Isolation and Phenotypic Detection of Metallo-Beta-Lactamase (MBL)-Producing Klebsiella Species from Cow Anal Swabs
Ejikeugwu Chika1*, Duru Carissa2, Eluu Stanley3, Oguejiofor Benigna1, Ezeador Chika4, Ogene Lilian1 and Iroha Ifeanyichukwu1
1Department of Applied Microbiology, Ebonyi State University, Abakaliki
2Department of Pharmaceutical Microbiology and Biotechnology, Nnamdi Azikiwe University, Awka, Nigeria
3Department of Biological Sciences (Biotechnology Programme), Ebonyi State University, Abakaliki
4Department of Microbiology and Parasitology, Nnamdi Azikiwe University Teaching Hospital (NAUTH), Nnewi, Nigeria
Submission: April 01, 2017; Published: May 30, 2017
*Corresponding author: Ejikeugwu Chika, Department of Applied Microbiology, Faculty of Science, Ebonyi State University, Abakaliki, P.M.B 053, Ebonyi State, Nigeria , Tel: +2348097684562; Email: ejikeugwu_chika@yahoo.com
Thassio Ricardo Ribeiro Mesquita, Department of Physiology, Federal University of Sergipe, Brazil, Tel: 55 79 2105 6642; Email: thassio.mesquita@gmail.com
How to cite this article: Ejikeugwu C, Duru C, Eluu S, Oguejiofor B, Ezeador C, Ogene L, Iroha I. Isolation and Phenotypic Detection of Metallo-Beta-Lactamase (MBL)-Producing Klebsiella Species from Cow Anal Swabs. Glob J Pharmaceu Sci 2(3) : 555586 (2017). DOI: 10.19080/GJPPS.2017.02.555586
Abstract
The production of β-lactamase enzymes such as metallo β-lactamases (MBLs) is one of the most distributed resistance mechanisms engineered by Gram-negative bacteria towards β-lactam antibiotics. MBLs confer on bacteria the ability to hydrolyze and become resistant to the carbapenems such as imipenem. This study was aimed at isolating and detecting the production of MBLs from Klebsiella species that emanated from anal swabs of cow in a local abattoir A total of 31 anal swab samples were bacteriologically analyzed for the isolation of Klebsiella species which were identified using standard microbiological techniques. Antimicrobial susceptibility testing was carried out using the modified Kirby- Bauer disk diffusion method on Mueller-Hinton agar plates. The isolated Klebsiella species was screened for MBL production by determining their susceptibility to the carbapenems and this was confirmed using the EDTA-inhibition-based assay for phenotypic detection of MBLs. A total of 18 Klebsiella species was isolated from the cow anal swab samples.
The Klebsiella species were resistant to oxacillin (100%), cefoxitin (77.8%), ceftriaxone (44.4%), cefotaxime (50%), and ceftazidime (38.9%). They also showed resistance to the carbapenems including ertapenem (88.9%), imipenem (22.2%) and meropenem (83.3%). However, the isolated Klebsiella species showed susceptibility to ciprofloxacin (38.9%), ofloxacin (72.2 %), gentamicin (88.9%) and amikacin (83.3%).Out of the 18 isolates, only 2 (11.1%) isolates of Klebsiella species were phenotypically confirmed to produce MBLs. The production of MBLs by Klebsiella species in the community portend serious public health concern since these organisms are resistant to the carbapenems (used as last line antibiotics for treating serious infections). Efficient detection and sustainable intervention protocol to control the emergence and spread of antibiotic resistant bacteria in the community are necessary to contain this dilemma.
Keywords: MBLs; Gram-negative bacteria; Antimicrobial resistance; Abattoir; Nigeria
Introduction
The current surge in the emergence and spread of microbial resistance in the community has impacted negatively on the most important discoveries of modern medicine. Antibiotics has over the years saved untold number of people from morbidity and mortality due to infectious diseases, but this important discovery of modern medicine is under threat due to microbial resistance which now occur in hospital and non-hospital environment. The emergence of carbapenem-resistant Gram-negative bacteria in both the community and hospital environments constitutes an alarming development in the field of infectious disease management and control [1-4]. This menace portends with major public health implications since they jeopardize the clinical significance of potent antibiotics used to treat serious infections. Metallo-β-lactamases (MBLs) which belong to class B beta-lactamase of Ambler classification are enzymes that hydrolyze and confer on bacteria the exceptional ability to resist the antimicrobial action of the carbapenems such as imipenem and meropenem [5].
They require divalent cations of zinc as cofactors for enzyme activity, and thus the activity of these enzymes are usually inhibited in vitro by chelating agents such as ethylenediamine tetra-acetic acid (EDTA). MBLs have potent hydrolyzing activity not only against the carbapenems but also against other (β-lactam antibiotics [6]. The increase in antibiotic resistance among bacteria, most notably Klebsiella species by the production of β-lactamases has led to the increased use of carbapenem antibiotics. However, most strains of this organism are resistant to the carbapenems since they produce MBLs. Klebsiella species typically cause opportunistic infections in soft tissues, wounds, urinary tract and in the blood stream [7]. They cause community and hospital acquired infections in human and therefore poses challenges to the public health. Antimicrobial resistance in Klebsiella species has become increasingly prevalent, and this has been partly attributed to the efflux pump, decreased outer membrane permeability and production of beta-lactamase enzymes by this organism.
Microbes develop resistance to antimicrobial agents following mutation and selective pressure imposed on them by the incessant use of antibiotics [8]. Resistance to carbapenems among these bacteria remains remarkably rare in most countries. However, the situation still remains unreported in many parts of the developing world where access to quality healthcare is still poor. More intensive efforts are urgently required to elucidate the epidemiological and infection control issues related to multidrug resistant organisms and to improve measures aimed at stopping their emergence and spread in any environment. The use of antibiotics in the rearing and production of livestock and poultry birds as well as in other veterinary purposes has contributed significantly to the emergence and spread of drug resistant bacteria in the community. In this study, attempts have been made to characterize phenotypically the occurrence of Klebsiella species producing MBLs from abattoir samples.
Methodology
Collection and processing of samples: A total of 31 samples were collected from the anal region of cows from a local abattoir in Abakaliki metropolis, Ebonyi State, Nigeria using sterile swab sticks. Samples were collected by inserting a sterile swab stick into the anal region of cow at a depth of 3cm deep and rotated at 360 °C. Samples were transported to the Microbiology Laboratory Unit of Ebonyi State University, Abakaliki, Nigeria for bacteriological analysis. Each of the samples were inserted into test tubes containing 5 ml double strength nutrient broth (Oxoid, UK) and the tubes were loosely covered with cotton wool, and incubated at 30oC for 18-24 hrs.
Bacterial isolation
Loopfuls of the turbid suspension in the nutrient broth tubes were aseptically cultured on freshly prepared eosin methylene blue (EMB) and MacConkey agar (Oxoid, UK) plates, and incubated at 30 °C for 18-24hrs. Culture plates showing bacterial growth were aseptically sub-cultured onto freshly prepared EMB and MacConkey agar plates for the isolation of pure cultures of Klebsiella species. Klebsiella species produces non-mucoid, non-lactose fermenting colonies on MacConkey agar and non- metallic sheen colonies on EMB. All isolated Klebsiella species was identified using standard microbiological methods [9].
Antibiotic susceptibility testing
Antibiogram was done on Mueller-Hinton (MH) agar (Oxoid, UK) plates by Kirby-Bauer disk diffusion method according to Clinical Laboratory Standard Institute (CLSI) criteria [10]. Single antibiotic disks (Oxoid,UK) comprising imipenem (10μg), meropenem (10μg), ertapenem (10μg), amikacin (30μg), ofloxacin (5μg), ceftazidime (30μg), ceftriaxone (30μg), cefotaxime (30μg), ciprofloxacin (30μg), oxacillin (1μg), cefoxitin (30μg), and gentamicin (10μg) were used for antimicrobial susceptibility testing. The susceptibility plates were incubated at 30°C for 18-24hrs and the zones of inhibition diameter was measured and reported according to CLSI criteria [10].
Screening for the presence of MBLs
The susceptibility of the test isolates to the carbapenems was evaluated by the Kirby-Bauer disk diffusion method as per the CLSI criteria. MBL-producing isolates was suspected when the test organism(s) was resistant to any of the carbapenems (imipenem and meropenem). As per the CLSI criteria, isolates showing inhibition zone diameter (IZD) of ≤23mm were suspected to produce MBL enzymes and these isolates were subjected to phenotypic confirmation test [3,10].
Phenotypic detection of MB
MBL production was phenotypically evaluated by the inhibition-based assay using EDTA [3]. The test bacteria isolates (adjusted to 0.5 MacFarland turbidity standards) were aseptically swabbed on MH agar plates, and imipenem (10|ig) and meropenem (10μg) disks impregnated with EDTA (0.5μg) was aseptically placed on the MH agar plates. Supplementary imipenem (10μg) and meropenem (10μg) disks without EDTA were also placed alongside antibiotic disks impregnated with EDTA. Antibiotic disks were placed 20mm apart, and all the plates were incubated at 30 °C for 18-24hrs. A difference of ≥7mm between the zones of inhibition of any of the carbapenem disks tested alone compared to disks with EDTA infers MBL production phenotypically [3,5].
Results

Key: + = positive, - = negative, n = number of isolates, % = percentage
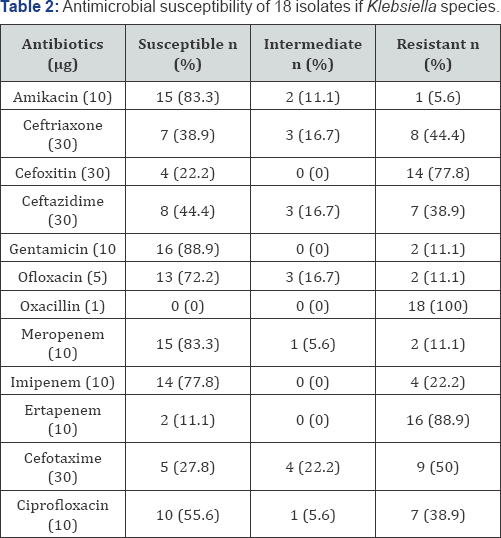
This study phenotypically evaluated the anal swabs of cow from a local abattoir for the presence of MBL-positive Klebsiella species. Bacterial isolation and biochemical characterization show that a total of 18 (58.1%) isolates of Klebsiella species was isolated from the 31 anal swab samples bacteriologically analyzed in this study (Table 1). The result of antimicrobial susceptibility testing of the Klebsiella species isolates to some commonly used antibiotics is shown in Table 2.
The isolated Klebsiella species showed high resistance to oxacillin (100%), ertapenem (88.9%), cefoxitin (77.8%), ceftriaxone (44.4%), imipenem (22.2%), cefotaxime (50%), ciprofloxacin (38.9%), and ceftazidime (38.9%). However, the isolated Klebsiella species showed susceptibility to meropenem (83.3%), ofloxacin (72.2%), gentamicin (88.9%) and amikacin (83.3%). Amikacin and gentamicin are both aminoglycosides, and they showed better inhibitory activity against the Klebsiella species isolated in this study (Table 2). Table 3 shows the frequency of MBL-positive Klebsiella species in this study. Out of the 18 isolates of Klebsiella species evaluated for the phenotypic production of MBLs, only 2 (11.1%) isolates of Klebsiella species produced MBLs by the inhibition-based assay used in this study.

Discussion
Antibiotic resistance within the non-hospital environment is an emerging public health threat that has put the efficacy of some available antimicrobial agents at risk and this phenomenon is due in part to the misuse of antibiotics for both human and non-human purposes. Klebsiella species is responsible for a handful of both community-acquired and nosocomial infections and this Gram-negative bacterium is notoriously resistant to some commonly available antibiotics. This study determined the antimicrobial susceptibility profile of Klebsiella species isolated from the anal swabs of cow from a local abattoir, and the production of metallo β-lactamases (MBLs) was phenotypically detected in the Klebsiella species isolates. Our bacteriological analysis shows that a total of 18 isolates of Klebsiella species was isolated from 31 anal swab samples analyzed in this study. Klebsiella species is a member of the Enterobacteriaceae family and they are commonly found in the gastrointestinal tract of warm blooded animals, from where they contaminate the environment through feacal contamination.
This organism is considered an important human pathogen and the number of reports on the isolation of species of this genus, from both hospital and a community sample is on the increase [11-14]. The isolated Klebsiella species in this study showed varying levels of susceptibility and resistance to the antibiotics tested. All the Klebsiella species were completelty resistant to oxacillin (100%). They also showed reduced susceptibility to the carbapenems, imipenem (22%) and ertapenem (88.9%). The Klebsiella species were also resistant to the cephalosporins used in this study including cefoxitin (77.8%), ceftriaxone (44.4%), cefotaxime (50%) and ceftazidime (38.9%). All the isolated Klebsiella species showed reduced susceptibility to the flouroquinolones, ciprofloxacin and ofloxacin at the rate of 38.9% and 72.2% respectively. However, meropenem, gentamicin and amikacin had better inhibitory activity against the Klebsiella species at the rates of 83.3%, 88.9%, and 83.3% respectively. Elsewhere, Klebsiella species has been reported to also show varying rates of resistance and susceptibility to the cephalosporins, flouroquinolones and the aminoglycosides [15].
The undue use of antibiotics for growth promotion and prevention of diseases in farm animals has impressed selective pressures that induce more resistance among bacteria in the community. Out of the 18 isolates of Klebsiella species phenotypically screened for the production of metallo β-lactamase (MBL), only 2 isolates were positive for the production of this enzyme. The other 16 isolates of Klebsiella species did not express MBL phenotypically by the method we used. The production of MBL is this study is similar to a previous study conducted by Yusuf et al. [16] in which MBL was detected in Klebsiella species from hospital origin. However, the prevalence of MBL-producing Klebsiella species in this study (11.1%) is lesser than that reported by Yusuf et al. [16].
Enwuru et al. [17] also reported a higher prevalence of MBL- producing Klebsiella species from both clinical and community samples in their study. Also in a recent study conducted in Japan, Okazaki et al. [18] reported the occurrence of K. pneumoniae positive for MBL production in a non-hospital environment. We have previously reported the occurrence of MBL-producing Klebsiella species from poultry birds - in which 5 (41.7%) isolates out of 24 isolates of Klebsiella species produced MBL phenotypically [19]. This shows the impact of antibiotic misuse in the community. We have demonstrated in this study that Klebsiella species from anal swabs of cow produce MBLs phenotypically. This organism is endowed with the ability to be resistant to the carbapenems. Therefore, the detection of multidrug resistant bacteria from community and hospital samples in this region should be given a boost in order to assuage the emergence and spread of MBL-producing bacteria through the food chain.
Conclusion
Conclusively, this study has presumptively reported the occurrence of Klebsiella species with multidrug resistance from anal swabs of cow. The Klebsiella species also expressed MBL phenotypically. The use of antibiotics unduly in the rearing and production of farm animals contributes a great deal to the emergence and spread of antibiotic resistant bacteria in the community through selective pressure. Antibiotic residues in food-producing animals can cause drug resistant bacteria to emerge, and be transmitted to humans via the food chain. It is therefore vital to curtail and if possible stop the use of antibiotics as growth promoting agents in agricultural practices.
References
- Walsh TR, Toleman MA, Poirel L, Nordmann P (2005) Metallo-ß- lactamases: the Quiet before the storm? Clin Microbiol Rev 18(2): 306-325.
- Bush K, Jacoby GA (2010) Updated functional classification of beta lactamases. Antimicrob Agents Chemother 54(3): 965-967.
- Ejikeugwu PC, Ugwu CM, Iroha IR, Eze P, Gugu TH, et al. (2014) Phenotypic Detection of Metallo-ß-Lactamase Enzyme in Enugu, Southeast Nigeria. American Journal of Biological, Chemical & Pharmaceutical Science 2(2): 1-6.
- Walsh TR (2010) Emerging carbapenemases: a global perspective. Int J Antimicrob Agents 36(3): S8- S14.
- Ejikeugwu C, Edeh C, Iroha I, Orji J, Eluu S, et al. (2016) Antibiogram and Detection of Metallo-Beta-Lactamase (MBL) positive Escherichia coli isolates from abattoir. Nature and Science 14(11): 65-69.
- Zurfluh K, Hächler H, Nüesch IM, Stephan R (2013) Characteristics of extended-spectrum beta-lactamase- and carbapenemase-producing
- Podschun R, Ullmann U (1998) Klebsiella spp. as nosocomial pathogens: epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin Microbiol Rev 11(4): 589-603.
- Chakraborty D, Basu S, Das S (2010) A study on infections caused by Metallo-Beta-Lactamase Producing Gram negative Bacteria in Intensive Care Unit Patients. Am J Infect Dis 6(2): 34-39.
- Cheesbrough M (2006) District Laboratory Practice in Tropical Countries (2nd edn) Cambridge University Press, UK, pp. 178-187.
- Clinical Laboratory Standard Institute, CLSI (2011) Performance standards for antimicrobial disk susceptibility test. Fifteenth informational supplement, CLSI document M100-S15, Wayne, USA.
- Chitanand MP, Kadam TA, Gyananath G, Totewad ND, Balhal DK (2010) Multiple antibiotic resistance indexing of coliforms to identify high risk contamination sites in aquatic environment. Indian J Microbiol 50(2): 216-220.
- Steve G, Helen H and Tim B (2005) Methods used in New Zealand diagnostic laboratories to identify and report extended spectrum beta- Lactamase producing Enterobacteriaceae, a report by the institute of Environmental Science and Research Limited ("ESR”) in New Zealand, p. 1-5.
- Liu PYF, Gur D, Hall LMC, Livermore DM (1992) Survey of the prevalence of Beta lactamases amongst 1000 Gram-negative bacilli, isolated consecutively at the Royal London Hospital. Journal of Antimicrobial Chemotherapy 30: 429-447.
- Paterson DL, Ko WC, Von Gottberg A, Mohapatra S, Casellas JM, et al. (2004) Antibiotic therapy for Klebsiella pneumoniae bacteremia: Implication of production of extended spectrum beta lactamases. Clin Infect Dis 39(5): 50-57.
- Aibinu I, Fashae K, Ogunsola F, Odugbemi T, Mee BJ (2004) Extended Spectrum Beta-Lactamase (ESBL) in Klebsiella pneumonia isolates from septicaemic children in Ibadan, Nigeria. Nigeria J Health Biomed Sci 3(2): 79-84.
- Yusuf I, Yusha M, Sharif AA, Getso MI, Yahaya H, et al. (2012) Detection of Metallo beta lactamases among Gram negative bacterial isolates from Murtala Muhammad Specialist Hospital, Kano and Almadina Hospital Kaduna, Nigeria. Bayero Journal of Pure and Applied Sciences 5(2): 84-88.
- Enwuru NV, Enwuru CA, Ogbonnia SO, Adepoju BAA (2011) Metallo- β-lactamase production by Escherichia coli and Klebsiella species isolated from hospital and community subjects in Lagos, Nigeria. Nat Sci 9(11): 1-5.
- Okazaki R, Hagiwara S, Kimura T, Tokue Y, Kambe M, et al. (2016) Metallo-β-lactamase-producing Klebsiella pneumoniae infection in a non-hospital environment. Acute Medicine and Surgery 3:32-35.
- Ejikeugwu C, Iroha I, Oguejiofor B, Orji OL, Eluu S, et al. (2017) Emerging Multidrug Resistant Metallo-β-lactamases (MBLs) positive Klebsiella species from cloacal swabs of poultry birds. Journal of Bacteriology and Parasitology 8(1): 1000305.






























