Effect of Different Physical Factors on Cassia Fistula Fruit Pulp Extract and their Herbal Formulation Efficacy
Deepa Hada* and Kanika Sharma
Department of Botany, Mohanlal Sukhadia University, India
Submission: October 05, 2017; Published: December 11, 2017
*Corresponding author: Deepa Hada, Department of Botany, Mohanlal Sukhadia University, India, Email: deepahada.hada52@gmail.com
How to cite this article: Deepa H, Kanika S. Effect of Different Physical Factors on Cassia Fistula Fruit Pulp Extract and their Herbal Formulation Efficacy. Glob J Pharmaceu Sci. 2017; 4(2): 555632. DOI: 10.19080/GJPPS.2017.04.555632
Abstract
In the present study, effect of different physical factors like sunlight, heat, pH and long-term storage on extract and herbal formulation has been studied to determine its stability under varying physical conditions. Extract and herbal formulation were exposed to varying conditions of the parameters chosen for a specific time period, and then observed the effect as a function of change in MIC of extract against the Alternaria solani. Active principles present in Cassia fistula are highly susceptible to change in physical environment. However it is found that extract and herbal formulation of Cassia fistula can be stored for 12 months, remains stable at alkaline pH, can stand with exposure to sunlight and high temperature. Hence a little favourable manipulation of physical conditions could improve its shelf life and can be used as fungicides for controlling microorganisms.
Keywords: Physical factor; Sunlight; Heat; Ph; Storage
Introduction
Amount of active constituents in the plant material affected by various physical factors, climatic conditions, geographical location of plants, seasonal variations, edaphic factors etc. Ashebir & Ashenafi [1]. Excess heating during extract preparation often affect biologically active substances such as flavonoids, essential oils and other heterogenous phytoconstituents present in the plant extract which might influence their respective activity Scalber [2]. Herbal formulation will be commercially viable, if its stability can be maintained at varying physical conditions. The prerequisite conditions for use of plant extracts in such formulations are that their physical and chemical properties should not undergo any drastic change due to change in temperature, pH or exposure to sunlight and it should have a long shelf life at least 6 months and there should not be any reduction in its antimicrobial activity. Hence, before using the plant extract for making any herbal formulations, detailed studies to check stability of extract in varying conditions of physical factors such as pH, temperature etc. and its effect on MIC should be conducted.
Several workers have checked the stability of extract in the presence of different physical factors. Gupta & Viswanathan [3] reported decrease in antimicrobial activity of garlic extract against B. cereus when stored at room temperature but when the same extract was stored at 8 °C the antimicrobial activity was not changed. Tyneca et al. [4] reported that antimicrobial activity of Allium ursinum juice decreases on storage above 4 °C. Moore & Atkins [5] suggested that inhibitory property of garlic extract was unaffected by storage temperatures. Shahi et al. [6] observed effect of pH on antidermatophytic activity of stored essential oils and found that efficacy of oils was enhanced at altered pH. Heat stable activity of combination of ethanolic extract of Cassia alata and Ocimum sanctum was reported by Ranganathan & Balajee [7]. Rath et al. [8] studied effect of high, temperature and 0.5M sucrose on the activity of turmeric oil. Effect of different temperature, autoclaving, illumination and pH values on ninety six plant extracts was investigated by Wang and Ke-Qiang [9] who reported that at higher temperature activity decreased while after steam sterilization there was no change in activity. Rong et al. [10] studied insecticidal activity of Ailanthus altissima extract and found that the activity is greater in light than in dark.
Patra et al. [11] studied effect of temperature and expiry of toxicity during storage of Citrus sinensis oil. Lee et al. [12] investigated heat and pH susceptibility of Chinese leaf extracts and found that heat treatment above 75 °C reduced the inhibitory activity while inhibitory activity is stable between pH 2.0 to 8.0. Similarly Di Mambro et al. [13] studied the combined effect of temperature and relative humidity on the antioxidant effect of different plant extracts. Doughari [14] reported significant increase in bioactivity of compounds of root extracts of Carica papaya L. directly proportional with increase in temperature and inversely proportional to increase in pH. Arabshahi et al. [15] studied the effect of pH, temperature and storage on the antioxidant activity of drumstick leaves (Moringa oleifera), mint leaves (Mentha spicata) and carrot tuber (Daucus carota).
Srinivasan et al. [16] reported that Allium sativum extract stored at room temperature showed inhibitory activity against the tested pathogens up to seven days. When the extract was stored at 4 °C, it exhibited moderate activity till 60 days and if the same extract was stored at 20 °C the antimicrobial activity decreased. He also reported that activity of the same extract decreased at alkaline pH. Mehrotra et al. [17] reported that bioactive components of ethanol extract of Syzizium aromaticum were stable over a wide range of pH values and temperatures. Magdy et al. [18] reported no change in activity of plant extracts exposed to different temperatures ranging from 4, 30, 60, and 90 °C. This showed that phytoconstituents are thermostable. Ghogare et al. [19] reported slight decrease in the antimicrobial activity of Z. officinale and A. sativum extracts on increasing the pH of the extract. Barpete et al. [20] observed that combination of low light intensity, phytagel as gelling agent and thidiazuron (TDZ)-a-naphthalene acetic acid (NAA) was very effective for high frequency shoot regeneration of Lathyrus sativus. It is very important to determine the effect of physical factors on extract as well as herbal formulation, to improve their storage condition and maintenance of efficacy for prolonged period. Hence, in the present study, effect of pH, storage, temperature, sunlight etc. on MIC of extract and herbal formulation has been studied to determine its stability under varying physical conditions. MIC and MFC of chloroform fraction of Cassia fistula fruit pulp was observed at 2.5mg/ml and 5mg/ml respectively.
Materials and Methods
Effect of physical factors such as heat, temperature, pH, sunlight etc. was studied by exposing the extract and herbal formulation to varying conditions of the parameters chosen for a specific time period, and then observing the effect as a function of change in MIC of extract against the test organism. Tubes containing MIC of extract, herbal formulation and extract free medium were maintained for comparison in each set of experiment against Al- ternaria solani. In the present study100% alcoholic crude extract and partially purified chloroform extract of Cassia fistula fruit pulp and best ratios (8, 12, 18, 22) of herbal formulation which is made by combining plant extract, elicitor (neem oil cake) and binder (Cow dung) were used for the experiments. All ingredients of herbal formulation were used in following ratio:
a. Formulation ratio no. 8 (100% alcohol crude extract (4ml): 100% neem oil cake (3ml): 100% cow dung (3ml)).
b. Formulation ratio no. 12 (100% alcohol crude extract (2ml): 100% neem oil cake (6ml): 100% cow dung (2ml)).
c. Formulation ratio no. 18 (Partially purified chloroform extract (3ml): 100% neem oil cake (3ml): 100% cow dung (4ml)).
d. Formulation ratio no. 22 (Partially purified chloroform extract (6ml): 100% neem oil cake (2ml): 100% cow dung (2ml)).
These extracts and herbal formulation were found to be most potent. Experiments were repeated thrice and three replicates were maintained.
Effect of sunlight
Effect of sunlight on the viability of extracts and herbal formulation was studied according to the method suggested by Wang & Ke-Qiang [9]. Sterile vials containing 5ml of 100% alcoholic crude extract, partially purified chloroform extract and herbal formulation (ratio no. 8,12,18,22) were placed in sunlight for 15h and 30h. After which effect on efficacy of extract and herbal formulation was assayed by tube dilution method.
Effect of heat
Efficacy of extract and herbal formulation was assayed according to the method suggested by Rath et al. [8]. Effect of dry heat was studied by exposing sterile glass vials containing 100% alcoholic crude extract, partially purified chloroform extract and herbal formulation (ratio no. 8,12,18,22) to 40 °C and 90 °C for 4h in hot air oven while in case of wet heat; extract and herbal formulation were kept at 50 °C and 100 °C in water bath for 4h. Effect on activity of extract and herbal formulation was then assayed by tube dilution method. One tube containing untreated extract as well as herbal formulation (room temperature) was maintained as control for comparison.
Effect of pH
Effect of varying pH i.e. 4, 7 and 9 on efficacy of extract and herbal formulation was studied by method suggested by Dixit et al. [21]. Natural pH of extract and herbal formulation is 7. 0. 0.1 N HCl and 0.1 NaOH were used to change the pH to 4 and 9 respectively. Culture medium was then added to tubes containing extract and herbal formulation and the tubes were inoculated with Alternaria solani. Inoculated tubes were incubated at 27±1 °C for 72 h and observed for change in herbal formulation and MIC of extract.
Effect of storage
Effect of storage on antifungal activity of extract and herbal formulation was assayed by method suggested by Rath et al. [8]. Extract and herbal formulation were stored at room temperature and change in their activity was assayed at regular intervals of 6 month up to 24 months by tube dilution method.
Results and Observations
The results of effect of different physical factors like sunlight, heat, pH and long-term storage on extract and herbal formulation of C. fistula fruit pulp are given in (Table 1-8).
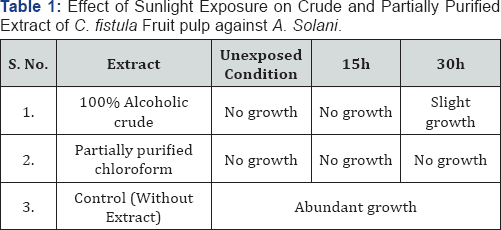
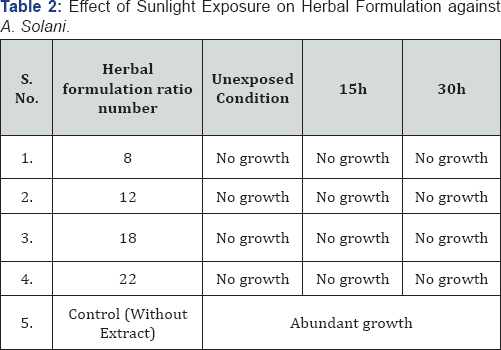

R.T. - Room Temperature
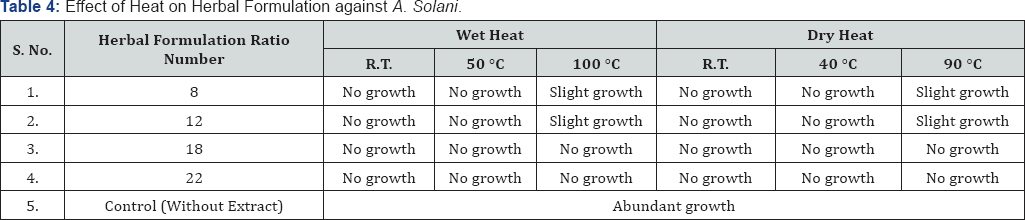
R.T. - Room Temperature
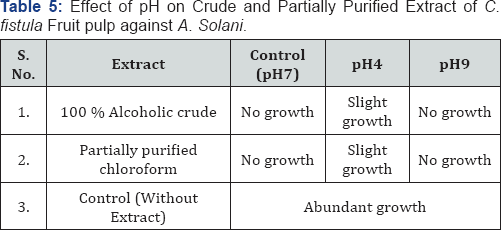
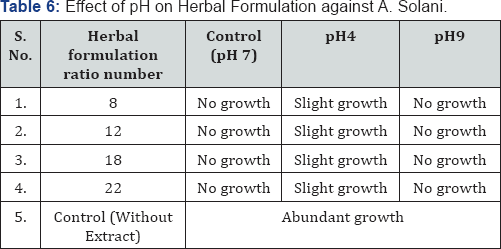

Table 1 & 2 shows that no changes in efficacy of chloroform extract and herbal formulation observed due to direct exposure to sunlight for 15h and 30h. In 100% alcoholic crude extract 15h exposure had no effect but after 30h exposure a slight decrease in activity was observed for A. solani. Table 3,4 depict the effect of wet as well as dry heat on extract and herbal formulation efficacy. Results indicate that 100% alcoholic crude extract and herbal formulation ratio number 8 and 12 up to 50 °C of wet heat and 40 °C of dry heat did not affect the activity of extract; however, heating at 100 °C of wet heat and 90 °C of dry heat for 4h resulted in slight decrease in extract and herbal formulation efficacy as a slight growth of test fungus was observed. Wet as well as dry heat treatment had no effect on activity of chloroform extract and herbal formulation ratio number 18 and 22.
Table 5 & 6 shows the result of effect of pH on the efficacy of plant extract as well as herbal formulation. There was no inhibitory effect observed on efficacy of extract and herbal formulation at neutral and alkaline pH up to 9 but there was decrease in antifungal activity at acidic pH (pH-4) against Alternaria solani.
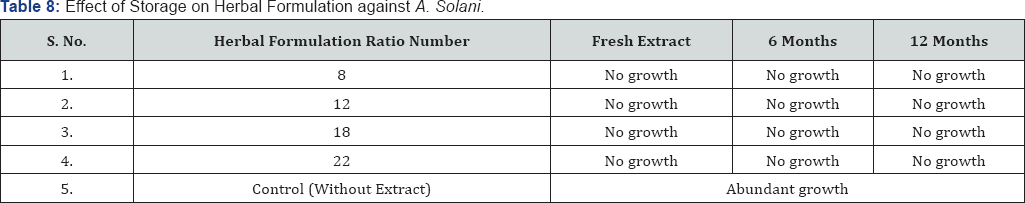
Table 7 & 8 shows the result of effect of long term storage of extract and herbal formulation at room temperature. Storage for 6 and 12 months had no effect on efficacy of extract and herbal formulation and the antifungal activity was same as the fresh extract.
Discussion
Herbal formulations using plant extracts are beneficial in the treatment of various diseases as they have no side effects as compared to synthetic antimicrobial drugs. Herbal formulations may be viable only when they have the ability to maintain stability and physical factors do not affect the activity of these formulations. Antimicrobial property of extract as well as herbal formulation may be affected by various physical factors such as pH, temperature, sunlight exposure etc. because all these factors are responsible for bringing the change in the chemical nature of compounds responsible for the antimicrobial activity.
Results showed that no change observed in antifungal activity of chloroform extract and herbal formulation after exposure to direct sunlight indicates that active principles of chloroform extract and herbal formulation are light stable and do not undergo photo oxidation. Whereas 100% alcoholic crude extract retained its antifungal potential up to 15h exposure of sunlight. Wang & Ke-Qiang [9] have reported similar results. Probably sunlight exposure do not destruct the active molecules of chloroform extract of Cassia fistula possess antifungal potential.
Effect of Heat on 100% alcoholic crude extract and herbal formulation ratio number 8 and 12 showed that the active principles can withstand the wet heat and dry heat up to 50 °C and 40 °C respectively. While prolonged exposure of extract with 100 °C wet heat and 90 °C dry heat destroyed its antifungal potential whereas it has no effect on chloroform extract and herbal formulation ratio number 18 and 22. Singh et al. [22] had also concluded the same for antifungal and antioxidative potential of Foeniculum vulgare volatile oil and its acetone extract. Magdy et al. [18] also reported that the activity of Cinnamomum cassia, Alium sativum, Syzygium aromaticum, Punica granatum, Citrus lemoniumn and Hibiscus sabdariffa plant extracts were not affected when exposed to different temperatures ranging from 4 °C, 30 °C, 60 °C and 90 °C. The temperature resistance studies indicate that the phytoconstituents are thermostable, but heating at 120 °C or beyond leads decrease/loss in the antimicrobial activity, this may be due to volatilization of components and/ or due to some physical and chemical changes in molecules of natural products during heating.
The antifungal activity of extract and herbal formulation of Cassia fistula fruit pulp was found to be stable at the pH 7 and 9. Decrease in the activity of the same at pH 4 was observed. These results suggest that the active principles of the extract are better active at neutral pH. Nishihara et al. [23] suggested that the presence of a high concentration of salt interfere with the binding of cationic peptides to the cell surface of B. subtilis, which are required for its growth. Yen & Duh [24] reported that a methanol extract from peanut hulls had a higher antioxidant activity at neutral and acid pH. Increase in activity of phytoconstituents in the presence of acidic medium has been reported by Doughari [14]. Azizah et al. [25] reported the antioxidant activity of different extracts from cocoa by-products was higher at alkaline pH.
Jeffery [26] also investigated the effect of various other physical factors like heat and temperature etc. on antimicrobial activity of pepper leaf extracts. Arabshahi et al. [15] suggested that antioxidant activity of extract of mint leaves, carrot and drumstick varies with the change in pH. Yang et al. [27] investigated the effect of pH on antibacterial activity of Propolis ethanol extract against Streptococcus mutans and reported that the active molecules are highly stable at acidic pH followed by neutral and then alkaline pH. Srinivasan et al. [16] also reported decrease in the antimicrobial activity of Allium sativum extract on increasing pH value and it was least at pH 9. Bayliak et al. [28] reported that antioxidant activity of aqueous extracts of Rosa canina, Rhodiola rosea, Hypericum perforatum and Gentiana lutea is decreased at alkaline pH while prooxidant activity increase at same pH.
Storage studies results suggest that there was no effect of long term storage on the efficacy of extract and herbal formulation. During storage combinations of physical factor not as much afflict the efficacy of extract as well as herbal formulation than individually affect. Arias et al. [29] investigated that aqueous and ethanolic extracts of Acacia aroma possess antibacterial activity against gram +ve and gram -ve bacteria and also evaluated that stored extracts have similar antibacterial activity as the fresh extracts.
Conclusion
The Results suggested that the active principles present in Cassia fistula are highly susceptible to change in physical environment. However it is found that it can be stored for 12 months, remains stable at alkaline pH, can stand with exposure to sunlight and high temperature. Hence a little favourable manipulation of physical conditions could improve its shelf life and can be used as fungicides for controlling microorganisms.
Acknowledgment
One of the authors (Deepa Hada) is thankful to University Grant Commission (UGC), New Delhi, India, for providing financial assistance.
References
- Ashebir M, Ashenafi M (1999) Assessment of the antibacterial activityof some traditional medicinal plants on some food-borne pathogens. Ethiopian J Health Dev 13(3): 211-216.
- Scalbert A (1991) Antimicrobial properties of tannins. Phytochem 30: 3875-3883.
- Gupta KC, Viswanathan R (1953) Combined action of streptomycin and chloramphenicol with plant antibiotics against Tubercle bacilli. Streptomycin and chloromphenicol with cepharanthene. II Streptomycin and allicin. Antibiot Chemother 5(1): 24-27.
- Tyneca Z, Syczesniak Z, Gelowniak (1993) the effect of variousenvironmental conditions on the antimicrobial activity of Allium ursinum. Planta Med 59: 701.
- Moore GS, Atkins RD (1977) The fungicidal and fungistatic effects of an aqueous garlic extract on medically important yeast-like fungi. Mycologia 69(2): 341-348.
- Shahi SK, Shukla AC, Dikshit A (1999) Antifungal studies of some essential oils at various pH levels for betterment of antifungal drug response. Curr Sci 77: 703-706.
- Rangnathan S, Balajee SAM (2000) Anticryptococcus activity of combination of extracts of Cassia alata and Ocimum sanctum. Mycoses 43(7-8): 299-305.
- Rath CC, Dash SK, Mishra RK, Charyulu JK (2001) Anti E. coli activity of turmeric (Curcuma longa L.) essential oil. Indian Drugs 38: 106-111.
- Wang S, Ke-Qiang C (2001) Studies on fungitoxic plant extract against Botrytis cinerea and other plant pathogens. Conference July 2-6 Agriculture research Institute Kromeriz Ltd., Czech Republic, Europe.
- Rong T, Frieda ER, Chris JP, Joel RC (2002) Plant growth regulatory effect and insecticidal activity of the extracts of the tree of Heaven (Ailanthus altissima L.). BMC Ecol 2: 1-7.
- Patra M, Shahi SK, Dikshit A (2003) Utilization of Pericarp of Citrus sinensis oil for development of natural antifungal against nail infection. Curr Sci 84(12): 1512-1515.
- Lee CF, Han CK, Tsau JL (2004) In vitro activity of Chinese leek extract against Campylobacter species. Int J Food Microbiol 94: 169-174.
- Di Mambro VM, Fonseca MJV (2005) Assays of physical stability and antioxidant activity of a topical formulation added with different plant extracts. J Pharm Biomed Anal 37(2): 287-295.
- Doughari JH (2006) Antimicrobial activity of Tamarindus indica Linn. Trop J Pharm Res 5(2): 597-603.
- Arabshahi SD, Devi DV, Urooj A (2007) Evaluation of antioxidant activity of some plant extracts and their heat, pH and storage stability. Food Chem 100: 1100-1105.
- Srinivasan D, Srinivasan S, Perumalsamy PL (2009) In vitro antibacterial activity and stability of garlic extract at different pH and temperature. Electro J Biol 5(1): 5-10.
- Mehrotra S, Srivastava AK, Nandi SP (2010) Comparative antimicrobial activities of Neem, Amla, Aloe, Assam Tea and Clove extracts against Vibrio cholerae, Staphylococcus aureus and Pseudomonas aeruginosa. J Med Plants Res 4(18): 2473-2478.
- Abu-Gharbia MA, M El-Maghraby O, El-Sayed M S, Abd El-Raheem WM, Emad AS (2014) Study of antimicrobial efficacy of some plant extracts against oral pathogens and comparative analysis of their efficiency against commercially available toothpastes and mouth rinses. J der Pharmazie Forschung 2(4): 6-19.
- Ghogare P, Anees Ahmed M, Srinivas R (2015) Studies on antimicrobial activity of spices and effect of temperature and ph on its antimicrobial properties. IOSR J Pharm Biol Sci 10(1): 99-102, 2015.
- Barpete S, Ozcan SF, Khawar KM, Ozcan S (2016) Effect of plant growth regulators and physical factors on in vitro high frequency regeneration of Grass pea. The J Anim Plant Sci 26(4): 1087-1093.
- Dixit A, Singh AK, Dixit, SN (1981) Effect of varying pH 4, 7 and 9 on efficacy of extract. J Antibact Antifung Agents 9: 9-10.
- Singh G, Maurya S, de Lampasona MP, Catalan C (2006) Chemical constituents, antifungal and antioxidative potential of Foeniculum vulgare volatile oil and its acetone extract. Food Control 17: 745-752.
- Nishihara T, Kosugi M, Matsue Y, Nishikawa J, Takasaki A, et al. (1992) Antimicrobial activity of positive colloids against food poisoning bacteria. J Antibac Antifung Agents 20: 241-245.
- Yen GC, Duh PD (1993) Antioxidantive properties of methanolic extracts from peanut hulls. J Am Oil Chem Soc 70(4): 383-386.
- Azizah AH, Ruslawati NM, Swee TT (1999) Extraction and characterization of antioxidants from cocoa by-products. Food Chem 64: 199-202.
- Jeffery AA (2006) Heat Stability of Pepper Leaf Extracts. J Amer Soc Hort Sci 131(1): 17-23.
- Yang HY, Ho WL, Chang CM, Chou CC (2007) Antibacterial activity of Propolis ethanol extract against Streptococcus mutans as influenced by concentration, temperature, pH and cell-age. J Food Drug Anal 15: 75-81.
- Bayliak MM, Burdyliuk NI, Lushchak VI (2016) Effects of pH on antioxidant and prooxidant properties of common medicinal herbs. Open Life Sci 11(1): 298-307.
- Arias ME, Gomez JD, Cudmani NM, Vattuone MA, Isla MI (2004) Antibacterial activity of ethanolic and aqueous extracts of Acacia aroma Gill. ex Hook et Arn. Life Sci 75: 191-202.






























