Miracle Pregnancy: Management of Poor Responder in In-Vitro Fertilization
Neeta Singh1 Ankita Sethi2*, Garima Patel2 and Monika Saini1
1 Division of Reproductive Medicine, Department of Obstetrics and Gynaecology, All India Institute of Medical Sciences, India
2 DM Reproductive Medicine Resident, Department of Obstetrics and Gynaecology, All India Institute of Medical Sciences, India
Submission: June 10, 2022; Published:June 28, 2022
*Corresponding author: Ankita Sethi, DM Reproductive Medicine Resident, Department of Obstetrics and Gynaecology, All India Institute of Medical Sciences, New Delhi, India
How to cite this article: Arzu Y, Nurettin T, Semih Zeki U. Effect of Prolonged GnRH Agonist Therapy Prior to Frozen Embryo Transfer on IVF-ET Outcome in Patients with and without Endometrioma Surgery. Glob J Reprod Med. 2022; 9(1): 555754. DOI: 10.19080/GJORM.2022.09.555754.
Abstract
Background:: Patient-Oriented Strategy Encompassing IndividualizeD Oocyte Number (POSEIDON) 3 and 4 subgroup is common nowadays. We hereby report a case of successful management of POSEIDON 4 patient, who underwent an individualized GnRH antagonist protocol.
Case Presentation:: A 36-year-old woman presented with primary infertility for 10 years. She had undergone multiple cycles of ovulation induction and intrauterine insemination previously. She had previous one IVF failure. Post COVID lockdown, patient was a POSEIDON 4 group patient who underwent second IVF cycle. Three grade 4 oocytes were retrieved after dual trigger. Two 8 celled grade B embryos were transferred on day 3. Patient conceived and had caesarean section at 37 weeks POG, 2.4 kg healthy male baby delivered.
Conclusion:: IVF with self-oocyte can be tried along with proper counselling to decrease the psychological and financial burden on the patient and family, therefore, ruling out oocyte donation as the only treatment option in these patients.
Keywords: POSEIDON 4; IVF; Individualised Treatment; GnRH antagoniste
Abbreviations: ESHRE: European Society of Human Reproduction and Embryology; POSEIDON: Patient-Oriented Strategies Encompassing Individualized Oocyte Number; GnRH: Gonadotrophin releasing Hormone; IVF: In Vitro Fertilization; FSH: Follicle Stimulating Hormone; HMG: Human Menopausal Gonadotrophins; rHCG: recombinant Human Chorionic Gonadotropin; E2: Estradiol; P4: Progesterone; UPT: Urine Pregnancy Test; COVID-19: CORONAVIRUS DISEASE-2019; AFC: Antral Follicle Count; DHEAS: Dehydroepiandrosterone Sulfate; ICSI: Intra-cytoplasmic Sperm Injection; AMH: Anti-Mullerian Hormone; r-hLH: Recombinant Luteinizing Hormone; POG: Period of Gestation; PGT: Preimplantation Genetic Testing
Introduction
Poor responders are often encountered by Reproductive medicine specialists around the world. A lot of research has been directed towards finding reasonable solutions for this group of patients. The lack of a universal definition has induced a considerable degree of heterogeneity to research on stimulation protocols in poor responders. Several definitions and methods have been proposed for the identification of poor responders, for individualization of their treatment, including the European Society of Human Reproduction and Embryology (ESHRE) Bologna criteria for poor responders, and the Patient-Oriented Strategies Encompassing Individualized Oocyte Number (POSEIDON) criteria [1,2]. Here we present a case of poor responder (POSEIDON 4) and her personalised/individualized successful management.
Case Presentation
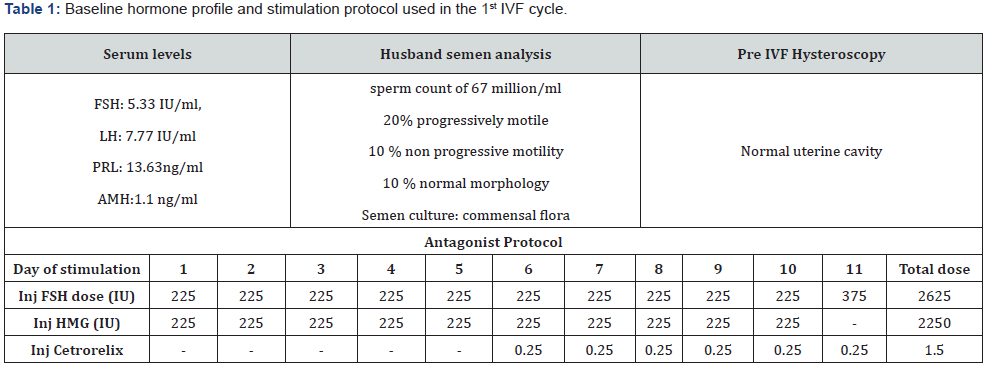
Patient X, a 36-year-old woman presented with primary infertility for 10 years. She had undergone multiple cycles of ovulation induction and intrauterine insemination previously. Patient was evaluated clinically with thorough history, examination, and investigations. She underwent her first Gonadotrophin releasing Hormone (GnRH) antagonist In-Vitro Fertilization (IVF) cycle in November 2019 as mentioned in protocol (Table 1). The dose of gonadotrophins used was 2625 IU rFSH (Follicle Stimulating Hormone) and 2250 IU HMG (Human Menopausal gonadotrophins), so total 4875 IU of gonadotrophins in first IVF cycle and total 11 days of stimulation. Six oocytes were retrieved after 36 hours of rHCG (recombinant human chorionic gonadotropin) 250 microgram trigger. Serum E2(estradiol) on day of trigger was 1495 pg/ml and P4 (progesterone) 0.9 ng/ml. Conventional IVF insemination was performed with two grade 1 and four grade 2 oocytes, which resulted two 2 celled grade Bembryo, which were transferred on day 2. Urine Pregnancy test (UPT) was done after 16 days which was negative and beta hCG <1.2 IU/ml.
Patient expressed her wish for second attempt and was postponed due to COVID-19 (CORONAVIRUS DISEASE-2019) pandemic. Therefore, DHEAS (Dehydroepiandrosterone sulfate) supplements were prescribed for 6 months till her next recruitment. Post lockdown, the initial AFC (antral follicle count) at start of cycle was low (right ovary=2, left ovary=3). Antagonist Protocol followed as mentioned in table 2. The total dose of gonadotrophins used was 3800 IU rFSH and 600 IU HMG, so total 4400 IU of gonadotrophins in second IVF cycle, total 10 days of stimulation. Three grade 4 oocytes were retrieved post 35 hours of dual trigger (Inj rHCG (Ovitrelle) 250 microgram and inj leuprolide 1 mg subcutaneously). Initially, ICSI (intracytoplasmic sperm injection) was planned because of poor fertilization rate in previous IVF cycle. However, conventional IVF was performed because of poor quality of oocytes. Fortunately, two oocytes fertilized (2 out of 3) and two 8 celled grade B embryos were available for transfer on day 3, much better quality as compared to first IVF cycle. Urine pregnancy test and beta HCG was done after 16 days, which was positive. Single live intrauterine fetus was documented at 6 weeks period of gestation and luteal phase support (Vaginal micronized progesterone 400 mg twice a day and injection micronized progesterone 100 mg intramuscularly OD) was continued throughout first trimester. Patient had caesarean section at 37 weeks POG, 2.4 kg healthy male baby delivered. We are reporting this to spread the message that POSEIDON 4 patients can have successful pregnancy outcomes with individualized/ personalized treatment and that oocyte donation is not the only alternative.

Discussion
The ESHRE Bologna criteria 2011 gave the definition of poor responders [2]. According to Bologna criteria, poor responders are patients from whom we would expect to retrieve 3 oocytes after conventional COS and markers for identifying poor responders are a previous poor response (three oocytes retrieved after COS), AMH (Anti-mullerian Hormone) <0.5 ng/mL or AFC < 3 [1,3]. Though it encompasses a broad, heterogeneous group of patients but it is not considered as clinically relevant for making informed treatment decisions [4-6]. Because it does not differentiate between oocyte quality and oocyte quantity [6]. Suboptimal responders are not widely recognized as a patient subgroup and were categorized either into normal responders or poor responders [1,7]. The POSEIDON Group bridged this gap and formulated these criteria by consensus that identify different groups of low prognosis patients [1]. These were developed specifically with the aim of guiding personalized treatment protocols for second and subsequent cycles, following unexpected suboptimal or poor response. POSEIDON criteria offer the best approach to personalization for poor/suboptimal ovarian responders, to assist the clinical decision-making, patient counselling and prognostication.
The biomarkers to identify suboptimal responders are: (a) previous history of suboptimal response (>4 to 9 oocytes retrieved after COS) (b) AMH > 0.5 to <1.2 ng/mL (c) AFC > 4 to 9. These constitutes 43.3% of patients undergoing IVF treatment according to study by Polyzos et al. [8]. A recent meta-analysis, shows that suboptimal responders and potentially other patient populations are likely to benefit from r-hLH (Recombinant Luteinizing hormone) supplementation with respect to implantation, pregnancy, and live birth rates [9-12]. Age is an important prognostic marker for IVF success, and it is expected that the younger patient will have a 4 times higher chance of live birth as compared to the older patient [13]. In POSEIDON group 3 (age < 35 years, AMH <1.2 ng/ml, AFC<5) patients, recommended protocols are either long GnRH agonist down-regulation or a “primed” GnRH antagonist co-treatment followed by stimulation with a maximum dose of 300 rFSH. In selected cases with a low oocyte yield and based on the estimate made by the ART (Assisted reproductive technology) calculator, DUO-Stim should be recommended for oocyte or embryo accumulation to shorten time to pregnancy [14,15]. With the increasing prevalence of POSEIDON group 4 patients (age >35 years, AMH <1.2 ng/ml, AFC <5) due to delay in age of childbearing [16], the dual effect of poor ovarian reserve (quantity) and age-related decline of quality makes this category of patients difficult to handle [17]. Though oocyte donation is a treatment option but there is a stigma with it especially in Indian society [18]. At the same time, despite poor prognosis, we cannot deny IVF with self-oocyte. Therefore, proper counselling is required explaining the chance of cycle cancellation and risk of empty follicle syndrome.
The case in discussion received starting dose of 225 IU rFSH and 225 IU HMG in first IVF cycle, but still patient had poor outcome in terms of poor quality of oocytes, poor fertilization rate and no clinical pregnancy. Therefore, in the second cycle, even though patient fits in POSEIDON 4 classification, we took the decision to administer 450 IU rFSH from day 2 for 5 days, followed by 300 IU rFSH and 150 IU HMG from day 6 when the antagonist was added. In first and second cycle, total dose and days of gonadotrophins used was 4825 IU and 11 days, 4400 IU and 10 days respectively. This is in complement to the literature that escalating the dose of gonadotrophins and increased duration of stimulation can negatively affect the IVF outcome [19]. Another change in treatment was dual trigger compared to HCG trigger in 1st cycle because it is associated with better IVF outcome in terms of number of total embryos and grade 1 embryos in poor responders’ patients [20]. Following this change in protocol, we had better oocyte yield, fertilization rate, embryo quality, positive urine pregnancy test and ongoing pregnancy at 22 weeks POG (period of gestation). As patient was 36 years old, PGT (Preimplantation Genetic Testing) could be considered to check the ploidy status of the embryos but was avoided due to deleterious effect of embryo loss and further hampering the chance of embryo implantation/viability on biopsy of day 3 embryos [21].
Conclusion
Therefore, would conclude that individualized/personalized treatment is required in POSEIDON 3 and 4 patients, and no fixed stimulation protocol can be followed. IVF with self-oocyte can be tried along with proper counselling to decrease the psychological and financial burden on the patient and family, therefore, ruling out oocyte donation as the only treatment option in these patients.
Declarations
Ethics approval and consent to participate: Not Applicable.
Consent for publication: Taken from the concerned patient .
Availability of data and material: Patient details are available.
Competing interests: No competing interests
Funding: No funding was required.
Authors’ Contributions:
NS, AS, GP were involved in clinical management of the patient and MS was the embryologist involved in the case, all authors were involved in writing and peer review of the final manuscript.
Acknowledgements:
We would like to thank the Division of Reproductive Medicine, Department of Obstetrics and Gynaecology, All India Institute of Medical Sciences, New Delhi.
References
- Alviggi C, Andersen CY, Buehler K, Conforti A, De Placido G, et al. (2016) A new more detailed stratification of low responders to ovarian stimulation: from a poor ovarian response to a low prognosis concept. Fertil Steril 105(6): 1452-1453.
- Ferraretti AP, La Marca A, Fauser BC, Tarlatzis B, Nargund G, et al. (2011) ESHRE consensus on the definition of “poor response” to ovarian stimulation for in vitro fertilization: the Bologna criteria. Hum Reprod 26(7): 1616-1624.
- Lehert P, Chin W, Schertz J, D’Hooghe T, Alviggi C, et al. (2018) Predicting live birth for poor ovarian responders: the PROsPeR concept. Reprod Biomed Online 37(1): 43-52.
- Younis JS, Ben-Ami M, Ben-Shlomo I (2015) The Bologna criteria for poor ovarian response: a contemporary critical appraisal. J Ovarian Res 8(1): 76.
- Venetis CA (2014) The Bologna criteria for poor ovarian response: the good, the bad and the way forward. Hum Reprod 29(9): 1839-1841.
- Ferraretti AP, Gianaroli L (2014) The Bologna criteria for the definition of poor ovarian responders: is there a need for revision? Hum Reprod 29(9): 1842-1845.
- Polyzos NP, Sunkara SK (2015) Reply: Is it necessary to recognize the sub-optimal responder. Hum Reprod 30(12): 2959.
- Polyzos NP, Sunkara SK (2015) Sub-optimal responders following controlled ovarian stimulation: an overlooked group? Hum Reprod 30(9): 2005-2008.
- Alviggi C, Conforti A, Esteves SC, Andersen CY, Bosch E, et al. (2018) Recombinant luteinizing hormone supplementation in assisted reproductive technology: a systematic review. Fertil Steril 109(4): 644-664.
- Conforti A, Esteves SC, Di Rella F, Strina I, De Rosa P, et al. (2019) The role of recombinant LH in women with hypo-response to controlled ovarian stimulation: a systematic review and meta-analysis. Reprod Biol Endocrinol 17(1): 18.
- Mochtar MH, Danhof NA, Ayeleke RO, Van der Veen F, van Wely M (2017) Recombinant luteinizing hormone (rLH) and recombinant follicle stimulating hormone (rFSH) for ovarian stimulation in IVF/ICSI cycles. Cochrane Database Syst Rev 5(5): CD005070.
- Santi D, Casarini L, Alviggi C, Simoni M (2017) Efficacy of Follicle-Stimulating Hormone (FSH) alone, FSH + Luteinizing Hormone, Human Menopausal Gonadotropin or FSH + Human Chorionic Gonadotropin on assisted reproductive technology outcomes in the “personalized” medicine era: a meta-analysis. Front Endocrinol (Lausanne) 8: 114.
- De Geyter C, Fehr P, Moffat R, Gruber IM, von Wolff M (2015) Twenty years' experience with the Swiss data registry for assisted reproductive medicine: outcomes, key trends, and recommendations for improved practice. Swiss Med Wkly 145: w14087.
- Alsbjerg B, Haahr T, Elbaek HO, Laursen R, Povlsen BB, et al. (2019) Dual stimulation using corifollitropin alfa in 54 Bologna criteria poor ovarian responders - a case series. Reprod Biomed Online 38(5): 677-682.
- Vaiarelli A, Cimadomo D, Trabucco E, Vallefuoco R, Buffo L, et al. (2018) Double stimulation in the same ovarian cycle (duostim) to maximize the number of oocytes retrieved from poor prognosis patients: a multicenter experience and swot analysis. Front Endocrinol (Lausanne) 9: 317.
- Conforti A, Esteves SC, Picarelli S, Iorio G, Rania E, et al. (2019) Novel approaches for diagnosis and management of low prognosis patients in assisted reproductive technology: the POSEIDON concept. Panminerva Med 61(1): 24-29.
- Esteves SC, Carvalho JF, Martinhago CD, Melo AA, Bento FC, et al. (2018) Estimation of age-dependent decrease in blastocyst euploidy by next generation sequencing: development of a novel prediction model. Panminerva Med 61(1): 3-10.
- Petersen KB, Hvidman HW, Sylvest R, Pinborg A, Larsen EC, et al. (2015) Family intentions and personal considerations on postponing childbearing in childless cohabiting and single women aged 35-43 seeking fertility assessment and counselling. Hum Reprod 30(11): 2563-2574.
- Sekhon L, Shaia K, Santistevan A, Cohn KH, Lee JA, et al. (2017) The cumulative dose of gonadotropins used for controlled ovarian stimulation does not influence the odds of embryonic aneuploidy in patients with normal ovarian response. J Assist Reprod Genet 34(6): 749-758.
- Maged AM, Ragab MA, Shohayeb A, Saber W, Ekladious S, et al. (2021) Comparative study between single versus dual trigger for poor responders in GnRH‐antagonist ICSI cycles: a randomized controlled study. Int J Gynecol Obstet 152(3): 395-400.
- Scott RT, Upham KM, Forman EJ, Zhao T, Treff NR (2013) Cleavage-stage biopsy significantly impairs human embryonic implantation potential while blastocyst biopsy does not: a randomized and paired clinical trial. Fertil Steril 100(3): 624-630.






























