99mTc Labeled L-Tryptophan Conjugated Macrocyclic Chelating Agent: Synthesis, Characterization and In Vitro & In Vivo Biological Studies
Ranjeet Kumar1, Ankur Kaul2, Anil K Mishra2 and Bachcha Singh1*
1Department of Chemistry, Banaras Hindu University, India
2Division of Cyclotron and Radiopharmaceutical Sciences, Institute of Nuclear Medicine and Allied Sciences, India
Submission: January 14, 2018; Published: March 28, 2018
*Corresponding author: Bachcha Singh, Department of Chemistry, Centre of Advanced Study, Institute of Science, Banaras Hindu University, India, Email: Indiabsinghbhu@rediifmail.com
How to cite this article: Ranjeet K, Ankur K, Anil K M, Bachcha S. 99mTc Labeled L-Tryptophan Conjugated Macrocyclic Chelating Agent: Synthesis, Characterization and In Vitro & In Vivo Biological Studies. Glob J Pharmaceu Sci. 2018; 5(3): 555661. DOI: 10.19080/GJPPS.2018.05.5556661
Abstract
Radiolabeled amino acids and their derivatives are an important class of imaging agents for PET and SPECT that target the increased levels of amino acid transport by many tumour cells. The novel amino acid (L-tryptophan methyl ester) based legend; 16Trp4A was successfully synthesized and well characterized through spectroscopic techniques and mass spectrometry which may be used as imaging agents. The pharmacokinetics of these compounds was analyzed by 99mTc labeled tracer methods. The labelling yield was found to be greater than 95%, as determined chromatographically by different solvent. Bio distribution studies in mice revealed significant uptake in blood, lung, liver, kidney, spleen and intestine initially, which decreases as time passes up to 24h. The maximum uptake of 99mTc -16Trp4A was found in liver 9.61+0.33, 5.95+0.29 and 3.66+0.19 %ID/g at 1,4 and 24h post injection, respectively, concluded that excretory pathway is hepatobiliary. Accumulation of activity in non-targeted organs and tissues (heart, brain, lungs, stomach, and intestine) was found<2%. The negligible activity in stomach (0.06+0.00, 0.05+0.00 and 0.25+0.01) %ID/g delineates very less availability of free 99mTcO4 confirming the stability of radiolabel complex in biological milieu.
Introduction
Target specific delivery of the radiolabeled compound to cancer cell to avoid possible uptake/damage to the surrounding normal healthy cells is an important aspect of tumour imaging and therapy. Fictionalisation of molecules with amino acid analogues has been proved a diverse and useful class of PET and SPECT tracers that records amino acid transportation and uptake exhibited by many tumour cells i.e. brain, neuroendocrine, prostate cancer [1-5].
Tumor cells show property of unlimited malignant growth and they require amino acid for proliferation thus amino acid transport generally increased malignant transportation. Increase rate for amino acid uptake is one of the earliest and most important events associated with proliferation. There are many evidences show that IDO (indoleamine 2, 3-dioxygenase) is over expressed in a variety of human tumors, including lung tumors resulting increase of abnormal tryptophan metabolism via the kynurenine pathway. L-Tryptophan is a substrate of IDO [5-8]. Intracellular IDO activity may result in the trapping of polar tryptophan (or their derivatives) metabolites or indirect accumulation via intracellular L-tryptophan depletion [9,10].
For the development of target specific amino acid based imaging system that can be utilized for imaging techniques viz. PET, SPECT and magnetic resonance imaging (MRI), the amino acids are conjugated with macrcyclic chelating agents which have ability to bind with metal ions to yield thermodynamically stable and kinetically inert metal chelates. For complexation with metal ions, macrocyclic polyamines, particularly tetraazacy cloalkanes, have gained a great deal of importance due to their ligating and biological properties [11-13]. Their strong affinity and selective binding with certain metals make them very useful in the field of radioactive diagnosis. An alternative to the conventional N-substituted macrocyclic frameworks is the functionalization of the carbon skeleton of the macro cycle which restores the reactivity of the secondary amines making them available for reaction with other coordinating groups [14].
99mTc is one of the most desirable radionuclide for imaging in diagnostic radiopharmaceuticals because of its optimal nuclear-physical properties and easy availability. The emission of gamma rays of optimal energy (140keV), a suitable halflife (6h), and availability from 99Mo-99mTc generator systems, excellent imaging properties, favourable dosimetry, and high specific activity makes 99mTc a logical choice for labelling [15]. Taking into account these features of the high stability and kinetic inertness and our interest for the development of radiolabeled complexes suitable for SPECT imaging, we have synthesized and characterized the tetraaza macrocyclic chelator 16-[methyl-3-(1H-indol-3-yl)-2 (2(amino)acetamido)] 3,6,9,12-tetraazabicyclo-[12.3.1] octadeca-1,14,16-triene-2,13- dione, 16Trp4A. Biological studies of this radiolabeled chelator were performed to explore their application as SPECT tumor imaging agent.
Experimental
Ligand synthesis and characterization
Synthesis of 16-amino-3,6,9,12-tetraazabicyclo[12.3.1] octadeca 1,14,16- triene 2,13-dione (1): Dimethyl-5- aminodiisophthalate (1.046g, 5mmol) was dissolved in 100ml of dry methanol and 1, 8-diamino-3, 6-diazaoctane (0.76ml, 5.1mmol) was diluted to 100ml by dry methanol. Both the precursors were added drop wise to three neck round bottom flask (1L) containing 450ml dry methanol with the help of dropping funnel with constant stirring under inert condition at room temperature. The reaction mixture was refluxed with constant stirring for 4 days. The solvent was evaporated under reduced pressure. The obtained white solid compound (1.04g, 71%) was washed with methanol and dried under vacuum (1). IR (KBr pellets, cm-1): 2953, 2854, 1653, 1565, 1435, 1361, 1261, 1246, 1128, 1016, 993, 894, 725, 752. 1H NMR (500MHz, CDCl3, TMS): 2.56( d, 4H, CH2 ), 3.81( s, 4H, CH2 ), 5.39 ( s, 2H, NH ), 5.73 ( s, 2H, NH2 ), 7.09 ( s, 1H, H-Ar ), 7.39 ( s, 1H, H-Ar ), 8.29 ( s, 2H, NH-CO ), 13C NMR (500 MHz, CDCl3, TMS): 41.23, 49.13, 50.18, 52.17 (CH2), 118.63, 120.40, 131.34, 146.82 (C-Ar), 166.43, 167.01 (CO). m/z (ESI) found 292.4 (M+ H+), calculated 291.17.
Synthesis of methyl-2-(2-chloro acetamido)-3-(1Hindol- 3-yl) propanoate (2): The methyl ester of L-tryptophan was dissolved in water (20mL). The chloro- acetylchloride (1.2eq.) in dichloromethane (20mL) and K2CO3 solution in water (1.2eq.) were added slowly to the stirring solution at 00C using a pressure equalized dropping funnel. The resulting reaction mixture was stirred at room temperature overnight. After completion of the reaction, the pale yellow viscous oil was washed with H2O (2x20mL), 0.1M HCl (2x20mL) and brine (2x20mL). The organic layer was dried over MgSO4, filtered and the solvent was removed under reduced pressure to yield pure chloroacetylated product (2). IR (KBr pellets, cm-1): 3366, 2953, 2854, 1633, 1555, 1435, 1361, 1261, 1246, 1128, 1016, 993, 894, 725, 752. 1HNMR (500MHz, CDCl3, TMS): 3.38 (d, 2H), 3.69 (s, 3H, -OCH3), 3.96 (s, 2H), 4.92-4.97 (m, 1H), 6.99 (s, 1H), 7.14-7.28 (m, 2H), 7.34 (d, 1H), 7.58 (d, 1H); 13C-NMR (500 MHz, CDCl3): 27.52, 42.47, 52.58, 53.17, 109.59, 111.32, 118.53, 119.80, 122.41, 122.78, 127.42, 136.12, 165.72 (CO), 171.68 (CO), m/z (ESI) found 296.0 (M+ 2H+), calculated 294.08.
Synthesis of 16-[methyl-3-(1H-indol-3-yl)-2-(2-(amino) acetamido) 3,6,9, 12-tetraazabicyclo-[12.3.1] octadeca- 1,14,16-triene-2,13-dione [16Trp4A](3): Compound 1 (0.29g, 1.0mmol), K2CO3 (0.51g, 3.60mmol) and methyl-2-(2- chloroacetamido)-3-(1H-indol-yl)propanoate (0.44 g, 1.5mmol) were dissolve in dry acetonitile at room temperature and then refluxed at 65-700 °C for overnight to give crude oily residue. The compound was purified by column chromatography (silica gel, 9:1 chloroform : methanol) to give the compound (0.46g, 83%) as a brown solid compound (3). 1HNMR (500MHz, D2O, TMS): 1.64-1.73 (m, 4H), 2.50-3.98 (m, 30H), 6.99-7.50 (m, 5H; indolyl protons) 7.14-7.28 (m, 2H)7.34 (d, 1H); 13CNMR (300 MHz, D2O): 26.88, 44.47, 45.68, 50.93, 51.22, 51.62, 54.36, 55.79, 56.20, 109.32, 111.94, 118.42, 119.52, 122.07, 124.58, 126.90, 135.82,145.93, 167.73(CO), 171.08(CO),172.27(CO). m/z (ESI) found 550.27 (M+ H+), calculated 549.62.
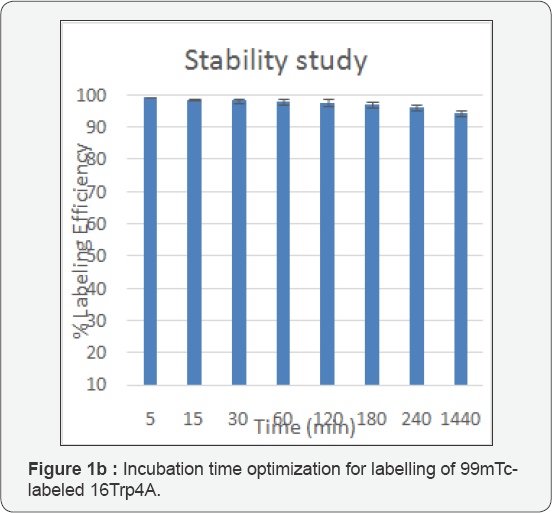
Radiolabeling of 16Trp4A with 99mTc (4): Compound 3 (1mg) was dissolved in a shielded vial and stannous chloride (300mL; 1mg dissolved in N2 purged 1mL of 10% acetic acid) was added followed by addition of freshly eluted (<1 h) 99mTechnetium pertechnetate (82MBq; 200mL). The pH of the reaction mixture was adjusted to 7 with 0.1M Na2CO3 and purged with N2, and shaken to mix. The vial was allowed to stand for 15min at room temperature (250C) (Figure 1).
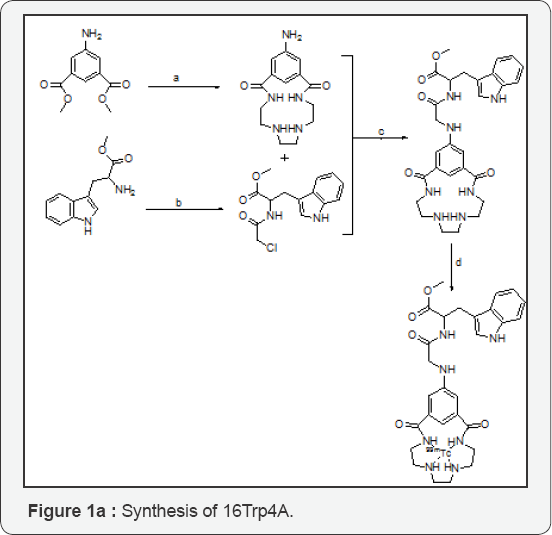
Scheme 1 Synthesis of 16Trp4A. Reagents and conditions: general synthetic pathway of 16Trp4A.
(a) K2CO3, CH3OH, 65-70 0C
(b) TEA, CH2Cl2: H2O, RT
(c) K2CO3, ACN, 65-70 0C
(d) SnCl2, pH=7.0(adjusted with 0.1M Na2CO3), 99mTcO4-*. *99mTcO-4+8H++3/2 Sn+2 ⥨ 99mTc4++3/2 Sn4++4H2O
In vitro cytotoxicity determination
Cell culture: The human cancer cell lines U-87 and U-373 (glioblastoma) were procured from the National Centre for Cell Science (NCCS), Pune, India. The cells (glioblastoma U-87, U373, cervical U756 and HeLa) were cultured and maintained in complete Dulbecco's modified Eagle's medium (DMEM, Invitrogen) as per supplier's specifications supplemented with 10% heat inactivated fetal bovine serum (FBS) and 1% antibiotic cocktail (Himedia, India) at 37°C in a humidified atmosphere with 5% CO2.
Cytotoxicity assay: MTT assay was performed to assess the cytotoxicity. For measurement of the cell proliferation, exponentially growing cells (7,000 cells/well) were seeded in 96-well microtitre plate in triplicate and cultured in complete Dulbecco’s modified Eagle's medium (DMEM). The cells were treated with different concentration of the ligand (TBPD and OPTT individually dissolved in deionized water) for cytotoxicity determination. Following exposure to the ligands, cell viability was assessed by using 200|ig/mL of 3-(4, 5-dimethyl-2- thiazoyl)-2,5-diphenyltetrazoliumbromide (Sigma-Aldrich) as per manufacturer’s protocol. For measurement of the cell proliferation, cells (U-87, U-373, SW756 and HeLa) were seeded in 96-well plates (7,000 cells/well) in triplicate and cultured in complete Dulbecco's modified Eagle's medium (DMEM). Cells were treated with varying concentrations (0.001mM to 1.0mM) of ligands for different time intervals (24,48,72h). After treatment, the cells were incubated with MTT for 4h. The yellow tetrazolium salt (MTT) is reduced in metabolically active cells to form insoluble purple formazan crystals, which were solubilised by the addition of DMSO. Absorbance was measured at 570nm in a multi-well plate reader (BIO-RAD model 680 micro plate reader version 1.70), and the absorbance values of treated cells were compared with the absorbance values of untreated cells.
Radio labeling: The compound was radiolabeled with 99mTc using SnCl2 as reducing agent. All the labeling parameters such as pH, concentration of reducing agent etc. were standardized, to achieve the maximum labeling efficiency. In vitro and in vivo stability of the labelled complexes was checked and the complexes were found to be stable for 24h under physiological conditions. The labelling yield was found to be greater than 95%, as determined chromatographically by different solvent systems. The reaction mixture was kept in saline for different time intervals to carry out in vitro stability studies. Percentage radio labeling was calculated for 0, 2, 4, 6, and 24h. The metal binding assay confirmed the radiochemical purity to be >99%. Radio chromatogram showed a single peak for the ligand which was found intact over 6h.
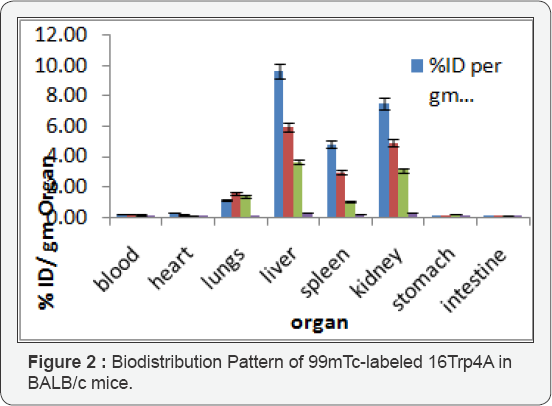
Biodistribution studies: The bio distribution studies were performed in BALB/c mice by intravenous injection of radio conjugates through tail vein. Mice were sacrificed at various time intervals ranging between 1, 4 and 24h after injection of 99mTc-[16Trp4A]. The uptake of the compound by various organs was determined by screening of the biological profile of mice. Highest uptake was shown by liver (9.61+0.33, 5.95+0.29 and 3.66+0.19 %ID/g at 1,4 and 24h post injection, respectively), which indicates that the major route of excretion was hepatobiliary. The uptake exhibited by kidney (7.51+0.31, 4.89+0.24 and 3.10+0.16%ID/g at 1, 4 and 24h) delineates partial excretion by renal route also. Accumulation of activity in non-targeted organs and tissues (heart, brain, lungs, stomach, and intestine) was found <2%. The negligible activity in stomach (0.06+0.00, 0.05+0.00 and 0.25+0.01) %ID/g delineates very less availability of free 99mTcO4 ,confirming the stability of radiolabeled complex in biological milieu Figure 2.
Results and Discussion
This work is focused on the development of the target specific macrocyclic vehicle, 16-[methyl-3-(1H-indol-3-yl)-2-(2-(amino) acetamido)] 3,6,9,12-tetraazabicyclo-[12.3.1] octadeca-1,14,16- triene-2,13-dione, 16Trp4A was synthesized as Scheme 1. All intermediates and final compounds are structurally elucidated by IR, 1H & 13C NMR spectroscopy, and ESI-MS spectrometry. The macro cyclic ligand was synthesized by high dilution method. Dimethyl-5-aminodiisophthalate and 1,8-diamino-3,6- diazaoctane were diluted separately to 100ml in dry methanol. Both the precursors were added drop wise to three neck round bottom flask (1L) containing 400ml dry methanol with the help of dropping funnel with constant stirring under inert condition at room temperature. The obtained white solid compound washed with methanol and dried under vacuum (1). Methyl- 2-(2-chloroacetamido)-3-(1H-indol-3-yl) propionate (2) was prepared by the reaction of L-methyl ester of tryptophan and chloroacetyl chloride in presence of base K2CO3, obtained as a colourless oil. These intermediate was treated with 1 in presence of base to give 16-[methyl-3-(1H-indol-3-yl)-2-(2(amino) acetamido)] 3,6,9,12-tetraazabicyclo-[12.3.1] octadeca-1,14,16- triene-2,13-dione, (3) .
The developed target specific biologically active molecule conjugated with macrocyclic ligand was labeled with 99mTc through a facile radio labeling protocol with greater than 95% labeling efficiency and high radiochemical purity. The compound was radiolabeled with 99mTc using SnCl2 as reducing agent. All the labeling parameters such as pH, concentration of reducing agent etc. were standardized, to achieve the maximum labeling efficiency. Biodistribution studies in mice revealed significant uptake in blood, lung, liver, kidney, spleen and intestine initially, which decreases as time passes up to 24h.
References
- Ke S, Wright JC, Kwon GS (2007) Intermolecular interaction of avidin and pegylated biotin. Bioconjugate Chem 18(6): 2109-2114.
- Hofmann K, Titus G, Montibeller JA, Finn FM (1982) Avidin binding of carboxyl-substituted biotin and analogues. Biochemistry 21(5): 978984.
- Green, NM (1966) Thermodynamics of the binding of biotin and some analogues by avidin. Biochem J 101(3): 774-780.
- Chaiet L, Wolf F (1964) The properties of streptavidin, a biotin-binding protein produced by streptomycetes. Arch Biochem Biophys 106: 1-5.
- Prakash S, Hazari PP, Meena VK, Jaswal A, Khurana H, et al. (2016) Biotinidase Resistant 68 Gallium-Radioligand Based on Biotin/Avidin Interaction for Pretargeting: Synthesis and Preclinical Evaluation. Bioconjugate Chem 27(11): 2780-2790.
- Langstrom B, Antoni G, Gulberg P (1987) Evaluation of D-Isomers of O-11C-Methyl Tyrosine and O-18F-Fluoromethyl Tyrosine as TumorImaging Agents in Tumor-Bearing Mice: Comparison with L- and D-11C-Methionine. J Nucl Med 28: 1037-1040.
- Ishiwata K, Vaalburg W, Elsinga PH, Paans AMJ, Woldring MG (1988) Comparison of L-[1-11C] methionine and L-methyl-[11C] methionine for measuring in vivo protein synthesis rates with PET J Nucl Med 29(8): 1419-1427.
- Chung JK, Kim YK, Kim SK, Lee YJ, Paek S, et al. (2002) Usefulness of 11C-methionine PET in the evaluation of brain lesions that are hypo- or isometabolic on 18F-FDG PET. Eur J Nucl Med Mol Imaging 29(2): 176-182.
- Singhal T, Narayanan TK, Jain V, Mukherjee J, Mantil J (2008) 11C-L- methionine positron emission tomography in the clinical management of cerebral gliomas. Mol Imag Biol 10(1): 1-18.
- Higashi K, Clavo AC, Wahl RL (1993) In vitro assesment of 2-fluoro-2- deoxy- D-glucose, L-methionine and thymidine as agents to monitor the early response of a human adenocarcinoma cell line to radiotherapy. J Nucl Med 34(5): 773-779.
- Meyer M, Dahaoui-Gindrey V, Lecomte C, Guilard R (1998) Conformations and coordination schemes of carboxylate and carbamoyl derivatives of the tetraazamacrocycles cyclen and cyclam and the relation to their protonation states. Coord Chem Rev 180: 1313-1405.
- Wainwrigh KP (1997) Synthetic and structural aspects of the chemistry of saturated polyaza macrocyclic ligands bearing pendant coordinating groups attached to nitrogen. Coord Chem Rev 166: 35.
- Izatt RM, Pawlak K, Bradshaw JS, Bruening RL (1995) Thermodynamic and Kinetic Data for Macrocycle Interaction with Cations, Anions and Neutral Molecules. Chem Rev 95(7): 2529-2585.
- Boschetti F, Denat F, Espinosa E, Lagrange JM, Roger G, et al. (2004) A powerful route to C-functionalised Tetraazamacrocycles. Chem Commun 5(2): 588-589.
- Maruk AY, Bruskin AB, Kodina GE (2011) Novel 99mTc Radiopharmaceuticals with Bifunctional Chelating Agents. Radiochemistry 53: 341-353.






























