Melioration of Fertility in High Fat Diet Induced Obese Male Wistar Rats using Polyherbal Formulation
Dileep Paruchuru and Goverdhan Puchchakayala*
Department of Pharmacology, Vaagdevi College of Pharmacy, India
Submission: July 13, 2017; Published: August 02, 2017
*Corresponding author: Goverdhan Puchchakayala, Department of Pharmacology, Vaagdevi College of Pharmacy, India.
How to cite this article: Dileep P, Goverdhan P. Melioration of Fertility in High Fat Diet Induced Obese Male Wistar Rats using Polyherbal Formulation. Glob J Pharmaceu Sci. 2017; 3(3): 555615.
Abstract
Background: increased incidence of infertility due to various other complications is not properly diagnosed. A safe and effective medication to treat such complications is much needed.
Objectives: To develop a Polyhedral Formulation (PHF) to treat obesity induced infertility in male wistar albino rats and to meliorate hyperlipidemia induced by High Fat Diet (HFD).
Methods: Extracts of W. Somnifera roots, A. Racemosus roots and A. Paniculata leaves were used for PHF. For the experiment 24 male wistar albino rats of 4 weeks age were taken and divided into 4 groups. Three of the groups were fed with HFD for 4 weeks. For the next 2 weeks two of the groups were treated with 200 and 400mg/kg PHF respectively. Parameters like complete lipid profile, body weights, Body Mass Index (BMI), spermiogram, Lee index, Serum testosterone levels, energy consumption and litter size and antioxidant activity were estimated.
Results: On 4th week, a significant (P< 0.0001) increase in the body weight was observed in the HFD group compared to the control group. PHF treated groups showed a significant (P < 0.05*, P< 0.01**, P< 0.001***) difference in all the parameters when compared to HFD group.
Conclusion: From the observations of the study performed, it could be predicted that PHF exerted a significant anti-obese activity and an improved fertility rates in obese rats.
Keywords: Infertility; Obesity; Lee index; Adiposity Index; Spermiogram; Testosterone
Abbreviations: PHF: Polyhedral Formulation; HFD: High Fat Diet; BMI: Body Mass Index; IUIs: Intrauterine Inseminations; IVF: In-Vitro Fertilization; ICSI: Intracytoplasmic Sperm Injection; ART: Assisted Reproductive Techniques; CI: Confidence Interval; OR: Odds Ratio; IAEC: Institutional Animal Ethical Committee; VLDLC: Very Low Density lipoproteins-cholesterol ; HDLC: high-density lipoprotein cholesterol; LDLC: Low Density Lipoprotein Cholesterol; NIBP: Non-Invasive Blood Pressure; TBA: ThioBarbituric Acid; MDA: Malondialdehyde; NCLAS: National Centre for Laboratory Animal Sciences
Introduction
Obesity is defined as a medical condition where excess body fat has accumulated to the extent that it may have an adverse effect on health (WHO 2000). Relative weight and body mass index (BMI) are nearly identical and are reasonably used as estimates of body fatness as measured by percentage body fat [1]. BMI up to 24.9 kg/m2 is considered as normal, between 25.0-29.9kg/m2 is overweight and above 30kg/m2 is classified as obese condition. ‘Infertility is the inability of a sexually active, non-contracepting couple to achieve pregnancy in one year’ (WHO, 2000). About 15% of couples do not achieve pregnancy within 1 year and seek medical treatment for infertility. Less than 5% remain unwillingly childless. Infertility in general is defined as the absence of pregnancy after 1 year of unprotected intercourse, affects 1 in every 13 couples in the world population [2].
Among all the cases of infertility male factor infertility constitutes 25 to 30%, along with female factors, to another 30%. Recognized etiological factors of male infertility include cryptorchidism, testicular torsion or trauma, varicocele, seminal tract infections, antisperm antibodies, hypogonadotropic hypogonadism, gonadal dysgenesis, and obstruction of the reproductive channels [3]. In case of presumed male factor infertility, couples are often offered intrauterine inseminations (IUIs) or in vitro fertilization (IVF) with intracytoplasmic sperm injection (ICSI). These therapies are expensive and can be beyond the reach of many affected couples. Approximately 15% of couples do not achieve pregnancy within one year of unprotected sexual intercourse [4]. A male infertility factor is identified in about 50% of these cases and is solely responsible in 20% of couples.
Male infertility has been attributed to a variety of causes including lifestyle factors, gonadotoxin exposure, hormonal dysfunction, chromosomal disorders, varicocele, testicular failure, ejaculatory disorders, and obstruction. Evaluation of male infertility is important to identify a cause and provide treatment if the etiology is correctable. If a specific treatment is not available or the origin of the male factor infertility is not correctable, other options such as assisted reproductive techniques (ART) may exist. The pharmacist plays a vital role in identifying medications that contribute to male infertility, counseling the couple on medications used to treat infertility, and promoting healthy lifestyles that minimize infertility factors. The purpose of this article is to provide a broad overview of the etiology, evaluation, and treatment of male infertility [5].
In the United States, approximately 8 million couples are affected by infertility. For healthy young couples, the probability of achieving pregnancy within the first year of fertility-focused sexual activity is 84%. Despite advances in the diagnosis and treatment of infertility, the conception rate remains stable. A recent increase in demand for infertility services has been attributed to a greater awareness of treatment options, increased acceptance of infertility, and a trend toward delayed marriage and childbirth leading to more fertility issues [6]. In a secondary analysis of the Agricultural Health Study, male BMI was associated with infertility with an odds ratio (OR) of 1.12 (95% confidence interval [CI], 1.01 to 1.25), after correction for female BMI, male and female age, smoking status, alcohol use, and exposure to solvents and pesticides. There was a doseeffect relation with maximal effect in the BMI class 32 to 43kg/ m2 [7]. A data extracted from the Danish National Birth Cohort of 47,835 women was studied, overweight (BMI: 25 to 29.99kg/ m2) and obese (≥30kg/m2) men were more likely to report infertility with ORs of 1.15 (95% CI, 1.09 to 1.22) and 1.49 (95% CI, 1.34 to 1.64), after correction for female BMI and the age of both partners [8].
A secondary analysis of the Norwegian Mother and Child cohort study, showed the OR for infertility was 1.19 (95% CI, 1.03 to 1.62) for overweight men (BMI: 25 to 29.9 kg/m2) and 1.36 (95% CI, 1.12 to 1.62) for obese men, after correcting for coital frequency, female BMI, male and female age, smoking status, and various risk factors for female infertility. In a study of 91 male Japanese auto workers, Ohwaki and Yano found an increased incidence of not fathering a child per year of marriage with increasing male BMI [9]. In men presenting with wives to the delivery suites or to a fertility clinic, a history of paternity was associated with lower BMI when compared with no history of paternity [10].
Although current BMI in men appears to be correlated to reduced fertility, increased weight during adolescence was also be correlated with reduced fertility in adulthood. Jokela et al. [11] studied the effect of increased weight in adolescence on future fertility in 583 male participants. The predicted number of future children was 1.46 for normal weight, 1.4 for overweight, and 0.99 for the obese male adolescents. The association between the adolescent’s skin fold thickness and number of future children confirmed the prediction. The semen analysis is the cornerstone of the laboratory evaluation of infertile men. At least two properly performed semen analyses after 2 to 7 days of abstinence are necessary to confirm the diagnosis. A semen analysis not only evaluates the number and quality of the sperm ejaculated but the function of the accessory glands as well. Semen analysis includes testing of semen volume, sperm concentration, sperm motility, and sperm morphology.
Material and methods
Plant Material
A. Paniculata– dried leaves from Tekumatla village, Adilabad District, Andhra Pradesh, India. A. Racemosus – dried roots from Tekumatla village, Adilabad District, Andhra Pradesh, India. W.Somnifera – dried roots from Chintapally forests, Khammam District, Andhra Pradesh, India. All the plants were authenticated by Dr. E. Narasimha Murthy, Department of Plant Sciences, School of Life Sciences, University of Hyderabad, Hyderabad, Andhra Pradesh, India.
Animals and Vivarium Conditions
Experiments were conducted at Vaagdevi College of Pharmacy, Hanamkonda. Permission for all procedures was granted from the Institutional Animal Ethical Committee (IAEC). Experiments were carried out with twenty four male Wistar rats weighing 40-70g, were obtained from Sanzyme Pvt Ltd., Hyderabad. The animals were maintained under controlled conditions of temperature (23±2 oC), humidity (50±5%) and 12-h light-dark cycles. All the animals were acclimatized for seven days before the study. The animals were housed individually in sanitized polypropylene cages (47cm×34cm×20cm) containing sterile paddy husk as bedding. The rats had free access to water. They were fed with 60g/cage standard pellet diet (Mahaveer enterprises, Hyderabad) once a day. All the studies were conducted according to prescribed guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Government of India. Standard high fat diet (20% fat) was procured from National Institute of Nutrition, National Centre for Laboratory Animal Sciences (NCLAS).
Acute (oral) toxicity study
The toxicity studies as per the official protocol OECD 420 guidelines. Single dose treatment of the PHF was done to the normal animals and the general observations such as skin and fur color, alteration in the color of the eyes, mucous membrane, behavior, salivation, sleep, body weight and incidences of tremors, coma, diarrhea, lethargy, are recorded up to 2 weeks. Dose of 2000mg/kg doesn’t show any toxic symptoms, so according to guideline; it is considered as a LD50 cutoff value.
Experimental procedure
The rats were randomly divided into four groups each consisting of 6 rats. Group 1 (normal control group, 𝑛 = 6) received vehicle orally and fed with 60gm of normal diet per cage daily. Group 2 (disease control group, 𝑛 = 6) was fed with 60gm of HFD per cage daily for a period of 6 weeks. Group 3 (𝑛= 6) and Group 4 (𝑛 = 7) were fed with HFD for 4 weeks and then treated with 200 and 400mg/kg PHF (p.o) respectively for 2 weeks.
Estimation of serum lipid profile
Serum triglyceride, cholesterol, low density lipoproteincholesterol (LDL cholesterol), high-density lipoprotein cholesterol (HDL-cholesterol) and Very Low Density lipoproteins-cholesterol (VLDL-cholesterol) were determined using an Autoanalyser (Turbochem 100, USA) and commercial kits ( identi kits, Jeev diagnostics PVT LTD., Chennai, India).
Estimation of serum SGOT and SGPT
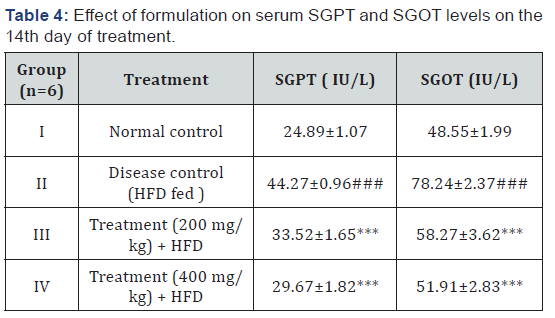
Effect of test formulation 1(200mg/kg) and test formulation 2 (400mg/kg) on serum SGPT levels in high fat diet induced metabolic syndrome in adult wistar rats. Samples are collected in the morning at the end of experiment (6th week) and analyzed using auto analyzer (CPC) using diagnostic kit.
Measurement of systolic blood pressure (SBP)
SBP was measured by using the tail-cuff method (IITC Life Science, Model 179 Non-Invasive Blood Pressure (NIBP) Multi Channel Blood Pressure System, USA). SBP was taken with the rats under conscious conditions at the beginning of the experiment and weekly. SBP values were the means of three measurements per rat. During acclimatization, the rats were placed in the restrainers for 10 to 15minutes at 30 °C daily to ensure the rats were comfortable with the restrainers, tail-cuff detector and warming chamber. Effect of high fat diet (HFD) and test formulation 1(2000mg/kg) and test formulation 2 (400mg/ kg) on SBP on high fat diet induced obese adult male wistar rats. The systolic blood pressure is recorded using NIBP apparatus in the morning (11 am).
Preparation of testicular homogenate
500mg of testes was homogenized in 5ml of ice-cold 1.15% KCl-0.01 M sodium, potassium phosphate buffer (pH 7.4). The homogenates (10%) were prepared by using Remi C24BL cooling centrifuge under cold conditions. The resultant homogenates were centrifuged at 12,000g for 30 min at 4 °C. The supernatant was collected and used for the experiments within 24 hours.
Thiobarbituric acid reactive substances (TBARS) assay
The reaction of thiobarbituric acid (TBA) with malondialdehyde (MDA), a secondary product of lipid peroxidation has been widely adopted as a sensitive assay method for measurement of lipid peroxidation in biological fluids. Aliquots of 0.5ml distilled water and 1 ml 10% TCA were added to a volume of 0.5ml brain homogenate, mixed well and centrifuged at 3000 rpm for 10min. To 0.2ml supernatant, 0.1ml TBA (0.375%) was added. The total solution was placed in a water bath at 80 °C for 40min and then cooled at room temperature. The absorbance of clear supernatant was measured at 532nm using spectrophotometer. Standard graph was plotted using TEPu (1, 1, 3, 3-tetra ethoxy propane).
Catalase Activity
Catalase activity was assessed by method of Luck, wherein the breakdown of Hydrogen peroxide is measured. In this 3ml of H2O2 phosphate buffer (0.05M. pH 7) was added to 0.05ml of the supernatant of tissue homogenate. The absorbance was measured after 1 minute at 240nm using UV spectophotometer. The results were expressed in 5 H2O2 activity calculated against blank.
DPPH (2, 2-diphenyl-1-picrylhydrazyl) assay
The free radical scavenging activity of the test drug was measured in-vitro by DPPH assay. In this measurement is made from bleaching of purple colored methanolic solution of DPPH. To 1ml of sample, 4ml of 0.004% w/v methanolic solution of DPPH was added and incubated for 30 minutes in dark. Then absorbance recorded at 517nm (Vogel, 2002). Inhibition of free radical by DPPH in % was calculated using the formula [% Inhibition = (A blank – A sample)/A sample × 100].
Estimation of serum testosterone: Serum was stored at -20 °C for analysis. The serum testosterone level was assayed using Coat- a - Count Radioimmunoassay kit (Active Testosterone RIA DSL-4000, Diagnostic System Laboratories Inc, Texas, USA) and expressed as ng/mL [12].
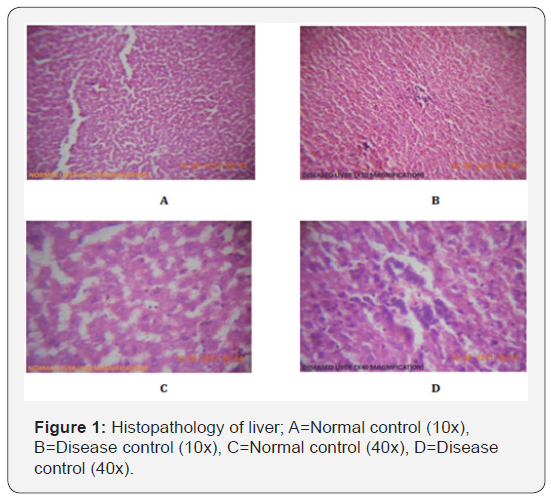
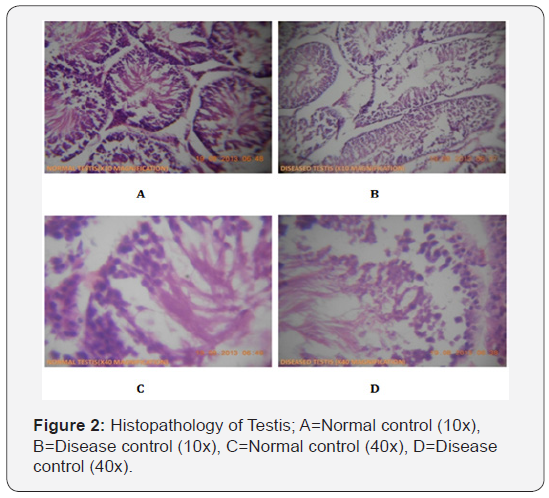
Histopathological studies: The animals were sacrificed by cervical dislocation on the 14th day of the treatment. The testes and epididymis were delivered via an abdominal section. While the testes were fixed in 10% formal saline solution, and appropriately processed for histological section using the Haematoxylin and Eosin staining techniques, the semen for seminal analysis was obtained from the caudal epididymis (Figure 1&2).
Statistical analysis
Statistical analysis was carried out using Graph Pad Prism software (version 6.03). The data was expressed as Mean±SEM. All the biochemical parameters were analyzed by one way ANOVA followed by Dunnet’s multiple comparison tests. A P value < 0.05 was considered as statistically significant.
Results
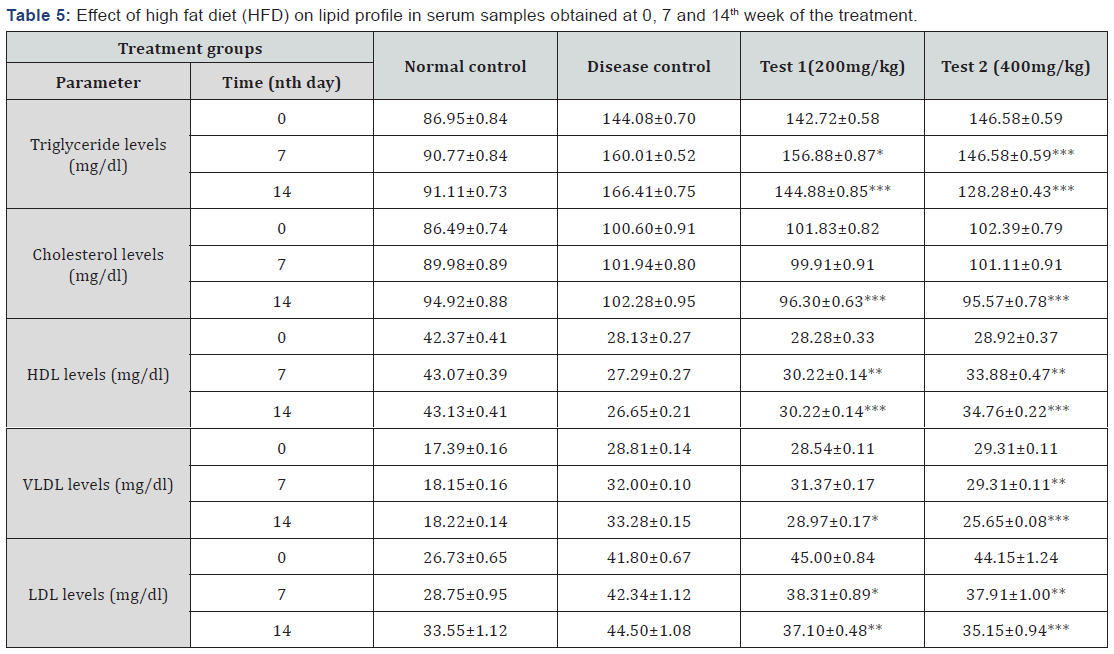
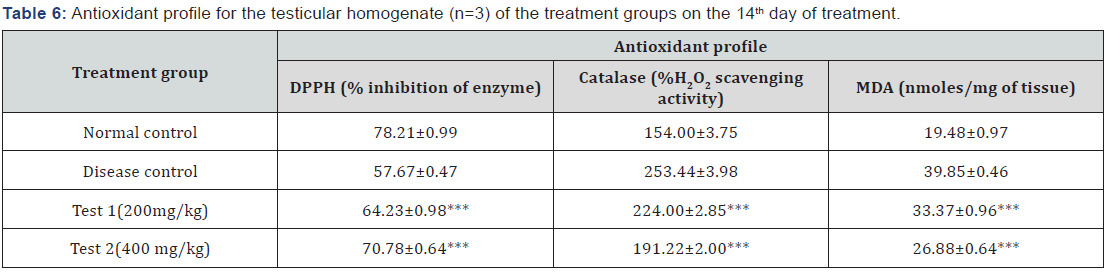
The mean body weight (gm) of rats in 4 groups was observed. Mean BMI (gm/cm2) and Lee index were calculated using the formulae (Body weight/nasoanal lenght2) and (Body weight0.33/ nasoanal length) [14]. The results were indicated in Table1. Systolic BP was recorded on the 4th week before treatment and 6th week after the treatment. All the results were noted as Mean±SEM in Table 2. At the end of 6th week, serum samples were collected and various serum parameters like Testosterone, liver function and lipid profile tests were performed and indicated as Table 3,4 & 5 respectively. Antioxidant profile of the testicular homogenate samples of 3 animals in each group was estimated on the last day. TBARS, Catalase and DPPH levels were measured and noted in Table 6.
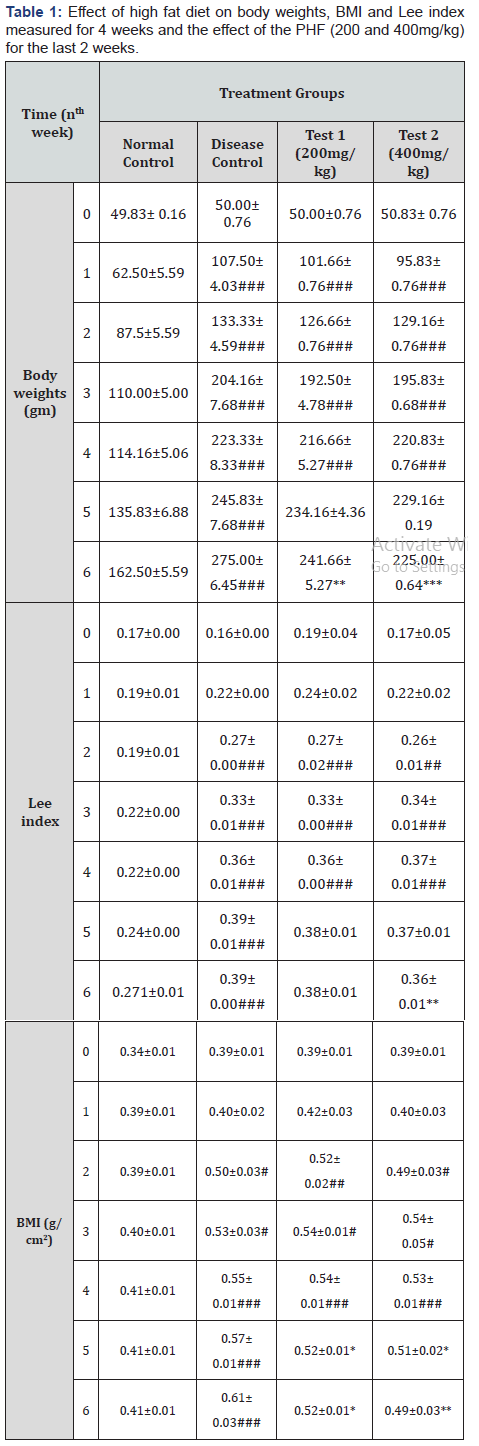
All the values are expressed in Mean and SEM of body weights (n=6). P < 0.05*, P< 0.01**, P< 0.001*** when compared to disease control and P < 0.05#, P< 0.01##, P< 0.001### when compared to normal control. Data was analyzed by one-way ANOVA followed by Dunnet’s test for multiple comparisons. BMI was calculated using the formula Body weight/ (Nasoanal length)2 and Lee Index as cube root (body weight/nasoanal length)/10.
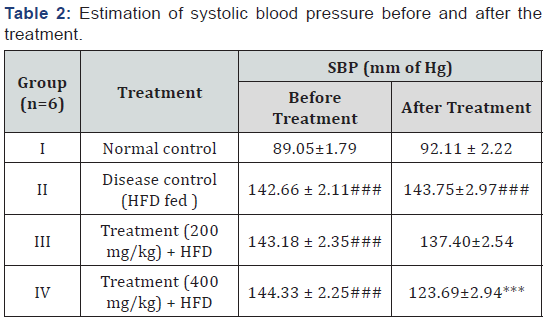
All the values are expressed in Mean and SEM of body weights (n=6). P <0.05*, P<0.01**, P<0.001*** when compared to disease control and P <0.05#, P<0.01##, P<0.001### when compared to normal control. Data was analyzed by one-way ANOVA followed by Dunnet’s test for multiple comparisons.
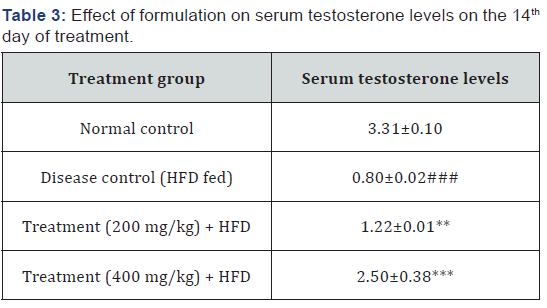
All the values are expressed in Mean and SEM of body weights (n=6). P <0.05*, P<0.01**, P<0.001*** when compared to disease control and P <0.05#, P<0.01##, P<0.001### when compared to normal control. Data was analyzed by one-way ANOVA followed by Dunnet’s test for multiple comparisons.
All the values are expressed in Mean and SEM of HDL-C levels (n=6). P <0.05*, P<0.01**, P<0.001*** when compared to disease control and P <0.05#, P<0.01##, P<0.001### when compared to normal control. Data was analyzed by one-way ANOVA followed by Dunnet’s test for multiple comparisons. . Data was analyzed by one-way ANOVA followed by Dunnet’s test for multiple comparisons.
All the values are expressed in Mean and SEM of triglyceride levels (n=6). P < 0.05*, P< 0.01**, P< 0.001*** when compared to normal control. Data was analyzed by one-way ANOVA followed by Dunnet’s test for multiple comparisons.
All the values are expressed in Mean and SEM of triglyceride levels (n=6). P <0.05*, P<0.01**, P<0.001*** when compared to normal control. Data was analyzed by one-way ANOVA followed by Dunnet’s test for multiple comparisons.
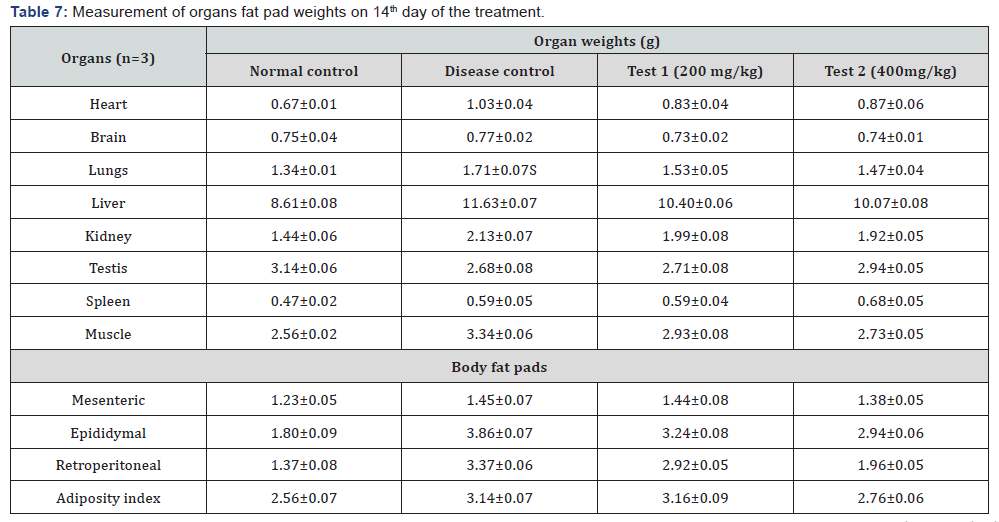
Various organ weights like brain, heart, lungs, liver, kidney, testis, spleen, muscle and fat pad weights were on the last day of the treatment and were noted in Table 7. Effect of test formulation 1(200mg/kg) and 2 (400mg/kg) on serum Testosterone levels in high fat diet induced metabolic syndrome in adult wistar rats. Samples are collected in the morning at the end of experiment (6th week) and analyzed using Count-a-count Radioimmunoassay kit.
Discussion
In the present work an attempt has been made to prepare a PHF from the respective plant extracts and evaluated the same for various parameters.
Acute toxicity studies
The observations indicated that the PHF did not produce any lethality and other signs of toxicity in normal animals. All three plants were used in India since ancient times. The extracts of W. Somnifera roots and A.Racemosus roots were already individually investigated and proved to have aphrodisiac activity. A.Paniculata leaves were proved to control the appetite and hence have anti obesity activity.
Effect of HFD on rats
High fat diet induced obesity model was selected for the study since it is a well established animal model for obesity which simulates the imbalance between energy intake and expenditure in humans [15]. At present, because of dissatisfaction with high costs and potentially hazardous side-effects, the potential of natural products for treating obesity is under exploration, and this may be an excellent alternative strategy for developing future effective, safe anti-obesity drugs. A variety of natural products, including crude extracts and isolated compounds from plants, can induce body weight reduction and prevent diet-induced obesity. Therefore, they have been widely used in treating obesity [16].
A growing body of evidence indicates that natural products having anti-obesity effects can be arranged into five categories based on their distinct mechanisms; they produce
I. Decreased lipid absorption,
II. Decreased energy intake,
III. Increased energy expenditure,
IV. Decreased pre-adipocyte differentiation and proliferation, or
V. Decreased lipogenesis and increased lipolysis [17].
Lipids are mostly consumed in the form of neutral fats, which are also known as triglycerides. The triglycerides are made up of a glycerol nucleus and free fatty acids. Triglycerides form major constituents in food of animal origin and much less in food of plant origin. Saturated fats increase blood cholesterol and thereby increase risk of atherosclerosis and coronary heart disease. Monounsaturated and polyunsaturated fats decrease blood cholesterol and reduce blood pressure. There is risk of obesity. Tran’s fats increase LDL and increase risk of atherosclerosis and coronary heart disease [18].
Immature male rats (low weight range) were selected as they have been shown more weight. Feeding with HFD caused significant increase in the body weight, body mass index, SGOT, SGPT, TC, LDL-C, VLDL, LDL and fall in HDL, serum testosterone, levels as compared to normal control animals.
Effect of PHF on body and organ weights
In the present study, a significant increase in body weight and BMI were observed in high fat diet groups. PHF treatment prevented the high fat diet induced adverse changes in testosterone levels suggesting its usefulness in metabolic syndrome and infertility. Studies using genetic models may further enhance understanding of the mechanism of action of the present PHF. The organ weights were measured in all the groups. This showed a significant reduction in the testicular weights in the diseased groups. There is no significant variation in the spleen weights in all the groups.
Effect of PHF on Lipid profile
The adiposity index was calculated as the sum of the weights of the fat pads divided by body weight; this quotient represented an estimation of body fat. High fat diet increases the expression of fatty acid catabolism related genes in the small intestine which is associated with development of obesity [19]. In the present study also, high fat diet increased the TC and TG levels and decreased the HDL-C levels. PFH decreased the TC and TG levels and increased the HDL-C levels as compared to high fat diet group. Serum SGOT, SGPT levels were altered (increased) in high fat diet group as compared to normal pellet diet group, PHF decreased the serum SGOT, SGPT levels.
Effect of PHF on Antioxidant profile
The antioxidant profile including DPPH, MDA and Catalase were estimated in the testicular homogenate of the rats on the 14th day of the treatment. The results observed were convincingly showing that the apoptotic rates in testis were significantly reduced in the HFD treated groups. This indicates that the spermatogenesis process gets accelerated leading to the increase of the sperm count and so reducing the infertility state in obese condition [20].
Effect of PHF on Blood Pressure
The NIBP results suggest that there is a significant increase in the blood pressure in the HFD group when compared to that of normal control group. PHF treatment reduced the hypertensive symptoms almost to the normal levels. Shalini et al. determined the antihypertensive activity of Ashwagandha [21].
Effect of PHF on the male reproductive parameters
In recent years, reproductive parameters in men were associated with the body mass index (BMI) in many studies, showing that increased BMI is related to poor semen quality, decreased sperm concentration [22] and decreased sperm cells motility [23]. On the other hand, some works showed little or no relation between obesity and sperm concentration, motility or morphology in men [24], even when serum reproductive hormone levels are altered. In the present observations, the sperm motility and concentration were increased significantly in the PHF treated groups when compared to that of the HFD fed diseased group. The mean litter size of the male rat treated with 400mg/kg PHF when mated with three normal female rats was higher than the normal male rats and that of the diseased rat was less than the normal male rats. Nadkarni explained that Ashwagandha infuses fresh energy and vigor in a worn-out system, due to any disease or from overwork and thus prevents premature ejaculation [25].
Histopathological studies
No significant change was observed in the histopathology of the liver slides of normal and the treatment groups. The Histopathological studies of the testis samples reveled that a significant destruction of seminiferous tubules was noted in the microscopic observation of the HFD fed group.
Conclusion
The present study demonstrated that the PHF had a therapeutic effect on improving the fertility and reducing the obesity state of the rats by increasing the testosterone levels and decreasing the lipid levels.
References
- Gray DS, Fujioka K (1991) Use of relative weight and Body Mass Index for the determination of adiposity. J Clin Epidemiol 44(6): 545-550.
- Stephen EH, Chandra A (2006) Declining estimates of infertility in the United States: 1982-2002. Fertil Steril 86(3): 516-523.
- Oehninger S (2000) Clinical and laboratory management of male infertility: an opinion on its current status. J Androl 21(6): 814-821.
- https://uroweb.org/wp-content/uploads/17-Male-Infertility_LR1.pdf
- American Urological Association (2010) The evaluation of the azoospermic male: AUA best practice statement. Revised 2010.
- Kim HH, Schlegel PN, Goldstein M (2010) Infertility: the male. In: Legato MJ. Principles of Gender-Specific Medicine (2nd edn) Amsterdam, Netherlands: Elsevier 366-80.
- Sallmén M, Sandler DP, Hoppin JA, Blair A, Baird DD (2006) Reduced fertility among overweight and obese men. Epidemiology 17(5): 520- 523.
- Ramlau-Hansen CH, Thulstrup AM, Nohr EA, Bonde JP, Sørensen TI, et al. (2007) Sub fecundity in overweight and obese couples. Hum Reprod 22 (6): 1634-1637.
- Ohwaki K, Yano E (2009) Body mass index as an indicator of metabolic disorders in annual health checkups among Japanese male workers. Ind Health 47(6): 611-616.
- Pauli EM, Legro RS, Demers LM, Kunselman AR, Dodson WC, et al. (2008) Diminished paternity and gonadal function with increasing obesity in men. Fertil Steril 90(2): 346-351.
- Jokela M, Kivimaki M, Elovainio M, Viikari J, Raitakari OT, et al. (2007) Body mass index in adolescence and number of children in adulthood. Epidemiology 18(5): 599-606.
- Dechaud H, Lejeune H, Garoscio-Cholet M, Mallein R, Pugeat M (1989) Radioimmunoassay of testosterone not bound to sex-steroid-binding protein in plasma. Clin Chem 35(8): 1609-1614.
- Nermine SN, Mona HM, Hala ME (2012) A Histological and Morphometric Study of Monosodium Glutamate Toxic Effect on Testicular Structure and Potentiality of Recovery in Adult Albino Rats. Res J Biol 2(2): 66-78.
- Rogers P, Webb GP (198) Estimation of body fat in normal and obese mice. Br J Nutr 43(1): 83-86.
- Sampey BP, Freemerman AJ, Zhang J, Kuan PF, Galanko JA, et al. (2012) Metabolic Profiling Reveals Mitochondrial-Derived Lipid Biomarkers that Drive Obesity-Associated Inflammation. PLoS One 7(6): 38812.
- Moro CO, Basile G (2000) Obesity and medicinal plants. Fitoterapia 71(1): 73-82.
- Srujana R, Mary Anne DF, Clifton AB (2008) Phytochemicals and regulation of the adipocyte life cycle. J Nutr Biochem 19(11): 717-726.
- Rajalakshmi R, Vijayavageesh Y, Nataraj SM, Muralidhar, Srinath CG (2012) Heart rate variability in Indian obese young adults. Pakistani J Physiol 8(1): 39-44.
- Shoko K, Hisashi Y, Tohru N, Michio K (2006) Interspecific variation in vessel size, growth and drought tolerance of broad-leaved trees in semi-arid regions of Kenya. Tree Physiol 26(7): 899-904.
- Dogan A, Bekir SP, Nihat U, Fikret E, Sahin K, et al. (2013) Weight loss and melatonin reduce obesity-induced oxidative damage in rat testis. Adv Androl 2013: 836121.
- Shalini K, Agatha B, Paramjit C (2012) Effect of Ashwagandha (W. Somnifera) Root Powder Supplementation in Treatment of Hypertension. Ethnomedicine 6(2): 111-115.
- Jensen TK, Andersson AM, Jorgensen N, Andersen AG, Carlsen E, et al. (2004) Body mass index in relation to semen quality and reproductive hormones among 1,558 Danish men. Fertil Steril 82(4): 863-870.
- Kort HI, Massey JB, Elsner CW, Mitchell-Leef D, Shapiro DB, et al. (2006) Impact of Body Mass Index Values on Sperm Quantity and Quality. J Androl 27(3): 450-452.
- Chavarro JE, Toth TL, Wright DL, Meeker JD, Hauser R (2010) Body mass index in relation to semen quality, sperm DNA integrity, and serum reproductive hormone levels among men attending an infertility clinic. Fertil Steril 93(7): 2222-2231.
- Nadkarni AK, Nadkarni KM (1976) Indian Materia Medica. Popular Prakashan Pvt. Ltd., Bombay, India 1: 1293-1294.






























