The Recent Trend in Bio-Similar Drugs Development
Sankha Bhattacharya1*, Shreya Naresh Naik, Prachi Jitendra Patel and Urvi Gopal Mav
Department of Pharmaceutics, Rofel Shri G M Bilakhia College of Pharmacy, India
Submission: March 04, 2018; Published: March 28, 2018
*Corresponding author: Sankha Bhattacharya, Department of Pharmaceutics, Rofel Shri G M Bilakhia College of Pharmacy, India, Email: sankhabhatt@gmail.com
How to cite this article: Sankha B, Shreya N N, Prachi J P, Urvi G M. The Recent Trend in Bio-Similar Drugs Development. Glob J Pharmaceu Sci. 2018; 5(2): 555660. DOI: 10.19080/GJPPS.2018.05.555660
Abstract
The patent expiry of various biological medicines has led to developed Bio-Similar drugs across the globe. This article reviews the various approved Bio-Similar drugs in India and the challenges of Bio-Similar drugs for getting marketing approval. USFDA guideline clearly said that Bio-Similar drugs are not generic medications nor identical to the innovator medicine and also it's not ensuring therapeutic equivalence with innovator drug. Getting Bio-Similar product marketing approval is a challenging task. To improve access of Bio-Similar drugs within the US market, US-FDA allows abbreviated pathway for their approval. Recently India is becoming a most preferable destination for Bio-Similar manufacturers, because of Make in India program. Introduction of recombinant technique to prepare Monoclonal antibody based Bio-Similar drug becoming popular within pharmaceutical manufactures because of many recent patent expiries of Biologics. The biologies are produced by cell culture method; hence, chances of variability's are more as comparable with the chemically synthesized conventional medicine. Therefor it is impossible to produce an identical copy of an innovator product; hence, Bio-Similar is not considered as generic drugs. These drugs are Twin but not a clone of the innovator drug. The Bio-Similar drugs always face challenges regarding verification of the similarity, the interchange ability, unique naming to differentiate the various Bio-Pharmaceutical products, commercial opportunities, IPR and public safety.
Keywords: Bio-similar; USFDA; European medicines agency; Insulin; Monoclonal antibody
Abbreviations : HGPRT: Hypoxanthine Guanine Phosphoribosyltransferase; EMEA: European Medicines Agency; PMS: Post Marketing Studies; IBSC: Institutional Biosafety Committee; ANDA: New Drug Application
Introduction
Bio-Similar is the fastest growing field of pharmaceutical industries. They were produced mainly by (Recombinant DNA technology-antibody/monoclonal body's technologies). This has provided many cures and prevention to some life Dangerous illness such as Diabetes, rheumatoid arthritis, cancer, inflammatory etc. This has produced a wide range of product like growth hormone, insulin, recombinant vaccines and gene cloning product.
What is Bio-Similar?
The development of Bio-Similar has reached its peak of success in recent times. The first product developed was HUMULIN by USFDA in 1982 that sales now account for US $92 and reached US $167 billion by 2015. Many countries like Japan, China and Europe are involved in it. Different countries claim them by different names; such as EUROPE- Bio-Similars. US, JAPAN- Follow-on pharmaceutical MEXICO- Bio comparables. Basically Bio-Similars are copies of biologic drugs, developed after the patient has expired on an originator drug. Bio-Similars is almost similar to the reference drug [1-3]. However, virtually they have never been the same. Bio-Similars are approved based on part on the studies done with the reference product. Meanwhile, the relative process is complex, i.e. introducing a generically equivalent of the chemical base drug. Bio-Similars exhibit high molecular complexity. Therefore, a small change in product will cause a major change in the process.
Advantages
The predominant advantage of biological drugs is higher specificity. For monoclonal antibodies (mAb), there are numerous ways that antibodies could be used for therapy. For example: mAb therapy can be applied to destroy malignant tumor cells or prevent tumor growth by blocking cell-specific receptors, or by delivering a radioactive molecule to a target cell, thereby delivering a lethal chemical dose to the target cell (radio immunotherapy). It is possible to produce a specific mAb to almost any extracellular/cell surface target, indicated by the ubiquitous amount of research and development currently being conducted to generate monoclonal antibodies against numerous diseases that are difficult to treat by drug therapy or surgery including rheumatoid arthritis, multiple sclerosis and different types of cancers [3-6].
Applications
A. Immuno-diagnostic application.
B. Immunodiagnosis of pregnancy.
C. Immunodiagnosis of bacterial and viral infection.
D. Immunodiagnosis of tumor.
E. Epidemiological studies.
F. Immunotherapy.
G. Cancer therapy.
H. Generation of Anti-Idiotypic antibodies.
Various standards required for approval of bio-similar product
a. Consistency of process.
b. Conforming manufacturing standard to appropriate regulations.
c. Innovator used should be standardized including.
d. Pharmacokinetics/pharmacodynamics and clinical data Table 1.
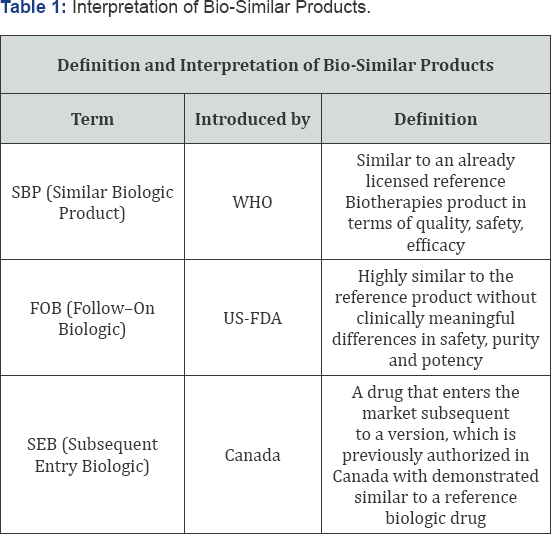
Bio-similar episode
The central drugs standard control organization and the department of biotechnology is the national regulatory body for the medical devices that released guidelines for the bio similar products. Bio-Similar product is similar in term of quality, safety and efficacy of the already authorized original biological medicinal product [7-9].
Bio-similar regulatory scenario-India [10-18]:
I. Preclinical studies are required
II. Phase II trials not required
III. Phase III, a non-inferiority trials against innovator is required for the specific
A. Indication
i. Regulatory process-Drug controller General of India
ii. Genetic engineering approval council GEAC
iii. Recombinant DNA Advisory Committee RDAC
iv. Review Committee on Genetic Manipulation RCGM
v. Institutional Biosafety Committee IBSC
vi. No separate approval process
B. Regulations & Guidelines
A. Guidelines for recombinant DNA safety, 1990
B. Guidelines for generating preclinical and clinical data for rDNA vaccines, diagnostics and other biologics, 1999
C. Guidelines for industry, 2008:
a) Submission of clinical trial application for safety and efficacy
b) Requirement for permission of new drug
c) Post approval changes in biological products
d) Preparation of the quality information for submission of new drug approval.
D. Guidelines for institutional Biosafety committees, 2011
E. Guidelines on similar biologic: regulatory requirements for marketing authorization in India, 2012.
F. Guidance on post marketing studies (PMS).
C. Following parameters for the post marketing protocol
I. Primary endpoint: safety
II. Secondary endpoint: Efficacy and immunogenicity
i. In PMS if clinical studies are evaluated, additional non- comparative immunogenicity studies are not required.
ii. The immunogenicity of the similar biological product should be evaluated with Morden methods using designed studies considering the potential impact on both safety and efficacy.
Selection of reference biologic
Reference biologic is an innovator product approved after evaluation of complete dossier is critical for the development of similar biologic. For selection of reference biologic, the following factors are considered:
A. The reference biologics should be approved in India or ICH countries and should be the innovator product. The reference biologics should be approved based on a safety, efficacy and quality data. Therefore, another similar biologics cannot be considered as a choice for reference biologic.
B. In case the reference biologic is not considered in India, the reference biologic should have been licensed in any ICH country. The reference biologic product can be imported from developing the similar biologic for quality, pre-clinical and clinical comparability.
C. The same reference biologic can be used throughout the Studies supporting the safety, efficacy, and quality of the product.
D. The dosage form, strength and route of administration of the similar biologics should be the same as that of reference biologic.
E. They are proteins derived from cell culture/ fermentation processes; the first example was recombinant human insulin. Other examples are cytokines, monoclonal antibodies, and other insulin's. Since a number of year's bio similar products are on the market for clinical usage, that is, "copies" of therapeutically used proteins e.g.: erythropoietin, somatropin. Bio-Similars are "a new biological product similar to an already authorized medicine. Similar but not identical to the biological reference medicine."
Insulin
It is composed of two polypeptide chains A and B linked via disulphide bonds. These chains are derived from pre-proinsulin, which is synthesized in the beta cells of the islets of Langerhans in the pancreas.
Manufacturing of insulin
The manufacture of therapeutic biomolecules is a complex and multistep process. The production of biological medicines utilizes recombinant DNA technology. Some of the main manufacturing steps associated with potential sources of variability are selection of suitable vector, influencing the process yield or degradation characteristics of soluble proteins and choice of appropriate production platform. DNA is isolated from human cells and modified before insertion into an appropriate vector and expression system (host cell) such as Bacterium Escherichia coli or yeast Saccharomyces cerevisiae. The expressed product is then recovered and refolded to form a pro-insulin-like molecule before C-peptide is removed and the final product purified and processed for storage. There are many steps in the production process where impurities may be introduced and complex testing is required to assure purity and stability. Purification can involve a number of techniques, such as centrifugation, filtration, or chromatography. The production process is very sensitive to physical conditions such as light and temperature and, as such, subtle changes in the manufacturing process can result in significant changes in the end product which can in turn affect its biological action [19-23].
Monoclonal Antibodies
Monoclonal antibodies are non-specific antibodies produced by identical B lymphocytes, called plasma cells, by inducing various conditions. The Mab acts against a specific epitope of the antigen, thereby it can bind specifically to an antigen in a mixture of antigens.
Production
Mab can be produced by including a plasma cell, life span of plasma cell is short, and hence the duration of secretion of antibody is short. It can be produced by employing hybridoma technology. Hybridoma cell clones are produced by the fusion of two deaf cells, the myeloma cell and the plasma cell. The spleen plasma cells are obtained from mouse or rabbit, which is already immunized with antigen of choice. The splenocytes contain polyclonal beta cells, which secrete antigen specific antibodies. The immortal myeloma cell is selected by growing in the culture containing toxic purine analog, 8-Azaguanine and then subjected to an enzyme inhibitor, aminopterin, a folic acid analog. These screens and choose myeloma cells are unable to produce and lack HGPRT (hypoxanthine, guanine, phosphoribosyltransferase) gene and cannot survive in HAT (Hypoxanthine, Aminopterin and Thymidine) medium.
Spleen cell and myeloma cell are fused by using polyethylene glycol as a fusion agent, which produced hybridoma cell. Cells are then transferred to HAT medium. In which the infused myeloma cell dies in HAT medium, the unfused beta cell dies naturally, only hybridoma cells are immortal and survive in HAT medium. In initial purification the 1st step is the separation of clones of hybridoma cells from each other. (Purification is done by chromatography method). Then the subcloning is done for the isolation of single clones. Then the large scale propagation of single clone in tissue culture medium to produced Mab.
Bio-similar expression methodology
Pharmacovigilance: There are many challenges during the research of Pharmacovigilance of Bio-Similars. Only clinical trials are insufficient to defeat the adverse effects. Therefore, the Pharmacovigilance report is required for the Bio-Similars. Same as biological medicines Pharmacovigilance report is important for the Bio-Similars. Post marketing study gives a safety data from the clinical trials. These clinical trials are applied for 2 situations.
a) Chemically synthesized molecule.
b) For Bio-molecule.
Biologicals are used very rarely for the disease. There is very limited information regarding to Pharmacovigilance. Biologicals are also used for multipurpose. It is difficult to give a Pharmacovigilance effect because some drugs have hidden pharmacodynamic or kinetic adverse effect. They are used as 2nd or 3rd line therapy in more severe disease. Bio-similes are given after the failure of the standard treatment. From the above data, we can say that the Pharmacovigilance of Bio-Similars have more challenges than the small molecules. The most difficult concern relating to Bio-Similars is immunogenicity. On the regulatory approval of a Bio-Similar, there is information for the reference biological. Small changes into the production and purification of Bio-Similars have implication on their safety and change into the immunogenicity profile. The adverse effect of the Bio-Similars is similar for the Bio-Similars and the reference original product. The production of the Bio-Similars have some ownership, manufacturer of the product will not be able to replicate of the protein product. Pharmacovigilance of BioSimilars is very important for safety profile. Bio-Similar product has the same requirement as their reference product. So in order to conduct proper marketing, Bio-Similars would have to submit a mix management plan and a periodic safety update regulatory [24-26].
Regular or Routine Pharmacovigilance
Routine Pharmacovigilance contains the reported adverse effect given by the professional's healthcare and patient. Small changes into the procedure of biological change in the safety profile. The new Pharmacovigilance legislators have an importance of traceability. They are concentrated to improve the traceability and also the collection of the name of the medicinal product and the batch number.
Pharmacokinetic
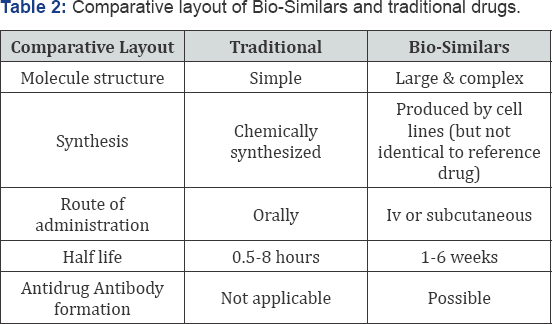
Study designs and analysis method would be enough to meet the objectives. After the long research, they found the successful design for the Bio-Similars. There is a table for comparing the Bio-Similars and traditional days Table 2. The route of administration of the Bio-Similars is a little bit more complex than the traditional drug. They are complex in nature. Due to their long half-line and potential to elicit on immunresponce. They are not suitable for the crossover designs so the parallel design is used. The absorption, half-life, AUC and clearance are also included in this. Antidrug antibodies have the potential to affect pharmacokinetics characteristics and should therefore be measured and their impact considered. The reference and test drug want some parameters according to its pharamacokinetic effect.
Indian Guidelines For Bio-Similars
The guidelines on Bio-Similars prepared by central drug standard control organization and the department of biotechnology. The guidelines of Bio-Similars are for the safety, quality and efficacy. It is also for the procedure of the BioSimilars. It is also included in the pre and post marketing study. CDSCD is the national regulatory authority in India for safety, quality of the drug in the country. The DBT is required for the development and preclinical study for the recombinant DNA. Nowadays, many companies produce the Bio-Similars. In India, the drug is approved by RCGM & CDSCD. The similar biologics are regulated as per the drug and cosmetic act, 1940 and Drug and Cosmetic rule; 1945. Various applicable guidelines for BioSimilars are following.
A. Recombinant DNA safety guidelines, 1990.
B. CDSCO guidelines for industry, 2008.
C. This guideline is for the-
i) Submission of clinical trials for safety.
ii) New drug for approval.
iii) Changes into the product.
iv) Preparation of quality information for drug submission.
Guidelines and handbook for institutional Bio safety committees, 2011.
Some authorities
Some authorities which are included into the process are given below:
A. Institutional Biosafety Committee (I.B.S.C): It is required for the handling of hazardous microorganism and/ or genetically engineered organism.
B. Review Committee on genetic manipulation: It is for the research and development, exchange of genetically engineered cell bank.
C. Genetic Engineering Approval Committee.
For modification in organism
a) Central drug standard control organization.
b) Responsible for the approval of the clinical trial.
c) The guidelines of the Bio-Similars are more focussed on to the post marketing Study. In the CDSCO, new guidance, the one of the major changes mentioned are
d) If the reference biological products are not marketed in India, it can be licensed in any ICH country.
e) The main two parameters are included in the marketing area
a) Safety
b) Efficacy & immunogenicity
c) Previous guidelines are given in to 2012.
d) As per the new guidelines some points are included in this and the most important point is the post marketing study. Regarding to CDSCO
f) In PMS, if clinical studies are evaluated additional noncomparative immunogenicity studies are not required.
g) The immunogenicity of the Bio-Similar product is evaluated with recombinant method using design study.
h) Pre approval studies conducted by the organization include more than 100 patients on the proposed similar biologic drug. In phase IV study, the number of patients can be reduced. Therefore, safety data include both phase III and phase IV is obtained from a minimum of 300 patients.
The primary aim of the phase IV study is safety, CDSCO says, noting that the primary point is for safety and 2nd for the efficacy and immunogenicity. The immunogenicity of the Bio-Similar should be evaluated using appropriately designed studies with state of the art methods. Taking into consideration, the potential impact on both safety and efficacy. Further, they added that if a product is found to be similar "In pre-clinical, in- vitro characterization having established PK method and a PD marker is of efficacy, the residual risk is significantly reduced in the phase I study if equivalent is demonstrated for both PK & PD. Phase III clinical trials of Bio-Similars were considered necessary, an appropriate single arm study in at least 100 evaluable subjects may be carried out in the most sensitive indications to address any residual uncertainly. As per CDSCO has altered the section on "Fermentation Process Development" which is now called "Upstream process Development".
Upstream process should be described in detail including media component used for cell growth. At least three batches of reproducible fermentation data at pilot scale. "Upstream process should be well controlled and monitored" CDSCO says. The regulator also says manufacturers should include details of "Upstream process kinetics data for consistency batches indicating the cell growth, product formation, PH, temperature, dissolved oxygen, major nutrients consumption pattern and agitation rate. " The Indian Bio-Similar market currently includes eight Bio-Similars, including one for Abbvie's blockbuster Humira and two Bio-Similars for Roche's breast cancer treatment, Herceptin which are not approved in any other country.
Regulatory aspects of bio-similars
It is very economical to prepare generic medicine from innovator drug. When the patent on a drug is expired. FDA has asked generic manufacturers to file an Abbreviated New Drug Application (ANDA) for marketing approval. Moreover, the Generic drug application was termed as "abbreviated" because to establish safety and effectiveness profile inclusion of preclinical and clinical data is not mandatory. In order to get generic product marketing approval, the generic manufactures need to demonstrate bio-equivalence and pharmaceutical equivalence between generic and innovator product. The biopharmaceutical products are collective large protein isoforms and not a single molecule entry in same fashion Bio-Similar are not identical but similar to the innovator product. The recent challenges in BioSimilar detection are non-availability of sophisticated analytical tool which could detect the difference between innovator and Bio-Similar product. Before taking marketing approval, it is necessary to carry out preclinical and clinical study. Recently European Medicines Agency (EMEA) has taken several steps to minimize the revocation of marketing approval of Bio-Similar product.
EMEA also issued a guideline called product-class- specification for the development of Bio-Similar product based on recombinant erythropoietin, human insulin, low molecular weight heparins, recombinant IFN-a. On 2006 USA approved Omnitrope; a Bio-Similar used in the treatment of Prader- Willi Syndrome. Hereafter US-FDA stopped giving approval to Bio-Similar products unless a proper regulatory guideline has been issued. Infect Australia, Canada, Turkey, Japan and many other countries are already working on regulatory hurdles for Bio-Similar medicines. The main objective of Bio-Similar drug propagation is to improve the therapeutic impact for millions of patients living with life threatening and chronic disease [27-30].
Importance of hospital pharmacist in Bio-Similar drug promotion:
It is very important for hospital pharmacist to know the noninterchange ability of innovator and Bio-Similar drug during patient Medicare. The pharmacist must understand Bio-Similar drugs are not generic and more safer when switch to clinical practice. Recently in Bio-Similar drugs, systematic checklist has been proposed. For example Pharmacist checklist for Retacrit (epoetin zeta) provides information related to manufacturer, protein product formulation, clinical safety information, tolerability, good handling practice. Retacrit, a potential BioSimilar introduced by the European Union, comprising of Erythropoietin's give opportunity to Pharmacist to treat anemia patient effectively.
Conclusion
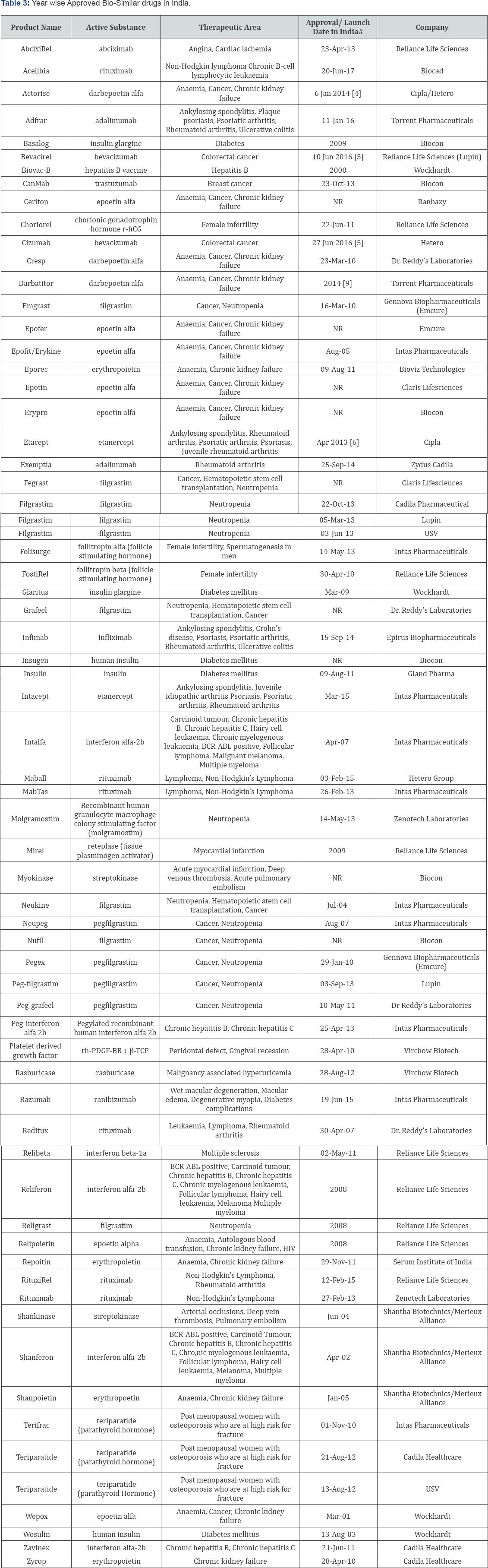
The Pharmacovigilance is an important soul for the extra knowledge and for the safety data for the Bio-Similar due to the limitation of the clinical trials. Traceability and proactive risk management is very important for the knowledge about the safety of the Bio-Similar and the immunogenicity is important. There is a specific need to use good design clinical trials to establish BioSimilar drugs. Most importantly due to complexity of Bio-Similar product it is necessary to identify specific product for a specific disease. The molecular complexity of Bio-Similar product, robustness of manufacturing process, structural resemblance to an innovator drug, mechanism of action, innovator experience and clinical data resemblance need to be considered before filing Bio-Similar for marketing approval. Another key concern is interchangeable with Biopharmaceuticals and Bio-Similar product. Nevertheless, physician awareness about the Bio - Similar product could enhance patient safety and efficacy. Most importantly labelling of Bio-Similar product should be the responsibility of a single authorized body and should be globally acceptable. EMEA and US-FDA should draft proper guideline for getting approval for Bio-Similar products. Table 3 depicts the approved Bio-Similar products in India.
References
- Dranitsaris G, Amir E, Dorward K (2011) Biosimilars of biological drug therapies: Regulatory, clinical and commercial considerations. Drugs 71(12): 1527-1536.
- Crommelin DJ, Storm G, Verrijk R, de Leede L, Jiskoot W, et al. (2003) Shifting paradigms: Biopharmaceuticals versus low molecular weight drugs. Int J Pharm 266(2): 3-16.
- Brockmeyer C, Seidl A (2009) Binocrit: assessment of quality, safety and efficacy of biopharmaceuticals. Eur J Hosp Pharm Pract 15(2): 3440.
- Schellekens H, Casadevall N (2004) Immunogenicity of recombinant human proteins: Causes and consequences. J Neurol 251(Suppl 2): II4- II9.
- (2012) The New India Guidelines on Similar Biologics.
- Roger SD (2006) Biosimilars: How similar or dissimilar are they? Nephrology Carlton 11: 341-346.
- Duerden M (2007) Prescribing advice needed for new biosimilar biological drugs. Prescriber 18: 1-2.
- Schellekens H, Ryff JC (2002) Biogenerics: The off-patent biotech products. Trends Pharmacol Sci 23(2): 119-121.
- Joshi SR (2009) Biosimilar insulins: Are they really similar? J Assoc Physicians India 57(3): 38-41.
- Weise M, Bielsky MC, De Smet K, Ehmann F, Ekman N, et al. (2011) Biosimilars-why terminology matters. Nat Biotechnol 29: 690-693.
- Malhotra H (2011) Biosimilars and non-innovator biotherapeutics in India: An overview of the current situation. Biologicals 39(5): 321-324.
- Rathore A (2011) Development and commercialization of biosimilars in India. BioPharm Int 24: 36-40.
- Haselbeck A (2003) Epoetins: Differences and their relevance to immunogenicity. Curr Med Res Opin 19: 430-432.
- Wadhwa M, Thorpe R (2009) The challenges of immunogenicity in developing biosimilar products. IDrugs 12(7): 440-444.
- Li J, Yang C, Xia Y, Bertino A, Glaspy J, et al. (2001) Thrombocytopenia caused by the development of antibodies to thrombopoietin. Blood 98(12): 3241-3248.
- Thomas TK (2008) Patent for biosimilar drugs may be made mandatory.
- Locatelli F, Del Vecchio L, Pozzoni P (2007) Pure red-cell aplasia epidemic -Mystery completely revealed? Perit Dial in 27(Suppl 2): S303-S307.
- Schellekens H (2004) Biosimilar epoetins: How similar are they? Eur J Hosp Pharm 10(3): 243-247.
- Keithi-Reddy SR, Kandasamy S, Singh AK (2008) Pure red cell aplasia due to follow-on epoetin. Kidney Int 74(12): 1617-1622.
- Yang J, Joo KW, Kim YS, Ahn C, Han JS, et al. (2005) Two cases of pure red-cell aplasia due to anti-erythropoietin antibodies. J Nephrol 18(1): 102-105.
- European Medicines Agency (2013) Marvel Lifesciences Ltd. withdraws its marketing authorization applications for insulin human rapid marvel, insulin human long marvel and insulin human 30/70 mix marvel.
- (2005) Guideline on Similar Biological Medicinal Products.
- (2006) Guideline on Similar Biological Medicinal Products Containing Biotechnology-Derived Proteins as Active Substance: non Clinical Issues.
- European Medicines Agency (2013) Committee for medicinal products for human use.
- European Medicines Agency (2012) Guideline on similar biological medicinal products containing biotechnology-derived proteins as active substance: quality issues (revision 1).
- European Medicines Agency (2014) Guideline on similar biological medicinal products containing biotechnology-derived proteins as active substance: non-clinical and clinical issues.
- European Medicines Agency (2006) Guidance on similar medicinal products containing recombinant erythropoietins.
- Pavlovic M, Girardin E, Kapetanovic L, Ho K, Trouvin JH (2008) Similar biological medicinal products containing recombinant human growth hormone: European regulation. Horm Res 69(1): 14-21.
- Hodgson J (2009) WHO guidelines presage US biosimilars legislation? Nat Biotechnol 27: 963-965.
- World Health Organisation (2009) Expert Committee on Biological Standardization. Guidelines on evaluation of similar biotherapeutic products (SBPs).






























