Effects of Ethanol Extract Leaves of nymphaea lotus (water lily) on Learning and Memory in CD-1 Mice
Nomso C and Aduema W*
Department of Human Physiology, Gregory University, Nigeria
Submission: December 21, 2017; Published: January 18, 2018
*Corresponding author: Wadioni Aduema, Department of Human Physiology, Gregory University, Uturu, Abia State, Nigeria, Tel: +234-08038046678; Email: Wadioniaduema@gmail.com
How to cite this article: Nomso C, Aduema W. Effects of Ethanol Extract Leaves of nymphaea lotus (water lily) on Learning and Memory in CD-1 Mice. Glob J Pharmaceu Sci. 2018; 4(3): 555637.DOI10.19080/GJPPS.2018.04.555638
Abstract
Nymphea lotusis a perennial and herbaceous aquatic plant, whose leaves floats or submerged in water across the regions of Africa. The plant has be reportedly used for various therapeutic purposes (such as aphrodisiac, antioxidant and antibacterial purposes) including the treatment of fever, diarrhea, urinary difficulties, enteritis, dyspepsia, bowel problems tumours, gastric ulcers and abnormal heart beat. The present study investigates the effect of the ethanolic extract of Nympheae lotus on learning and memory in mice. The Morris water maze test was used to evaluate the neuro-cognitive effects of this plant. Before the neurobehavioral parameters were assessed, the LD50, Photochemical and acute toxicological screenings of the plant were determined. The median lethal dose was above 5000mg/kg orally in mice for the ethanol extract. Twenty seven (27) Swiss mice were randomly assigned into 3 groups; control (normal saline), low dose (200mg/kg) and high dose (400mg/kg) for fourteen days (14).
The results showed that on days 1, 2 &3 of acquisition training mice from low dose and high dose learned equally when compared to the control. In the reversal training, memory was improved in the low dose and high dose treated mice when compared to control (P<0.01, P<0.001). During the probe trial, the swim duration in the South-East quadrant was significantly higher for high dose and low dose compared to control (p<0.05). However, during the visible platform task, the swim latencies for the low dose and high dose group were also significantly lower compared to control.In conclusion, these findings suggest that the ethanol extract of Nympheae lotus enhances cognitive functions and may be useful in the treatment of retrograde amnesia and neuro-cognitive disorders.
Keywords: Nymphaea lotus; Morris water maze; memory; mice; Post-traumatic stress disorder (PTSD); Neuro-behavioral disorders (NBD)
Introduction
The link between the human behavior and neural control by the brain had been established by early researches in Neuroscience. Certain parts of the cortical and sub cortical structures control the actions of the body system. Neuro-behaviour such as emotions, thoughts, cognition, representational knowledge and motor function have been found to be controlled by certain areas of the cerebral cortex such as prefrontal cortex, caudate nucleus, amygdala and basal ganglia. Hippocampus has also been found to play a critical role in learning and memory, which is a complex biological process including the acquisition, consolidation and retrieval of information [1]. These neuro-behaviors are controlled by the action of neurochemicals (cathecolamines) and neuropeptides such as Galanin, Nociceptin, and Dynorphins [2]. The transmission of impulses to these Neurobehavior- control areas of the brain through the release of neurochemicals (dopamine, non-epinephrine, and acetyl-choline) leads to the generation of coordinated functions otherwise called executive functions.
Neuro-behavioral difficulties involve two categories; cognitive decline including memory problems and dementia, and Neuropsychiatric disorder including neurasthenia, depression, post-traumatic stress disorder (PTSD) and suicidal tendencies. Other Neurobehavioral disorders associated with motor difficulties characterized by problems such as weakness, tremors, involuntary movement in-coordination and gait/ walking abnormalities. Amongst the cognitive decline associated with aging, Executive function is most affected (e.g., Tariot & Pierre [3], with even more severe decline associated with mild cognitive impairment and Alzheimer's disease. Executive function is compromised across a large number of psychiatric illnesses, including schizophrenia, bipolar disorder, depression, substance use disorders, and attention deficit hyperactivity disorder (e.g., Willcutt et al. [4]). Asperger's disorder also presents a special case of NBD. Ghaziuddin et al. [5], in a review of the literature of Asperger’s Disorder, it was found that about 50% of the studies referred to impaired motor functioning such as clumsiness and uncoordinated movement patterns. 26-36 cases per 10,000 children suffer from Asperger's Disorder, and 4-5 cases per 10,000 suffer from Autism [6].
A lot of funding in form of research grants and scientific awards has been pumped towards understanding the science of neuro-behavioral disorders (NBD). Patients with multiple systems atrophy, progressive supranuclear palsy and Huntington’s disease Robbins et al. [7], Lawrence et al. [8] show significant deficits in neuro-behavioral function. Numerous neuropsychological studies have shown that Parkinson's disease is characterized by executive impairments [9,10], which are evident early in the course of the disorder when pathology is confined to basal ganglia regions, and are seen in patients who are unmediated.
Neuro-behavior deficit has been linked to many causes ranging from substance abuse to neuron degeneration as a result of trauma, to stress to malnutrition and so on. More causative factors are still being discovered. A study by Heffelfinger & Newcomer [11] and Reus & Walkowitz [12] indicated that increased glucocorticoid exposure in humans, including endogenous glucocorticoids (cortisol) associated with stress, decreases memory and learning function and in impairment of memory and hippocampal atrophy. A study done by Ohayon & Roth [13] in their study of 14,915 subjects aged 15-100 years representative of the general population of the United Kingdom, Italy, Germany and Portugal found 28% of subjects with insomnia had a current diagnosis of mental disorders and 25.6% had a psychiatric history, and, insomnia appeared before (>40%), or at the same time (>22%) as, mood disorder symptoms amongst most of the subjects with mood disorders. They also found that with anxiety disorders, insomnia appeared mostly at the same time as (>38%), or after (>34%), the anxiety disorder symptoms.
In Nigeria, Ogunremi [14] found insomnia, (of the sleep onset type) present among 13.9% of his subjects. Children with sleep disorders have been found to have poor academic performances and cognitive disabilities [15]. Neurobehavioral disorders have also been associated with cigarette smoking and substance abuse. Abbott et al. and Burke et al. [16,17] observed in their studies that prenatal cigarette smoke exposure from mothers result in aetiopathologies of certain features seen in children. Some features which include neurobehavioral changes such as attention deficits, hyperactivity and impulsivity; perceptual deficits; altered learning and memory; lowered IQ and impaired intellectual development, and an increased propensity for conduct, behavioral, or psychological disorders. Moreover, Heavy alcohol consumption and hard liquor drinkers likewise exhibit neurological deficit and cognitive decline [18].
Toxic chemicals such as lead and mercury which occur in many forms of land and water pollutions show neurotoxicity effects and lead to varieties of neurobehavioral disorders such as cognitive disabilities, motor in-coordination, executive function deficits etc. pointed out that on exposure to mercury chloride, there is degeneration and reduction of pyramidal cells and neural cell fibers of the pyramidal cell layer of the hippocampus leading to learning and memory function deficits [19]. Aging is one of the inevitable processes of life. World-wide researches are being done in order tackle neuro-cognitive disorders and behavioral decline associated with aging. Oxidative damage as a result of Oxidative stress which characterizes aging process has been linked to a number of neurobehavioral disorders such as Alzheimer’s disease and dementia [20]. Although there is no permanent cure for these NBDs, Antioxidants therapy is utilized to mop up the free radicals and reactive oxygen species that results in the process of aging. Diets rich in antioxidants such as vitamin A, C, E and bioflavonoid (such as flavones, tannins, and anthocyanins) provide ameliorative effects to these NBDs related with aging [21]. There is considerable evidence of significant effect of poverty on neurobehavioral development. Bergen Ippolita CC & Shukitt-Hale B [22] found that mental retardation occurs in three to five per 1000 individuals in high- income countries but occurs in as much 22 out of 1000 individuals in developing countries. Noble et al. [23] found significantly lower brain volume in the hippocampus and amygdala in children from lower-income households compared to children from higher income household. Furthermore, Neuro-cognitive disorders have also been linked to poor academic performances in students. Children with executive function deficits have difficulties in coping with the Academia [24].
Manifestations of these disorders in academics are inadequate educational coping strategies, poor organizational and study skills, and inefficient time management. The use of Functional magnetic resonance imagining and advanced neuro-imaging techniques has been employed in the detection and studies of patients with Neurobehavioral disorders. These are aimed at understanding and assessing the effects of these disorders on executive functions. Brain scans and readings have been analyzed and has been noted that neural activities with the brain of affected subjects differ significantly from those who do not have these NBD. Executive functions are compromised in an array of clinical disorders; patients with various neurological and psychiatric complaints, as well as those with trauma to the frontal lobes, exhibit executive deficits.
Neuro-imaging has provided a means for exploring these deficits, as well as developing an understanding of recovery of executive function frequently seen in patients suffering traumatic brain injury or stroke [25]. In addition to neuropsychological studies, there are several human neuro-imaging studies indicating that the basal ganglia are involved in learning skills and habits. Activation in the caudate nucleus has been observed while subjects are learning the skill of reading mirror-reversed text [26]. Interestingly, the striatal involvement may only be present during learning, with highly skilled performance primarily activating cortical circuitry [27]. In another study, positron emission tomography (PET) was used to measure dopamine release while participants played a video game [28]. As subjects played the game and improved their performance, there was decreased binding of radio-labeled Raclopride (a dopamine antagonist) in dorsal and ventral striatum in comparison to a control condition. These data suggest that endogenous dopamine release increased in the neostriatum during practice, consistent with research in lower animals demonstrating a role for striatal dopamine in S-R habit learning (e.g., Packard MG & White NM [29]).
The provision of complete treatment of neurobehavioral disorders still remains elusive. Therapies and Management of the condition is still very crucial. This is achieved through pharmacologic and non-pharmacologic interventions. In pharmacologic interventions, two major type of drugs are used in the management of Neurobehavioral disorders such as Alzheimer, Parkinsonism and dementia: these are Acetylcholinesterase Inhibitors & N-methyl-D-aspartate (NMDA) Receptor antagonists. Non-drug therapies: Simple non-drug treatments like Reminiscence therapy and Social Interaction may prevent need for drugs. Example of treatment includes Massage, CBT, music and dance therapies with drugs; however immediate results should not be expected [30]. Non- Pharmacological interventions may be promising for a number of reasons older adults may prefer non-pharmacological strategies to maintain cognitive function and community independence. Non-pharmacological interventions have less risk of side effects. Recommendations include-Cognitive intervention, Physical activity, Psycho-social intervention [31]. Extensive researches on different plants are underway worldwide. Plant extracts have rather more therapeutic benefits and fewer side effects and are more economical [32].
The use of medicinal plants as a prophylactic or therapeutic agent has existed over the course of history and man has explored the plant kingdom for compounds which are of medicinal value [33]. This has been done in order to identify chemical constituents responsible for biological effects isolated or purified in the search for new single entities as pharmaceutical ingredients or characterized and standardized in the search for new multicomponent botanical products [34]. Regardless of the fact that full understanding of neurobiology of neurobehavior has not yet been achieved and mysteries about its disorders still remain unsolved, researchers and scientists are constantly working, searching for solutions. Many researchers have resorted to traditional medicines in order to find answers. Therefore, it will be worthwhile to research in to some of our local plants which may be beneficial in the management of behavioral disorders such as the ever increasing incidence of learning, emotional or behavioral problems that are common in the world including Nigeria; hence this study is designed to solve this issue.
Materials and Methods
Plant material
Nympheae lotus (water lily) Leaves were collected and identified in the Department of Biological sciences, Ahmadu Bello University, Zaria, and a voucher specimen was deposited in the herbarium of the Department (V/N 1689).
Preparation of extract
Nympheae lotus leaves were first separated from the stalk, rinsed with water to remove dirt, the air-dried leaves 200g of Nympheae lotus were pulverized and soaked in ethanol (2L) for 72hrs. The solutions were filtered and the filtrate concentrated with a rotary evaporator to give a semisolid residue. The product was kept in the refrigerator for further use.
Acute toxicity test (LD50)
The determination of the toxicity levels were done through oral route and intra-peritoneal route. The mice were divided into 3 groups in two phases (low dose & high dose, 1 & 2 respectively) for each route of administration; both routes has phase 1which consist of 3 groups of 3 mice each and Phase 2 consist of 3 groups of 1mouse each. Each group of mice administered orally were given a different dosage of the extract (10mg/kg, 100mg/kg, 1000mg/kg) for phase 1 (low dose) and 1600mg/kg, 2900mg/ kg and 5000mg/kg for phase 2 (high dose). For the mice groups administered through intra-peritoneal route, 10mg/kg, 100mg/ kg, 1000mg/kg was given as low dose and 1000mg/kg, 1600mg/ kg and 2900mg/kg for high dose. The number of deaths in oral route for phase 1 was n=0 for all groups and for phase 2 also n=0 for all groups. For intra-peritoneal route, the number of deaths (n) recorded in phase 1 was n=1 and n=1 for high dose was recorded within 24-72 hours.
The LD50 was calculated using probit kill of the dose proposed by Lorke [35].
For oral route=√(minimum tolerated dose x minimum toxic dose)
LD50>5000mg/kg
LD50 for IP route= √(minimum tolerated dose x minimum toxic dose)
=√1600mg/kgx2900mg/kg
=2154mg/kg
Care and management of animals
Thirty Swiss mice weighing between 14g and 17g were purchased and used for this experiment. The rats were housed in the animal holding of the Department of Anatomy and physiology, Abia state University, Abia state. Animals were housed in groups of 3 (control, low dose and high dose) in plastic cages, maintained under standard dark-light cycle Food and water was available Ad libitum. All rules applying to animal safety and care were observed.
Experimental animals/grouping
Thirty [27] Swiss mice weighing between 14g and 17g were randomly assigned into three groups A, B, and C of 9 mice each. Group A was the control; groups B and C are low dosed and high dosed respectively. Animals in group A were administered normal saline, group B animals received a solution of Nympheae lotus at a dose of 200mg/kg and group C, received ethanol extract of Nympheae lotus at 400mg/kg. Extracts were administered via an oral cannula daily for a period of 14 days.
Apparatus and Experimental Protocols
Morris water maze

Experimental procedure using morris water maze: (Figure 1a), A Morris water maze designed by Morris [36] modified by Paylor [37] was used for this study. The maze was constructed out of a circular polypyroplene pool measured 110cm diameter and 20cm in depth. The pool was filled to depth of 14cm with room temperature tap water. The water was made opaque with the addition of milk to ensure camoflaouge of the white escape platform. The platform was submerged 1cm below the water surface. The pool was divided into four quadrants: Northwest, northeast, southwest and southeast Boundaries of the quadrant was marked on the edge of the point with masking tape and labeled; North, south, east and west. A wooden platform (13.75cmx9cm diameter) was used as an escape platform in the maze. The level of water in the pool was adjusted to 1cm above or below the platform creating a visible or invisible platform respectively. The pool was placed in a laboratory room whose walls were mounted several posters to act as visual cues. Furniture and machine equipment was also used as visual cues. During test, the room was dimly lit. Testing of the Morris water lasted for eight (8) days. Mice were carried into the test room in their home cages. Each mouse was picked by the base of its tail and placed in the pool. The first three days was the acquisition training with and invisible platform and the next three days was the reversal training with invisible platform. On the seventh day, a probe trial was conducted with no escape platform. On the eight day, 4 trials were conducted with a visible platform. During the test period, each mice was placed in a clean empty cage (bedding paper tower was torn and place on the bottom of the cage to allow the mice to dry more quickly after each trial). This paper towel was replaced when it becomes completely wet. Mice were then run in squads of 6with 3 minutes between each trial for each mouse.
During the acquisition training, the water was adjusted appropriately such that the platform was placed in the center of the northeast quadrant. Each animal received 4 trials of 60 seconds (max) per day. The starting position was predetermined, which prevented 2 trials from being repeated by the same animals during other days. Start positions were located at the boundaries of each quadrant. Each mouse was removed from its holding using a small, clean 70cl plastic container to minimize stress. The animal was placed into the water at the appropriate start position, facing the center of the pool. The mouse was then permitted to explore the pool and to search for hidden escape for 60 seconds. When the animal located the platform the timer was stopped and the animal was removed from the plastic container and placed into the holding cage. Once in the platform the mouse was permitted to visually explore their surrounding for 20 seconds at which point, they were picked up and placed on their appropriate holding cage. The next mouse was then placed in the pool and same procedure was repeated. Each mouse completed 4trials per day over 3days. This amounted for 12 trials for acquisition training.
Reversal training period began on the 4th day. The invisible platform was moved to the opposite quadrant (Southwest) and mice were again assigned to appropriate start position. The same procedures as in acquisition training were carried out during the reversal training. Each of the animals completed 4 trials per day for 3 days for a total of 12 trials for reversal training. A probe trial was conducted at day 7. At this time, there was no escape platform in the maze. Each animal completed one trial of 60 seconds. Each animal was placed in the maze from one of the four possible positions and allowed to explore the pool. The duration in each quadrant and the frequency of entry form the northeast and southwest quadrants was noted. The visible platform task was conducted on day 8. The visible platform was placed in the Southwest quadrant of the pool. The same procedure as in acquisition training and reversal training was carried out and mice completed 4 trials.
Behavior scored
The behaviors scored during the Morris water maze were in acquisition training and reversal, visible platform test. These include:
1. Swim latency (time taken to locate and mount the platform)
2. Thigmotaxic frequency (DFTB)
3. Swim path error (SPE) is measured as the duration of the mouse from a relatively direct path from the start position to the location of the hidden platform.
4. Proximity to platform (PTP)
5. Average velocity (AV)
6. Swim Distance (SD)
Whereas during the probe trial, the duration in each quadrant were recorded (NE, SE, NW, and SW) including the annulus acquisition crossing (i.e. the number of times the mouse crosses the location of the platform during acquisition training) and annulus reversal crossing (i.e. the number of times the mouse crosses the location of the platform during reversal training) [38].
Statistical Analysis
Data Obtained from the experiments were statistically analysed using Microsoft excel, with factorial ANOVA/T-test in the statistics programme start view version for windows or Mac. Post-hoc comparison was also done using the student ± Newman-keuls design. Values were represented as Mean±SEM and a "P" value less than 0.05 & P<0.001 was considered as significant.
Results
Acquisition training
On day 1, the swim latencies obtained showed that mice, fed with control, low dose and high dose diets of Nymphea lotus, scored 188.50±15.76; 189.25±7.29 and 163.50±4.62 seconds respectively. In day 2, the swim latencies were 78.00±15.13; 79.25±13.64 and 82.75±9.52 seconds for mice fed with control, low dose and high dose treated diets respectively. For day 3 of the acquisition training, the swim latencies were 100.50±6.94; 82.00±7.58 and 84.75±5.49 seconds for mice fed with control, cooked, low dose and high dose treated diets respectively. The swim latencies for the low and high dose treated groups were not significantly different compared to the control group during the 3 days of training. This is as shown in Figure 1b.
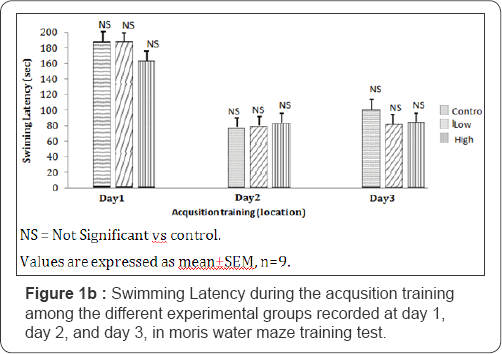
Reversal training
On day 1, of the reversed training, the swimming latency for mice fed with control, low dose and high dose treated diets of Nymphea lotus were 81.50±5.78; 42.00±4.02 and 41.75±5.49 seconds respectively. During the second day of reversal training, the swimming latency were 45.25±2.05; 25.00±4.74 and 25.25±3.81 seconds for mice fed control, low dose and high dose treated diets respectively. For day three (3) of the reversal training, the swimming latency were 38.00±3.34; 21.25±6.14 and 18.50±1.32 seconds for mice fed with control, low dose and high dose diets of Nymphea lotus respectively. The swim latencies for the low and high dose treated groups were significantly lower during the reversal training (Figure 2) compared to the control (p<0.001; P<0.01). However, the low dose treated group was also significantly shorter (P<0.05) compared to control group.
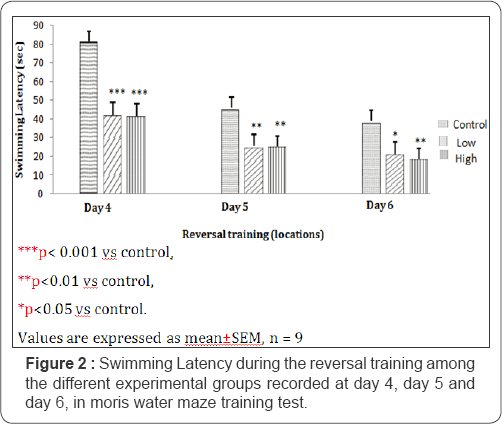
Retention quadrant
Figure 3 compares the quadrant duration during the probe trial in the Morris water maze between the three experimental groups of mice which were, 16.56±2.40; 27.09±1.28 and 28.14±2.34. The duration of stay in each quadrant is shown in Figure 4. During the trial, the three groups had more preference to the South-East quadrant (that bears the platform during the reversal training when compared to control). The preference to the SE quadrant for the mice fed with low dose and high dose treated diets were significantly higher (P<0.01) compared to control. However, the high dose and the low treated groups were not significantly different when compared.
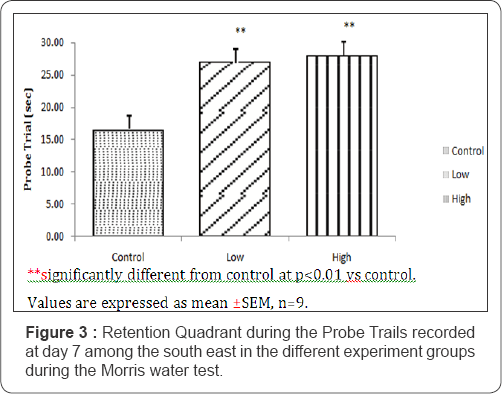
Annulus acquisition & annulus reversal crossings
Figure 4 compares the annulus acquisition and reversal crossings during the probe trial task. The values for the annulus acquisition crossings are 33.00±5.50; 51.66±12.57 and 45.00±5.77 seconds for mice fed with control, low dose and high dose treated diets respectively. The values for the annulus reversal crossings are 20.00±2.88; 42.00±1.73 and 44.66±5.48 seconds for mice fed control, low dose and high dose diets of Nymphea lotus respectively. There was no significant difference among the groups in the annulus acquisition crossings. For the annulus reversal crossings, as shown in Figure 4, the low dose and high dose treated groups were significantly higher compared to control (P<0.01).
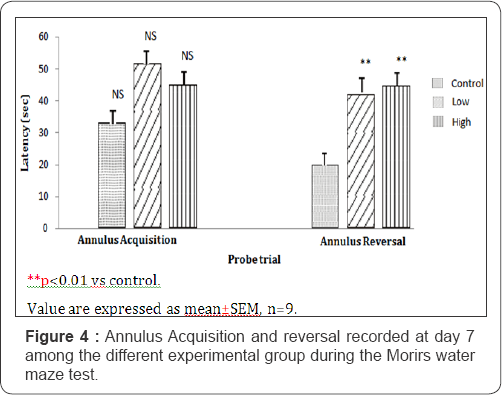
Visible platform task
The swimming latency in the visual platform task was 51.50±4.62; 33.75±2.59 and 30.50±1.32 seconds for mice fed with control, low dose and high dose diets of Nymphea lotus respectively. During visible platform task, the swim latencies of the low and high dose treated groups was significantly lower (P<0.01 and P<0.001) compared to control (Figure 5).
Discussion
This study was set out to determine the effect of the ethanolic extract of Nympheae lotus on learning and memory (i.e. On Neuro-cognition) in Swiss white mice using the Morris water maze. The hippocampus is the part of the brain responsible for visuo-spatial learning and memory which is assessed with the hidden platform test. This process is impaired if the hippocampus is injured. Visible platform on the other hand is a non-hippocampal task dependent on the dorsal striatum of the basal ganglia [39]. The hidden platform uses extra maze cues while the visible platform uses intramaze cues that are placed at the location of the escape platform. The time it took each mouse to locate and mount the escape platform defines the swim latency. The shorter the swim latency betters the learning process. Mice that learnt faster located the platform quicker than the others. During the probe trial, when no escape platform was placed, the longer the time the mice stayed in the quadrants that had the escape platform, the better the memory. So the mice that had better memory will be expected to spend more time in those quadrants where the escape platform was placed during the acquisition and reverse training. Therefore, the probe trial was to evaluate memory, while the acquisition and reversal training was for learning.
The result showed that the swim latencies for the first three days during acquisition training showed no significant difference in the swim latencies of the low dose group and high dose treated group compared to the control. This means that the mice in the three groups learned equally. During reversal training, the swim latencies for the low and high dose treated group was lower compared to the control group; though this was significant, which means that this groups of mice were able to locate the hidden platform faster and so, learn better than the control group of mice that was administered normal saline. Also, the histogram chart of the swim latencies indicates low and high dose mice learnt better than the control. During the probe trial, the low dose and high dose mice spent significantly more time than the control exploring the retention quadrant. This showed that they had better memory than the control group of mice. Also, from the result, high dose mice may have better memory than the low dose control group as the retention quadrant is significantly higher in the high dose group than in the low dose group.
The cued version of the Morris water maze assesses cued learning and visual integrity of the animals tested. Impairments in performance in the hidden platform model may be due to some brain lesions or drugs which may affect the motivation to escape, or sensory motor factors rather than spatial learning. This cueing procedure, in which the escape platform protrudes above the water surface, provides a control for this [36,40]. Here, the swim latencies were also used for the comparisons. Shorter swim latencies in the visible platform task indicate improved cued learning while longer swim latencies indicate poor cued learning. Low dosed and high dosed group had significantly lower swim latencies compared to the mice in the control. This means that the Nympheae lotus (low and high dose) treated mice improved cued learning. Summarily, Learning and memory processes were improved in the mice treated with the low dose and high dose of the extract but however, better in the high dose group.
Conclusion
This research has shown that the ethanol extract of Nympheae lotus (water lily) improves learning and memory in Swiss mice. This may probably be attributed to the presence of neuro-stimulatory phytochemicals which could cause an increased release of neurotransmitters and synaptic connection in the hippocampus and other areas of the cerebral cortex thereby, facilitating neuro-cognition. Further study is suggested on determining the particular phytochemical responsible for this effect and clinical trials carried out to determine its efficacy on humans subjects with neuro-cognitive disorders.
Acknowledgment
We acknowledged Mr. Uchenna Azunna and Mr. Munachi Igbojekwe for their priceless support.
References
- Hou Q, Gao X, Zhang X, Kong L, Wang X, et al. (2004) SNAP-25 in hippocampal CA region is involved in memory consolidation. Eur J Neurosci 20(6): 1593-1603.
- Alberini CM (2005) Mechanism of memory stabilization: are consolidation and reconsolidation similar or distinct processor. Trends in neuroscienced 28(1): 51-56.
- Tariot, MD, Pierre N (1998) Efficacy and tolerability of carbamazepine for agitation and dementia.Am j psychiatry 155(1): 54-61.
- Willcutt EG, Doyle AE, Nigg JT, Faraone SV, Pennington BF (2005) Validity of the executive function theory of attention deficit/ hyperactivity disorder: A meta-analytic review. Biological Psychiatry 57(11): 1336-1346.
- 5 Ghaziuddin M, Butler E, Tsai L, Ghaziuddin N (1994) Is clumsiness a marker for Asperger's syndrome? Journal of Intellectual Disability Research 38(5): 519-527.
- Fombonne E (1999) The epidemiology of autism: A review Psychological Medicine 29(4): 769-786.
- Robbins TW, James M, Owen AM (1994) Cognitive deficits in progressive supranuclear palsy, Parkinson's disease and multiple systems atrophy in tests sensitive to frontal lobe dysfunction. J NeurolNeurosurg Psychiatry 57(1): 79-88.
- Lawrence AD, Sahakian BJ, Hodges JR (1996) Executive and mnemonic functions in early Huntington's disease. Brain 119(5): 1633-1645.
- Taylor AE, Saint-Cyr JA, Lang AE (1986) Frontal lobe dysfunction in Parkinson's disease. Brain 109(5): 845-883.
- Owen AM, James M, Leigh PN (1992) Fronto-striatal deficits at different stages of Parkinson's disease. Brain 115(6): 1727-1751
- Hutchinson J, Dalziel JM (2001) Flora of West Africa. The Crown agents for Colonies: London, UK.
- Weinberger DR, Gallhofer B (2002) Cognitive function in schizophrenia. Int Clinical Psychopharmacol 2: 111-1344.
- Ohayon M, Roth T (2003) Place of chronic insomnia in the course of depressive and anxiety disorders. J Psychiatry Res 37(1): 9-15.
- Ogunremi OO (1978) The subjective sleep patterns and dreams of Nigerians. Afr J Psychiatry 3: 113-119.
- Gozal D (1998) Sleep disordered breathing and school performance in children. J Pediatrics 102(3): 616-620.
- Abbott LC Winzer-Serhan UH (2012) Smoking during pregnancy: Lessons learned from epidemiological studies and experimental studies using animal models. Crit Rev Toxicol 42(2): 279-303.
- Burke H, Leonardi-Bee J, Hashim A, Pine-Abata H, Chen Y, et al. (2012) Prenatal and passive smoke exposure and incidence of asthma and wheeze: Systematic review and met analysis. Pediatrics 129(4): 735744.
- Heymann D, Stem Y, Tatarina-Nulman O, Dorrejo J, Gu Y (2016) The association between alcohol use and the progression of Alzheimer's disease. Current Alzheimer research 13(12): 1356-1362.
- Sadeeq AA, Ibegbu AO, Taura MG, Timbuak JA, Adamu LH, et al. (2013) Studies on the effects of mercury exposure on spatial learning and memory of adult wistar rats. International Journal of Pharmaceutical Science Invention ISSN (Online): 2319-6718.
- Fallarini, S, Miglio, G, Paoletti U (2009) Clovanoide and rosmarinic acid induce neuro-protective effects in in-vitro models of neuronal death. Br J pharmacol 157(6): 1072-1084.
- Bergen C (2008) Effects of poverty on cognitive functioning: a hidden neurologic epidemic. Neurology 7(6): 447-451.
- Ippolita CC, Shukitt-Hale B, Joseph JA (2000) Neurobehavioral aspects of antioxidants in aging. IJ of Developmental neurosci 18 (4-5): 367381.
- Noble KG, Houston SM, Kan E, Sowell ER (2012) Neural correlates of socioeconomic status in the developing human brain. Developmental science 15 (4): 516-527.
- Rogawski MA, Loscher W (2004) The neurobiology of anti-epileptic drugs. Nature reviews neuroscience 5 (7): 553-564.
- Elliott R, Sahakian BJ (1995) The neuropsychology of schizophrenia: relations with clinical and neurobiological dimensions. Psychol Med 25: 581-594.
- Dong Y, Fukuyama H, Honda M, Okada T, Hanakawa T (2000) Essential role of the right superior parietal cortex in Japanese kana mirror reading: an fMRI study. Brain 123(4): 790-799.
- Poldrack RA, Gabrieli JDE (2001) characterizing the neural mechanisms of skill learning and repetition priming: evidence from mirror reading. Brain 124(1): 67-82.
- Koepp MJ, Gunn RN, Lawrence AD, Cunningham VJ, Dagher A (1998) Evidence for striatal dopamine release during a videogame. Nature 393(6682): 266-268.
- Packard MG, White NM (1991) Dissociation of hippocampus and caudate nucleus memory systems by post training intracerebral injection of dopamine agonists. Behav Neurosci 105(2): 295-306.
- Davidson RJ, Kabat-Zinn J, Schumacher J, Rosenkranz, M, Muller D, et al. (2003) Alterations in brain and immune function produced by mindfulness meditation. Psychosomatic Medicine 65(4): 564-570.
- Fayol P (2003) Non-pharmacological treatment of neuro-behavioral disorders following severe traumatic brain injury. A commend literature review. Ann Readapts Med Phys 46(2): 97-103.
- Jivad N, Rabiei Z (2014) A review study on medicinal plants used in the treatment of learning and memory impairments. Asian Pacific Journal of Tropical Biomedicine 4(5): 112-124.
- Gogtay NJ, Bhatt HA, Dalvi SS, Kshirsagar NA (2002) The Uses and Safety of non-allopathic Indian Medicines. Drug safety 25(14): 10051019.
- Gedif T, Hahn HJ (2003) The Uses of Medicinal Plants in self-care in Rural Central Ethopia. Journal of Ethnopharmacology 87(2-3): 155161.
- Lorke D (1983) A new approach to practical acute toxicity test. Arch Toxicol 54(4): 275-286.
- Morris R (1984) Developments of a water-maze procedure for studying spatial learning in the rat. Journal of Neuroscience Methods 11(1): 47-60.
- Paylor R, Baskall-Baldini L, Yuva L, Wehner JM (1996) Developmental differences in place-learning performance between C57BL/6 and DBA/2 mice parallel the ontogeny of hippocampal protein kinase C. Behavioural Neuroscience 110(6): 1415-1425.
- Wishaw I, Jarrard L (1995) Similarities vs. differences in place learning and circadian activity in rats after fimbria-fornix section or ibotenate removal of hippocampal cells. Hippocampus 5(6): 595-604.
- Mcdonald RJ, White NM (1994) Parallel information processing in water maze: Evidence for independent memory systems involving dorsal striatum and hippocampus. Behaviour Neural Biology 61(3): 260-270.
- Aduema W (2016) Neurobehioural Effects of Chronic Consumption of Uncooked Black Eye Beans on Spatial Learning and Memory in Mice. Curr Trends Biomedical Eng & Biosci 1(2): 555556.






























