles might be due to the diffusion of drug that was adsorbed on the surface of nanoparticles.
Determination of Release Rate Kinetics
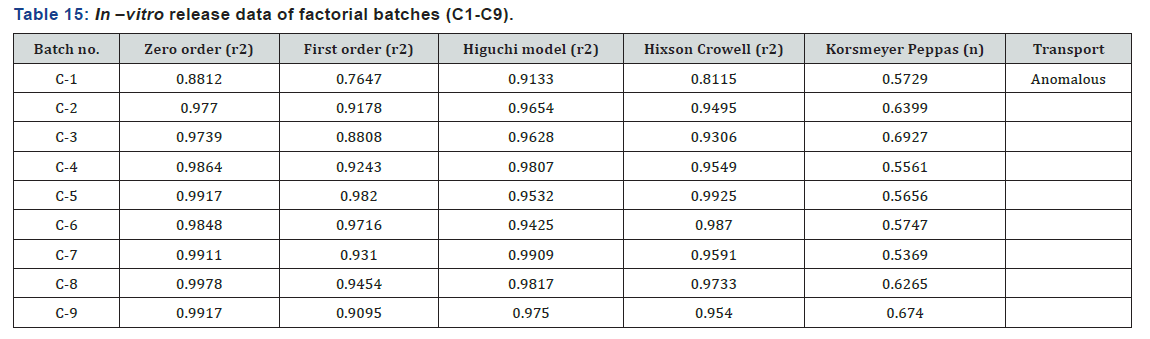
The r2 value is considered as the tool for repressing the best fitting kinetic model. The value of regression correlation coefficient for most of the formulations was highest in case of zero order release, so drug release from nanoparticles followed zero order release (Table 15). Drug release mechanisms of the nanoparticles were evaluated using the Korsmeyer -Peppas model. In this model, the value of n identified the release mechanism of drug. The n value for most of the batches was found between 0.5 and 1, which confirmed that mechanism of drug release follows an anomalous transport.
Statistical Analysis and Factor Influence Study
Statistical analysis was carried out for the data of particle size, % entrapment efficiency and in vitro release study. These three factors were considered as dependent variables for the study. Analysis and optimization were carried out by using design expert 8.0.7.1 software.
ANOVA (Analysis of Variance) Analysis for Particle Size
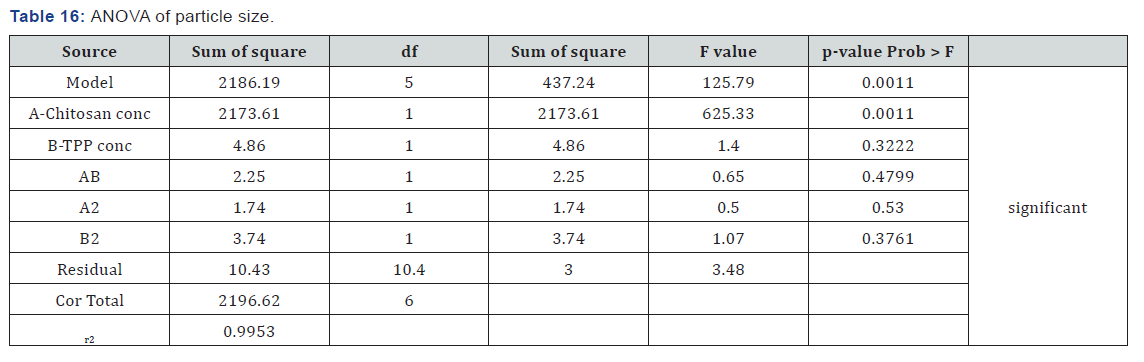
ANOVA for response surface quadratic model for particle size was found to be significant as p-value for the model is 0.0011 which is less than 0.05 (Table 16). Both the independent factors are having p-value less than 0.05 indicating the significant effect of the factors on the response. Interaction p-value was 0.4799 which indicates that there was no significant interaction between factors. r2 value was found to be 0.9953 indicating the linearity of the model. Above equation represents the quantitative effect of the independent factors on the particle size written in terms of coded factors. Polynomial equation obtained indicated that both the factors have same effects on the particle size. It showed that factor A (concentration of chitosan) have positive effect on particle size i.e. as the A increases particles size also increases. Factor B (concentration of TPP) also have positive effect on the particle size i.e. as the B increases particle size also increases.Factor B (concentration of TPP) also have positive effect on the particle size i.e. as the B increases particle size also increases.
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Polynomial Equation
The response (Y1) obtained at various levels of the 2 independent variables (X1 and X2) were subjected to multiple regression to yield a second-order polynomial equation (full model). Equation clearly reflects the wide range of values for response (Y1).
Particle size = +182.56 +19.03 * A +0.90 * B +0.75 * A*B -0.93 * A2 +1.37 *B2
The positive effect of concentration of chitosan & TPP on particle size i.e. increases as concentration increases.
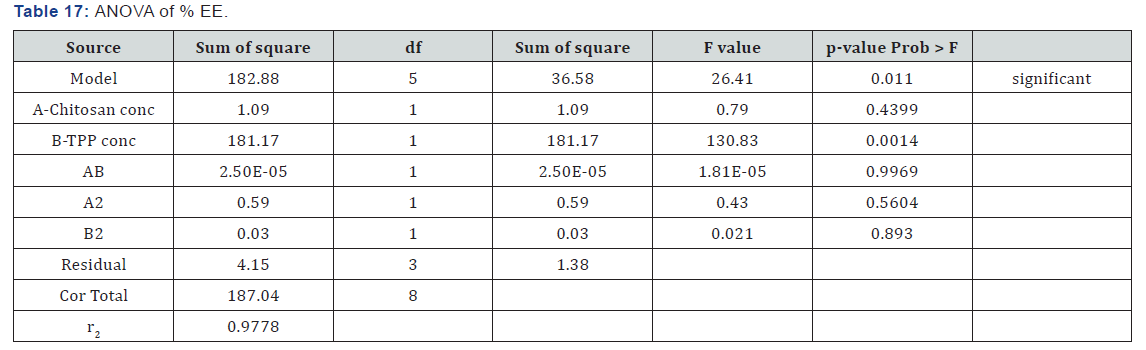
ANOVA for response surface quadratic model for % entrapment efficiency was found to be significant as p-value for the model is 0.0110 which is less than 0.05 (Table 16). Factor A (concentration of chitosan) have p-value of 0.4399 which indicated that factor A has insignificant effect on the % entrapment efficiency. While p-value for factor B was 0.0014 which indicated that it has significant effect on the % entrapment efficiency. Interaction p-value was 0.9969 which indicates that there was no significant interaction between factors. r2 value was found to be 0.9778 indicating the linearity of the model. Above equation represents the quantitative effect of the independent factors on the % entrapment efficiency written in terms of coded factors (Table 17).
Polynomial Equation
The response (Y1) obtained at various levels of the 2 independent variables (X1 and X2) were subjected to multiple regression to yield a second-order polynomial equation (fullmodel). Equation clearly reflects the wide range of values for response (Y2).
% EE = +72.54 +0.43 * A +5.50 * B +2.500E-003 * A*B +0.54 * A2 -0.12 *B2
Polynomial equation obtained indicated that only factors B have effect on % entrapment efficiency. It showed that factor B (concentration of TPP) have positive effect on % entrapment efficiency i.e. as the B increases % entrapment efficiency also increases.
ANOVA (Analysis of Variance) analysis for in vitro release
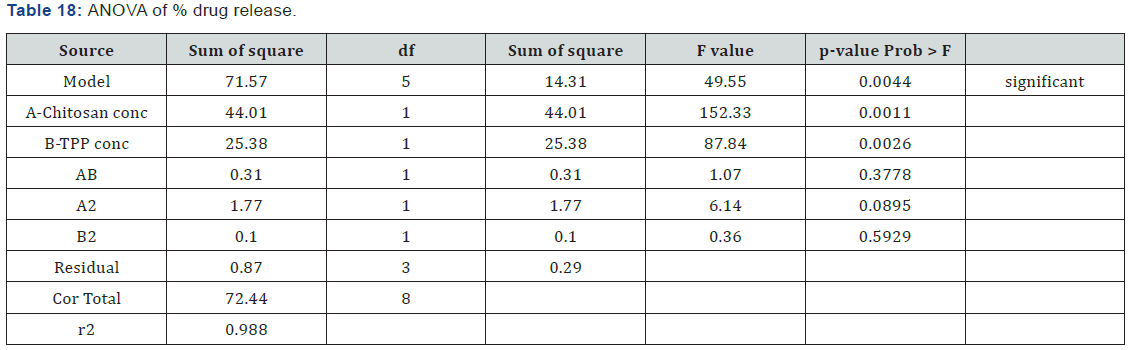
ANOVA for response surface quadratic model for In vitro release of drug was found to be significant as p-value for the model is 0.0044 which is less than 0.05 (Table 17). Both the independent factors are having p-value less than 0.05 indicating the significant effect of the factors on the response. Interaction p-value was 0.3778 which indicates that there was no significant interaction between factors. r2 value was found to be 0.9880 indicating the linearity of the model. Above equation represents the quantitative effect of the independent factors on the in vitro release written in terms of coded factors (Table 18).
Polynomial Equation
Polynomial equation obtained indicated that both the factors have same effects on the in vitro release. It showed that factor A (concentration of chitosan) have negative effect on in vitro release i.e. as the A increases in vitro release decreases. Factor B (concentration of TPP) also have negative effect on the in vitro release i.e. as the B increases in vitro release decreases
In vitro release = +71.34 -2.71 * A -2.06 * B +0.28 * A*B -0.94 * A2 -0.23 *B2
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Optimization of Formulation
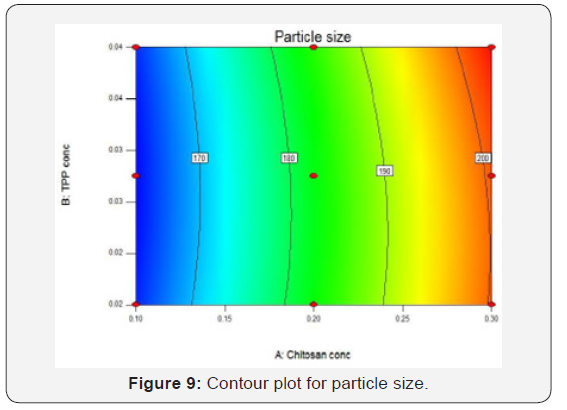
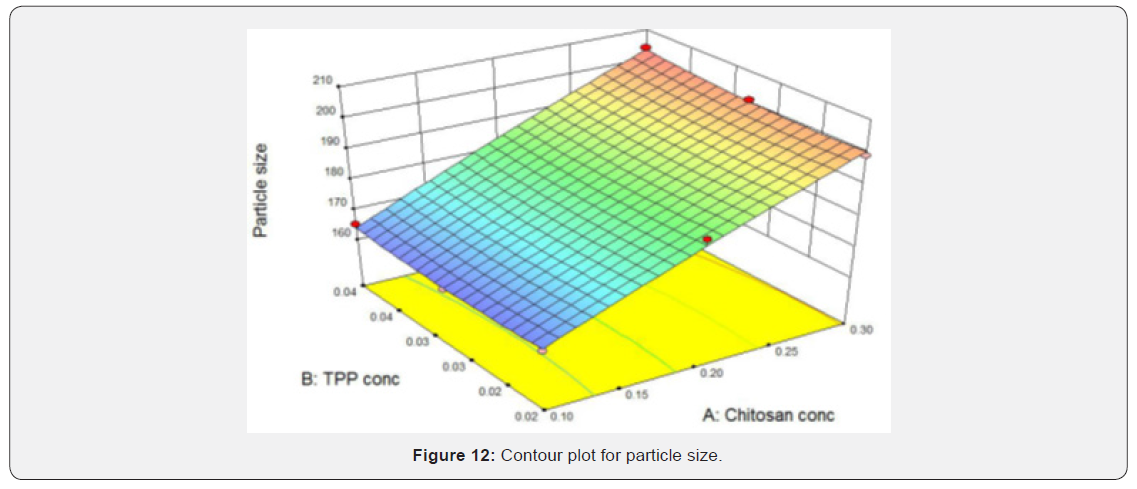
Contour & Surface Response Plot for Particle Size
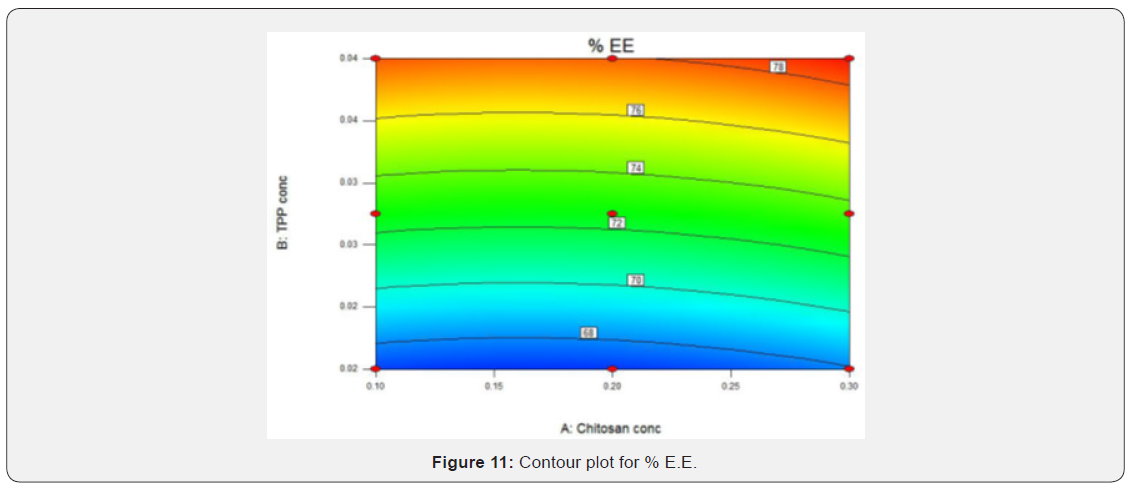
(Figures 9 & 10) shows that as the concentration of chitosan increases particle size also increased and as the concentration of TPP increases particle size found to be increased slightly means it has very less effect on particle size. The smallest particle size area is corresponds to blue region of the graphs which represents lowest concentration of chitosan and TPP. Contour plot and response surface plot for % Entrapment efficiency: As the concentration of TPP increased entrapment efficiency also increased. Concentration of chitosan does not have any effect on the entrapment efficiency. Red region in the graph shows the highest entrapment efficiency which corresponds to higher concentration of TPP (Figures 11 & 12).
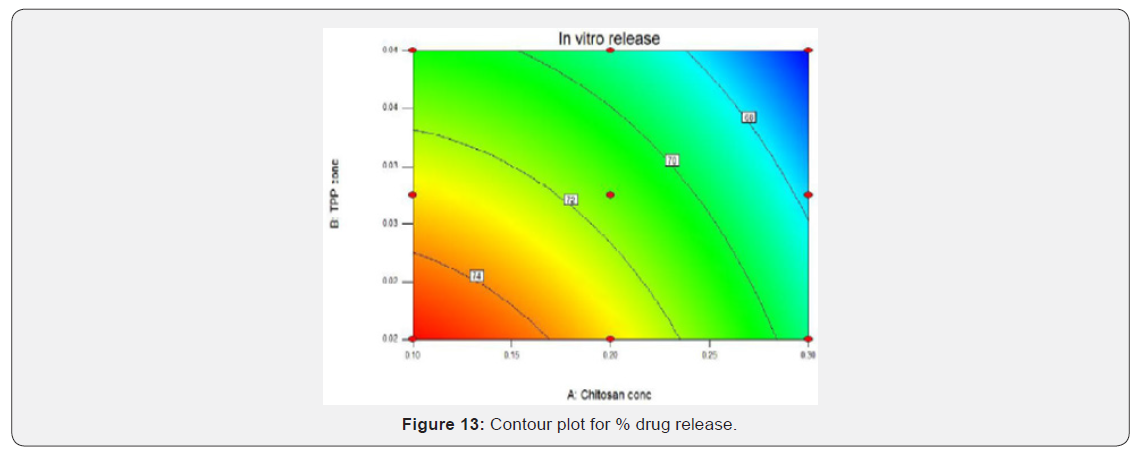
Contour plot and response surface plot for % Drug release: As the concentration of chitosan increases in vitro release is decreasing and as the concentration of TPP increased in vitro release was found to be decreasing. This shows that as concentration of chitosan and TPP increased it sustained the release of drug for longer duration of time because of cross linking between polymer and cross-linking agent. The slower release of drug is corresponding to blue region of the graphs which represents highest concentration of chitosan and TPP (Figures 13 & 14).


- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Overlay Plot
Overlay plot was obtained by superimposing the critical response contours on a contour plot. Graphical optimization displays the area of feasible response values in the factor space. Regions that do not fit the optimization criteria are shaded. The yellow region indicates the area in which optimized formulation can be formulated. The yellow portion covered one point that near to (-1,+1) value that means formulation C-7 (Figure 15).
Evaluation of Check Point Batch


To determine, whether the selected model was correct or not, check point batch was prepared. Quantity of the ingredients was chosen from the Design- Expert version 8.0.7.1 software. It provides theoretical results. Same quantities of ingredients were taken and check point batch was formulated and evaluated for the desired responses (Figures 16 & 17) (Table 19).
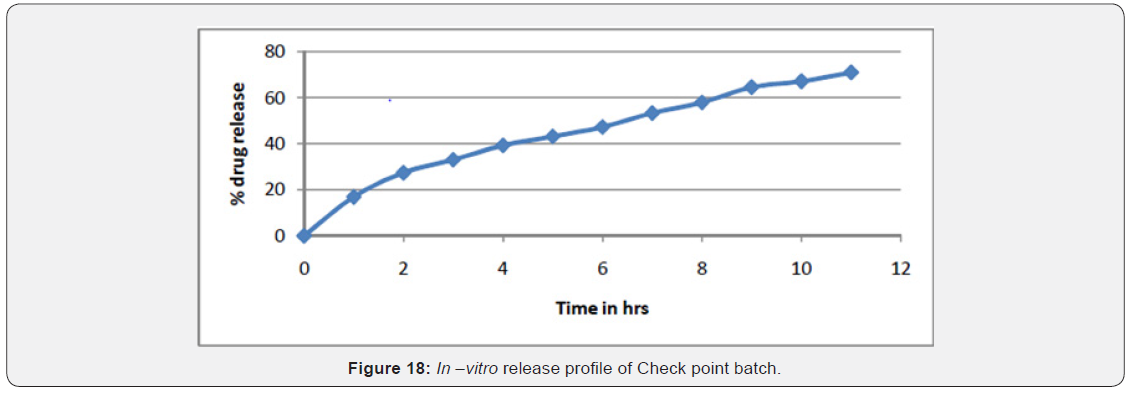
In Vitro Release of Optimized Batch

In vitro release of 70.80% drug in 11 hr from check point batch which indicate sustained release of drug for longer duration of time (Figure 18) (Table 20).
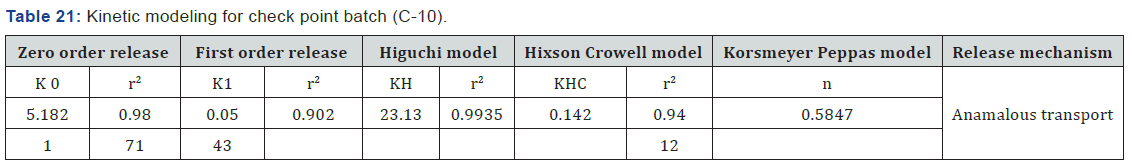
Release Kinetic Modeling
According to this data formulation follow higuchi model and release mechanism is anomalous transport, which means release of drug is by erosion and diffusion mechanism (Table 21).
SEM Study


Shape and surface morphology were investigated using scanning electron microscopy (Figures 19 & 20) of check point batch indicates that the cross-linked chitosan nanoparticle possessed a nearly smooth surface and spherical shape.
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
Conclusion
Prednisolone loaded chitosan nanoparticles were prepared by ionic gelation method. In this method chitosan was cross linked with tripolyphosphate. The nanoparticles were spherical in shape. The optimized formulation showed particle size around 168 nm with good entrapment efficiency. In vitro evaluation shows that the nanoparticles seem to be a sustained dosage form of Prednisolone for inflammatory bowel disease. Factorial design indicates that higher concentration of chitosan leads to increase in particle size and sustained release of drug and higher concentration of tripolyphosphate leads to higher entrapment efficiency and sustained release of drug from nanoparticles.
- Research Article
- Abstract
- Introduction
- Materials & Methods
- Spectrophotometric Analysis of Prednisolone
- Formulation of Chitosan Nanoparticles (Preliminary Screening)
- Optimization of Formulation by 32 Full Factorial Designs 32 Factorial Designs
- Characterization of Nanoparticles
- Results and Discussion
- Evaluation of Factorial Batches of Prednisolone Nanoparticles
- Mucoadhesion Study
- Polynomial Equation
- Optimization of Formulation
- Overlay Plot
- Conclusion
- References
References
- Rang HP, Dale MM, Ritter JM (2003) Pharmacology (5th edn), Elsevier science limited, pp. 377-378.
- Tripathi KD (2004) Essential of medical pharmacolody (5th edn), Jaypee brother’s medical publishers Ltd. pp. 661-663.
- http://en.wikipedia.org/wiki/Inflammatory_bowel_disease
- Bernstein C (2009) Inflammatory bowel disease: a global perspective.
- Edward F (2010) Researchers Discover Successful Probiotic Treatment for Inflammatory Bowel Disease.
- Mohanraj VJ, Chen Y (2006) nanoparticles. Trop J of Pharm 5(1): 561- 573.
- Patrick C, Catherine D, Irene B (2002) Nanoparticles in cancer therapy and diagnosis. Adv. Drug Del 54(5): 631-651.
- Anton S, Maruthi G, Manavalan AR (2011) Nanoparticles. J Adv Sci 2: 12-19.
- Hosseinzadeh H, Atyabi F, Dinarvand R, Naser Ostad S (2012) Chitosan– Pluronic nanoparticles as oral delivery of anticancer gemcitabine: preparation and in vitro study. Int. J. of Nanomed 7: 1851–1863.
- Seda R, Pulat M (2012) 5-Fluorouracil Encapsulated Chitosan Nanoparticles for pH-Stimulated Drug Delivery: Evaluation of Controlled Release Kinetics. J of Nanomaterials 10: 1-10.
- Rabiskova M (2012) Coated chitosan pellets containing rutin intended for the treatment of inflammatory bowel disease: In vitro characteristics and in vivo evaluation. Int. J of Pharmaceutics 422(1-2): 151-159.
- Yan W, Fan W, Xu Z, Ni H (2012) Formation mechanism of monodisperse, low molecular weight chitosan nanoparticles by ionic gelation technique. colloids and surfaces B: Biointerfaces 90: 21-27.
- Indian Pharmacoepia (2007) Government of India, Ministry of health and family welfare, published by Indian pharmacopeia commission, Ghaziabad 1: 244.
- Nayak S, Patel H, Kesarla R, Murthy R (2011) Colon delivery of 5 – Fluoro uracil using cross- linked chitosan microspheres coated with eudragit S 100. Int. J. of Drug Del 3: 260-268.
- Guan J (2011) Optimized Preparation of Levofloxacin-loaded Chitosan Nanoparticles by Ionotropic Gelation. Int. Conf. on Phy. Sci. and Tech 22: 163-169.
- Jose S, Prema M, Chacko A, Thomas A, Souto E (2011) Colon Specific Chitosan Microspheres for Chronotherapy of Chronic Stable Angina. Colloids and Surfaces B: Biointerfaces 83(2): 277-283.
- Hosseinzadeh H, Atyabi F, Dinarvand R, Naser Ostad S (2012) Chitosan– Pluronic nanoparticles as oral delivery of anticancer gemcitabine: preparation and in vitro study. Int. J. of Nanomed 7: 1851–1863.
- Valle B, Omwancha W, Mallipeddi R, Neau S (2013) Chitosan as a pore former in coated beads for colon specific drug delivery of 5-ASA. Int. J. of Pharmaceutics 441(1-2): 343-351.
- Anitha A, Deepagan VG, Divya Rani VV, Menon D, Nair SV et al. (2011) Preparation, characterization, in vitro drug release and biological studies of curcumin loaded dextran sulphate–chitosan nanoparticles. Carbohydrate Polymers 84: 1158–1164.
- Goracinova K, Dodov M, Crcarevska M (2008) Chitosan coated Ca–alginate microparticles loaded with budesonide for delivery to the inflamed colonic mucosa. European J of Pharm and Biopharm 68: 565- 578.
- Dash S, Murthy P, Nath L, Chowdhury P (2010) Kinetic Modeling on Drug Release from Controlled Drug Delivery. Systemsacta Poloniae Pharmaceutica N Drug Research 67(3): 217-223.
- Merchant HA, Shoaib HM, Yousuf RI (2006) Once-daily tablet formulation and in vitro release evaluation of cefpodoxime using hydroxypropyl methylcellulose: A technical note. AAPS Pharm Sci Tech 7(3): E178-E183.
- Higuchi T (1961) Rate of release of medicaments from ointment bases containing drugs in suspension. J Pharma Sci 50: 874-875.
- Korsemeyer R, Gurny R, Peppas N (1983) Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm 15: 25-35.
- Peppas NA (1985) Analysis of Fickian and non-Fickian drug release from polymers. Pharm Acta Helv 60(4): 110-111.
- Ritger PL, Peppas NA (1987) Simple equation for solute release. Part 1. Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or disks. J Control Rel 5(1): 23-26.






























