Spurious and Substantial-Solid Pseudo-Papillary Neoplasm-Pancreas
Anubha Bajaj*
AB Diagnostics, India
Submission: February 22, 2023; Published:February 24, 2023
*Corresponding author: Anubha Bajaj, AB. Diagnostics, New Delhi, 110027, India Anubha Bajaj, AB. Diagnostics, New Delhi, 110027, India
How to cite this article: Anubha Bajaj. Spurious and Substantial-Solid Pseudo-Papillary Neoplasm-Pancreas. Glob J Reprod Med. 2023; 9(4): 555767. DOI: 10.19080/GJORM.2023.09.555767.
Keywords: Neoplasm; Pancreas; Pseudo-papillary neoplasm
Editorial
Solid pseudo-papillary neoplasm pancreas configures as a potential low malignant tumour demonstrating an undefined cell of origin. The neoplasm was incorporated into contemporary classification of pancreatic tumours as designated by World Health Organization (WHO) in 1996. The neoplasm incriminates young females and characteristically exemplifies a solitary tumour mass confined to body or tail of pancreas. Tumour is additionally designated as solid pseudo-papillary tumour, papillary epithelial neoplasm, papillary cystic neoplasm, solid and papillary neoplasm, low grade papillary neoplasm and Hamoudi or Frantz tumour. Of obscure pathogenesis, tumefaction is posited to be derived from pluripotent stem cell component of genital ridges which adhere to pancreas during embryogenesis. The exceptional solid pseudo-papillary neoplasm configures ~ 2% of pancreatic neoplasms. A female predominance is encountered with female to male proportion of 10:1 [1,2]. Tumefaction commonly emerges within third decade to fourth decade with mean age of disease emergence at 35 years. An estimated 30% of pancreatic tumours arising in females below < 40 years are comprised of solid pseudo-papillary neoplasm. Tumours confined to male subjects appear in older individuals and manifest an aggressive biological behaviour. Occasionally, solid pseudo-papillary neoplasm may concur with familial adenomatous polyposis [1,2]. Solid pseudo-papillary neoplasm may comprehensively incriminate the pancreas although neoplasms occurring in adults frequently emerge within body or tail of pancreas. In contrast, paediatric tumours appear confined to head of pancreas. Exceptionally, extra-pancreatic sites such as mesocolon, omentum, ovary or testis may be implicated [1,2]. Solid pseudo-papillary neoplasm pancreas preponderantly (>90%) demonstrates point mutation within exon 3 of β catenin gene (CTNNB1). Subsequently, genetic mutations induce intracytoplasmic accumulation of β catenin with the configuration of β catenin Tcf / Lef complex. Consequently, Wnt signalling pathway activates diverse oncogenic genes such as MYC and cyclin D1. Exceptionally, genetic mutations within APC gene are documented [2,3].
Solid pseudo-papillary neoplasm commonly demonstrates cogent clinical symptoms as abdominal pain and a palpable, non-tender tumefaction confined to upper abdomen. Besides, symptoms contingent to an intra-abdominal mass effect as abdominal discomfort, nausea, vomiting or early satiety may ensue [2,3].
Upon gross examination, the neoplasm varies from 0.5 centimetre to 34.5 centimetres magnitude with a mean tumour diameter of 6 centimetres. Tumefaction is well defined, encapsulated and exhibits variable quantities of solid and cystic tumour patterns. Miniature lesions appear as solid, minimally circumscribed neoplasms. Enlarged tumours appear encompassed with a fibrous pseudo-capsule. Cut surface is variegated and friable. Foci of cystic degeneration and haemorrhage are commonly encountered. Exceptionally, tumefaction may extend into adjacent structures as the duodenum [2,3].
Cytological examination depicts cellular smears with configuration of delicate, papillary fronds. Neoplastic cells appear bland, uniform and imbued with moderate quantities of cytoplasm, clear perinuclear vacuoles of variable magnitude, intracytoplasmic eosinophilic hyaline globules, spherical or elliptical nuclei with grooves and finely granular nuclear chromatin. Additionally, features such as cercariform cells, cytoplasmic vacuoles, reniform nuclei or hyaline globules may be discerned. Foci of cellular or stromal degeneration as cholesterol crystals, calcification, foamy macrophages or giant cells are commonly encountered within solid pseudo-papillary neoplasm, in contrast to acinar cell carcinoma or neuroendocrine tumour of pancreas [2,3].
Upon microscopy, tumour appears heterogeneous and demonstrates a variable admixture of solid and pseudo-papillary zones. Solid areas are constituted of uniform neoplastic cells intermingled with vascular articulations akin to capillaries.
Pseudo-papillae are configured due to dissociation of tumour cells from vascular configurations wherein pseudo-papillae articulate fibro-vascular stalks or rosette-like structures. Tumour cells are incorporated with moderate quantities of eosinophilic cytoplasm, perinuclear vacuoles and intracytoplasmic hyaline globules which appear stainable by periodic acid Schiff’s (PAS+) stain with diastase resistance and are immune reactive to alpha-1-antitrypsin [2,3]. Nuclei of carcinoma cells appear uniform and permeated with finely textured nuclear chromatin, inconspicuous nucleoli and characteristic longitudinal grooves. Mitotic activity is exceptional. Circumscribing stroma displays variable hyalinization or evidence of stromal degeneration along with foci of haemorrhage, accumulated foamy macrophages, calcification and cholesterol clefts. Although the neoplasm is well circumscribed upon gross examination, microscopic infiltration into encompassing pancreatic tissue is commonly discerned. Neoplasms depicting cogent histological features as a diffuse pattern of tumour evolution, sarcomatoid features, extensive necrosis, significant nuclear atypia or elevated mitotic activity comprised of 35 mitotic figures to 70 mitotic figures/50 high power fields are infrequently associated with markedly aggressive clinical behaviour [2,3]. Tumefaction may be composed of papillary fronds localized upon myxoid or hyalinised vascular stalks coated with loosely cohesive, uniform neoplastic cells demonstrating nuclear grooves. Solid and cystic tumour areas appear immune reactive to beta catenin within cytoplasmic or nuclear zones [2,3]. Solid pseudo-papillary neoplasm pancreas expounds distinctive variants as clear cell, oncocytic and pleomorphic. Upon ultrastructural examination, cellular cytoplasm appears mitochondria rich and permeated with electron dense granules along with rough endoplasmic reticulum (Figure 1 & 2) [2,3].
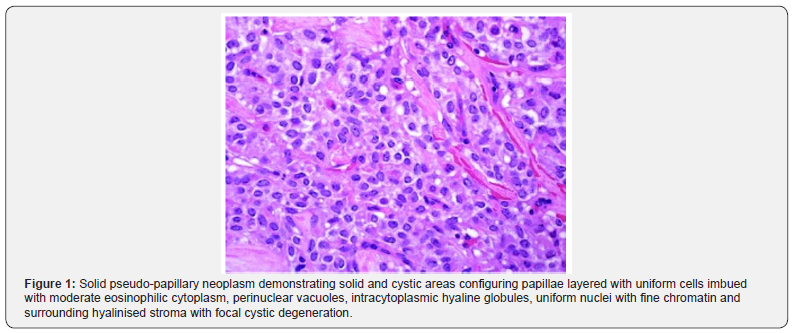
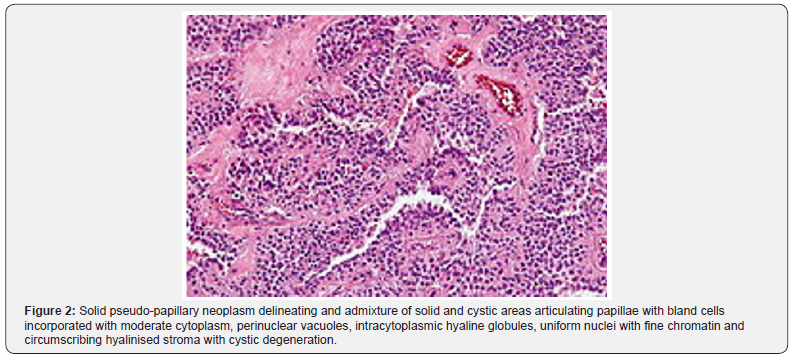
Solid pseudo-papillary neoplasm pancreas appears immune reactive to beta catenin, E-cadherin, p120, alpha-1- antichymotrypsin, alpha-1-antitrypsin, vimentin, cyclin D1, CD10, SOX11, androgen receptor, TFE3, LEF1, FUS, progesterone receptor, claudin7, claudin5, CD56, neuron specific enolase, synaptophysin, cytokeratin or CD99. Solid pseudo-papillary neoplasm pancreas is immune non-reactive to chromogranin A, carcinoembryonic antigen (CEA) or oestrogen receptor [4,5] (Table 1).
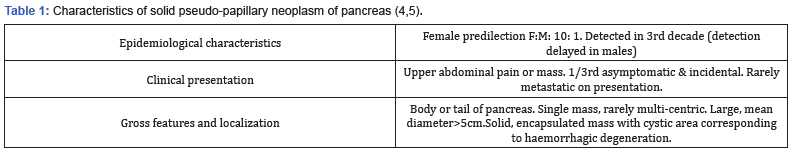
Solid pseudo-papillary neoplasm pancreas requires segregation from neoplasms such as pancreatic neuroendocrine tumour, acinar cell carcinoma, mucinous cystic neoplasm of pancreas, serous micro-cystic adenoma, neuroendocrine tumour with cystic features as cystadenoma or cystadenocarcinoma, intraductal papillary mucinous neoplasm (IPMN) or pancreatoblastoma [4,5]. Majority of tumours are discovered incidentally upon imaging. Upon imaging, a solid or solid and cystic tumour mass appears confined within body or tail of pancreas. Young women are preponderantly incriminated. Upon computerized tomography (CT) and magnetic resonance imaging (MRI), a well circumscribed, encapsulated, heterogeneous pancreatic lesion with cystic degeneration is observed [4,5]. Aspirated cyst fluid is frequently haemorrhagic. Fluid levels of carcinoembryonic antigen (CEA) and amylase appear decimated. Cogent cytological and histological evaluation is pre-requisite for appropriate confirmation of the neoplasm [4,5]. Surgical resection of solid pseudo-papillary neoplasm pancreas appropriately alleviates the neoplasm in a majority (>95%) of instances. Factors contributing to inferior prognostic outcomes are tumour magnitude > 5 centimetres, incrimination of male subjects, occurrence of tumour necrosis, cellular atypia, vascular invasion, perineural invasion or tumour infiltration into adjacent anatomical structures [4,5].
References
- Paluri RK, Babiker HM (2022) Solid and Papillary Epithelial Neoplasm. Stat Pearls International Treasure Island, Florida.
- Sibio S, Di Carlo S (2022) Current highlights on solid pseudopapillary neoplasm of the pancreas. World J Hepatol 14(1): 300-303.
- Tasar P, Kilicturgay SA (2022) Solid pseudopapillary neoplasms of the pancreas: Is there a factor determining the prognosis? Experience of a single institution. Medicine (Baltimore) 101(34): e30101.
- Maimaijiang A, Wang H, Li W, Yaqi Wang (2022) Diagnosis and treatment of solid pseudopapillary neoplasm of the pancreas in children: A report of 18 cases. Front Pediatr 10: 899965.
- Cruz MAA, Moutinho-Ribeiro P, Costa-Moreira P, Macedo G (2021) Solid Pseudopapillary Neoplasm of the Pancreas: Unfolding an Intriguing Condition. GE Port J Gastroenterol 29(3): 151-162.






























