Comparative In-Vitro Equivalence Evaluation of Fexofenadine Hydrochloride 120mg Generic Tablets Marketed in Bangladesh
Sabreena Chowdhury Raka1*, Arifur Rahman2 and BSM Bodiuzzaman1
1Department of Pharmacy, Daffodil International University, Bangladesh
2Department of Pharmacy, BRAC University, Bangladesh
Submission: May 26, 2017; Published: June 22, 2017
*Corresponding author: Sabreena Chowdhury Raka, Department of Pharmacy, Daffodil International University, Bangladesh, Tel: +8801676033062; Email: raka.pharmacy@diu.edu.bd
How to cite this article: Sabreena C R, Arifur R, B Bodiuzzaman. Comparative In-Vitro Equivalence Evaluation of Fexofenadine Hydrochloride 120mg Generic 000022 Tablets Marketed in Bangladesh. Glob J Pharmaceu Sci. 2017; 3(1): 555602. DOI: 10.19080/GJPPS.2017.03.555602
Abstract
Fexofenadine hydrochloride, a piperidine derivative, is indicated to relieve signs and symptoms related with seasonal allergic rhinitis, such as rhinorrhoea, sneezing nose, throat and itchy eyes. The aim of my study is to establish the pharmaceutical equivalence of different brands of fexofenadine hydrochloride 120mg film coated tablets available in Bangladesh. The quality control parameters which are studied are weight variation test, thickness, friability, hardness, disintegration, dissolution and assay specified by British and United State Pharmacopoeia. The assay value was determined by HPLC. Out of the tablets from six evaluated pharmaceutical companies, the potency of fexofenadine hydrochloride tablets from four companies was found to be satisfactory and two was poor of the range.
The in-vitro dissolution studies of fexofenadine hydrochloride 120mg tablets were carried out 0.1N HCl for 10 and 30minutes using USP- II method. Three samples showed more than 60% drug release within 10 minutes and more than 80% drug release within 30minutes. During this evaluation, no major problem was found in weight variation, thickness and friability. Among six samples, five showed hardness within range but one sample did not meet up the hardness level. The % RSD of weight variation of six brands of fexofenadine hydrochloride were in the range of ≤2. Disintegration time for all brands was within range 30minutes and complies with the BP/USP recommendation. From the investigation, we can conclude that 70% of the pharmaceutical companies in the urban area of Bangladesh produce standard drugs, whereas 30% companies provide substandard drugs.
Keywords: Substandard; H1-receptor antagonist; Friability; Dissolution; Resistance; Pharmacopoeia
Introduction
The WHO has been tracking and documenting the incidences of substandard drugs. The records show that problems of substandard and counterfeit drugs are on increase as 50% of all reported cases occurred in the period 1993 to 1997 [1]. Most of these incidences (70%) were reported in developing countries. The report identifies that the causes of the poor quality of drugs in about 50% of all the cases the formulations did not contain any drug, 20% contained the wrong active ingredient and 10% the wrong amount of the active ingredient. Only in 5% of the reported incidences, the drugs did contain the right active ingredient in the correct amounts but were judged substandard by failing other quality tests [2]. 9% of pharmaceutical sales in Bangladesh have been traced to counterfeit or low-quality drugs, openly sold in pharmacies [3].
Fexofenadine is a long lasting H1receptor antagonist (antihistamine) which has a selective and peripheral H1- antagonist action [4]. This study helps to evaluate and compare the quality control parameters of oral fexofenadine hydrochloride 120mg tablets of six pharmaceutical companies in Bangladesh. Among those two were top ranked pharmaceutical companies (urban), two medium ranked pharmaceutical companies (urban, rural), one multi-national pharmaceutical company (urban) and one low ranked pharmaceutical company (rural) marketed in Bangladesh. This present study provides a comprehensive knowledge about the hardness, friability, weight variation, disintegration, dissolution, percentage of potencies of fexofenadine hydrochloride 120mg tablets and compares these values with their specifications according to the respected pharmacopoeias. One-Way ANOVA was applied and results were assessed. This study will help both health practitioners and consumers to select quality products. Also, this study can provide some information for Drug Control Authority of Bangladesh to evaluate the overall quality status of fexofenadine hydrochloride marketed in Bangladesh.
Materials and Methods
Sample collection
The samples for the investigation where purchased from different retail pharmacies from the following areas (Farmgate, Kolabagan, Sobhanbag, etc.) of Dhaka city. All the purchased samples were stored in the favourable conditions and away from direct sunlight. The samples were taken and verified before carrying out any type of experiment on them.
Physical parameters
All physical tests parameters for the tablets are done according to USP (United States Pharmacopeia) and BP (British Pharmacopeia) [5]. The parameters are shape, color, appearance and size (Table 1).
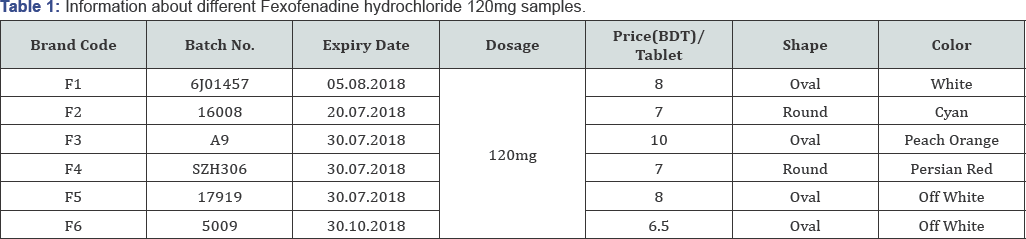
Diameter and thickness
The thickness of a tablet is the only dimensional variable related to the process. Tablet thickness is consistent batch to batch or within a batch only if the tablet granulation or powder blend is adequately consistent in particle size and size distribution, if the punch tooling is of consistent length, and if the tablet press is clean and in good working order. Tablet thickness should be with in ±5% variation of standard value [6].
Hardness
Tablets need a certain amount of hardness and resistance to friability, to withstand mechanical shocks of handling during manufacture, packaging and shipping [7]. Monitoring of tablet hardness is particularly important for drug products. Because it can possess real or potential bioavailability problems or that are sensitive to altered dissolution release profile.
Friability
Friability is the loss of weight of tablet in the container due to removal of fine particles from the surface during transportation or handling. The laboratory friability tester was known as Roche friabilator. Conventional compressed tablets that lose less than 0.5 to 1.0% of their weight are generally considered acceptable [8]. The percent friability was calculated by the following formula:
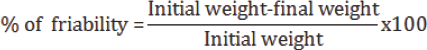
Weight variation
Weight variation test is carried out in order to ensure uniformity in the weight of tablets in a batch. A fundamental quality attribute for all pharmaceutical preparations is the requirement for a constant dose of drug between individual tablets. 10 tablets of each batch were taken and weighted individually by an analytical balance. The average weight of the tablets was calculated. Then % of weight variation is calculated by using following formula [9]
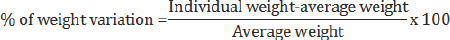
In this way, the weight variation for 6 different brands of tablets was measured and the observed value for each sample was recorded.
Disintegration
The first important step toward solution is breakdown of the tablet into smaller particles or granules a process known as disintegration. The time that it takes a tablet to disintegrate is measured in a device described in the USP [10].
In-vitro dissolution study
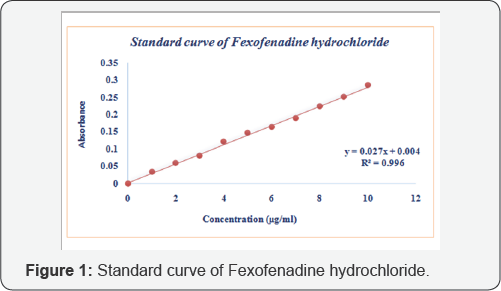
Release rate of fexofenadine hydrochloride tablet was carried out according to the general procedure of United States Pharmacopoeia (USP). Samples of dissolution fluid were withdrawn and analyzed by UV Spectrophotometer [11].The amount of drug present in the sample was calculated with the help of straight-line equation obtained from the calibration curve of Fexofenadine hydrochloride (Figure 1).
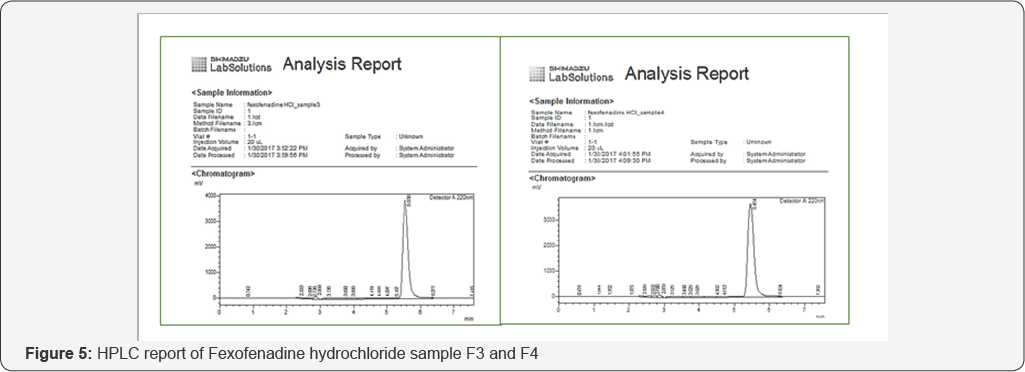
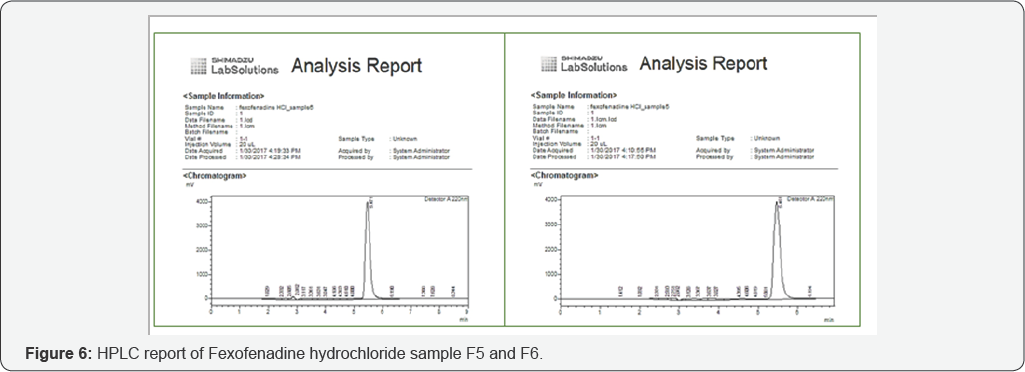
Assay of fexofenadine hydrochloride by HPLC
An accurately weighed quantity of USP fexofenadine hydrochloride RS was dissolved in diluent to attain a solution having a known concentration of about 1.0mg per ml. A standard and sample solution was injected consequently into suitable column of HPLC [12]. The content of drug present in each sample was calculated by comparing both the peak areas of active fexofenadine hydrochloride present in the standard preparation and prepared sample [13].
To calculate the quantity the formula was,

Results and Discussion
Price variation
Price fluctuation of different brands of fexofenadine hydrochloride was investigated during medicine collection. The 100 highest price variation was found for brand F3 with a maximum price of 10 taka per tablet and minimum of 6.5 taka per tablet for brand F6 (Figure 2) [14,15]. To prevent possible problems related to tablet weight and content uniformity, determination of the diameter and thickness of the tablets are very vital at an early stage. Among six brands, brand F1 had the highest average diameter (7.86mm) whereas brand F6 had the lowest average diameter (5.62mm) which makes it difficult for patient's to swallow. The average thickness was found to be between the ranges of 6.06-5.02mm (Table 2) [16].

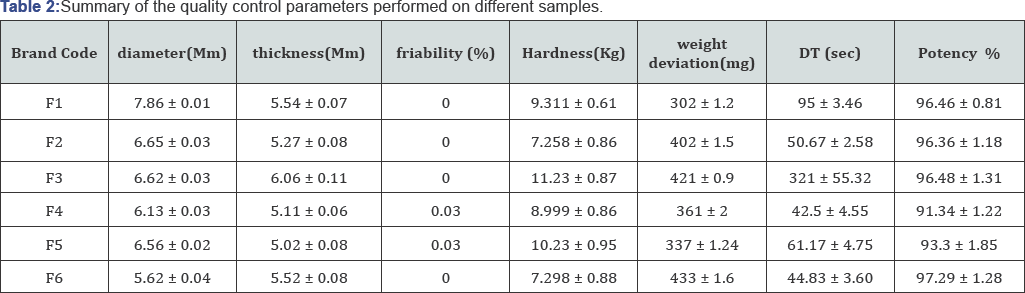
*Values are expressed as mean ± SD.
Hardness test
Materials used, amount of binder and pressure applied between the upper and lower punches during the process of compression give rise the hardness of a tablet. Hardness testing plays an essential role in product development and subsequently in quality control. High hardness value indicates increased disintegration time and decreased dissolution times, and vice versa [17-25]. Hardness of a tablet is not a reliable indicator for measuring tablet strength as formulations vary among different manufacturers and considered as non-compendial test. Tablet hardness was found between 11.23-7.26kg. Brands F1, F4 and F5 were satisfactory for hardness but brands F2, F3and F6 did not comply with this requirement (Figure 3).
Friability test
Friability testing is considered to assess the capability of the tablet to withstand abrasion during packaging, handling and shipping which can lead to capping, chipping, abrasion or even breakage of the tablets [26]. It is now included in the USP [27] was found that 6 different brand of fexofenadine tablets were in as a compendial test and the specification for friability is 1%. It accordance with the stated U.S.P guideline (Table 2).
Weight variation test
The weight uniformity test is a satisfactory method of determining the drug content uniformity in tablets. Among them, brand F6 did not meet the specification of ±10% because of its minimum deviation of 1.6%, but the other brands have average weight within the acceptable limit (Figure 4).
Disintegration test
If a product fails to disintegrate, it will presumably fail dissolution criteria because the disintegration tests do serve as a component in the overall quality control of tablets manufacturing. According to BP specification, film coated tablets should disintegrate within 30min, while the USP specifies that both uncoated and film coated tablets should disintegrate within 30min [28]. All brands of fexofenadine hydrochloride tablets were immediate release tablets and maximum time for disintegration was found 321sec in case of brand F3.
Dissolution test
To comply with USP standard for Fexofenadine hydrochloride at least 60% of must be dissolved within 10 minutes and at least 80% must be dissolved within 30minutes [29]. Interbrand comparison showed that brand F1 and F5 had maximum drug release within the first 10minutes (90.11%, 95.86%) in dissolution test, while brand F2 released only 34.13% of drug after this time. After the 30minutes interval, brand F1 and F5 showed maximum drug release (95.86%, 106.58%) and brand F2 exhibited minimum drug release (43.51%). From the data, it can be assumed that variation in dissolution profile of different brands due to manufacture by different companies using different excipients in different ratio [30,31] (Table 3).
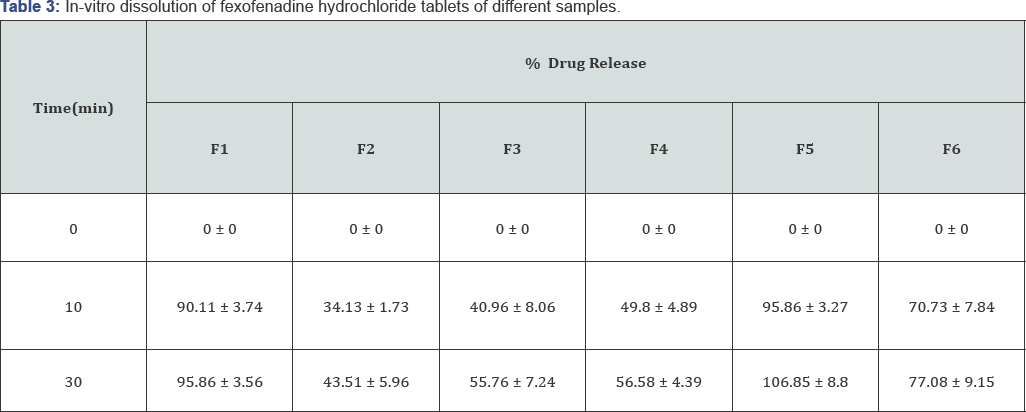
*Values are expressed as mean ± SD.
Assay of different brands of fexofenadine hydrochloride by HPLC
Conclusion
Although Fexofenadine has been manufactured in generic form since 2011, it is the most popular choice of antihistamine used for the treatment of allergy symptoms, such as hay fever, nasal congestion, and urticarial. Close monitoring of different process in pharmaceutical industries will reduce the production time and cost, as well as will improve the quality of the produce. Out of the tablets of six evaluated pharmaceutical companies, the potency of fexofenadine hydrochloride tablets from four companies was found to be satisfactory and two was poor of the range. Finally from the testing, we can conclude that 70% of the companies in the urban area of Bangladesh are of standard quality, whereas 30% companies provide substandard drugs. It was noted that some of the local companies has very low price as compared to the multi-national companies having the same good quality but some local companies has nearly the same price as that of multi-national with low price.
The drug administration should make new policy and drug acts to stop adulteration and control substandard drugs. Extend their regulatory mandate to cover manufacturers of marketed pharmaceutical product particularly with regard to inspection of GMP compliance, and impose rigorous requirements for labelling and certificates of analysis for consignments moving in international commerce. Introduce regulations that require APIs and all other starting materials intended for the formulation of medicinal products to be clearly labelled "for pharmaceutical use" or with a suitable pictogram. However, products of some less known local pharmaceutical industry needs developments in quality and should be strictly monitored for manufacturing of substandard drugs.
References
- Holgate ST, Canonica GW, Simons FE, Taglialatela M, Tharp M, et al. (2003) Consensus Group on New-Generation Antihistamines (CONGA): present status and recommendations. Clin Exp Allergy 33(9): 1305-1324.
- Church DS, Church MK (2011) Pharmacology of Antihistamines. World Allergy Organ J 4(3): S22-S27.
- Meltzer EO (1997) Prevalence of allergic rhinitis in the United States. J Allergy Clin Immunol 99(6): S808-S814.
- Haye R, Høye K, Berg O, Frønes S, Ødegård T (2005) Morning versus evening dosing of desloratadine in seasonal allergic rhinitis: a randomized controlled study [ISRCTN23032971]. Clin Mol Allergy 3(1): 3.
- Devillier P, Roche N, Faisy C (2008) Clinical pharmacokinetics and pharmacodynamics of desloratadine, fexofenadine and levocetirizine: a comparative review. Clin Pharmacokinet 47(4): 217-230.
- Yasir M, Asif M, Kumar A, Aggarval A (2010) Biopharmaceutical Classification System: An Account. International Journal Pharm Tech, Research 2(3): 1681-1690.
- Dahan A, Miller JM, Amidon GL (2009) Prediction of solubility and permeability class membership: Provisional BCS classification of the world's top oral drugs. AAPS J 11(4): 740-746.
- Etman MA, Gamal M, Nada AH, Shams EMA (2014) Formulation of Desloratadine Oral Disintegrating Tablets. Journal of Applied Pharmaceutical Science 4(11): 054-061.
- Hibaezzathamed, Kamal BA (2014) Preparation and in-vitro evaluation of desloratadine floating tablets. International Journal of Pharmacy and Pharmaceutical Sciences 6(9): 154-159.
- Buck ML (2011) Loratadine and desloratadine use in children. Pediatr Pharm 17(5): 1-4.
- Kreutner W, Hey JA, Anthes J, Barnett A, Young S (2000) Preclinical pharmacology of desloratadine, a selective and nonsedating histamine H1 receptor antagonist. 1st communication: receptor selectivity, antihistaminic activity, and antiallergenic effects. Arzneimittelforschung 50(4): 345-352.
- Molimard M, Diquet B, Benedetti MS (2004) Comparison of pharmacokinetics and metabolism of desloratadine,fexofenadine, levocetirizine and mizolastine in humans. Fundam Clin Pharmacol 18(4): 399-411.
- Purohit A, Melac M, Pauli G, Frossard N (2003) Twenty-four hour activity and consistency of activity of levocetirizine and desloratadine in the skin. Br J Clin Pharmacol 56(4): 388-394.
- Vuurman EF, Rikken GH, Muntjewerff ND, de Halleux F, Ramaekers JG (2004) Effects of desloratadine, diphenhydramine and placebo on driving performance and psychomotor performance measurements. Eur J Clin Pharmacol 60(5): 307-313.
- Simons FER, Fraser TG, Maher J, Pillay N, Simons KJ (1999) Central nervous system effects of H1- receptor antagonists in the elderly. Ann Allergy Asthma Immunol 82(2): 157-160.
- Kreutner W, Hey JA, Chiu P, Barnett A (2000) Preclinical pharmacology of desloratadine, a selective and nonsedating histamine H1 receptor antagonist. 2nd communication: lack of central nervous system effects. Arzneimittelforschung 50(5): 441-448.
- Banfield C, Padhi D, Glue P, Herron JM, Statkevich P, et al. (2000) Electrocardiographic effects of multiple high doses of desloratadine. Journal of Allergy and Clinical Immunology 104(1): S383.
- Posner J, Griffin JP (2011) Generic substitution. Br J Clin Pharmacol 72(5): 731-732.
- Bano R, Gauhar S, Naqvi SBS, Mahmood S (2011) Pharmaceutical evaluation of different brands of levofloxacin tablets (250 mg) available in local market of Karachi (Pakistan). International Journal of Current Pharmaceutical Research 3: 15-22.
- Mendis S, Abegunde D, Yusuf S, Ebrahim S, Shaper G, et al. (2005) WHO study on Prevention of REcurrences of Myocardial Infarction and StrokE (WHO-PREMISE). Bull World Health Organ 83(11): 820-828.
- Du J, Hoag SW (2003) Characterization of excipient and tableting factors that influence folic acid dissolution, friability, and breaking strength of oil- and water soluble multivitamin with mineral tablets. Drug Drug Dev Ind Pharm 29(10): 1134-1147.
- United State Pharmacopoeia (2014) United States Pharmacopoeial Convention. Maryland, USA.
- British Pharmacopeia (BP) (2013) Her Majesty's Stationary Office, London, UK.
- Akgeyik E, Kaynak MS, Jelebier m, Altinoz S, Çahin S (2016) Evaluation of pharmaceutical quality of conventional dosage forms containing paracetamol and caffeine available in the Turkish drug market. Dissolution Technology 23(2): 36-41.
- Banker GS, Anderson NR (2013) In the Theory and Practice of Industrial Pharmacy. In: Lachman L, Lieberman HA, Kanig JL (Eds) Philadelphia, USA: Varghese Publishing house, USA, pp. 293-345.
- Pisek R, Korselj V, Vrecer F (2002) Comparison of direct rotor pelletization (fluid bed) and high shear pelletization method for pellet production. Pharm and Biopharm 53: 327-333.
- Allen LV, Popovich NG, Ansel HC (2011) Ansel's pharmaceutical dosage forms and drug delivery systems (Edn. 8th) Lippincott Williams & Wilkins, Philadelphia, USA, pp. 225-256.
- US Pharmacopeia National Formulary USP 23/NF 18 (1995) United States Pharmacopeial Convention. Inc., Rockville, MD, USA.
- Oishi TS, Nimmi I, Islam SMA (2011) Comparative in vitro Bioequivalence Analysis of Some Generic Tablets of Atorvastatin, a BCS Class II Compound. Bangladesh Pharmaceutical Journal 14(1): 61-66.
- Lachman L, Lieberman H, Kanig J (1987) The theory and practice of industrial pharmacy (3rd Edn), Varghese publication house, USA, pp. 293-345.
- Waiver of in-vivo bioavailability and bioequivalence studies for immediate-release solid oral dosage forms based on a biopharmaceutics classification system; guidance for industry; US (2015) Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), US Government Printing Office: Washington, USA.






























