Strategies to Improve Outcomes of Ovulation Induction and Assisted Reproductive Technology (ART): The Iraqi Consensus
Fadia Al-Izzi1, Manal Al-Obaidi2, Eman Sabah Ahmed3, Sarmad Sami Khunda4, Safiya Abdulkareem Wahd5, Maida Yousif Shamdeen6, Amanj Rahim Kadir7, Mahboubeh Mohammadzadeh8* and Juan García Velasco9
1Al-Mustansiriyah Medical College, Iraq
2High Institute for Infertility Diagnosis and Assisted Reproductive Technologies, Al-Nahrain University, Iraq
3Azadi General Hospital, Iraq
4Baghdad University, Iraq
5Hawler Medical University, Iraq
6Duhok Medical College, Iraq
7Royal IVF Center, Iraq
8Merck Serono Middle East, United Arab Emirates, an affiliate of Merck KGaA Darmstadt, Germany
9Rey Juan Carlos University, Spain
All authors are equally contributed
Submission: January 31, 2023; Published:March 01, 2023
*Corresponding author: Mahboubeh Mohammadzadeh, Merck Serono Middle East, United Arab Emirates, an affiliate of Merck KGaA Darmstadt, Germany
How to cite this article: Fadia Al-Izzi, Manal Al-Obaidi, Eman Sabah Ahmed, Sarmad Sami Khunda, Safiya Abdulkareem Wahd, et al. Strategies to Improve Outcomes of Ovulation Induction and Assisted Reproductive Technology (ART): The Iraqi Consensus. Glob J Reprod Med. 2023; 9(4): 555770. DOI: 10.19080/GJORM.2023.09.555770.
Abstract
The ESHRE 2019 Guidelines provide clinicians with valuable evidence-based recommendations to optimize ovarian induction and ovarian stimulation in in vitro fertilization (IVF). However, data from such guidelines are primarily based on randomized controlled trials with highly selected populations that are conducted under very controlled conditions. To complement evidence from clinical guidelines and further improve treatment outcomes, seven Iraqi experts gathered to evaluate opinions on the specific approaches during an IVF cycle in Iraq and developed 15 statements agreed upon: The Iraqi Consensus. A Delphi consensus was conducted to formulate expert opinion on how assisted reproductive technology outcomes could be improved. Step 1 involved the scientific Board, comprising the scientific coordinator, who developed the initial statements and supporting references, which were discussed/amended by seven experts, and refined the final statements and references. Step 2 involved 30 Iraqi experts voted on their level of agreement/disagreement with each statement. Consensus was reached if the proportion of participants agreeing/disagreeing with a statement was >66%. Step 3 data was collected and analyzed then communicated to participating experts. Consensus was achieved for 15 statements with high level of agreement in >85% of statements, tackling follicular development/gonadotropins; pituitary suppression; final oocyte maturation triggering; luteal-phase support. This represents the collective point of view of the experts that took part in the consensus development and are based on local practice. This Delphi consensus provides a real-world clinical perspective from Iraqi experts and complements the international guidelines, and may help to further improve treatment outcomes in Iraq and the region.
Conclusion: The social isolation caused by the COVID-19 pandemic did not affect most of the self-reported symptoms, quality of life and self-care attitudes of the analyzed women.
Keywords: Delphi consensus; ART; Ovarian induction; Ovarian stimulation; IVF
Abbreviations: AFC: Antral Follicle Count; AMH: Anti-Müllerian Hormone; cAMP: Cyclic AMP; CLBR: Cumulative Live Birth Rate; EMT: Endometrial Thickness; ESHRE: European Society of Human Reproduction and Embryology; ET: Embryo Transfer; FET: Frozen-Thawed Embryo Transfer; FNPO: Follicular Number Per Ovary; FSH: Follicle‐Stimulating Hormon; GnRH: Gonadotropin-Releasing Hormone; hMG: Human Menopausal Gonadotropin; HRT: Hormone Replacement Therapy; ICMART: International Committee for Monitoring ART; IM: Intramuscular; IVF: In-Vitro fertilization; LBR: Live Birth Rates; LHCGR: Luteinizing Hormone/Choriogonadotropin Receptor; LHR: LH-Receptor; LPS: Luteal Phase Support; MAR: Medically Assisted Reproduction; mNC-FET: Modified Natural Cycle-Frozen-Thawed Embryo Transfer; NC-FET: Natural Cycle-Frozen-Thawed Embryo Transfer; OA: Ovarian Area; OHSS: Ovarian Hyper Stimulation Syndrome; OI: Ovulation Induction; OV: Ovarian Volume; PCOM: Polycystic Ovary Morphology; PLC: Phospholipase C; POR: Poor Ovarian Response; PROsPeR: Poor Responder Outcome Prediction; RCTs: Randomized Controlled Trials; r-hFSH: Recombinant Follicle‐Stimulating Hormone; SC: Subcutaneous
Introduction
Infertility is defined as the inability to reach a clinical pregnancy after a year of regular and unprotected sexual intercourse. This worldwide condition affects 8-12% of reproductive-aged couples, whether males or females. Overall, one-third of infertility cases are caused by male reproductive issues, one-third by female reproductive issues, and one-third by both male and female reproductive issues or by unknown factors [1]. Infertility rates can reach up to 30% in some regions of the world, including the Middle East [2]. The primary causes of female infertility are ovulatory disorders, decreased ovarian reserve, abnormalities of the reproductive system (anatomical, endocrine, genetic, functional, or immunological), chronic illnesses, and sexual conditions incompatible with coitus [3]. The therapy used generally targets the diagnosed etiology to increase the chances of achieving clinical pregnancy in infertile couples. Three principal therapeutic solutions are currently available, i.e., pharmacological treatment, surgical treatment, and medically assisted reproduction (MAR).
Over 40 years ago, Louise Brown, the first test-tube baby, was born, bringing hope for couples desperately trying to conceive. Techniques have considerably evolved since then, and assisted reproductive technology (ART) has now made it possible to treat successfully previously untreatable cases of infertility [4,5]. The International Committee for Monitoring ART (ICMART) declared in the European Society of Human Reproduction and Embryology (ESHRE) 2018 meeting the evolution of in vitro fertilization (IVF) techinques, and estimated over 8 million babies born worldwide from IVF, since 1978 [6]. Although ART has improved over the years, it still has some limitations. First, since it was believed that multiple oocytes retrieval was essential to improve the chances of pregnancy, a high wastage of embryos has been observed [7]. Second, women undergoing ART are also at increased risk of developing Ovarian Hyper Stimulation Syndrome (OHSS), an iatrogenic complication presenting in the luteal phase or early pregnancy [8].
To complement evidence from clinical guidelines and further improve pregnancy outcome of ovulation induction and ART while managing the treatment-related adverse events that might emerge, 7 Iraqi experts gathered to evaluate opinions on the specific approaches during an IVF cycle in Iraq and developed 15 statements agreed upon: The Iraqi Consensus. This consensus discussed the main steps of ART treatment, including follicular development and stimulation with gonadotropins, pituitary suppression, final oocyte maturation triggering, and luteal phase support.
Assessment of statements according to the Delphi consensus process [9].
The Iraqi consensus included three steps (Figure 1) and involved A scientific board comprising the scientific coordinator from Spain and seven Iraqi experts, as well as an extended panel of 30 Iraqi experts. The scientific coordinator initially drafted the 15 statements and supporting references based on an evaluation of the latest scientific literature. In Step 1, the Scientific Board discussed these statements, argued, and suggested modifications to reflect the Iraqi practice. The agreed upon statements were then distributed to the extended panel in Step 2 and the 30 Iraqi experts voted on their level of agreement/disagreement with each statement using a 5-point Likert scale: 1 (absolutely disagree), 2 (disagree), 3 (agree), 4 (more than agree), and 5 (absolutely agree). Data was collected and analyzed in Step 3.
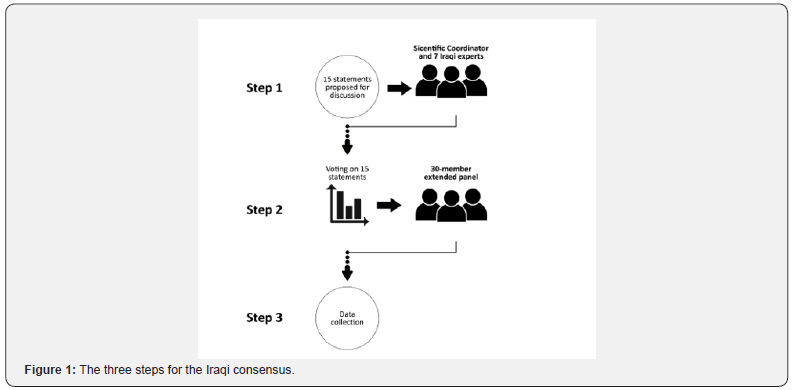
A consensus was considered reached if the proportion of participants either disagreeing (responding 1 or 2) or agreeing (responding 3, 4 or 5) with a statement exceeded 66%. If the proportion of participants either agreeing or disagreeing with a statement did not exceed 66%, this statement would be revised according to the feedback received, and another survey initiated, including only the statement(s) not reaching consensus. This process was repeated with the revised statements until consensus was reached for every statement.
Results of the consensus and recommendationsIn Step 1, seven statements were agreed upon without modification, and eight were modified and subsequently approved. These 15 approved statements were then voted on in Step 2 by the 30-member extended panel (Table 1) and distributed as follows: follicular development and gonadotropins (9 statements), luteal phase support (2 statements), pituitary suppression (2 statements), and final oocyte maturation triggering (2 statements).
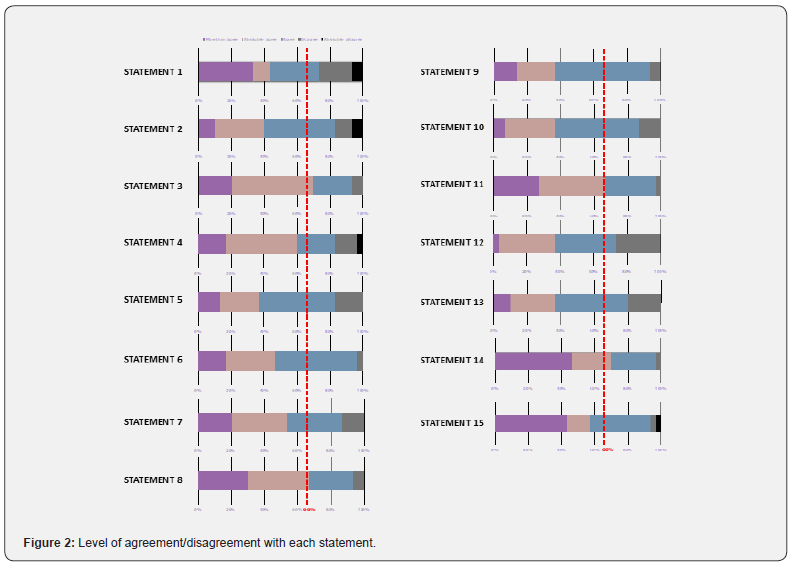
The consensus was reached after the first vote and all 15 statements obtained over 66% of participants’ agreement. A high level of agreement (>80%) was reached in >85% of statements, exceeding 90% in almost 50% of the statements (7/15). The maximum level of disagreement was 7%, obtained in only two statements: two experts voted “totally disagree” in statements 1 and 2, and one expert totally disagreed with Statements 4 and 15. The 15 statements grouped according to their area of focus (follicular development and stimulation with gonadotropins, pituitary suppression, final oocyte maturation triggering, and luteal phase support), together with their associated references, are discussed below.
Statement 1: Rotterdam criteria are recommended for the diagnosis of PCOS (clinical and/or biochemical hyperandroginaemia, oligo or amenorrhoea and/or ultrasound of the ovary showing ≥12 AFC/ovary and /or ovarian volume > 10cc in at least one ovary).
This statement received 73% total agreement from the extended panel (Figure 2). The physicians explained that they base PCOS diagnosis more on the ultrasound than the Anti-Müllerian hormone (AMH) or hormones, and the new cut-off ≥12 antral follicle count (AFC) instead of >10 per ovary (AFC) recommended by the new guidelines was added.
PCOS is a frequent endocrine disease in women of reproductive age. It involves several features, which made it hard to set general guidelines to come up with the diagnosis. Over the years, several consensus seminars about PCOS have been performed. The first one took place in Rotterdam, the Netherlands, in 2003 and came up with the diagnostic criteria for PCOS, where patients should fit two out of three of the following criteria: clinical or biochemical hyperandrogenism, oligomenorrhoea or oligo-ovulation, and polycystic ovaries on ultrasound. These guidelines are frequently adopted. The second consensus was in Thessaloniki, Greece, in 2007 and focused on infertility management in PCOS. Finally, in October 2010, the third consensus was held in Amsterdam, the Netherlands, and reported updated knowledge (and breaches in knowledge) in health aspects of women with PCOS. It included several characteristics of this disease, such as hirsutism, contraception, menstrual cycle abnormalities, quality of life, sexual health, pregnancy complications, among others [10]. In 2018, an international evidence-based guideline for PCOS assessment and management recommended the Rotterdam PCOS diagnostic criteria in adults. It focused on the fact that ultrasound is not required if other features are present. It also described the new tightened imaging criteria with advancing technology [11].
Regarding imaging techniques and polycystic ovary morphology (PCOM), several features are considered in women with this condition: AFC, follicular number per ovary (FNPO), ovarian volume (OV), ovarian area (OA), ovarian blood flow, and the ratio of stroma to total ovarian size. Following a consensus opinion in 2003, the threshold for PCOS was set at ≥12 follicles measuring 2-9 mm in diameter. The ESHRE PCOS guideline group 2018 suggested a cut-off of FNPO ≥20 in one or both of the ovaries and/or OV>10mL, without the inclusion of dominant follicle or corpus luteum or any cysts using endovaginal ultrasound transducers with a frequency bandwidth that includes 8MHz. FNPO of 12 or more or OV> 10 mL is required for ultrasound machines of older technology [11,12]. The new threshold led to underdiagnosed cases of PCOS: 48.2% of women with PCOS features were excluded from the diagnosis. These patients presented worse metabolic and hormonal symptoms than the control group and similar criteria with the group of approved PCOS [13].
Statement 2: In PCOS patients, the first-line treatment for follicular development is clomiphene/letrozole, and a maximum of 4 to 6 cycles should be considered. Metformin 500-2000mg may be added after three failed cycles or CC resistance.
This statement received 83% total agreement from the extended panel (Figure 2). During the Scientific Board meeting, experts decided to put a cut-off of 4-6 cycles for the use of gonadotropins and the time to start them (according to several factors, including BMI) and agreed that approaches differ between guidelines and real-life daily practice and from country to country. PCOS leads to ovulatory dysfunction resulting in infertility. Several treatments are proposed to handle anovulatory infertility. A review ranked the treatments based on the success rate, complexity, and adverse effects and suggested solutions from lifestyle modification to IVF and surgery. Insulin sensitizers like metformin (1500-2500mg per day divided into 2 or 3 doses), selective estrogen receptor modulators like CC (50-150mg per day for 5 days), and aromatase inhibitors like letrozole (2.5-5mg for 5 days) are suggested as first-line treatment. The best ovulation and live birth rates are observed with letrozole and a combination of CC and metformin [14].
An individual participant data meta-analysis compared letrozole alone and CC plus metformin, as compared to CC alone, as ovulation induction (OI) agents, in women with PCOS suffering from infertility. In contrast with CC, letrozole showed higher live birth and clinical pregnancy rates with lower time-to-pregnancy. The combination (metformin + CC) presented increased clinical pregnancy and reduced time-to-pregnancy with no evidence on live birth, compared to CC alone [15]. A randomized clinical trial among 666 women with six prior failed cycles with CC studied the endometrial thickness (EMT) as a biomarker to determine whether to continue treatment with CC or switch to gonadotropins. The suggested EMT cut-off was 7 mm in the sixth cycle. If EMT ≤ 7 mm, switching to gonadotropins improved live birth rates (LBR) over continuing treatment with CC at a high extra cost. If the EMT > 7 mm, LBR were similar among continuing CC treatment or switching to gonadotrophins; thus, it was advised to continue with CC without extra costs [16].
Statement 3: If CC/letrozole fails after 4-6 cycles, gonadotropins are recommended next, and the step-up protocol (low dose or chronic low dose protocol) is effective and safer than other protocols, with a starting dose of 37.5-75 IU and a 50% increase after 7 days (if no follicles recruitment >10mm).
This statement received 93% total agreement from the extended panel (Figure 2). One panelist stated that there are two protocols for PCOS patients: the step-up protocol and the chronic low-dose step-up protocol, where treatment continues for 14 days instead of 7 days. However, this doctor uses the step-up protocol as she believes that waiting for seven days is better. Panelists then agreed that the literature shows that physicians can wait up to 14 days but that this would be difficult to apply in real-life practice when confronted with a patient with no recruitment of follicles > 10 mm after 7 days. In most centers, the anti-estrogen CC is the first line of treatment for ovulation induction, resulting in ovulation in 75-80% of women. For patients who do not conceive after CC, exogenous gonadotropins have been used with variable success (overall pregnancy rate ~30%) [17]. Indeed, only half of the women ovulating on CC conceive within six months of treatment. In women who experience CC failure, CC or letrozole treatment is often changed to second‐line OI with gonadotrophins [18]. The major problem associated with this treatment is the development of multiple follicles leading to multiple pregnancies and OHSS [17].
A comparative randomized multicentric study comparing the step-up versus step-down protocols in women with CC resistant polycystic ovaries showed that the step‐up protocol using recombinant follicle‐stimulating hormone (r-hFSH) is more efficient in obtaining a monofollicular development and ovulation than the step‐down protocol in this category of women. Although the duration of stimulation is longer in the step-up protocol (15.2±7.0 days in step‐up versus 9.7±3.1 in step‐down, P < 0.001), the rate of OHSS is much lower [19]. A prospective randomized study has assessed the efficacy and safety of a decremental follicle‐stimulating hormone (FSH) dose regimen, applied once the leading follicle was 10-13 mm in diameter among women treated for WHO Group II anovulation using a chronic low-dose (CLD; 75 IU FSH for 14 days with 37.5 IU increment) step-up protocol. The results confirmed that maintaining the same FSH starting dose for 14 days before increasing it in a step-up regimen is critical to control the risk of over-response [20]. A randomized, double‐blind, dose‐finding study aiming to determine the optimal regimen of r-hFSH for OI in Japanese women with amenorrhea or anovulatory infertility concluded that a starting dose of 75 IU of r-hFSH (compared to a starting dose of 37.5 IU and 150 IU) is associated with favorable results and safety profile [21].
Statement 4: In PCOS patients, due to the higher precision, recombinant FSH is more recommended than biosimilars that are not approved by the EMA or FDA as they are not comparable with r-hFSH with regards to efficacy and safety.
This statement received 83% total agreement from the extended panel (Figure 2). A review of the available evidence comparing the efficacy, safety, and cost-effectiveness of r-hFSH follitropin alpha originator with its biosimilars found that most IVF Centers are aware of the availability of FSH biosimilars on the market, but almost all require more information on these products to use them, thus, limiting their use. The inadequate number of observations made it impossible for the authors to reach any conclusion about the equivalence of biosimilars to the originator with respect to LBR and OHSS incidence [22]. The experts agreed that r-hFSH is more precise and effective than its biosimilars but then discussed the price difference, a relevant factor to consider. They also agreed not to use the biosimilars currently available in Iraq because they are not EMA or FDA approvedlack proper registration process and are not registered in every country, and do not have similar standards of scientific evidence with regards to efficacy and safety compared to biosimilars approved by EMA.
Statement 5: Progestational therapy (vaginal or other routes) for luteal phase support may be used in OI cycles and is recommended in Gonadotropin OI cycle for up to 10-12 weeks.
This statement received 83% total agreement from the extended panel (Figure 2). Luteal phase deficiency is a condition in which women have insufficient progesterone secretion to maintain a normal secretory endometrium and allow for normal embryo implantation and growth. This condition is a cause of infertility, and its pathophysiology is not very well understood. Progesterone supplementation is a frequently used treatment [23]. In case of corpus luteum deficiency, the vaginal administration of progesterone (in the form vaginal tablets, creams, or suppositories) is the preferred and most used route to maintain adequate luteal phase support (LPS), despite the alternatives available, such as oral, rectal, intramuscular (IM), and subcutaneous (SC) medications [24]. Interestingly, the route of administration does not seem to impact the ART outcome; therefore, patients’ preferences can be considered [25].
Statement 6: Exogenous FSH alone is probably sufficient for follicular development in normogonadotropic patients aged < 35 years.
This statement received 97% total agreement from the extended panel (Figure 2). It is well established that FSH is essential for the development of small follicles, while the development of granulosa cells for larger follicles requires both FSH and estradiol. However, the factors responsible for follicle growth and maturation are mainly FSH and growth factors, such as IGF-1 and TGF-ß [26]. In 1985, Shaw and al. performed the first successful IVF pregnancy using gonadotropin-releasing hormone (GnRH) analogs only combined with pure urinary FSH [27].
A 33-year-old G2P0A2 patient first received 100 μg of intranasal LHRH agonist analog ‘Buserelin’ 5 times a day starting on day 22 of her cycle to achieve pituitary desensitization. When adequate suppression of pituitary activity was achieved, she was administered daily injections of two ampoules of Metrodin®, which contains 150IU of FSH and only 2IU of LH, setting the day of the first injection as day 0. Four follicles >19mm diameter were detected on her ultrasound on day 12. Buserelin and FSH were discontinued and 5000IU of hCG were administered. Four mature oocytes were recovered, three of them fertilized and transfered and two fetuses with beating hearts were detected 6 weeks later. By week 16 (time of study publication), the pregnancy was progressing well. Since then, thousands of IVF pregnancies using recombinant FSH stimulation have taken place and have shown that recombinant FSH is as effective as urinary human menopausal gonadotropin (hMG) with a better tolerability [27], in normogonadotropic and hypergonadotropic women.
A randomized control trial compared the efficacy of r-hFSH vs r-hFSH:r-hLH in women of 2 age groups undergoing their first or second IVF cycle. They found that, while r-hLH administration significantly increase the implantation rate in patients aged 36 to 39 years, it was not beneficial in patients younger than 36 years [28]. A retrospective cohort study on 465 patients, which compared the embryo outcomes of IVF/ICSI with a GnRH antagonist protocol (FSH alone vs. FSH-hMG), showed that in patients aged <35 years, the number of oocytes retrieved in the FSH only group was significantly higher than the one in the FSH‐hMG group (13.7 vs. 9.2, P = 0.04). Hence, it concluded that exogenous FSH alone is probably enough for the development of follicles and that hMG may not improve the embryo profile in a GnRH antagonist protocol [29], while r-hLH was not beneficial in patients younger than 36 years [28].
Statement 7: There is a strong relationship between the number of oocytes and LBR following fresh IVF cycles and a positive linear association with cumulative LBR (in fresh and frozen).
This statement received 87% total agreement from the extended panel (Figure 2). A retrospective multicenter, multinational analysis, including approximately 15,000 women, intended to evaluate the association between the number of oocytes retrieved and the cumulative live birth rate (CLBR). It showed a significant progressive increase of CLBR with the number of oocytes, suggesting that ovarian stimulation might not have a damaging effect on the quality of the oocyte/embryo in women less than 40 years old [30]. In the UK, 400 135 IVF cycles were analyzed to determine whether there was an association between the number of eggs retrieved following IVF and the number of live births and the optimal number of eggs needed to obtain a positive outcome. The results showed a strong relationship between the number of eggs and LBR. Moreover, this relationship was not linear and the optimal number of eggs to reach clinical success was approximately 15 [31].
A systematic review also examined the association between the number of oocytes retrieved and the number of oocytes associated with the higher success rate. It showed that cumulative LBR increases with the number of oocytes retrieved and that 12- 18 oocytes are associated with the maximal cumulative LBR [32].
In conclusion the number of oocytes and high CLBR have a positive linear association; however, patient safety is a priority and targeting a high number of oocytes should be done in accordance with the number of cycles (fresh or frozen) to be done and the patient’s desire while avoiding OHSS.
Statement 8: When compared to treatment with r-hFSH alone, r-hFSH:r-hLH treatment might improve the ongoing pregnancy rate in some groups of low/poor prognosis patients.
This statement received 93% total agreement from the extended panel (Figure 2). Studies evaluating the effect of recombinant-human LH supplementation on ovarian response and pregnancy outcome showed that LH supplementation seems to have a beneficial effect on the maturity and fertilizability of oocyte [28,33]. A literature review concluded that poor responders and patients 35 years old and older might benefit from exogenous LH, which increases the number of mature oocytes, the quality of zygotes, and the implantation rates [34].
A retrospective multicenter controlled study also questioned the benefit of adding LH during COS. Out of 9787 ovarian stimulations, 5218 were treated with both LH and FSH and 4569 were only treated with FSH. Poor Ovarian Response (POR) was defined according to the ESHRE Bologna criteria, and classified into three categories (Mild, Moderate and Severe) according to the Poor Responder Outcome Prediction (PROsPeR) score. They found that the benefit of combining LH and FSH depended on the patient’s initial severity cathegory and that the combination therapy was indeed beneficial in increasing CLBR in patients in moderately (CLBR : 14.3% vs. 11.3%) and severely (CLBR : 9.8% vs. 4.4%) poor ovarian responders [35].
Statement 9: When compared to treatment with hMG, the effects observed across studies show a tendency towards an improved embryo number and pregnancy rate with r-hFSH:rhLH treatment. Nevertheless, further studies are needed before drawing a firm conclusion on the comparison between hMG and r-hFSH:r-hLH preparations.
This statement received 93% total agreement from the extended panel (Figure 2). One of the panelists declared that this statement is true in the case of hypogonadotropic hypogonadism. A systematic review and meta-analysis reviewing the efficacy of the r-hFSH-r-hLH combination protocol versus hMG protocol in COS reported that the r-hFSH:r-hLH combination might have better efficacy than hMG alone in COS [36]. An observational retrospective cohort trial on 4828 IVF cycles of different low prognosis patients, aiming to determine the potential role of LH addition to FSH, found that poor prognosis patients are usually treated with both LH and FSH and that the addition of LH to FSH retrieved a relative higher oocytes number compared to hMG [37].
Another randomized controlled trial comparing IVF outcomes in ovarian stimulation protocols with r-hFSH:r-hLH versus hMG in 122 patients also showed that both groups were comparable in pregnancy rate, implantation rate, oocytes, and embryo quality, with a higher OHSS risk in the r-hFSH:r-hLH group. The high OHSS risk might be explained by the fact that the group receiving r-hFSH:r-hLH had significantly more oocytes (7.8±1.1 total oocytes retrieved) than the group receiving hMG (4.1±1.2 total oocytes retrieved). It should be noted that this trial was conducted in 2010 in Italy and the insemination of 3 oocytes only was then allowed [38].
Statement 10: LH and hCG are characterized by specific molecular and biochemical features; they interact with different binding sites of the same receptor, resulting in a lower dissociation rate by hCG than LH binding. Recombinant LH has a shorter terminal half-life (around 10hrs) compared to hCG (terminal half-life 28-31hrs). Downstream effects of gonadotropin signaling consist of LH-related proliferative and anti-apoptotic signals vs. high steroidogenic potential and pro-apoptotic activity of hCG in vitro.
This statement received 87% total agreement from the extended panel (Figure 2). Both LH (26-32kDa) and hCG (37kDa) are heterodimeric glycoproteins [39], composed of a common α subunit combined with distinct 1β subunits that confers receptor binding specificity and intracellular signaling cascades in ovarian cells [39,40]. The LH-Receptor (LHR) also serves as the hCG receptor, but both hormones interact differently with the U-shaped portion of the hinge region [41,42]. The CG β subunit derives from duplications of the LH β subunit gene and is characterized by 24 additional amino acids at the C-terminus and a higher degree of glycosylation, resulting in an extended circulatory half-life [43].
Signaling pathways are different among r-hLH and HCG, LH mainly activates phospholipase C (PLC) (Gq pathway) and hCG mainly activates cyclic AMP (cAMP) production (Gs pathway) . In vitro, the onset of LH action (cAMP increase over baseline) was more rapid than the onset of hCG action; and the action of hCG on the receptor was prolonged, while the action of LH is transient [44]. First, the terminal half-life of the β subunit of r-hLH is estimated to be 9-12 hours, while that of the β subunit of HCG is estimated to be 28-31 hours [9]. Second, details of the luteinizing hormone/choriogonadotropin receptor (LHCGR) signaling are still not fully understood, with several mechanisms and interfaces not yet defined [45]. Moreover, a study compared these molecules in vitro and showed that LH and hCG result in different effects on the viability of cells. In particular, LH treatment increases levels of ERK1/2, especially AKT phosphorylation, anti-apoptotic XIAP gene expression, and increased cell viability over three days. In contrast, hCG regulates procaspase-3 cleavage and decreases cell viability over three days, explaining the pro-apoptotic effect of hCG [46].
Statement 11: The LBR using the long GnRH-agonist and multiple-dose GnRH-antagonist are comparable but pound for efficacy, the incidence of any grade of OHSS, and cycle cancellation due to it, is significantly lower after pituitary downregulation with a GnRH antagonist compared to pituitary downregulation with a GnRH agonist.
This statement received 97% total agreement from the extended panel (Figure 2). Pituitary suppression regimens are frequently used in ART. Two comparable protocols are applied to block the pituitary LH secretion, i.e., the GnRH agonist and GnRH antagonist. The GnRH agonist was developed first and played an essential role in increasing the number of retrieved oocytes and pregnancy rates and decreasing the number of cycle cancellations, which might result in many complications, including OHSS.
Several years later, the GnRH antagonist was discovered, and its rapid action of blocking GnRH receptors led to fewer side effects and shorter treatment duration [47]. Many studies compared both methods on different groups of women. A meta-analysis included 6 studies comparing LBR in GnRH antagonist and agonist groups among women with a normal ovarian reserve and concluded no significant difference between the two protocols [47].
A retrospective cohort study among women of different ages and various ovarian reserves who underwent ART between 2015 and 2018 evaluated the two protocols and measured CLBR. The results showed 45.3% of CLBR in the antagonist group vs. 50% in the long agonist group [48]. A 2016 updated Cochrane review shed light on the reduced risk of OHSS with the GnRH antagonist protocol (between 6 and 9%) compared to the GnRH agonist (11%). Moreover, the GnRH antagonist showed a lower incidence of cycle cancellation related to the high risk of OHSS but a higher incidence of cycle cancellation due to poor ovarian response [49].
Statement 12: After the administration of GnRH analogues, a functional state of hypogonadotropic hypogonadism with LH deficiency could be achieved, potentially affecting folliculogenesis, steroidogenesis, and oocyte competence, impinging on clinical outcomes (achieving good oocyte quality for pregnancy). The concept of LH dynamic changes following GnRH analogue employment as an indicator for the LH threshold should be pursued instead of a single serum LH evaluation.
This statement received 73% total agreement from the extended panel (Figure 2). During the meeting of the seven experts, it was stated that, in the Iraqi practice, LH concentration is not measured during the stimulation, patients are followed with ultrasound, and ESHRE guidelines do not recommend following the LH activity.
FSH and LH play an essential role in follicular maturation in both natural cycles and women with hypogonadotropic hypogonadism. Supplementation of LH to FSH is necessary for folliculogenesis, steroidogenesis, and clinical outcome. Recombinant LH and FSH are used in ART, and their utilization with GnRH analogues is still debatable [50]. GnRH agonists and antagonists have different modes of action . GnRH agonists, after an initial increase in LH and FSH secretion (flare up), induce downregulation of the GnRH receptor Conversely, GnRH antagonists competitively block the GnRH receptor thereby preserving pituitary gland responsiveness, so that gonadotropin levels are restored within a few hours of discontinuing suppression. Usually, the residual circulating levels of LH are sufficient to support steroidogenesis in theca cells, and recombinant human FSH (r-hFSH) is sufficient for OS. However, LH levels much lower than baseline can negatively affect MAR outcomes in some women [51].
It is not clear to which extent serum LH levels are predictive of reproductive outcome in GnRH agonist cycles. However, it should be noted that different LH threshold values were used in different studies to define low LH groups (i.e. <1.5–0.5 IU/l LH). It has been suggested that the severity of the LH deficiency caused by GnRH antagonist treatment is not linked to absolute LH serum levels but rather to the magnitude of suppression over time versus the baseline [51]. Eight systematic reviews and metaanalyses examined the GnRH analogues protocols in ART. They found a positive outcome for pregnancy achievement of r-hLH supplementation in the GnRH agonist method among women over 35 years old with a poor ovarian response but couldn’t conclude regarding the GnRH antagonist method. On the other hand, in the infertile population, r-hLH supplementation is not beneficial in the GnRH antagonist method and is still controversial in the GnRH agonist protocol. Finally, the LH threshold must be followed to decide which group of women should benefit from supplementation during GnRH analogs protocols [50].
GnRH analogues are employed in several IVF methods. The main disadvantage for GnRH antagonist administration is the decline in LH and E2 hormones, which leads to low IVF success rates due to the mediocre quality of embryos and the low number and poor quality of oocytes saved. A prospective, randomized, cross matched study was performed on 40 patients undergoing a GnRH antagonist treatment cycle to analyze the ideal timing and dose of r-hLH supplementation to improve IVF success. The results showed an improved ovarian response when r-hLH was administered at 150 IU/day starting from GnRH-antagonist administration [52].
Statement 13: In the modified natural cycle of frozen embryo transfer in normoresponder patients with regular ovulatory cycles, the HCG trigger demonstrates controversial efficacy compared to monitoring the LH peak or other therapeutic approaches such as hormone replacement therapy to optimize natural ovulation in endometrial preparation.
This statement received 80% total agreement from the extended panel (Figure 2). During the meeting of the seven experts, it was declared that hormone therapy is the most frequently used technique, fresh cycles are more common than frozen cycles in the Iraqi practice, frozen ones are used in PCOS anovulatory patients to avoid potential OHSS, and no significant difference exists between hCG triggering and LH monitoring in normoresponder patients.
IVF techniques have evolved, from fresh embryo transfer (ET) treatment to frozen-thawed embryo transfer (FET) cycles, which presents several benefits and is frequently applied nowadays. A successful pregnancy is based on two pillars, i.e., a good quality embryo and a receptive endometrium to synchronize together. Therefore, numerous techniques are used to prepare the endometrium for FET: Hormone Replacement Therapy (HRT) with or without GnRH analog, a ‘pure’ natural cycle based on the detection of the endogenous LH-surge in the blood (NC-FET) or a modified natural cycle (mNC-FET), using hCG for final oocyte maturation [53]. A systematic review conducted in 2016 compared the effectiveness of multiple endometrium preparation methods in 18 RCTs for FET in 3815 women. It showed no preference between NC-FET, mNC-FET, HT-FET alone or with GnRHa suppression, and HMG alone or with clomiphene [54]. Furthermore, Lawrenz et al., reported a higher risk of hypertensive diseases or preeclampsia during pregnancy after FET reproduction, likely due to multiple corpora lutea or the absence of a corpus luteum induced by ART methods. The pathogenesis of hypertension may be associated with the absence of corpus luteum, which contains relaxin, a vasodilator hormone. Prospective randomized controlled trials are necessary to confirm these findings since no published data support this hypothesis [53].
Statement 14: In a GnRH antagonist protocol, a GnRH agonist trigger is recommended for final oocyte maturation in women at risk of OHSS.
This statement received 97% total agreement from the extended panel (Figure 2). Oocyte maturation is a process where the oocyte gains the competence for fertilization necessary for the success of IVF techniques. This phenomenon needs the intervention of LH to happen. Many LH-like molecules can play this role and are used in ART, such as hCG, GnRH agonist, and r-hLH (although not common in clinical use). Furthermore, OHSS is a dangerous complication that could occur after extended exposure to LH-like molecules, especially seen with hCG as a trigger for oocyte maturation; it is due to induction of angiogenesis and increase in VEGF [55]. GnRH agonist is a safer option since it reduces exposure to LH-like molecules. Triggering with GnRH agonist aims to utilize its flare up effect and by that to induce endogenous release of LH and oocyte maturation with low to non- OHSS related adverse events. To mature follicles and prepare for ovulation, two main protocols are used: the “long” GnRH agonist and the “short” GnRH antagonist. The short protocol consists of competitive antagonist with inhibitory effect; it avoids premature ovulation. This protocol can be overcome with GnRH agonist. The short protocol, which allows better flexibility, permits GnRH agonist to induce final oocyte maturation. However, hCG or r-hLH can be employed in both short and long protocols [56].
This cotreatment is the safest option to alleviate OHSS risk in women [55]. A prospective randomized controlled study compared GnRH agonist trigger after GnRH antagonist cotreatment with hCG trigger and a dual pituitary suppression protocol in 66 women (<40 years old) at high risk of OHSS. According to the same study, high risk of OHSS patients are young women with PCOS, or with PCOS on ultrasound and patients with previous high response to gonadotropins [57].
The research showed a lower OHSS risk in the GnRH agonist protocol group: none of the patients developed OHSS versus 31% in the HCG trigger group. Moreover, there were no significant differences in implantation, clinical pregnancy, and ongoing pregnancy rates among both groups [57]. A systematic review and meta-analysis of randomized trials counting five studies of 859 patients also compared GnRH agonist trigger followed by luteal LH activity support and hCG trigger in IVF patients undergoing fresh embryo transfer. OHSS was reported in a total of 4/413 cases in the GnRH agonist group vs. 7/413 in the hCG group; there were no significant differences in LBR among groups and a slight but non-significant increase in miscarriage rates in the GnRH agonist triggered group compared to the hCG group [58].
Statement 15: The route of progesterone administration does not influence outcomes according to GPP. Vaginal progesterone therapy represents the most common approach for luteal phase support after IVF/ICSI.
This statement received 93% total agreement from the extended panel (Figure 2). A systematic qualitative review explained the role of LPS after IVF/ICSI. Since the luteal function is compromised by stimulated IVF, luteal support is fundamental in avoiding luteal insufficiency and its detrimental influence on early pregnancy. It also concluded that the optimal time for LPS would be between 24-72 hours after oocyte-retrieval and should continue at least until a positive pregnancy test is achieved. Several routes of progesterone administration are employed and comparable, the most frequent one being vaginal progesterone. Moreover, oral and subcutaneous progesterone are innovative ways with similar pregnancy rates and pregnancy loss rates to vaginal and intramuscular progesterone [59].
Another review reported the importance of LPS after IVF was conducted with either GnRH agonists or GnRH antagonists and showed comparable pregnancy rates between HCG and exogenous progesterone administration. Although hCG is associated with higher OHSS risk, a low-dose luteal hCG supplementation showed several advantages in GnRH antagonist cycles where GnRH agonists are used for the final maturation trigger. In addition, the multiple administration routes of progesterone presented the same efficacy [60]. A prospective randomized clinical trial involving 186 women compared different routes of progesterone administration for LPS in IVF. Patients were divided into three treatment regimens: rectal pessaries, vaginal pessaries, and vaginal capsules. The results showed no difference in implantation and clinical pregnancy rates per transfer among the three groups with the same perineal irritation rate as a side effect [61]. A randomized clinical trial, including 1031 subjects assigned to receive either oral dydrogesterone (n=520) or micronized vaginal progesterone MVP (n=511), showed no significant difference between the two groups [62].
Discussion
This Iraqi consensus provides a real-world clinical perspective from a group of experts on the specific approaches during the main steps of ART treatment in Iraq. The consensus has several strengths, including the fact that it used the Delphi technique [9]. It enabled the inclusion of more topics than would typically be addressed in a systematic review or a guideline approach, usually based on strict methodology limiting the scope of investigations. Besides using all types of published studies (systematic reviews, meta-analyses, randomized controlled trials, cohort studies, and case reports), this consensus benefited from the opinion of experts in the field who work with these patients daily. The consensus results are mainly in line with the ESHRE recommendations but often provide additional specific considerations. The consensus has some limitations. It only represents the collective point of view of the experts included and are based on local practice. Furthermore, not all statements reached 100% agreement, with some reaching consensus even though some participants disagreed with them.
Conclusion
This Iraqi consensus offers expert opinion on specific methods during ART treatment, including follicular development and stimulation with gonadotropins, pituitary suppression, final oocyte maturation triggering, and luteal phase support. All 15 statements reached consensus after the first vote, with a high level of agreement in over 85% of statements. Clinical perceptions in this consensus complement clinical guidelines and policies and help improve treatment results.
Acknowledgements
Authors would like to thank Science PRO sarl, Lebanon for their support in the medical writting and editing of the article.
Disclosure Statement
The consensus concept was initiated and funded by Merck KGaA, Darmstadt, Germany. The sponsor was involved early in the process, defining the overarching topic to be discussed, but did not participate in the development of the statements or in any of the meetings or discussions involved in developing the Delphi consensus. The statements were, therefore, developed independently of the industry sponsor. The authors from Merck KGaA were only involved in the development of the manuscript, critically revising it for important intellectual content, especially in the Introduction, Results and Discussion sections, but could not alter the consensus statements in any way.
Conflicts of Interest
All authors had received honoraria from Merck for participating in this consensus.
References
- How common is male infertility, an what are its causes? (2022)
- Mélodie Vander Borght , Christine Wyns (2018) Fertility and infertility: Definition and epidemiology. Clinical Biochemistry 62: 2-10.
- Sandro C Esteves, Peter Humaidan, Matheus Roque, Ashok Agarwal (2019) Female infertility and assisted reproductive technology. Panminerva Med 61(1): 1-2.
- Craig Niederberger, Antonio Pellicer (2018) Introduction: IVF’s 40th world birthday. Fertility and sterility 110(1): 4.
- Szamatowicz M (2016) Assisted reproductive technology in reproductive medicine — possibilities and limitations. Ginekologia Pol 87(12): 820-823.
- https://www.eshre.eu/Annual-Meeting/Barcelona-(2018)/ESHRE-(2018)-Press-releases/De-Geyter.
- J Ryan Martin, Jason G Bromer, Denny Sakkas, Pasquale Patrizio (2010) Live babies born per oocyte retrieved in a subpopulation of oocyte donors with repetitive reproductive success. Fertil Steril 94(6): 2064-2068.
- Bradley JVV (2006) Outcomes from assisted reproductive technology. Obstetrics and gynecology 107(1): 183-200.
- Raoul Orvieto, Christos A Venetis, Human M Fatemi, Thomas D'Hooghe, Robert Fischer, et al. (2021) Optimising Follicular Development, Pituitary Suppression, Triggering and Luteal Phase Support During Assisted Reproductive Technology: A Delphi Consensus. Frontiers in Endocrinology 12: 675670.
- Bart C J M Fauser, Basil C Tarlatzis, Robert W Rebar, Richard S Legro, Adam H Balen Fauser, et al. (2012) Consensus on women’s health aspects of polycystic ovary syndrome (PCOS): the Amsterdam ESHRE/ASRM-Sponsored 3rd PCOS Consensus Workshop Group. Fertility and sterility 97(1): 28-38.
- Helena J Teede, Marie L Misso, Michael F Costello, Anuja Dokras, Joop Laven, et al. (2018) Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Fertility and sterility 110(3): 364-379.
- Rao P, Bhide P (2020) Controversies in the diagnosis of polycystic ovary syndrome. Therapeutic advances in reproductive health 14: 2633494120913032.
- Sylvia Kiconco, Joop S E Laven, Helena J Teede (2020) Impact of the newly recommended antral follicle count cutoff for polycystic ovary in adult women with polycystic ovary syndrome. Human reproduction 35(9): 2166-2167.
- Tom Tanbo, Jan Mellembakken, Sverre Bjercke, Eva Ring, Thomas Åbyholm, et al. (2018) Ovulation induction in polycystic ovary syndrome. Acta Obstetricia et Gynecologica Scandinavica 97(10): 1162-1167.
- Rui Wang, Wentao Li, Esmée M Bordewijk, Richard S Legro, Heping Zhang, et al. (2019) First-line ovulation induction for polycystic ovary syndrome: an individual participant data meta-analysis. Human reproduction update 25(6): 717-732.
- EM Bordewijk, NS Weiss, MJ Nahuis, J Kwee, AF Lambeek, et al. (2020) Gonadotrophins or clomiphene citrate in women with normogonadotropic anovulation and CC failure: does the endometrium matter? Human reproduction (Oxford, England) 35(6): 1319-1324.
- DM White, DW Polson, D Kiddy, P Sagle, H Watson, et al. (1996) Induction of ovulation with low-dose gonadotropins in polycystic ovary syndrome: an analysis of 109 pregnancies in 225 women. The Journal of clinical endocrinology and metabolism 81(11): 3821-3824.
- Nienke S Weiss, Elena Kostova, Marleen Nahuis, Ben Willem J Mol, Fulco van der Veen, et al. (2019) Gonadotrophins for ovulation induction in women with polycystic ovary syndrome. The Cochrane Database of Systematic Reviews 1(1): CD010290.
- S Christin-Maitre, JN Hugues, Recombinant FSH Study Group (2003) A comparative randomized multicentric study comparing the step‐up versus step‐down protocol in polycystic ovary syndrome. Human Reproduction 18(8): 1626-1631.
- JN Hugues, I Cédrin-Durnerin, C M Howles, FSH OI Study Group, M Amram, et al. (2006) The use of a decremental dose regimen in patients treated with a chronic low-dose step-up protocol for WHO Group II anovulation: a prospective randomized multicentre study. Human Reproduction 21(11): 2817-2822.
- Yuji Taketani, Eduardo Kelly, Yasunori Yoshimura, Hiroshi Hoshiai, Minoru Irahara, et al. (2009) Recombinant follicle‐stimulating hormone (follitropin alfa) for ovulation induction in Japanese patients with anti‐estrogen‐ineffective oligo‐ or anovulatory infertility: results of a phase II dose-response study. Reproductive Medicine and Biology 9(2): 91-97.
- Loredana Bergandi, Stefano Canosa, Andrea Roberto Carosso, Carlotta Paschero, Gianluca Gennarelli, et al. (2020) Human Recombinant FSH and Its Biosimilars: Clinical Efficacy, Safety, and Cost-Effectiveness in Controlled Ovarian Stimulation for In Vitro Pharmaceuticals 13(7): 136.
- Mesen TB, Young SL (2015) Progesterone and the Luteal Phase. Obstetrics and gynecology clinics of North America 42(1): 135-151.
- Crinone (2011) Summary of Product Characteristics.
- Lawrenz, Barbara, Coughlan, Carol, Fatemi, Human M (2019) Individualized luteal phase support. Current Opinion in Obstetrics & Gynecology 31(3): 177-182.
- Zafeiriou S, Loutradis D, Michalas S (2000) The role of gonadotropins in follicular development and their use in ovulation induction protocols for assisted reproduction. The European journal of contraception & reproductive health care 5(2): 157-167.
- RW Shaw, G Ndukwe, D A Imoedemhe, A G Bernard, G Burford (1985) Twin pregnancy after pituitary desensitisation with LHRH agonist and pure FSH. Lancet 2(8453): 506-507.
- Chisa Tabata, Toshihiro Fujiwara, Miki Sugawa, Momo Noma, Hiroki Onoue, et al. (2015) Comparison of FSH and hMG on ovarian stimulation outcome with a GnRH antagonist protocol in younger and advanced reproductive age women. Reproductive medicine and biology 14(1): 5-9.
- Bosch E, Elena Labarta, Juana Crespo, Carlos Simón, José Remohí, et al. (2011) Impact of luteinizing hormone administration on gonadotropin-releasing hormone antagonist cycles: an age-adjusted analysis. Fertility and Sterility 95(3): 1031-1036.
- Nikolaos P Polyzos, Panagiotis Drakopoulos, Jose Parra, Antonio Pellicer, Samuel Santos-Ribeiro, et al. (2018) Cumulative live birth rates according to the number of oocytes retrieved after the first ovarian stimulation for in vitro fertilization/intracytoplasmic sperm injection: a multicenter multinational analysis including ∼15,000 women. Fertil Steril 110(4): 661-670.
- Sesh Kamal Sunkara, Vivian Rittenberg, Nick Raine-Fenning, Siladitya Bhattacharya, Javier Zamora, et al. (2011) Association between the number of eggs and live birth in IVF treatment: an analysis of 400 135 treatment cycles. Human reproduction 26(7): 1768-1774.
- Yin Jun Law, Ning Zhang, Efstratios M Kolibianakis, Michael F Costello, Elena Keller, et al. (2021) Is there an optimal number of oocytes retrieved at which live birth rates or cumulative live birth rates per aspiration are maximized after ART? A systematic review. Reproductive biomedicine online 42(1): 83-104.
- Antonio Pezzuto, Bruno Ferrari, Francesco Coppola, Giovanni Battista Nardelli (2010) LH supplementation in down-regulated women undergoing assisted reproduction with baseline low serum LH levels. Gynecological Endocrinology: The Official Journal of the International Society of Gynecological Endocrinology 26(2): 118-124.
- Micah J Hill, Gary Levy, Eric D Levens (2012) Does exogenous LH in ovarian stimulation improve assisted reproduction success? An appraisal of the literature. Reproductive Biomedicine Online 24(3): 261-271.
- Philippe Arvis, Nathalie Massin, Philippe Lehert (2021) Effect of recombinant LH supplementation on cumulative live birth rate compared with FSH alone in poor ovarian responders: a large, real-world study. Reproductive Biomedicine Online 42(3): 546-554.
- Yuning Wang, Ling Li, Ke Deng, Jiali Liu, Yanmei Liu, et al. (2020) Comparison of the combination of recombinant follicle-stimulating hormone and recombinant luteinizing hormone protocol versus human menopausal gonadotropin protocol in controlled ovarian stimulation: A systematic review and meta-analysis. Journal of evidence-based medicine 13(3): 215-226.
- Paolo Emanuele Levi-Setti, Irene Zerbetto, Annamaria Baggiani, Elena Zannoni, et al. (2019) An Observational Retrospective Cohort Trial on 4,828 IVF Cycles Evaluating Different Low Prognosis Patients Following the POSEIDON Criteria. Frontiers in Endocrinology 282.
- Alessandro Pacchiarotti, Marco Sbracia, Antonio Frega, Helmy Selman, Leonardo Rinaldi, et al. (2010) Urinary hMG (Meropur) versus recombinant FSH plus recombinant LH (Pergoveris) in IVF: A multicenter, prospective, randomized controlled trial. Fertility and sterility 94(6): 2467-2469.
- Livio Casarini, Daniele Santi, Giulia Brigante, Manuela Simoni (2018) Two Hormones for One Receptor: Evolution, Biochemistry, Actions, and Pathophysiology of LH and hCG. Endocrine Reviews 39(5): 549-592.
- KC Mcfarland, Rolf Sprengel, Heidi S Phillips, Martin Köhler, Nora Rosemblit, et al. (1989) Lutropin-choriogonadotropin receptor: an unusual member of the G protein-coupled receptor family. Science 245: 4917.
- Paul Grzesik Annika, Kreuchwig Claudia Rutz, Jens Furkert, Burkhard Wiesner, Ralf Schuelein, et al. (2015) Differences in Signal Activation by LH and hCG are Mediated by the LH/CG Receptor’s Extracellular Hinge Region. Frontiers in Endocrinology 6.
- X Jiang, M Dreano, DR Buckler, S Cheng, A Ythier, et al. (1995) Structural predictions for the ligand-binding region of glycoprotein hormone receptors and the nature of hormone-receptor interactions. Structure 3(12): 1341-1353.
- Britta Troppmann, Gunnar Kleinau, Gerd Krause, Jörg Gromoll (2013). Structural and functional plasticity of the luteinizing hormone/choriogonadotrophin receptor. Human Reproduction Update 19(5): 583-602.
- Livio Casarini, Monica Lispi, Salvatore Longobardi, Fabiola Milosa, Antonio La Marca, et al. (2012) LH and hCG Action on the Same Receptor Results in Quantitatively and Qualitatively Different Intracellular Signalling PLoS ONE 7(10).
- Janet Choi, Johan Smitz (2014) Luteinizing hormone and human chorionic gonadotropin: origins of difference. Molecular and cellular endocrinology 383(1-2): 203-213.
- Livio Casarini, Laura Riccetti, Francesco De Pascali, Lisa Gilioli, Marco Marino, et al. (2017) Estrogen Modulates Specific Life and Death Signals Induced by LH and hCG in Human Primary Granulosa Cells In Vitro. International journal of molecular sciences 18(5): 926.
- Ruolin Wang, Shouren Lin, Yong Wang, Weiping Qian, Liang Zhou (2017) Comparisons of GnRH antagonist protocol versus GnRH agonist long protocol in patients with normal ovarian reserve: A systematic review and meta-analysis. PloS one 12(4): e0175985.
- Wanlin Zhang, Duo Xie, Hengde Zhang, Jianlei Huang, Xifeng Xiao, et al. (2020) Cumulative Live Birth Rates After the First ART Cycle Using Flexible GnRH Antagonist Protocol vs. Standard Long GnRH Agonist Protocol: A Retrospective Cohort Study in Women of Different Ages and Various Ovarian Reserve. Frontiers in endocrinology 11: 287.
- Hesham G Al-Inany, Mohamed Afm Youssef, Mohamed Aboulghar, Frank Broekmans, Monique Sterrenburg, et al. (2011) Gonadotrophin-releasing hormone antagonists for assisted reproductive technology. The Cochrane database of systematic reviews 11(5).
- Johnny S Younis, Neri Laufer (2018) Recombinant luteinizing hormone supplementation to recombinant follicle stimulating hormone therapy in gonadotropin releasing hormone analogue cycles: what is the evidence? Current medical research and opinion 34(5): 881-886.
- E Bosch, C Alviggi, M Lispi, A Conforti, A C Hanyaloglu, et al. (2021) Reduced FSH and LH action: implications for medically assisted reproduction. Human Reproduction 36(6): 1469-1480.
- Salvatore Gizzo, Alessandra Andrisani, Marco Noventa, Serena Manfè, Alessandra Oliva, et al. (2015) Recombinant LH supplementation during IVF cycles with a GnRH-antagonist in estimated poor responders: A cross-matched pilot investigation of the optimal daily dose and timing. Molecular medicine reports 12(3): 4219-4229.
- Barbara Lawrenz, Carol Coughlan, Laura Melado, Human M Fatemi (2020) The ART of frozen embryo transfer: back to nature! Gynecological endocrinology : the official journal of the International Society of Gynecological Endocrinology 36(6): 479-483.
- Tarek Ghobara, Tarek A Gelbaya, Reuben Olugbenga Ayeleke (2017) Cycle regimens for frozen-thawed embryo transfer. The Cochrane database of systematic reviews 7(7).
- Ali Abbara, Sophie A Clarke, and Waljit S Dhillo (2018) Novel Concepts for Inducing Final Oocyte Maturation in In Vitro Fertilization Treatment. Endocrine reviews 39(5): 593-628.
- Deepika Krishna, Snehal Dhoble, Gautham Praneesh, Suvarna Rathore, Amit Upadhaya, et al. (2016) Gonadotropin-releasing hormone agonist trigger is a better alternative than human chorionic gonadotropin in PCOS undergoing IVF cycles for an OHSS Free Clinic: A Randomized control trial. Journal of Human Reproductive Sciences 9(3): 164-172.
- Lawrence Engmann, Andrea DiLuigi, David Schmidt, John Nulsen, Donald Maier, et al. (2008) The use of gonadotropin-releasing hormone (GnRH) agonist to induce oocyte maturation after cotreatment with GnRH antagonist in high-risk patients undergoing in vitro fertilization prevents the risk of ovarian hyperstimulation syndrome: a prospective randomized controlled study. Fertility and sterility 89 (1): 84-91.
- Thor Haahr, Matheus Roque, Sandro C Esteves, Peter Humaidan (2017) GnRH Agonist Trigger and LH Activity Luteal Phase Support versus hCG Trigger and Conventional Luteal Phase Support in Fresh Embryo Transfer IVF/ICSI Cycles-A Systematic PRISMA Review and Meta-analysis. Frontiers in endocrinology 8: 116.
- Vlatka Tomic, Miro Kasum, Katarina Vucic (2019) The role of luteal support during IVF: a qualitative systematic review. Gynecological endocrinology : the official journal of the International Society of Gynecological Endocrinology 35(10): 829-834.
- Yanushpolsky, E.H. (2015) Luteal phase support in in vitro Seminars in reproductive medicine 33(2): 118-127.
- Mohamed Khrouf, Soufiene Slimani, Myriam Razgallah Khrouf, Marouen Braham, Maha Bouyahia, et al. (2016) Progesterone for Luteal Phase Support in In Vitro Fertilization: Comparison of Vaginal and Rectal Pessaries to Vaginal Capsules: A Randomized Controlled Study. Clinical medicine insights. Women’s health 9: 43-47.
- Herman Tournaye, Gennady T Sukhikh, Elke Kahler, Georg Griesinger et al. (2017) A Phase III randomized controlled trial comparing the efficacy, safety and tolerability of oral dydrogesterone versus micronized vaginal progesterone for luteal support in in vitro fertilization. Human reproduction (Oxford, England) 32(5): 1019-1027.






























