Development and Validation of A RP- HPLC Method for the Simultaneous Estimation of Valsartan and Sacubitril in Rat Plasma
N Anjaneyulu*, R Naga Kishore, M Ravi Kumar and G Sneha
Department of Pharmaceutical Analysis, Geethanjali College of Pharmacy, India
Submission: December 12, 2018; Published: December 21, 2018
*Corresponding author: N Anjaneyulu, Professor, Department of Pharmaceutical Analysis, Geethanjali College of Pharmacy, Cheeryal(V), Keesara(M), Medchal (Dist), Telangana, India
How to cite this article: N Anjaneyulu, R Naga K, M Ravi K, G Sneha. Skipping Breakfast Everyday Keeps Well-Being Away. Glob J Pharmaceu Sci. 2018; 6(5): 555697. DOI: 10.19080/GJPPS.2018.06.555697.
Abstract
A basic precise selective, sensitive isocratic technique was developed and valid for the quantitative synchronic estimation of Sacubitril and Valsartan drug in rat plasma was by RP- HPLC system. The chromatographic activity separation was administrated out on Intersil C18 (250 x 4.6mm, 5μm) column with a mix of acetonitrile: di-potassium hydrogen phosphate, pH 3.0 adjusted with Phosphate buffer (30:70%v/v) as mobile part. The analytes were eluted with a rate of flow of 0.8ml/min and at a wavelength of 371nm of UV detection. The strategy was valid for precision, accuracy, linearity, Limit of detection, Limit of Quantification, Ruggedness following the ICH guidelines.
The retention time was 10.725min and 15.366min and the system suitableness results was 99.95% and 100.24% for sacubitril and valsartan respectively. Linearity contemplate was completed between 100-500μg/ml and 5μg-25μg/ml, linear regression coefficient was observed to be 0.999 and the percentage recovery varies from 98-102% of Sacubitril and valsartan. No interference from any part of bulk and pharmaceutical dosage form was determined. All the parameters of validation are found to be at intervals within the vary that confirms the quality of the strategy for the determination of Sacubitril and Valsartan.
Keywords: Sacubitril; Valsartan; RP-HPLC; Method development; Validation
Introduction
High Performance Liquid Chromatography
Pittcon paper, originally indicated the proven fact that prime air mass was wont to generate the flow required for liquid natural action in packed columns. Among the beginning, pumps only had a pressure capability of 500 psi. This was known as air mass liquid natural action, or HPLC. New HPLC instruments could develop up to 6,000 psi of pressure, and incorporated improved injectors, detectors, and columns [1-6]. With continued advances in performance throughout this time (smaller particles, even higher pressure), the descriptor HPLC remained an analogous, but name was changed to high performance liquid natural action.
Reversed Phase Chromatography
Reversed phase mode is the most prevalent mode for scientific and preparative partitions of mixes of worry in biological products, pharmaceutical plans and API’s, substance substances, nourishment and biomedical designing. The stationary stage is non-polar hydrophobic pressing with octyl and octadecyl useful gathering attached to silica gel and the mobile stage is a polar dissolvable, regularly a mostly or completely watery versatile stage [7-11]. Polar substances lean toward the versatile stage and elute first. Maintenance increments as the hydrophobiccharacter of the solutes expands, by and large, the lower the extremity of the versatile stage, higher is the eluent quality.
Drug Profile
Sacubitril is chemically 4-{[(2S,4R)-1-(4-Biphenylyl)-5-ethoxy-4-methyl-5-oxo-2-pentanyl] amino}-4-oxobutanoic acid which is an antihypertensive drug used in combination with valsartan for the treatment of heart failure. Sacubitril could be a prodrug that’s activated to sacubitril at (LBQ657) by de-ethylation via esterases. Sacubitril at inhibits the catalyst neprilysin, that is accountable for the degradation of chamber and brain symptom organic compound, a pair of blood pressure-lowering peptides that employment within the main by reducing blood volume. Valsartan is chemically (2S)-3-methyl-2- [pentanoyl- [[4- [2-(2H-tetrazol-5- yl) phenyl] phenyl] methyl] amino] butanoic acid. Valsartan is Associate in Nursing man of affairs that by selection inhibits the binding of Hypertensin to AT1, that’s is found in several tissues like tube-shaped structure sleek muscle and in addition the adrenal glands [8-12].
This effectively inhibits the AT1-mediated agent vasoconstrictive and aldosterone-secreting effects of Hypertensin and finally ends up during a decrease in tube-shaped structure resistance, and force per unit area. Valsartan is selective for AT1 and has regarding affinity for AT2. Inhibition of mineralocorticoid secretion might inhibit metallic element and water organic process among the kidneys whereas decreasing excretion. The primary matter of valsartan, valeryl 4-hydroxy valsartan, has no medicine activity. Literature search reveals that only two analytical methods were reported for simultaneous estimation of sacubitril and valsartan from rat plasma using LC-MS/MS and from a synthetic mixture using HPLC [12-20]. Hence a simple, rapid, sensitive and accurate stability indicating HPLC method was developed for the simultaneous estimation of sacubitril and Valsartan from rat plasma (Figures 1 & 2).
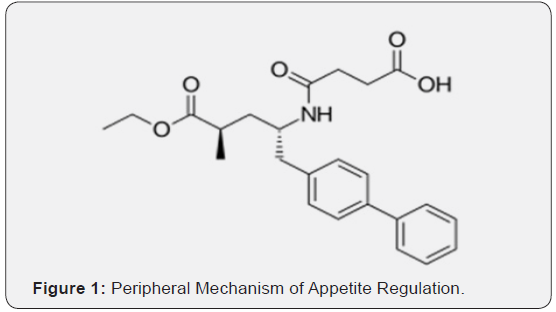
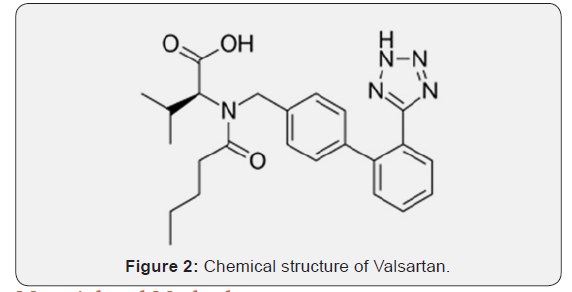
Material and Methods
Materials and Reagents
Sacubitril and Valsartan, KH2PO4, Water and Methanol for HPLC, Acetonitrile for HPLC, di-potassium hydrogen phosphate, Ortho phosphoric Acid.
Equipment
HPLC-WATERS, software: Empower, 2695 separation module, PDA detector, UV/VIS spectrophotometer LABINDIA UV 3000+, pH meter, Weighing machine.
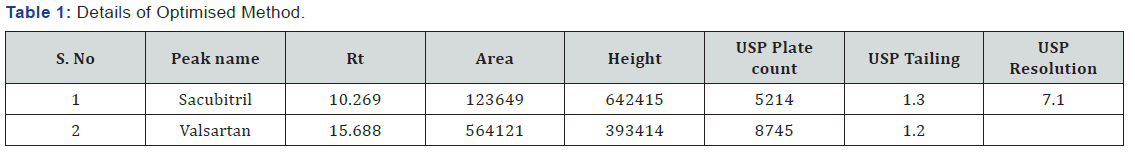
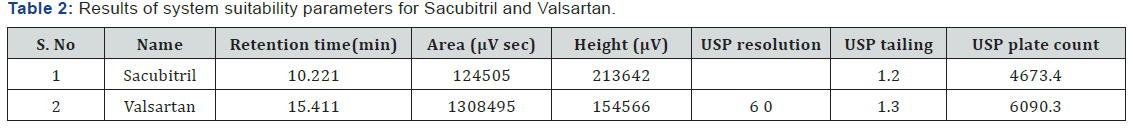
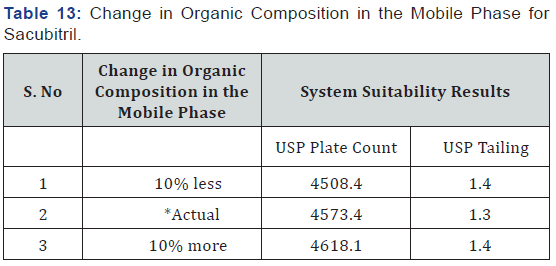
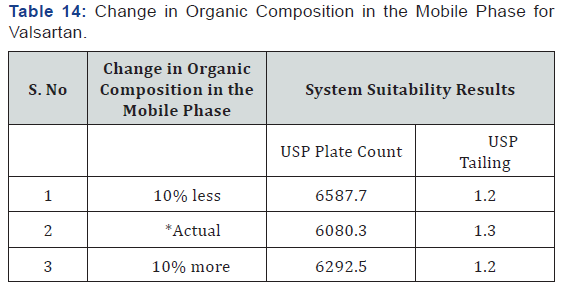
(Table 1) The separation was good, peak shape was good, so we conclude that there is no required for reduce the retention times of peaks, so it is taken as optimized method. The developed Method was validated for linearity, precision, accuracy, robustness and is applied for forced degradation studies as per the ICH guidelines.
Preparation of The Sacubitril & Valsartan Standard & Sample Solution
Standard Solution Preparation
10 mg Amount of standard was mixed with 10 ml acetonitrile to +2ml of rat plasma (untreated) then vertically shaked for 30 min then centrifuged at 5000rpm for 1 hr. Then it was filterated using membrane filters to get clear organic solution. Then it was filled in to the sample vials of HPLC and loaded on to HPLC for Run.
Sample Solution Preparation
Blood samples are collected from the animal’s rats and then centrifuged at 5000rpm for 1 hr to separate the plasma from blood. Then the separated was mixed with acetonitrile then loaded on to the HPLC for Run.
Results and Disscusion
Method Development
Chromatographic Conditions (OptimisedMethod) (Figure 3)
Mobile phase : Phosphate buffer pH 3.0: Methanol (30:70%v/v)
Column : Inertsil C18 5μm (4.6x250mm)
Flow rate : 0.8 ml/min
Wavelength : 371 nm
Column temp : Ambient
Sample Temp : Ambient
Injection Volume: 10 μl
Method Validation
The described method has been validated which include parameters like system suitability, linearity, accuracy, precision, robustness, LOD (limit of detection) and LOQ (limit of quantification).
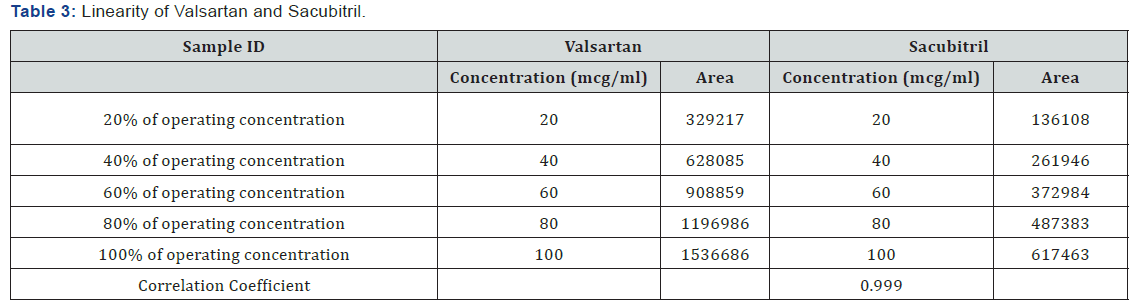
Linearity
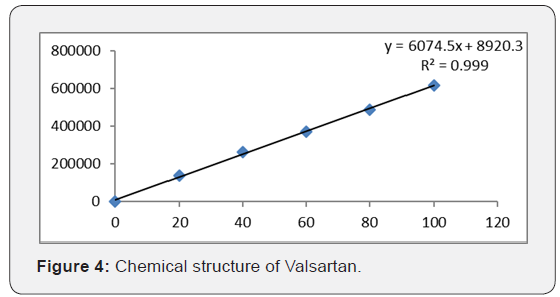
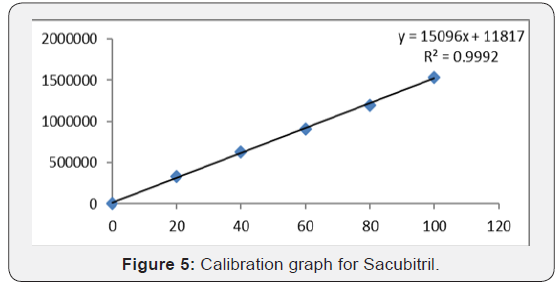
Accurately weigh 10 tablets crush in mortor and pestle and transfer equivalent to 10 mg of Sacubitril and Valsartan (marketedformulation) sample into a 10ml clean dry volumetric flask add about 7ml of Diluent and sonicate to dissolve it completely and make volume up to the mark with the same solvent (Tables 3 & 4 ) (Figures 4 & 5).
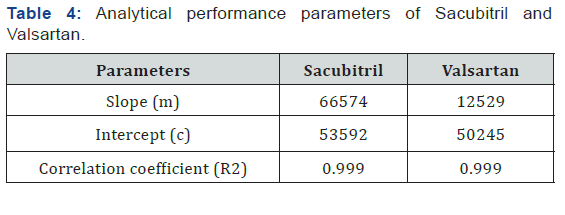
System Suitability
System suitability and chromatographic parameters were validated such as resolution, theoretical plates, and tailing factor was calculated (Table 2).
Accuracy
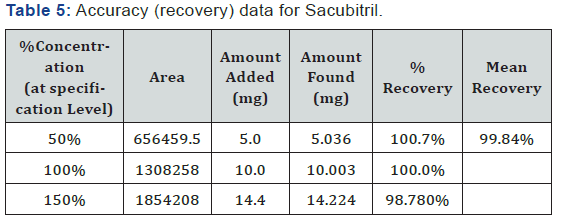
Accurately weigh and transfer 10 mg of Sacubitril and Valsartan10mg of working standard into a 10ml& 100ml clean dry volumetric flask add about 7ml of Diluent and sonicate to dissolve it completely and make volume up to the mark with the same solvent (Tables 5 & 6).

Precision
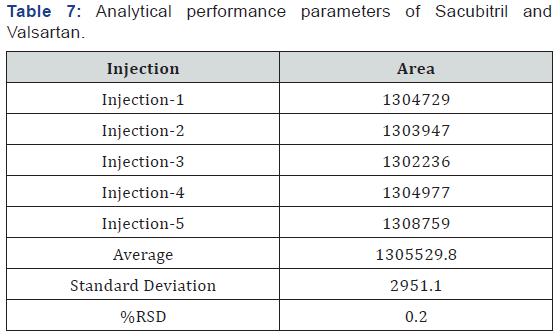
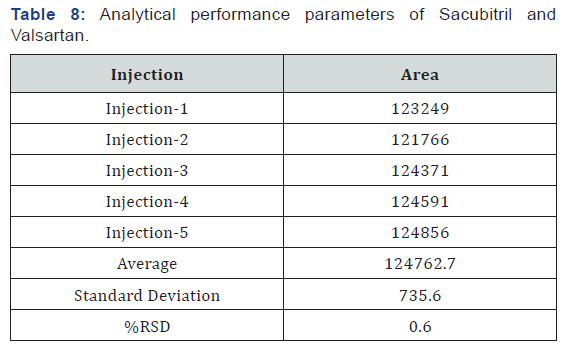
Accurately weigh and transfer 25 mg of Sacubitril and Valsartan working standard into a 10ml clean dry volumetric flask add about 7ml of Diluent and sonicate to dissolve it completely and make volume up to the mark with the same solvent (Tables 7 & 8).
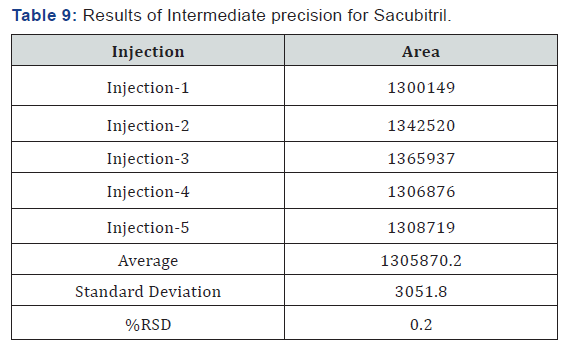
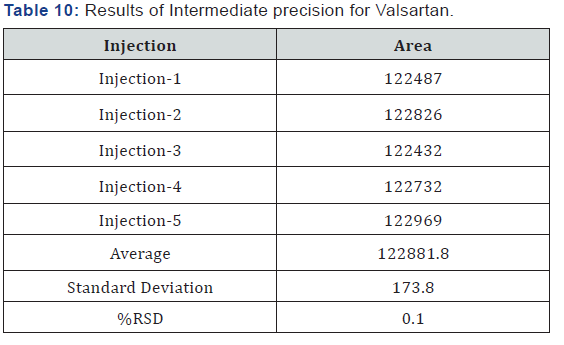
Intermediate Precision/Ruggedness
To evaluate the intermediate precision (also known as Ruggedness) of the method,
Precision was performed on different day by using different make column of same dimensions (Tables 9 & 10).
Robustness
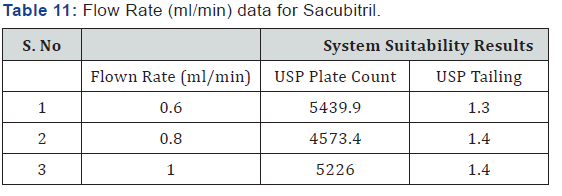
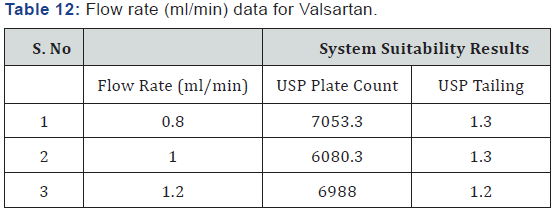
As part of the Robustness, deliberate change in the Flow rate, Mobile Phase composition, Temperature Variation was made to evaluate the impact on the method (Tables 11-14).
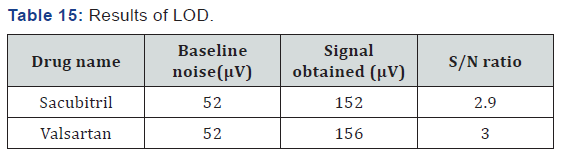
Limit of Detection
Limit of detection: (For Sacubitril)
Accurately weigh and transfer 10 mg of Sacubitril working standard into a 10ml clean dry volumetric flask add about 7ml of Diluent and sonicate to dissolve it completely and make volume up to the mark with the same solvent.
Limit of Detection (For Valsartan)
Accurately weigh and transfer 10mg of Valsartan working standard into a 100ml clean dry volumetric flask add about 7ml of Diluent and sonicate to dissolve it completely and make volume up to the mark with the same solvent (Table 15).
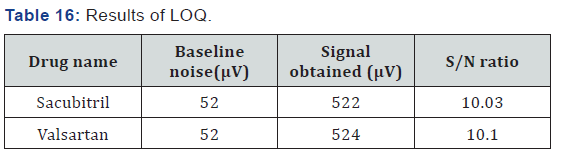
Limit of Quantification
Limit of Quantification (for Sacubitril)
Accurately weigh and transfer 10 mg of Sacubitril working standard into a 10ml clean dry volumetric flask add about 7ml of Diluent and sonicate to dissolve it completely and make volume up to the mark with the same solvent.
Limit of Quantification (for Valsartan)
Accurately weigh and transfer 10mg of Valsartan working standard into a 100ml clean dry volumetric flask add about 7ml of Diluent and sonicate to dissolve it completely and make volume up to the mark with the same solvent (Table 16).
Summary and Conclusion
The proposed HPLC method was found to be simple, specific, precise, accurate, rapid and economical for simultaneous estimation of valsartan and sacubitril in bulk and tablet dosage form. Thus the validated economical method was applied for forced degradation study of valsartan and sacubitril tablet. High performance liquid chromatography is at present one of the most sophisticated tools of the analysis. The estimation of Sacubitril and Valsartan was done by RP-HPLC in rat plasma. The Phosphate buffer was pH 3.0 and the mobile phase was optimized with consists of ACE: di-potassium hydrogen phosphate mixed in the ratio of 70:30 % v/ v.
Inertsil C18 column C18 (4.6 x 150mm, 5m) or equivalent chemically bonded to porous silica particles was used as stationary phase. The detection was carried out using UV detector at 371 nm. The solutions were chromatographed at a constant flow rate of 0.8 ml/min. the linearity range of Sacubitril and Valsartan were found to be from 100-500 g/ ml of Sacubitril and 1-5g/ml of Valsartan. Linear regression coefficient was not more than 0. 999.The values of % RSD are less than 2% indicating accuracy and precision of the method. The percentage recovery varies from 98-102% of Sacubitril and valsartan. LOD and LOQ were found to be within limit.
References
- GR Chatwal, SK Anand (2002) Text book of Instrumental Methods of Chemicaln Analysis. (5th edn). Himalaya Publishing House, pp. 2566- 2570.
- GW Ewing (2011) Text book of Instrumental Methods of Chemical Analysis. (5th edn). Mc Graw-Hill Book Company, pp. 375-385.
- BK Sharma (2011) Textbook of Instrumental Methods of Chemical Analysis. (23rd edn) GOEL publishing house, Meerut, pp. 288-289.
- G Vidyasagar (2009) Textbook of Instrumental Methods of Drug Analysis. Pharmamed Press, pp. 106-120.
- HH Willard, LL Merritt, JA Dean, FA Settle (1986) Textbook of Instrumental Methods of Analysis. (7th edn). CBS publishers and distributors, New Delhi, pp. 592-596.
- HH Tackett, JACripe, GDyson (2008) Positive displacement reciprocating pump fundamentals-power and direct acting types, Proceedings of the twenty-fourth international pump user’s symposium, p. 45-58.
- DA Skoog, FJ Holler, SR Crouch (2007) Textbook of Instrumental Analysis, Brooks/Cole, Cengage Learning India Private Limited, PP. 900-906.
- RE Schirmer (1990) Textbook of Modern Methods of Pharmaceuticals. (2nd edn). CRC press, pp. 242-244.
- LR Snyder, JJ Kirkland, LG Joseph (1997) Practical HPLC Method Development (2nd edn). Wiley Inter Science, New York, p. 1-56.
- Ranjith singh (2013) HPLC Method Development and Validation- an Overview, J Pharm. Educ. Res. 4, p. 26-33.
- ICH: Q2B (1996) Analytical Validation – Methodology.
- LRD Bhavani (2015) Method Development and Validation of HPLC for Detremination of Levetiracetan and Valsartan in their Formulations RRJPA 4(2).
- Muznah AlKhani, Antoun Al-Laham, Mohammed Amer Al-Mardini (2016) Rapid HPLC-UV Method for Quantification of Valsartan in Plasma and Intestinal Perfusate for Pharmacokinetic Studies Int. J. Pharm. Sci. Rev. Res 39(2): 225-229.
- Venkata Suresh, Rama Rao Nadendlaa, BR Challa (2013) Bio- analytical method development and validation of Valsartan by precipitation method with HPLC-MS/MS: Application to a pharmacokinetic study Journal of Chemical and Pharmaceutical Research 5(7): 7-20.
- Ahmed M, Shetty AS, Ahmed M, Krishna V, Aradhya C, et al. (2017) Stability indicating RP-HPLC method for simultaneous estimation of sacubitril and valsartan in bulk and combined pharmaceutical dosage form. World J. Of Pharmacy and Pharm. Sci 6(4): 1714-1728.
- Gopal NM, Sridhar C (2017) RP-UPLC method for simultaneous estimation of sacubitril and valsartan in its bulk and tablet dosage form with force degadation studies. Int. J. of ChemTech Res 10(4): 279-87.
- Patel C, Mishra S, Patel M (2016) Simultaneous estimation of sacubitril and valsartan in pharmaceutical dosage form by development and validation of stability indicating chromatographic method. World J. Pharm. Pharm. Sci 8(6): 1434-1448.
- Patel KH, Luhar SV, Narkhede SB (2016) Simultaneous estimation of sacubitril and valsartan in the synthetic mixture by RP-HPLC method. J Pharm Sci Biosci Res 6(3): 262-269.
- Chunduri RH, Dannana GS (2016) Development and validation of a reliable and rapid LC‐MS/MS method for simultaneous quantification of sacubitril and valsartan in rat plasma and its application to a pharmacokinetic study. Biomedical Chromatography 30(9): 1467- 1475.
- Pedersen-Bjergaard S, Rasmussen KE (2005) Bioanalysis of drugs by liquid-phase microextraction coupled to separation techniques. Journal of Chromatography B 817(1): 3-12.






























