Development of Analytical Method for Simultaneous Estimation of Adapalene and Benzoyl Peroxide in Gel Formulation by RP-HPLC
Harikishor Barange*, Suhail Asghar and Ujami Sheikh
Unijules Life Sciences Ltd, India
Submission: August 07, 2017; Published: January 22, 2018
*Corresponding author: Harikishor Barange, Unijules Life Sciences Ltd, Maharashtra, India, Tel:-07118-271010, 271008; Email: ard@unijules.com
How to cite this article: Harikishor B, Suhail A, Ujami S. Development of Analytical Method for Simultaneous Estimation of Adapalene and Benzoyl Peroxide in Gel Formulation by RP-HPLC . Glob J Pharmaceu Sci. 2018; 4(3): 555639.DOI10.19080/GJPPS.2018.04.555639
Abstract
A simple, accurate, rapid and precise isocratic high-performance liquid chromatographic method has been developed and validated for simultaneous estimation of Adapalene and Benzoyl Peroxide in pharmaceutical gel formulation. The chromatographic separation was carried out at Analytical Technology HPLC instrument (Software: HPLC Work Station) equipped with Deuterium lamp as detector, HPLC pump and manual injecting facility programmed at 20μL capacity per injection was used. The stationary phase was Zodiac Column C8 (250mmx4.6mm, 5μm) at ambient temperature. Mobile phase containing Acetonitrile: Methanol (90:10). The mobile phase flow rate 1.0mL/min, Injection volume 20μL and detection was carried out at 245nm using UV-Detector. The retention time was found to be 3.7±0.1mins and 5.8±0.1mins Benzoyl Peroxide and Adapalene respectively.
The assay values of both were found to be well within the limits that is 98.42% and 99.19% for Adapalene and Benzoyl Peroxide respectively.The linearity of the proposed method was investigated in the range of 1.9-4.4μg/ml and 48-112μg/ml for Adapalene and Benzoyl Peroxide, respectively. Mean percentage recoveries were 98.94% for Adapalene and 99.28% for Benzoyl Peroxide. The LOD of Adapalene and Benzoyl Peroxide was found to be 0.3μg/mL and 4μg/mL whereas the LOQ was 1.2μg/mL and 20μg/mL respectively. Percentage relative standard deviation of percent assay values for replicate sample preparation was 0.25% for Adapalene e and 0.40% for Benzoyl Peroxide. The method was robust with respect to change in flow rate, temperature and composition of mobile phase. These analytical methods are also applicable in ordinary laboratories. It can also be adopted for quality control tests for these drugs in gel formulation.
Keywords: Adapalene; Benzoyl peroxide; Simultaneous Estimation; RP-HPLC
Introduction
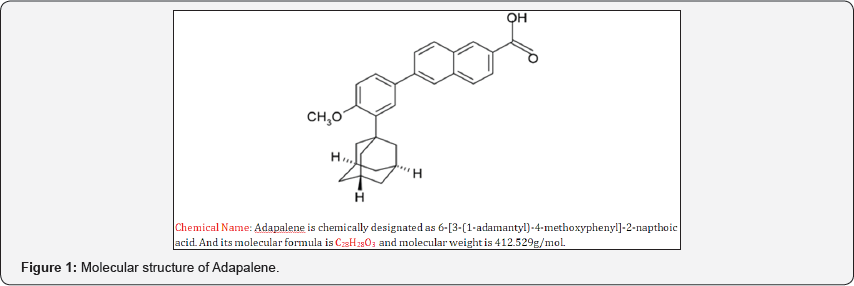
Adapalene is a third-generation synthetic topical retinoid used in the treatment of mild-moderate acne and is also used to treat keratosis pilaris as well as other skin conditions. It is effective against acne conditions where comedones are predominant. It is a highly lipophilic compound, derived from napthoic acid, has both exfoliating and anti-inflammatory effects. Topical retinoids are a group of medicines derived from vitamin A. These compounds result in the proliferation and reduced keratinisation of skin cells independent of their functions as a vitamin [1-4]. Adapalene in small concentrations is a moderator of cellular differentiation, keratinization, and inflammatory processes. It has both exfoliating and anti- inflammatory effects. The exact mode of action of Adapalene is unknown [5]. The molecular structure of the drug is given in Figure 1.
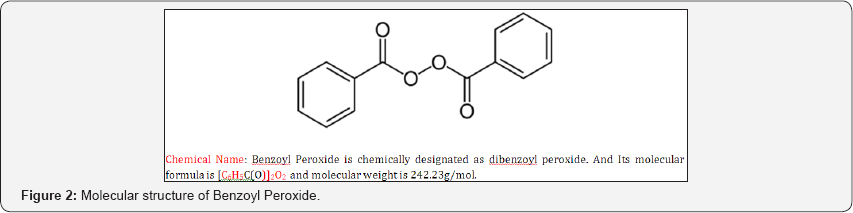
Benzoyl Peroxide is organic peroxide consist of two Benzoyl groups bridged by a peroxide link. Benzoyl Peroxide is an antibacterial agent with demonstrated activity against propionibacterium acnes. This action combined with the mild keratolytic effect of Benzoyl Peroxide is believed to be responsible for its usefulness in acne. It is available in concentration from 2.5-10% [6-8]. Benzoyl Peroxide works as a peeling agent. It increases skin turnover, clearing pores, and reducing the bacterial count as well as acting directly as an antimicrobial [9,10]. The molecular structure of the drug is given in Figure 2.
Combination therapy with a topical retinoid and an antimicrobial agent, which addresses the majority of the causative factors of acne, is considered a first-line treatment option for almost all patients. Adapalene has also been shown to retain its efficacy when applied at the same time as Benzoyl Peroxide due to its more stable chemical structure [11-15]. A detailed literature survey for Benzoyl Peroxide and Adapalene revealed that the determination of an individual compound or in combination with other drugs has been reported using HPLC [16-21], LC-MS, and spectrophotometric techniques [22,23]. But there is no method which describes the simultaneous determination of Adapalene and Benzoyl Peroxide and from gel dosage form meant for external application. The objective of this investigation was to develop simple accurate and economical procedures for simultaneous estimation of Adapalene and Benzoyl Peroxide from a gel dosage form.
Experimental Work
Simultaneous estimation of Adapalene and Benzoyl Peroxide by HPLC
Reagents and material
Adapalene RS, Benzoyl Peroxide RS, Acetonitrile (HPLC grade), Tetra Hydrofuran, Methanol
Instrumentation and chromatographic condition
Chromatographic separation was performed on an Analytical technology HPLC instrument (Software: HPLC Work Station) equipped with Deuterium lamp as detector, HPLC pump and manual injecting facility programmed at 20μL capacity per injection was used. Detection was carried out at 245nm using UV Detector. The separation was achieved on the ODS Zodiac Column C8 (250mmx4.6mm, 5μm) at ambient temperature. The elution was carried out isocratically at flow rate of 1mL/min.
Preparation of mobile phase
Mobile phase was prepared by mixing 90 volumes of Acetonitrile and 10 volumes of Methanol. The mobile phase was ultrasonicated, filtered through 0.45μm membrane filter, and degassed.
Diluent
Use the mixture of Acetonitrile and Tetrahydrofuran (55:45).
Preparation of Standard Stock Solution
A. Standard stock solution for adapalene:
Weigh and transfer about 10.0mg Adapalene RS in 50mL volumetric flask sonicate to dissolve and make up the volume with the diluent to mark. Further dilute 5mL in 25mL with diluent.
B. Standard stock solution for benzoyl peroxide:
Weigh accurately transfer about 25.0mg Benzoyl Peroxide RS in 25mL volumetric flask sonicate to dissolve and make up the volume with the diluent to mark.
Combined standard solution
Mix 2mL each of above standard stock solution in to 25mL volumetric flask and dilute to mark with diluent.
Sample solution: Weigh accurately gel about 1.0g in to a 25.0mL volumetric flask, and add 15.0mL of diluent, The solutions were sonicated for 20 minutes on ultra-sonicator, dilute to volume with diluent. Take 2mL from above solution in 25mL volumetric flask and dilute to mark with diluent.
Procedure: Filter both Sample and Standard Solution with 0.2μ filter paper and inject 20μL (Figure 3a-3d).
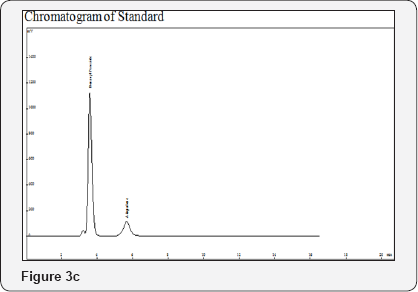
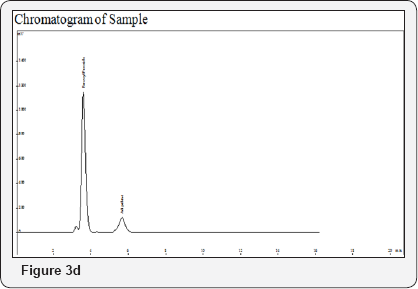
Method Validation
As per ICH guideline the method was validated and following parameters were evaluated, along with Ruggedness [24-29]. Analysis of sample was carried out using the above method and the result are tabulated in Table 1.
System Suitability Studies
System suitability testing is an integral part of many analytical procedures. The tests are based on the concept that the equipment, electronics, analytical operations and samples to be analyzed constitute an integral system that can be evaluated as such. In that the column efficiency, resolution and peak tailing factor were calculated for the standard solutions (Table 2). The values obtained demonstrated the suitability of the system for the analysis of this drug combination.
Linearity
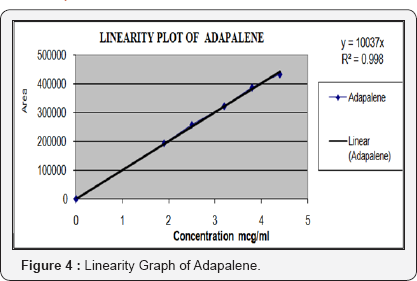
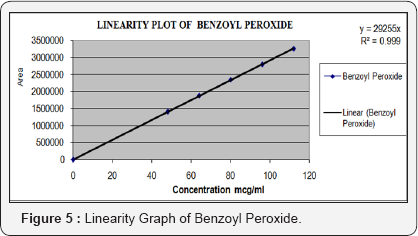
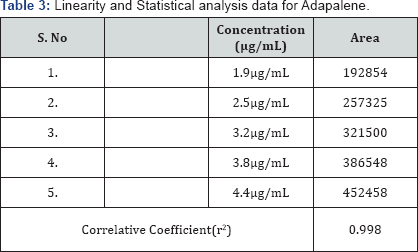
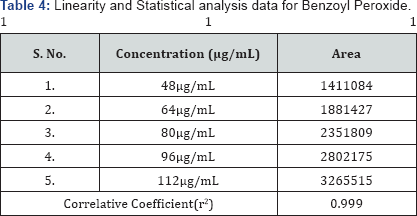
Linearity of the method was established by analysis of combined standard solution. The range of an analytical procedure is the interval between the upper and lower concentrations (amounts) of analyse in the sample (including these concentrations) for which it has been demonstrated that the analytical procedure has a suitable level of precision, accuracy and linearity. Linearity of the proposed method was established by using series of standard solutions of Adapalene and Benzoyl Peroxide these studies are repeated in triplicate with different stock solutions. The curve obtained by concentration on X-axis and peak area on Y-axis against showed linearity in the concentration range of 1.9 to 4.4μg/mL for Adapalene and 48 to 112μg/mL for Benzoyl Peroxide and its correlation coefficient is 0.998 and 0.999, and linearity graph is shown in Figure 4 & 5 (Table 3 & 4).
Limit of Detection (LOD) and Limit of Quantification (LOQ)
The limit of detection and limit of quantification of the developed method were determined by injecting progressively low concentration of the standard solutions using the developed RP-HPLC method. The LOD of Adapalene and Benzoyl Peroxide was found to be 0.30μg/mL and 4.0μg/mL respectively. The LOQ is the smaller concentration of the analyte response that can be quantified accurately the LOQ was 1.2μg/mL and 20μg/mL respectively.
Recovery Studies
To study the accuracy and reproducibility of the proposed method recovery experiments were carried out. A fixed amount of pre-analyzed sample was taken and standard drug was added at 80%, 100% and 120% levels. Each level was repeated three times. The contents of Adapalene and Benzoyl Peroxide found by proposed method is shown in the Table 5. The mean recoveries of Adapalene and Benzoyl Peroxide were 99.08% and 98.94% respectively which shows there is no interference from excipient (Table 6).
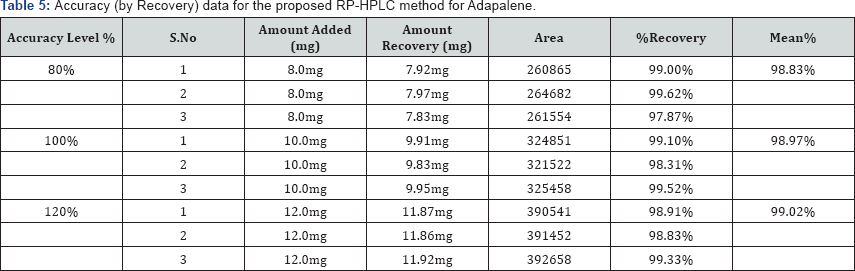
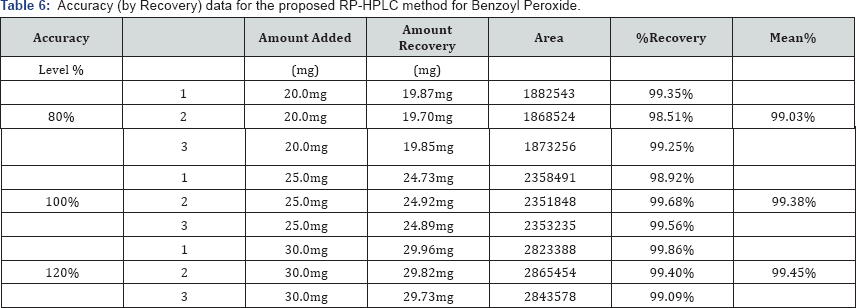
Precision Studies
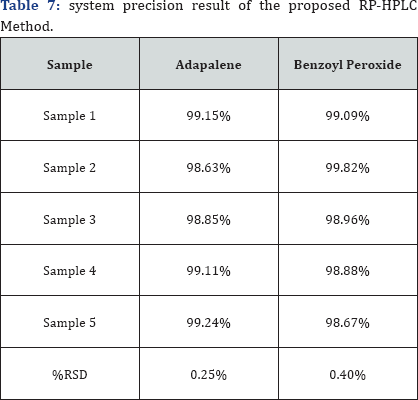
Precision of method was studies by analysis of multiple sampling of homogeneous sample. The precision of analytical procedure expresses the closeness of agreement (degree of scatter) between a series of measurements obtained from multiple sampling of the same homogenous Sample under the prescribed conditions. Precision may be considered at three levels: repeatability, intermediate precision and reproducibility. Precision should be investigated using homogenous authenticated sample. Precision Expressed as % RSD is given in Table 7 which should be less than 2%.
Robustness and Ruggedness of the Method
Robustness of the methodj
Robustness is a measure of its capacity to remain unaffected by small but deliberate variations in the chromatographic method parameters and provides an indication of its reliability. This was done by small deliberate changes in the chromatographic conditions at 3 different levels and retention time of Adapalene and Benzoyl Peroxide was noted. The factor selected were flow rate, Column Temperature and % Acetonitrile in the mobile phase. It was observed that there were no deliberate changes in the chromatogram, which demonstrated that the RP-HPLC method developed, are robust. Results describe in Table 8.
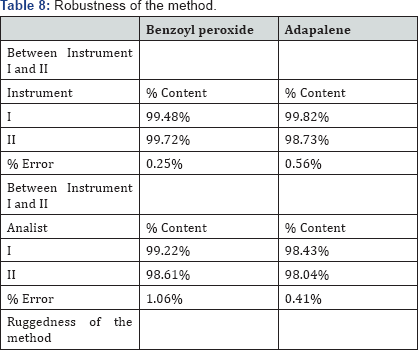
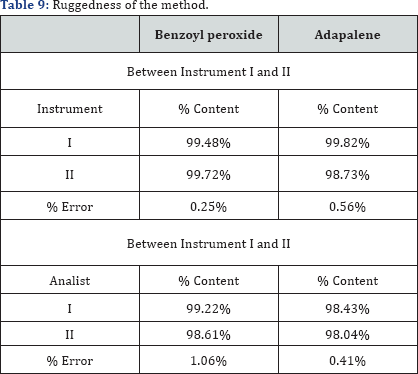
The USP guideline defines ruggedness as "the degree of reproducibility" of the test result obtained by the analysis of the same samples under a variety of normal test condition such as; different Laboratory, different analyst, different instrument etc. Here this was done by changing the instrument and analyst. Results, presented in the Table 9 that indicates the selected factors are remained unaffected by small variations of this parameter.
Conclusion
Based on the results, it is concluded isocritic RP-HPLC method was successfully developed for the assay of Adapalene and Benzoyl Peroxide in topical pharmaceutical formulation. The developed method is selective, precise, accurate, linear and robust. The forced degradation data proved that the method is specific for the analytes and free from the interference of the placebo and degradation products. Moreover, it may be applied for the individual and simultaneous determination of Adapalene and Benzoyl Peroxide compounds in a pharmaceutical drug product and substance. It can be utilized for the determination of assay, blend uniformity, and content uniformity of pharmaceutical products. The developed methods were validated based on ICH guidelines and gave comparable results.
References
- Zaenglein AL (2008) Topical retinoids in the treatment of acne vulgaris. Semin Cutan Med Surg 27(3): 177-182.
- Jain GK, Ahmed FJ (2007) Adapalene pretreatment increases follicular penetration of clindamycin: In vitro and in vivo studies. Indian J Dermatol Venereol Leprol 73(5): 326-329.
- Wolf JE, Kaplan D, Kraus SJ, Loven KH, Rist T, et al. (2003) Efficacy and tolerability of combined topical treatment of acne vulgaris with adapalene and clindamycin: a multicentre, randomized, investigator- blinded study. J Am Acad Dermatol 49(3): S211-S217.
- Zhang JZ, Li LF, Tu YT, Zheng J (2004) A successful maintenance approach in inflammatory acne with adapalene gel 0.1% after an initial treatment in combination with with clindamycin topical solution 1% or after monotherapy with clindamycin topical solution 1% . J Dermatol 15(6): 372-378.
- 5 Mailvelan R, Selvamani P, Rameshkumar T, Raviraj T (2013) HPLC method development and validation for the estimation of Adapalene in pharmaceutical formulations. Asian Journal of Pharmaceutical Analysis and Medicinal Chemistry 1(3): 166-171.
- Brevoxyl G (2017) Aqueous Base Acne Gel for Topical Use.
- Julie C, Harper MD (2010) Benzoyl peroxide development, pharmacology, formulation and clinical uses in topical fixed combinations. J Drug in Dermatol 9(5): 482-487.
- 8. Yi-Cheng C, Pi-Ju T, Yaw-Bin H, Pao-Chu W (2015) Optimization and validation of High-performance chromatographic condition for simultaneous determination of Adapalene and Benzoyl peroxide by Response Surface Methodology. plos one 10(3): 1-9.
- http://en.wikipedia.org/wiki/Benzoyl_peroxide
- Savage LJ, Layton AM (2010) Treating Acne Vulgaris: Systemic, Local and Combination Therapy. Expert Rev Clin Pharmacol 3(4): 563-580.
- http://en.wikipedia.org/wiki/Adapalene/benzoyl_peroxide
- (2018) User Reviews for Adapalene / benzoyl peroxide.
- (2017) Adapalene and Benzoyl Peroxide.
- http://www.emedicinehealth.com/drug adapalene and_benzoyl_ peroxide_topical/article_em.
- Epiduo (2007) Adapalene-Benzoyl Peroxide Gel With Pump.
- Behnam D (2017) Benzoyl peroxide gel monograph. US pharmacopeia-29. Pharmacopeial Forum 30(4):1165.
- Adapalene M (2012) The United States Pharmacopeial Convention. Rockville 2012: 2343-2345.
- Adapalene M (2009) European pharmacopoeia 7.0.
- Martins LA, Meneghini LZ, Junqueira CA, Ceni DC, Bergold AM (2011) A Simple HPLC-DAD Method for Determination of Adapalene in Topical Gel Formulations. J Chromatogr Sci 49(10): 796-800.
- Barrios JG, D'Avila Farias G, Roggia I, Cadore Peixoto S, Pons FR, et al. (2011) Validation of analytical method by HPLC for determination of Adapalene in suspension of nanocapsules. Quimica Nova 34(8):1464- 1467.
- Deo SS, Inam F, Karmarkar NP (2013) Analytical Method Development for Determination of Performance of Adapalene in Adapalene 0.1% Gel Formulation Using Manual Diffusion Cell. Chem Sci Trans 2(1): 251257.
- Abe-Onishi Y, Yomota C, Sugimoto N, Kubota H, Tanamoto K (2004) Determination of benzoyl peroxide and benzoic acid in wheat flour by high-performance liquid chromatography and its identification by high-performance liquid chromatography-mass spectrometry. J Chromatogr A 1040: 209-214.
- Adhikari L, Jagadev S, Sahu S, Moitra SK, Murthy PN (1993)Derivative spectrophotometric. Proceedings of the International Conference on Harmonization. Geneva, Switzerland.
- Stability Testing of New Drug Substances and Products (1993) Proceedings of the International Conference on Harmonization. ICH, Q1A, Geneva, Switzerland.
- Test On Validation of Analytical Procedures (1994) Proceedings of the International Conference on Harmonization. ICH, Q2A, Geneva, Switzerland.
- Tripartite Guideline and Validation of Analytical Procedure: Methodology (1996) Proceedings of the International Conference on Harmonization. ICH, Q2B, Geneva, Switzerland.
- ICH Guidance on Analytical Method Validation (2002) Proceedings of the International Convention on Quality for the Pharmaceutical Industry. Toronto, Canada.
- Wilson TD, Forde MD (1990) Stability of milrinone and epinephrine atropine sulfate, lidocaine hydrochloride, or morphine sulfate, lidocaine hydrochloride, or morphine sulfate infection. Am j Hosp Pharm 47(11): 2504-2507.
- Levine B, Blanke R, Valentour J (1983) Gas Chromatographic Analysis of Lidocaine in Blood and Tissues Journal of Analytical Toxicology 7(3): 123-124.






























