Protective Effect of Purple Grape Juice on Homocysteine - Induced Oxidative Stress
Elif Kağa, Ayhan Vurmaz and Ahmet Kahraman*
Department of Biochemistry, Afyon Kocatepe University, Turkey
Submission: February 13, 2018; Published: April 04, 2018
*Corresponding author: Ahmet Kahraman, Department of Biochemistry, Afyon Kocatepe University, Turkey, Tel: +90-272-2463301/5103; Fax: +90-272-2463300; Email: ahmetkah@aku.edu.tr
How to cite this article: Elif K, Ayhan V,Ahmet K. Protective Effect of Purple Grape Juice on Homocysteine - Induced Oxidative Stress.Glob J Pharmaceu Sci. 2018; 5(2): 555663. DOI: 10.19080/GJPPS.2018.05.5556663
Abstract
The objective of this study was to evaluate the protective role of purple grape juice against oxidative damage induced by hyperhomocysteinemia in rats. For this purpose, rats were divided into 4 groups: Rats in the control group (n=8) received tap water with gavage and intraperitoneal (i.p.) saline solution (lml/kg/day) for 30 days. Rats in the purple grape juice group (PGJ) (n=8) received purple grape juice (10ml/kg/day) with gavage and saline solution (lml/kg/day). Rats in the homocysteine group (HCY) (n=8) received i.p. homocysteine (lml/kg/day) and tap water (lOml/kg/day) with gavage. Rats in the purple grape juice+homocysteine group (PGJ+HCY) (n=8) received i.p. homocysteine (lml/kg/day) and grape juice (lOml/kg/day) with gavage. During the study, plasma malondialdehyde (MDA), plasma carbonyl were assessed as indicators of oxidative stress; erythrocyte reduced glutathione (GSH), plasma sulfhydryl (SH) levels and erythrocyte catalase (CAT) and erythrocyte superoxide dismutase (SOD) activities were assessed as indicators of antioxidant capacity. Plasma MDA levels were significantly higher in the homocysteine group compared to control group, but they were significantly lower in the PGJ group (p<0.05). Plasma carbonyl levels were significantly higher in the HCY group compared to the control group (p<0.05). Plasma -SH levels were significantly lower in the HCY group compared to the control group and significantly higher in the PGJ+HCY group compared to the HCY group (p<0.05, p<0.05 respectively). Erythrocyte GSH levels were significantly higher in the PGJ group compared to the control group and they were significantly higher in the PGJ+HCY group compared to the HCY group (p<0.05, p<0.01 respectively). Erythrocyte CAT activities were significantly higher in the PGJ group compared to the control group and they were significantly higher in the PGJ+HCY group compared to the HCY group (p<0.05, p<0.01 respectively). Erythrocyte SOD activities were significantly lower in the HCY group compared to the control group (p<0.05). In conclusion, with this study, it was shown that purple grape juice had a protective role against the oxidative stress induced by homocysteine, as it decreased MDA levels through its increasing effect on the plasma-SH, erythrocyte GSH levels and erythrocyte CAT activities.
Keywords: Homocysteine; Oxidative stress; Antioxidant; Flavonoids; Purple grape juice
Abbreviations : HCY: Homocysteine Group; PGJ: Purple Grape Juice; GSH: Glutathione; SOD: Superoxide Dismutase; CAT: Catalase; SH: Sulfhydryl; HCY: Homocysteine; MDA: Malondialdehyde; PCO: Protein Carbonyl; MTHFR: Methyltetrahydrofolate Reductase; BHMT: Betaine-Homocysteine Methyl-Transferase; CBS: Cystathionine Beta-Synthase; ROS: Reactive Oxygen Species.
Introduction
Homocysteine (HCY) is one of the sulfur-containing essential amino acids and it is produced as a by-product during the methionine metabolism. Although methionine can be synthesized from aspartate in bacteria and plants, it is an essential amino acid for humans since adequate homocysteine and methyl groups cannot be supplied [l]. Both homocysteine and methionine are precursors of each other and the destruction of one of them builds the synthesis phase of the other [2]. 50% of the normal levels of intracellular homocysteine are converted again to methionine after the addition of a methyl group through remethylation [3]. Gradually increasing data shows that high plasma or serum levels of homocysteine is correlated with the atherosclerotic vascular disorders as a risk factor [4].
HCY is easily oxidized through autoxidation and free radicals emerge, which derive from oxygen and increase the lipid peroxidation during the oxidation of the sulfhydryl groups [5,6]. Free radicals are molecules with unpaired electron emerging through the biochemical redox reactions during the cell metabolism [7,8]. They play an important role in the process of several disorders [9,l0]. Under physiological conditions, the adverse effects of the free radicals are neutralized by the cellular defense system [ll]. Fruits and especially compounds prepared from purple grape (vitis vinifera) were used in the traditional medicine for centuries [l2]. These substances, which are plenty in purple grape, are phenolic compounds, flavonoids, anthocyanins and proanthocyanidins [l3]. Flavonoids are commonly found in the human diet and have multiple biological and pharmacological functions [14,15]. Flavonoids are the major scavengers of free radicals [16,17].
The objective of this study was to investigate the protective effect of purple grape juice against oxidative damage induced by high homocysteine levels. For this purpose, the plasma malondialdehyde (MDA) levels were evaluated as an indicator of the lipid peroxidation and plasma protein carbonyl (PCO) levels as an indicator of protein oxidation. Moreover, erythrocyte reduced glutathione (GSH) and plasma sulfhydryl (-SH) levels were determined as an indicator of the non-enzymatic antioxidant capacity and erythrocyte superoxide dismutase (SOD) and erythrocyte catalase (CAT) enzyme activities were determined as an indicator of the enzymatic antioxidant capacity.
Materials and Methods
In this study, we used male Sprague-Dawley rats with an average weight of 200-300g. In order to let them adapt to the environment, they were enrolled into study after one week of their transfer to Application and Research Center for Experimental Animals in the Afyon Kocatepe University. They were housed in special cages designed for rats under suitable temperature and ideal 12-hour lighting conditions. They were fed with standard rat food and tap water. During the study, which lasted 30 days, standard rat food and water were provided every day at 17:00. They were allowed to eat and drink as much as they wanted [18-20].
Rats were distributed in the following four groups:
a. Control group (n=8): This group received tap water with gavage and intraperitoneal (i.p.) saline solution (1ml/ kg/day) for 30 days.
b. Purple grape juice group (PGJ) (n=8): This group received daily 10ml/kg purple grape juice (100% grape juice) once a day with gavage and 1ml/kg intraperitoneal saline solution.
c. Homocysteine group (HCY) (n=8): This group received daily 1ml/kg homocysteine (i.p.) and 10m/kg tap water with gavage.
d. Purple grape juice + homocysteine (PGJ+HCY) group (n=8): This group received daily 1ml/kg intraperitoneal homocysteine and 10ml/kg PGJ with gavage.
After 24 hours of the last doses, an anesthetic solution containing 0.4ml ketamine and 0.6ml xylazine was administered in doses of 1.6ml/kg intraperitoneally to all rats. After the establishment of deep anesthesia, blood samples were drawn by cardiac puncture into heparinized tubes. Blood samples were centrifuged at 2500rpm and 4 °C for 10 minutes and plasma and blood cells were separated. The cells were washed 3 times with saline solution. Samples were stored at -20 °C until the time of analysis. Erythrocytes were ruptured with repeated freeze- dissolve process and a diluted stock solution was prepared after the samples were diluted with distilled cold saline solution in 1/1 ratio. Shimadzu UV-1601 spectrophotometer was used for all spectrophotometric measurements. All chemical substances used were of analytical purity.
Hemoglobin assessment was done with the Drapkin's method. Plasma MDA levels were measured according to the method described by Ohkawa et al. [21]. Plasma protein carbonyl groups were evaluated with the modified spectrophotometric method of Levine et al. [22]. Erythrocyte GSH levels were determined with the spectrophotometric Beutler method [23]. Plasma -SH groups were assessed with the spectrophotometric method of Koster et al. [24]. The spectrophotometric method of Winterbourn et al. [25] was used for the measurement of the erythrocyte SOD activities [25]. Erythrocyte CAT activities were determined with the Beutler spectrophotometric method [26]. The total phenol quantity in the purple grape juice was measured with the colorimetric method of Folin-Ciocalteu [27].
Statistical analysis
The results were analyzed with the Mann-Whitney U test using the package software SPPS v10.0 (Chicago, IL, USA). The results were given as mean ± SEM. The accepted limit of significance was p<0.05.
Results
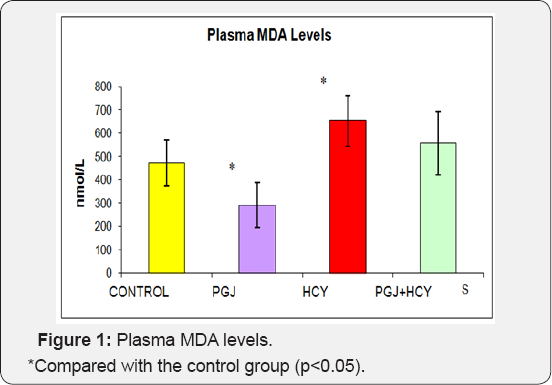


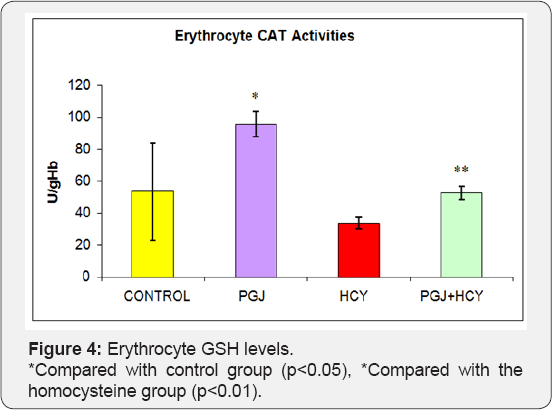
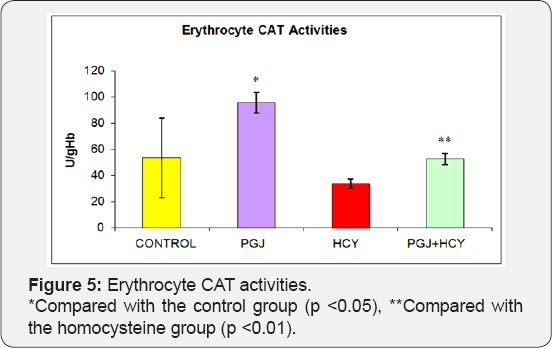
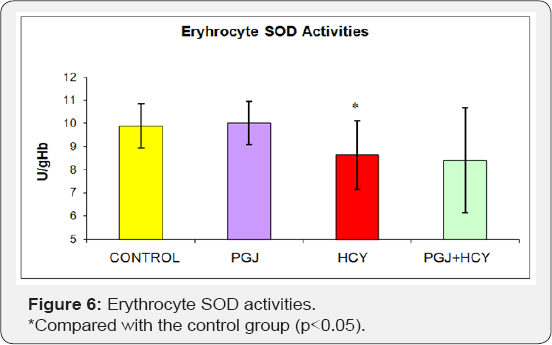
Plasma MDA levels were significantly higher in the homocysteine group and significantly lower in the PGJ group compared to the control group (Figure l) (p<0.05). Plasma carbonyl levels were significantly higher in the homocysteine group compared to the control group (p<0.05) (Figure 2). As shown in Figure 3, plasma -SH levels were significantly lower in the homocysteine group compared to the control group, but significantly higher in the PGJ+HCY group compared to the homocysteine group (p<0.05, p<0.05 respectively). Erythrocyte GSH levels were significantly higher in the PGJ group compared to the control group and significantly higher in the PGJ+HCY group compared to the homocysteine group (p<0.05, p<0.0l respectively) (Figure 4). Erythrocyte CAT activities were significantly higher in the PGJ group compared to the control group and significantly higher in the PGJ+HCY group compared to the homocysteine group (p<0.05, p<0.0l respectively) (Figure 5). Erythrocyte SOD activities were significantly higher in the homocysteine group compared to the control group (p<0.05) (Figure 6). Total phenolic substance quantity assessed with the Folin-Ciocalteu method in the PGJ was 720mg/ml.
Discussion
Homocysteine is a sulfur-containing amino acid synthesized in the human organism and used in the synthesis of several substances. It is produced as a by-product of the methionine metabolism. The intracellular homocysteine metabolism depends on the availability of methionine, re-methylation of homocysteine to methionine and cysteine transsulfuration. Several vitamins play role as cofactor and substrate in the homocysteine-methionine metabolism. They serve as regulators in the metabolic pathways, which were catalyzed by folic acid, cyanocobalamin, methyltetrahydrofolate reductase (MTHFR) and methionine synthetase enzymes [28]. The synthesis and metabolism of homocysteine occur mostly in liver. Methionine is metabolized predominantly in the liver. The synthesis of the enzymes betaine-homocysteine methyl-transferase (BHMT) and cystathionine beta-synthase (CBS) takes place in liver. Therefore, homocysteine metabolism might be affected in cases of liver injury [29]. There are gradually increasing data that confirm the correlation of high homocysteine levels with clinical atherosclerotic vascular disorders as a risk factor [28]. There is a general consensus regarding the effect of the increased plasma homocysteine level on the development of cardiovascular diseases [30].
Free radicals are produced during the oxidation process in the organism. Atoms and molecules with unpaired electron trigger certain reaction chains, which cause damage in cells. Although the reactive oxygen species (ROS) play a role as the intermediator in low doses, in high doses they might cause the oxidative stress and damage in the cells [3l]. These by-products of metabolism, which have oxidant and mutagenic effects, destroy DNA, protein and other macromolecules and they initiate even chronic disorders by causing cell death [32].
Purple grape (vitis vinifera) has a powerful antioxidant effect due to the high phenolic compounds in its seeds and flavonoids originating from these phenolic compounds. Purple grape, thanks to its antioxidant properties, possess several pharmacological features and provides protection against oxidative stress [33].
In their study, Kazem et al. [34] determined a vasorelaxation effect of the grape leaf extracts on the isolated rat aortas. Phenolic substances behave as antioxidant compounds due to their partial reactivity depending on the OH-groups in the aromatic rings. The major metabolism of the antioxidant activity is based on scavenge of radicals as a result of hydrogen atom release [33]. It was determined that high homocysteine concentrations caused an increase in free oxygen radicals and lipid peroxidation [35]. Homocysteine is easily oxidized as a result of autoxidation and free oxygen radicals emerge during the oxidation of the sulfhydryl groups, which initiate lipid peroxidation [36].
Tan et al. [37] in their study conducted with Asian adults concluded that hyperhomocysteinemia was an independent risk factor for the ischemic stroke and it increased the risk of stroke in the large artery strokes due to its proatherogenic effect. In the study conducted by Baydas et al. [36], chronic methionine administration increased the plasma homocysteine levels in rats and lipid peroxidation was stimulated in the groups receiving homocysteine. Boushey et al. [38] demonstrated that an increase of 5μmol/L in homocysteine level increased the coronary artery disorder risk 1.7 times, cerebrovascular disorder risk 1.5 times and peripheral artery disorder risk 6.8 times [39-41].
One of the late-stage products of lipid peroxidation is MDA. MDA is synthesized through the peroxidation of fatty acids containing three or more double bonds. MDA affects the ion exchange in the cell membranes and leads the substances in the membrane to build cross-bonds and causes adverse results such as the modification of the ion permeability and enzyme activity. Because of these features and as MDA might react with nitrogenous bases of DNA, it is genotoxic and carcinogenic for the mutagenic cell cultures [42].
Yuce et al. [43] investigated the plasma and tissue MDA values of rats receiving homocysteine and reported that the MDA values increased significantly in the rats of the homocysteine group compared to the control group. They also stated that this result emerged because the homocysteine decreased glutathione peroxidase (GSH-Px) activity which prevented the production of the lipid peroxidation.
Cavalca et al. [44] conducted a study on patients with coronary artery disorder and found out that the plasma homocysteine concentrations of the patients with cardiovascular disease were significantly higher than the patients in the control group. They used MDA as the parameter of the free radical production in the cardiovascular disease patients and evaluated the probable role of homocysteine. They determined that the plasma MDA concentrations of these patients were twice higher than the patients in the control group. They also reported that high MDA concentrations pointed at the increased lipid peroxidation and to the potential progression of the oxidative stress regarding all patients. In our study, the plasma MDA levels were significantly higher in the rats of HCY group (p<0.05). Furthermore, the plasma MDA levels in the rats of the PGJ group were significantly lower than the control group rats (p<0.05).
Although the high MDA values in HCY group indicated that homocysteine induced the oxidative stress, the low MDA values in PGJ+HCY group might be related to the protective effect of the phenolic substances in the PGJ composition against oxidative stress. Duarte et al. [44] observed that after the administration of quercetin, which is a flavonoid, for 5 weeks, the blood pressure decreased, glutathione peroxidase activity increased and plasma and hepatic MDA levels decreased in the spontaneously hypertensive rats. In this genetic model of hypertension, quercetin showed antihypertensive and antioxidant characteristics. Quercetin is one of the most important components of purple grape juice
Sreemantula et al. [45] showed the antioxidant activity of the purple grape extract with the Hydroxyl Radical Assay Method. They determined that the antioxidant activity of the vitis vinifera extract was 8 times higher than ascorbic acid. James et al. [1] reported that short-term administration of purple grape juice to the patients with coronary artery diseases improved the endothelial functions and decreased the LDL oxidation sensitivity. They also suggested that a potential mechanism provided by the flavonoids in the purple grape was regressed the endothelial vasodilation and prevented LDL oxidation. Intraperitoneal homocysteine administration caused intimal hyperplasia and cell proliferation in the carotid arteries. It is known that there is a positive correlation between the total plasma homocysteine concentrations and the wall thickness of the carotid arteries [46]. Serafinowicz et al. [47] reported that the products of the lipid peroxidation and the carotid intimal media thickness were increased in the rats with hyperhomocysteinemia. In their study, Hollman et al. [48] found out that feeding with quercetin resulted in the inhibition of the lipid peroxidation and the antioxidant capacity was significantly increased in the rats fed a diet containing 0.2% quercetin compared to the control group.
Free radical scavenging and inhibition of lipid peroxidation are important functions of phenolic acids, anthocyanins and other flavonoids. The comparable antioxidant activities of red and purple grapes are well known. All of these fruit extracts have high activity against the superoxide radicals, which are released through various chemical pathways. Another feature of these extracts is the inhibitory effect on the xanthine oxidase enzyme, which initiates the production of the free radicals within the cell [49]. The primary modification reactions cause the synthesis of the protein carbonyl derivatives. These reactions consist of the oxidative modification of the a-carbon atoms or side chains of the amino acids and following this modification cleavage of the reactive oxygen-mediated peptide bonds. As a result of the interaction of the reactive oxygen derivatives with the proteins, protein carbonyl products emerge due to the oxidative damage, which occurs on the peptide backbone of several amino acids like histidine, proline, arginine and lysine or proteins. The measurement of the protein carbonyl levels is a sensitive and widely used method for the determination of the protein oxidation [50,5l].
ROS causes oxidative damage in protein, carbohydrate and lipid molecules. Lipid peroxidation and carbohydrate oxidation, which occur with the effect of ROS, invoke modifications in the amino acid content of the proteins and an increase in the plasma protein carbonyl composition. Horakova et al. [52] investigated the antioxidant activity of standardized flavonoid (EGb 761, pycnogenol) extract, trolox and stobadine and produced oxidative damage from Fe+3/H2O2/AA and HOCl oxidant systems on the sarcoplasmic reticulum in the skeletal muscles of rabbits. They found out that the protein carbonyl levels of the oxidant + antioxidant groups were significantly lower compared to the protein carbonyl levels of the oxidant groups.
In our study, we obtained comparable results to the study mentioned above regarding the plasma carbonyl levels (PCO). The plasma carbonyl levels in the rats of the homocysteine group increased significantly compared to the control group (p<0.05). Although we observed a decrease in the plasma carbonyl levels in PGJ and PGJ+HCY groups compared to the homocysteine group, the difference was not significant. The decreasing effect of PGJ on the carbonyl levels might depend on its protective effect against oxidative stress and on its scavenging activity on free radicals, which arise due to the high homocysteine values. Molina et al. [53] reported that plasma protein carbonyl levels were increased in the rats, in which oxidative stress was induced by chronic alcohol administration compared to the control group. They also determined that protein carbonyl levels were decreased in the rats, which received both quercetin and ethanol.
The thiol (-SH) group of the cysteine is quite sensitive to oxidative damage and thyl (-S) radical, which emerges with different mechanisms from the -SH groups, lead to the production of the disulfide bonds in the proteins. The conversion of the -SH groups to the oxidized derivatives such as disulfides and oxyacids is a sign of the protein oxidation originating from the radicals and can be observed at the early stage. Another oxidation form of the thiol groups occurs after the binding of the 4-hydroxynonenal to the -SH groups of the proteins with a thioether bond as a result of the Michael reaction. Michael reaction is a reversible reaction [22,54].
In our study, the plasma -SH levels of the homocysteine groups, which were exposed to the oxidative stress with the homocysteine administration, were significantly lower than in the control group (p<0.05). However, the plasma -SH levels in the PGJ+HCY group were significantly higher compared to the HCY group (p<0.05). GSH is the most important antioxidant in the organism with no enzymatic properties. It reacts with the reactive oxygen products and thus protects the cells against the oxidative stress. Moreover, it maintains the reduction of the -SH groups in the proteins and in this way protects these groups against oxidation, which prevents also the inactivation of the proteins and enzymes [35]. Homocysteine induces the intracellular inactivation of the glutathione antioxidant defense system and reduces probably the GSH synthesis.
Yüce et al. [43] conducted a study regarding the changes in the oxidant-antioxidant system and coronary arteries caused by homocysteine and compared the male rats among themselves. This comparison displayed that the GSH levels in the rats in the homocysteine group were lower than the rats in the control group. In this study, the investigators also observed that early-stage atherosclerosis, which was characterized by the detachment of the endothelial cells in the cardiac vessels, developed in the male rats of the homocysteine group compared to the control group. In our study, the erythrocyte GSH values in the homocysteine group were significantly lower compared to the control group (p<0.05). Erythrocyte GSH values were significantly higher in the purple grape group compared to the control group (p<0.05). Erythrocyte GSH values in PGJ+HCY group was significantly higher compared to the homocysteine group (p<0.01).
Rajdl et al. [55] suggested in their study that consumption of an appropriate amount of white wine had cardioprotective and antioxidant effects. In this study, considering the increase in the intracellular GSH levels, they determined that white wine provided a reasonable reduction against reactive oxygen molecules. Morrison et al. [56] reported that along with the high plasma total homocysteine levels, the GSH levels were lower in patients with coronary artery disease compared to the control group. Hultberg et al. [57] determined significantly decreased GSH concentrations in the human cell walls exposed to 250 mmol/L homocysteine. Upchurch et al. [58] found out that GSH activity was significantly decreased in the bovine aortic endothelial cells, which were exposed to 50-200mmol/L homocysteine [59].
Fiorani et al. [60] reported that quercetin protected glutathione against the glutathione consumption, which was induced by the dehydroascorbic acid. Homocysteine prevents the binding of SOD to the endothelial cell membrane in arteries as it destructs the heparan sulfate proteoglycans, which enable the binding of SOD to the endothelial cell surface. High levels of homocysteine decrease the SOD amount, which is released to the surrounding by the fibroblasts. As a result, homocysteine decreases the defensive ability of the endothelial cells against free radicals particularly against superoxide radicals [40]. Yüce et al. [47] conducted a study on rats, in which oxidative stress was induced by homocysteine administration and found out that plasma SOD and CAT activities of the rats in homocysteine group were lower compared to the rats in the control group.
The inhibition of the SOD activity is one of the oxidative stress damage mechanisms induced by homocysteine. SOD has three isoenzymes. In their oxidative stress study, which was induced by homocysteine in the vascular smooth muscle cells of rats, Chang et al. [61] demonstrated that homocysteine caused a dose- dependent inhibition of the mitochondrial Mn-SOD activity. In a study, it was reported that homocysteine produced homocystine, mixed disulfides and homocyteine after the rapid autoxidation in the plasma. During the autoxidation of homocysteine, potent reactive oxygen molecules like superoxide and hydrogen peroxide emerge. Particularly hydrogen peroxide was blamed for the vascular toxicity related to hyperhomocysteinemia. It was demonstrated that the emergence of hydroxyl radicals in relation to superoxide anion radicals initiates the lipid peroxidation because of an effect at the level of endothelial plasma membrane and intracellular lipoprotein particles.
In our study, erythrocyte SOD activities were significantly lower in the homocysteine group compared to the control group (p<0.05). There was no significant difference between other groups. These results suggest that SOD enzyme in homocysteine- treated rats is one of the indicators of oxidative damage. Reshma et al. [62] investigated the effects of ocimum flavonoids on the erythrocyte antioxidants in oral cancer and found out - similar to our study - that SOD activity was higher in the flavonoid group compared to the control group. Kahraman et al. [32] investigated the antioxidative and antihistaminic activity of quercetin in the gastric lesions induced by alcohol and determined that SOD activities decreased significantly with the administration of alcohol and increased significantly with the administration of quercetin. The distribution of SOD enzyme in the living beings should be evaluated with CAT because the product, which emerged as a result of the reaction catalyzed by SOD, was one of the toxic compounds of oxygen and its accumulation was prevented by CAT. CAT is a hemoprotein, which contains four hem groups and is found with different concentrations in general cell types. It is mostly encountered in the peroxisomes. With hydrogen peroxide, CAT has a reductive activity on small molecules such as methyl and ethyl hydroperoxides and it has no effect on large molecule lipid hydroperoxides. It is found in large amounts in blood, bone marrow, mucous membranes, liver and kidneys [23].
Lin et al. [63] conducted a study on rabbits in order to investigate the protective activity of grape suspension on the oxidative effects of bilateral ischemia and reported that the protective activity of the grape suspension accompanied the regulation activity of SOD and CAT. They also determined that the SOD and CAT activities were increased in the groups received grape. In our study, erythrocyte CAT activities were significantly higher in the quercetin and purple grape juice groups compared to the control group (p<0.05). The CAT activity in the homocysteine group was significantly lower compared to the control group (p<0.05). Comparison of the erythrocyte CAT activities of the homocysteine + quercetin group and homocysteine group revealed that CAT activity values were significantly higher in the homocysteine + quercetin group (p<0.05). Similarly, the comparison of the homocysteine + purple grape juice group and homocysteine group displayed that the erythrocyte CAT activities were higher in the homocysteine + purple grape juice group (p<0.05). The reason for this finding might be the antioxidant effect of the flavonoids found in the grape juice against the free radicals induced by homocysteine.
In conclusion, the increases in lipid peroxidation and protein oxidation and the decreases in the plasma-SH and erythrocyte SOD levels caused by hyperhomocysteinemia were an indicator of the oxidative stress induced by ROS. We concluded that in PGJ+HCY group, the decrease of plasma MDA levels but the increase of erythrocyte GSH and CAT values showed the suppressive effect of purple grape juice on the peroxidation products and on the antioxidant enzyme activity. We believe that this study conducted with rats should be confirmed with human studies.
References
- James DF, John JM (2000) Homocysteine. Int J Biochem Cell Biol 32(4): 385-389.
- Brattström L, Lindgren A, Israelsson B, Andersson A, Hultberg B (1994) Homocystein and cystein: Determinants of plasma levels in middle- aged and elderly subjects. J Intern Med 263(6): 633-641.
- Koehler KM, Romero LJ, Stauber PM, Pareo-Tubbeh SL, Liang HC, et al. (1996) Vitamin suplementation and other variables affecting serum homocysteine and methylmalonic acid concentrations in elderly men and women. J Am Coll Nutr 15(4): 364-376.
- Malinow R, Andrew G (1999) Homocysteine, Diet and Cardiovascular Disease. Circulation 99: 178-182.
- Jara-Prado A, Ortega-Vazquez, Martinez-Ruano L, Rios C, Santamaria A (2003) Homocysteine-induced brain lipid peroxidation: effects of NMDA receptor blockade, antioxidant treatment, and nitric oxide synthase inhibition. Neurotox Res 5(4): 237- 243.
- Dayal S, Arning E, Bottiglieri T, Liu ML, Arning E, et al. (2004) Cerebral vascular dysfunction mediated by superoxide in hyperhomocysteinemic mice. Stroke 35(8): 1957-1962.
- Yamamato M, Hara H, Adachi T (2000) Effects of homocysteine on the binding of extracellular-superoxide dismutase to the endothelial cell surface. FEBBS Lett 486(2): 159-162.
- Freeman BA, Crapo ID (1982) Biology of disease-free radicals and tissue injury. Lab Invest 47(5): 412-426.
- Lunec J, Blake DR (1985) Copper, iron, free radicals and arthritis. Br J Rheumatol 24(2): 123-125.
- AkkuŞ I, Saglam NI, Caglayan O, Vural H, Kalak S, et al. (1996) Investigation of erythrocyte membrane lipid peroxidation and antioxidant defense systems of patients with coronary artery disease (CAD) documented by angiography. Clin Chim Acta 244(2): 173-180.
- Potapovich AI, Kostyuk VA, Kostyuk TV, de Luca C, Korkina LG (2013) Effects of pre- and post-treatment with plant polyphenols on human keratinocyte responses to solar UV. Inflamm Res 62(8): 773-780.
- Koyama K, Kamigakiuchi H, Iwashita K, Mochioka R, Goto-Yamamoto N (2017) Polyphenolic diversity and characterization in the red-purple berries of East Asian wild Vitisspecies. Phytochemistry 134: 78-86.
- Toscano LT, Tavares RL, Toscano LT, Silva CS, Almeida AE, et al. (2015) Potential ergogenic activity of grape juice in runners. Appl Physiol Nutr Metab 40(9): 899-906.
- Morand C, Crespy V, Manach C, Besson C, Demigné C, et al. (1998) Plasma metabolites of quercetin and their antioxidant properties. Am J Physiol 275: 212-219.
- Manach C, Morand C, Crespy V, Demigné C, Texier O, et al. (1998) Quercetin is recovered in human plasma as conjugated derivatives which retain antioxidant properties. Febbs Lett 426(3): 331-336.
- Gebhardt R (2003) Variable influence of kaempferol and myricetin on in vitro hepatocellular cholesterol biosyn thesis. Planta Med 69(12): 1071-1074.
- Li BH, Tian WX (2004) Inhibitory effects of flavonoids on animal fatty acid synthase. J Bioche 135(1): 85-91.
- Knekt P, Jarvinen R, Reunanen A, Maatela J (1996) Flavonoid intake and coronary mortality in Finland: a cohort study. Br Med J 312(7029): 478-481.
- Kuo SM (1997) Dietary flavonoid and cancer prevention: Evisence and potential mechanism. Crit Rev Oncog 8(1): 47-69.
- Hollman PCH, Gaag MVD, Mengelers MJB, van Trijp JMP, de Vries JHM, et al. (1996) Absorption and disposition kinetics of the dietary antioxidant quercetin in man. Free Radic Biol Med 21(5): 703-707.
- Okhawa H, Ohishi N, Yagi K (1979) Assay for Lipid Peroxidase in Animal Tissues By Thiobarbituric Acid Reaction. Anal Biochem 95(2): 351-358.
- Levine RL, Garland D, Oliver CN (1990) Determination of Carbonyl Content in Oxidatively Modified Proteins. Method Enzymol 186: 464478.
- Beutler E, Robson MJ, Buttenvieser E (1957) The Glutathione Instability of Drug Sensitivity Red Cells. J Lab Med 49(1): 84-95.
- Koster JF, Biemond P, Swaak JG (1986) Intracellular and Extracellular Sulphydryl Levels in Romatoid Arthritis. Annals of the Rheumatic Disease 45(1): 44-46.
- Wintenbourn CC, Hawkin RE, Brian M, Corell RW (1975) The estimation of red cell superoxide dismutase activity. J Lab Clin Med 85(2): 337-341.
- Beutler E (1973) Red Cell Metabolism, A Manuel Of Biochemical Methods. Grune & Stratton Inc, New York, USA.
- Gamez-Meza N, Noriega-Rodriguez J, Medina-Juarez L, Ortega-Garcia J, Cazarez-Casanova R, et al. (1999) Antioxidant activity in soybean oil of extracts from Thompson grape bagasse. JAOCS 76: 1445-1447.
- £elik N, Vurmaz A, Kahraman A (2017) Protective effect of quercetin on homocysteine-induced oxidative stress. Nutrition 33: 291-296.
- McCully KS (1969) Vascular pathology of homocysteinemia. Am J Pathol 56(1): 111-128.
- De la Vega MJ, Santolaria F, Gonzalez-Reimers E, Alemán MR, Milena A, et al. (2001) High prevalence of hyperhomocysteinemia in chronic alcolism: the importance of the thermolabile form of the enzyme methylene tetrahydrofolate reductase MTHFR. Alchol 25(2): 59-67.
- Martin KR, Barret JC (2002) Reactive oksijen species as double-edged swards in cellular processes: low-dose cell signaling versus high-dose toxicity. Hum Exp Toxicol 21(2): 71-75.
- Kahraman A, Inal ME (2002) Protective effects of quercetin on ultraviolet A light-induced oxidative stress in the blood of rat. J Appl Toxicol 22(5): 303-309.
- Yu H, Zhao X, Xu G, Wang SE (2003) Effect of grape seed extracts on blood lipids in rabbits model with hyperlipidemia. Wei Sheng Yan Jiu 31(2): 114-116.
- Kazem M, Naseri G, Heidari A (2006) Bronchodilatory Activity of Vitis vinifera Leaf Hydroalcoholic Extract in Rat Iranian. Biomedical Journal 10(2): 79-83.
- Qasim M, Bukhari SA, Ghani MJ, Masoud MS, Huma T, et al. (2016) Relationship of oxidative stress with elevated level of DNA damage and homocysteine in cardiovascular disease patients. Pak J Pharm Sci 29(6): 2297-2302.
- Baydas G, Özer M, Yajar A, Koz ST, Tuzcu M (2006) Melatonin Prevents Oxidative Stress and Inhibits Reactive Gliosis Induced by Hyperhomocysteinemia in Rats. Biochemistry 71: 91-95.
- Tan NC, Venketasubramanian N (2002) Hyperhomocysteinemia and risk of ischemic stroke amoung Asian adults. Stroke 33(8): 1956-1962.
- Boushey CJ, Beresford SA, Omenn GS, Motulsky AG (1995) A quantitative assessment of plasma homocysteine as a risk factor for vascular disease. Probable benefits of increasing folic acid intakes. J Am Med Assoc 274(13): 1049-1057.
- Yamamoto Y, Oue E (2006) Antihypertensive Effect of Quercetin in Rats Fed with a High-Fat High-Sucrose Diet. Biosci Biotechnol Biochem 70(4): 933-939.
- Juzwiak S, Wojcicki J, Mokrzycki K, Marchlewicz M, Biaiecka M, et al. (2005) Effect of quercetin on experimental hyperlipidemia and atherosclerosis in rabbits. Pharmacological Reports 57(5): 604-609.
- Cao L, Tan C, Meng F, Liu P, Reece EA, et al. (2016) Amelioration of intracellular stress and reduction of neural tube defects in embryos of diabetic mice by phytochemical quercetin. Sci Rep 6: 21491.
- Pan X, Zhu L, Lu H, Wang D, Lu Q, et al. (2015) Melatonin Attenuates Oxidative Damage Induced by Acrylamide In vitro and In vivo. Oxid Med Cell Longev 2015: 703709.
- Cavalca V, Cighetti G, Bamonti F, Loaldi A, Bortone L, et al. (2001) Oxidative Stress and Homocysteine in Coronary Artery Disease. Clinical Chemistry 47(5): 887-892.
- Duerte J, Perez-Palencia R, Vargas F, Ocete MA, Pérez-Vizcaino F, et al. (2001) Antihypertensive effects of the flavonoid quercetin in spontaneously hypertensive rats. Br J Pharmacol 133: 117-24.
- Sreemantula S, Nammi S, Kolanukonda R, Koppula S, Boini KM, et al. (2005) Adaptogenic and nootropic activities of aqueous extract of Vitis vinifera (grape seed): an experimental study in rat BMC Complementary and Alternative Medicine 5: 1.
- Stein JH, Keevil JG, Wiebe DA, Aeschlimann S, Folts JD (1999) Purple Grape Juice Improves Endothelial Function and Reduces the Susceptibility of LDL Cholesterol to Oxidation in Patients With Coronary Artery Disease Circulation 100(10): 1050-1055.
- Serafinowice A, Kukula K, Cieciura T, Shaibani B, Baczkowska T, et al. (2000) Homocysteine and lipid peroxidation products: Impotant atherosclerosis risk factors in renal all graft recipients. Transplant Proc 32(6): 1367-1368.
- Hollman PCH, de Vries JHM, van Leeuwen SD, Mengelers MJB, Katan MB (1995) Absorption of dietary quercetin glycosides and quercetin in healthy ileostomy volunteers. Am J Clin Nutr 62(6): 1276-1282.
- Cai X, Bao L, Ren J, Li Y, Zhang Z (2016) Grape seed procyanidin B2 protects podocytes from high glucose-induced mitochondrial dysfunction and apoptosis via the AMPK-SIRT1-PGC-1a axis in vitro. Food Funct 7(2): 805-815.
- Erden Inal M, Kahraman A (2000) The protective effect of flavonol quercetin against ultraviolet a induced oxidative stress in rats. Toxicology 154(1-3): 21-29.
- Kahraman A, Erkasap N, Köken T, Serteser M, Aktepe F, et al. (2003) The antioxidative and antihistaminic properties of quercetin in ethanol- induced gastric lesions. Toxicology 183(1-3): 133-142.
- Horakova L, Strosova M, Skuciova M (2005) Protective effects of some antioxidant on oxidatively modified sarcoplazmic reticulum from rabbit skeletal muscle Biologia. Bratislava 17: 131-134.
- Berlett BS, Stadtman ER (1997) Protein oxidation in aging, disease, and oxidative stress. J Biol Chem 272: 20313-20316.
- Mosharow E, Cranford MR, Banerjee R (2000) The quantatively important relationship between homocysteine metabolism and glutathione syntesis by the transsulphuration pathway and its regulation by redox changes. Biochemistry 39(42): 13005-13011.
- Rajdl D, Racek J, Trefil L, Siala K (2007) Effect of White Wine Consumption on Oxidative Stress Markers and Homocysteine Levels. Physiol Res 56(2): 203-212.
- Morrison JA, Jacobsen DW, Sprecher DL, Robinson K, Khoury P, et al. (1999) Serum glutathione in adolescent males predicts parenteral coronary heart disease. Circulation 100(22): 2244-2247.
- Hultberg B, Andersson A, Isaksson A (2000) Hypomethylation as a cause of homocysteine-induced cell damage in human cell lines. Toxicology 147(2): 69-75.
- Upchurch GR, Welch GN, Fabian AJ, Freedman JE, Johnson JL, et al. (1997) Homocysteine decreases bioavailable nitric oxide by a mechanism involving glutathion peroxidase. J Biol Chem 272: 17012- 17027.
- Zhang YM (2005) Protective effect of quercetin on aroclor 1254-induced oxidative damage in cultured chicken spermatogonial cells. Toxicol Sci 88(2): 545-550.
- Fiorani M, De Sanctis R, Menghinello P, Cucchiarini L, Cellini B, et al. (2001) Quercetin prevents glutathione depletion induced by dehydroascorbic acid in rabbit red blood cells. Free Radic Res 34(6): 639-648.
- Chang L, Xu J, Zhao J, Pang Y, Tang C, et al. (2004) Taurine antagonized oxidative stres injury induced by homocysteine in rat vascular smooth muscle cells. Acta Pharmacol Sin 25(3): 341-346.
- Reshma K, Ashalatha VR, Dinesh M, Vasudevan D (2005) Effect of ocimum flavonoids as a radioprotector on the Erythrocyte antioxidants in oral cancer Indian Journal of Clinical Biochemistry 20(1): 160-164.
- Lin AD, Mannikarottu A, Kogan BA, Whitbeck C, Leggett RE, et al. (2007) Effect of bilateral in vivo ischemia/reperfusion on the activities of superoxide dismutase and catalase: response to a standardized grape suspension. Mol Cell Biochem 296(1-2): 11-16.






























