Results of Xenform® Matrix Repair in Treatment of Pelvic Organ Prolapse- A Four Year Study
Amita Singla*, Sujana Molaktalla and Priti Pradhan
Consultant, Obstetrics and Gynaecology, The Queen Elizabeth Hospital and Calvary Hospital, Adelaide
Submission: February 18, 2019; Published: May 03, 2019
*Corresponding author: Dr. Amita Singla. Department of Gynaecology The Queen Elizabeth Hospital 28, Woodville Road, Woodville SA 5011, Australia
How to cite this article: Amita Singla, Sujana Molaktalla, Priti Pradhan. Results of Xenform® Matrix Repair in Treatment of Pelvic Organ Prolapse- A Four Year Study. Glob J Reprod Med. 2019; 6(5): 555696. DOI:10.19080/GJORM.2019.06.555696.
Abstract
Abbrevations: WHI: Women’s Health Initiative; POP: Pelvic Organ Prolapse; FDA: Food and Drug Administration; PDS: Polydiaxanone Sutures; TEDS: Thrombo-Embolic Deterrent Stockings; IUGA/ICS: International Urogynaecological Association/International Continence Society
Introduction
Pelvic organ prolapse is a common gynaecological disorder with a 11 % life time risk of requiring at least one surgery to correct prolapse. Approximately, one third of these women face the risk of needing surgery for recurrent prolapse [1]. The Women’s Health Initiative (WHI) study demonstrated post hysterectomy prevalence of cystocoele of 32.9% and rectocele of 18.3% [2]. Similar rates were shown to be prevalent in women with uterus and rate of uterine prolapse was 14.2% [2]. There has been an ongoing clinical need for various materials to reinforce the native tissues to optimise the prolapse repair with particular emphasis to reduce the recurrence [3-30].
Commonly used terms to describe specific sites of female genital prolapse include
i. Anterior compartment prolapse–Hernia of anterior vaginal wall often associated with descent of the bladder (cystocele)
ii. Posterior compartment prolapse–Hernia of the posterior vaginal segment often associated with descent of the rectum (rectocele)
iii. Enterocele – Hernia of the intestines to or through the vaginal wall.
iv. Apical compartment prolapse (uterine prolapse, vaginal vault prolapse) – Descent of the apex of the vagina into the lower vagina, to the hymen, or beyond the vaginal introitus. The apex can be either the uterus and cervix, cervix alone, or vaginal vault, depending upon whether the woman has undergone hysterectomy. Apical prolapse is often associated with enterocele.
v. Uterine procidentia -Hernia of all three compartments through the vaginal introitus.
The terms anterior vaginal wall prolapse and posterior vaginal wall prolapse are preferred to cystocele and rectocele because vaginal topography does not reliably predict the location of the associated viscera in Pelvic organ prolapse (POP) [3,4].
Over the last decade, there has been an exponential increase in use of synthetic mesh for pelvic organ prolapse repair in all the three anatomic compartments. According to industry estimates from manufacturer data in United States, mesh was used approximately in one out of three pelvic organ prolapse (POP) surgeries, and three out of four of the mesh POP procedures were performed by trans-vaginal route [5,16,18,24-30]. Due to large number of complications secondary to mesh use in transvaginal repair like infection, pelvic pain and mesh erosion, The Food and drug administration (FDA) has re-classified the devices from Class II (moderate risk) to Class III (high risk) [6,13,14,17,20,25]. The FDA update in 2011 stated that mesh related complications were no longer considered rare [6]. It has given specific recommendations to clinicians for careful selection of patients for the use of mesh in prolapse repair as well as strong emphasis on counselling.
Various biological grafts have been used to avoid synthetic mesh related complications like infection, vaginal erosions or fistulas [7,8,10,11]. Xenform® Matrix is an acellular collagen based graft derived from foetal bovine dermis. A growing number of reconstructive surgeons advocate for biologic grafts [32] in which the host tissue repopulates the graft with blood vessels, fibroblast infiltration, and subsequent collagen deposition, creating a new tissue layer that lasts after the graft resorbs. There are three potential outcomes of a biologic graft after surgical implantation: absorption, encapsulation and remodeling. The first potential outcome is simple absorption of the material. This may occur if the host tissue does not recognize the graft as remodelable and simply forms scar tissue in response to insertion of the graft. This response is similar to what might happen if an absorbable synthetic mesh material, such as polyglycolic acid was implanted. A potential response to a biologic material, especially one that has been cross-linked with some chemical, such as glutaraldehyde, is encapsulation. The body will attempt to wall off the material by forming a fibrous capsule around the material. The most desirable outcome, however, is remodeling, whereby the host tissue sees the graft as a material in which it may re-populate with blood vessels, blood cells, collagen, and growth factors. The anticipated result is that after the biologic graft reabsorbs, the patient is left with a stronger layer of “fascia” beneath the vaginal epithelium [7,10,15,21,23]. Due to its origin from foetal dermis, it has higher type III collagen content than type I collagen. Type III collagen has less inflammatory cells with higher metalloproteinase to tissue derived inhibitor ratio that favours remodeling over collagen accumulation [8].
Non crossed linked intact porcine- derived acellular dermal matrix is designed to perform as surgical matrix for soft tissue repair while serving as a scaffold for the rapid ingrowth of host cells, collagen and blood vessels [9]. The length of remodelling is variable and based on patient characteristics. In a rat model, by 12 weeks, there was no inflammatory response and population by host fibroblasts was noted which is typical of wound healing [12]. The aim of this study is to determine the success and complication rates of Xenform® matrix in the treatment of pelvic organ prolapse over a three year period from 2011-2014 [10]
Materials and Methods
This is a retrospective study which included fifty seven (57) women who underwent pelvic organ prolapse repair with Xenform augmentation by single operator. The ethics committee at The Queen Elizabeth Hospital, Lyell McEwin Hospital and Modbury Hospital approved this retrospective audit. The ethics review reference number was Q20151101.
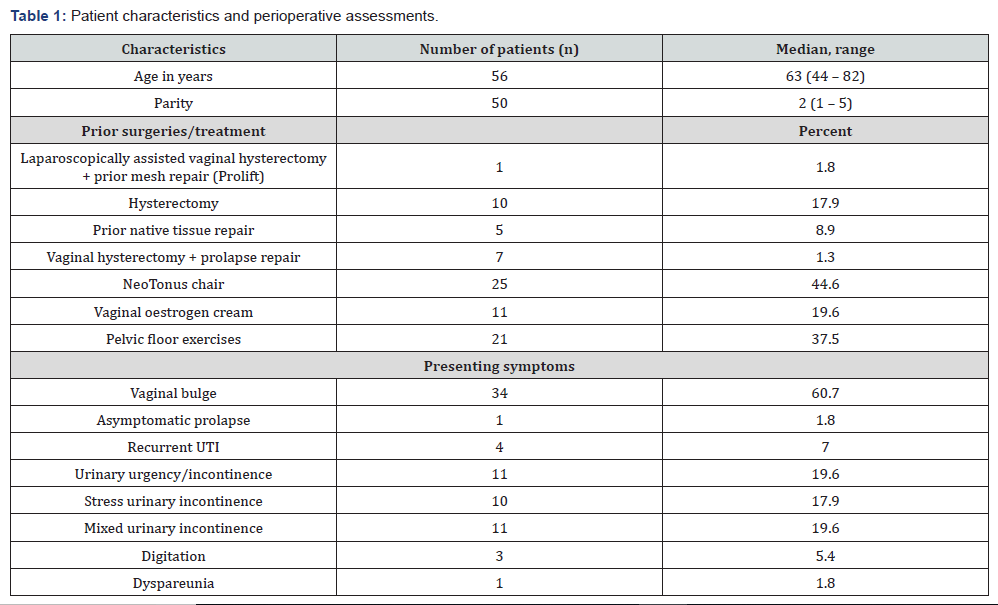
Women with Grade II or greater pelvic organ prolapse (as per POP Q classification) were treated with Xenform during primary surgery or during secondary surgery for recurrent prolapse. Xenform was placed either in anterior or posterior compartments as a single procedure, with traditional native tissue repair or for either compartment if clinically indicated. The POP Q scoring system was used to assess the degree of prolapse to maintain consistency in the pre- operative and post- operative findings. Demographic criteria included age, parity, previous conservative management such as Neo-Tonus chair (Extracorporeal Magnetic Innervation), vaginal oestrogen cream and pelvic floor exercises as well as previous native and mesh repair surgeries (Table 1).
Concomitant surgeries such as sub urethral sling procedures were performed according to the clinical indication and associated urinary stress incontinence after outpatient urodynamic studies.
Procedure
Informed consent was obtained after thorough consultation in the clinic. Intravenous antibiotics were administered as per hospital protocol. An indwelling catheter was inserted and local anaesthesia was infiltrated in the vaginal mucosa. A midline incision was made on the anterior vaginal wall. Endo pelvic fascia was separated with sharp dissection. Blunt dissection was also performed laterally to expose ischial spines. Three interrupted absorbable polydiaxanone sutures (PDS) were placed on each side in the sacrospinous ligaments with the help of Capio© suture capturing device (Boston Scientific). Xenform mesh was trimmed to 10 cm size to accommodate the inter spinous distance and 4 cm perpendicular to the horizontal dimension and then secured to sutures. Sutures were tightened to reduce prolapse and trimmed. The anterior vaginal wall was then closed with absorbable 2-0 Polydiaxone suture. Cystoscopy and rectal examination were performed to exclude any injuries to the bladder and rectum at the end of procedure. At the conclusion of surgery an estrogen soaked pack was inserted in the vagina. The indwelling catheter and vaginal pack was then left in for 24 hours post operatively and any non- steroidal anti- inflammatory drugs or low molecular weight heparin for thrombo-prophylaxis were avoided. Mechanical thrombo-prophylaxis in form of Thrombo-embolic deterrent stockings (TEDS) were used.
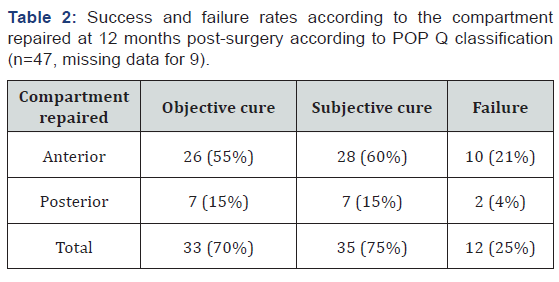
Patients were followed up at 6 weeks, 3 months, 6 months and 12 months after the procedure. At each post-operative visit, review of clinical symptoms and assessment of grade of prolapse as per the POP Q classification was undertaken. Objective success was defined as no or Grade 1 prolapse at 12 months of follow up and subjective success was defined as no prolapse symptoms or no bulge reported by patients at 12 months. Surgical failure was described in terms of subjective and objective assessments (Table 2). Graft failure was determined at 6 weeks with recurrence of prolapse.
The intra-operative complications were recorded as followingureteric injury, bladder injury, vaginal mucosal injury (button hole tear), urethral injury, bowel injury, vascular injury, neurological injury, perioperative blood transfusion and blood loss > 500ml. Peri-operative complications such as unplanned return to theatre for procedure related event within 72 hours, prolonged catheterization for more than 10 days post operatively and return to hospital within 30 days, with procedure related event were recorded.
Post-operative complications include de novo dyspareunia, recurrence of prolapse at the same site requiring conservative treatment, recurrence at same site requiring surgery, recurrent or new incontinence requiring surgery, vaginal narrowing secondary to mesh retraction. Complications were classified and documented according to the International Urogynaecological Association/ International Continence Society (IUGA/ICS) [17]. The case notes were reviewed for data collection. This retrospective audit was approved by Hospital Ethics Commit.
Results
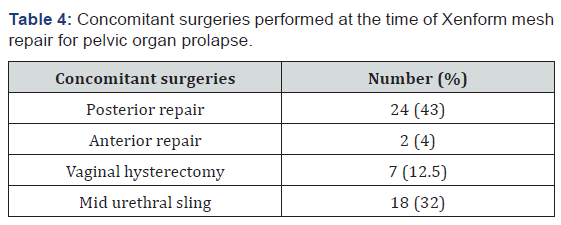
Of the 56 women included in the study,, 12 % had prior repair done. 18 % had had undergone prior hysterectomy and at least one compartment prolapse. 45 % of the patients had been treated with neo-tonus chair and one third had been commenced on pelvic floor exercises and hence, failed non-surgical management of prolapse (Table 1). Out of the 56 women included in study, approximately 12 month follow up data was available for 47 patients due to loss of 9 patients for follow up in clinic especially in public hospital. Anterior compartment prolapse was the most common condition requiring surgery and success rate was 75% at 12 months after surgery, with 25 % failure rate thus having a subjective cure rate of 75% (Table 2 &3, Figure 1). Other concomitant procedures done were anterior repair (4%), Posterior repair (43%), Vaginal hysterectomy (12.5%) and mid urethral sling (32%) (Table 4).


There were no major surgical complications in our group
In our series, there were no vaginal erosions, contractures and need to revise the mesh during the follow up. One case with secondary hemorrhage needed return to theatre and need for re-suturing of vaginal wall (Table-3). There was one case of symptomatic granulation tissue and vaginitis that required conservative management with oral anti-biotics. One patient had de novo dyspareunia that was reported at 6 months follow up and was not associated with any vaginal erosion and stenosis or recurrent prolapse (Table 3). Out of the 12 patients of recurrence group with same site prolapse, 3 required interventions with native tissue repair and one patient underwent second Xenform implantation in same compartment. Out of the recurrence group, 2 had different site prolapse (posterior compartment and uterine prolapse), and one patient underwent native tissue posterior repair.
Discussion
Our retrospective study was aimed to determine the success rates and complications associated with Xenform matrix use for reinforcement in trans-vaginal prolapse repair. Our study shows very promising results with the success rate of 75 % and no major complications or other unique problems associated with the use of synthetic mesh [17]. Thus, this audit demonstrates that Xenform matrix use in prolapse repair shows promising results.
There were fewer minor complications like urinary retention, vaginitis. The case of de novo dyspareunia wasn’t associated with vaginal erosion or contracture. Even though our study demonstrated failure of repair in 12 cases, only 4 cases of same site recurrence required second procedure.
Only other prospective cohort study by Goldstein et al reported 88% success rate based on mean Ba and Bp points at one year follow up. This group of 43 demonstrated no erosions and significant improvement in PDFI-20(pelvic floor distress inventory score) [8,22]. Xenform Soft tissue repair matrix is derived from fetal dermal bovine tissues [7-12] that, per World Health Organisation and European designation, has no known detectable prion infectivity. It is solely derived from cattle obtained in compliance with US and European regulatory requirements.
Xenform Matrix is intended for use as a soft tissue patch and is indicated for [17] repair of colon, rectal, urethral, and vaginal prolapse; reconstruction of the pelvic floor for procedures such as sacrocolpo suspension and urethral sling. This is a retrospective study of a small group of patients (N=56), with initial follow up for a period of 12 months. The study includes all patients operated by a single practitioner with consistent surgical procedure; the general application to other practitioners would involve learning curve and requires appropriate training. The lack of longerterm follow up could potentially miss late failures due to myxoid degeneration of the graft.
Nevertheless, our study is the largest retrospective study available regarding the Xenform use in pelvic organ prolapse repair as prior studies were in relation to use in dura-mater repair and urological procedures [30]. Hence, the emphasis should be on long term follow up with standardized questionnaires to assess quality of life and sexual function after prolapse repair.
References
- Olsen AL, Smith VJ, Bergstrom JO, Colling JC, Clark AL (1997) Epidemiology of surgically managed pelvic organ prolapse and urinary incontinence. Obstet Gynecol 89(4): 501-506.
- Hendrix SL, Clark A, Nygaard I, Aragaki A, Barnabei V, et al. (2002) Pelvic organ prolapse in the Women's Health Initiative: gravity and gravidity. Am J Obstet Gynecol 186(6): 1160-1166.
- Bump RC, Mattiasson A, BøK, Brubaker LP, DeLancey JO, et al. (1996) The standardization of terminology of female pelvic organ prolapse and pelvic floor dysfunction. Am J Obstet Gynecol 175(1): 10-17.
- Kenton K, Shott S, Brubaker L (1997) Vaginal topography does not correlate well with visceral position in women with pelvic organ prolapse. Int Urogynecol J Pelvic Floor Dysfunct 8(6): 336-339.
- Chaliha C, Khalid U, Campagna L, Digesu GA, Ajay B, et al. (2006) SIS graft for anterior vaginal wall prolapse repair-a case-controlled study. Int Urogynecol J 17: 492-497.
- FDA (2011) Centre for devices and radiological health (CDRH) Urogynecologic Surgical Mesh: Update on the Safety and Effectiveness of Transvaginal Placement for Pelvic Organ; FDA Safety Communication.
- Stuart Fox, MD Wheaton, Franciscan Health Care St. Mary’s Regional Hospital Racine (2009) Xenform® Soft Tissue Repair Matrix Posterior Compartment Repairs Results Wisconsin© Boston Scientific Corporation.
- Goldstein HB, Maccarone J, Naughton MJ, Aguirre OA, Patel RC (2010) A multicentre prospective trial evaluating foetal bovine dermal graft (Xenform® Matrix) for pelvic reconstructive surgery. BMC Urol 10: 21.
- Golla D, Russo CC (2014) Outcomes following placement of non-cross linked porcine- derived acellular dermal matrix in complex ventral hernia repair. Int Surg 99 (3): 235-240.
- Cornwell KG, Landsman A, James KS (2009) Extracellular Matrix Biomaterials for soft tissue repair. Clin Podiatry Med Surg 26(4): 507-523.
- Wilson CM, Williams BJ, Bilello S, Gomelsky A (2008) Bovine dermis: a novel biologic substitute for autologous tissue in sling surgery. Int Urogynecol J Pelvic Floor Dysfunct 19(12): 1671-1676.
- Adelman DM, Selber JC, Butler CE (2014) Bovine versus Porcine Acellular Dermal Matrix: A Comparison of Mechanical Properties. Plast Reconstr Surg Globe Open 2(5): e155.
- Jacquetin B, Cosson M (2009) Complications of vaginal mesh: our experience. Int Urogynecol J 20(8): 893-896.
- Peter L Dwyer (2006) Evolution of biological and synthetic grafts in reconstructive pelvic surgery. Int Urogynecol J17: S10-S15.
- Aguirre OA (2002) Foetal Bovine Dermal Graft (Xenform Matrix) in Pelvic Floor Reconstruction The Centre for Female Pelvic Medicine, Englewood, Colorado, USA.
- Vivian W Sung, Rebecca G Rogers, Joseph I Schaffer, Ethan M Balk, Katrin Uhlig, et al. (2008) For the Society of Gynecologic Surgeons Systematic Review Group, Graft Use in Transvaginal Pelvic Organ Prolapse Repair A Systematic Review 2008 by The American College of Obstetricians and Gynecologists.
- Bernard T Haylen, Robert M Freeman, Steven E Swift, Michel Cosson, G Willy Davila, et al. (2011) An International Urogynaecological Association (IUGA)/International Continence Society (ICS) joint terminology and classification of the complications related directly to the insertion of prostheses (meshes, implants, tapes) & grafts in female pelvic floor surgery, Int Urogynecol J 22: 3-15.
- Deprest J, Zheng F, Konstantinovic M, Spelzini F, Claerhout F, et al. (2006) The biology behind fascial defects and the use of implants in pelvic organ prolapse repair. Int Urogynecol J 17: S16-S25.
- Willy Davila, Robert D Moore (2006) Innovations in the Treatment of Vaginal Prolapse, Supplement to OBG Management.
- Marcus-Braun N, von Theobald P (2010) Mesh removal following transvaginal mesh placement: a case series of 104 operations. Int Urogynecol J 21: 423-430.
- Mouritsen L, Kronschnabl M, Lose G (2010) Long-term results of vaginal repairs with and without xenograft reinforcement. Int Urogynecol J 21(4): 467-473.
- Grimes CL, Tan-Kim J, Whitcomb EL, Lukacz ES, Menefee SA (2012) Long-term outcomes after native tissue vs. biological graft-augmented repair in the posterior compartment. Int Urogynecol J 23(5): 597-604.
- Nguyen JN, Jakus-Waldman SM, Walter AJ, White T, Menefee SA (2012) Perioperative Complications and Reoperations After Incontinence and Prolapse Surgeries Using Prosthetic Implants. Obstet Gynecol 119(3): 539-546.
- Wong KS, Nguyen JN, White T, Menefee SA, Walter AJ, et al. (2013) Adverse Events Associated With Pelvic Organ Prolapse Surgeries That Use Implants. Obstet Gynecol 122(6): 1239-1245.
- (2008) Surgical repair of vaginal wall prolapse using mesh Interventional procedure guidance.
- Sara Armitage, Elvis I Seman, Marc JNC Keirse (2012) Clinical Study Use of Surgisis for Treatment of Anterior and Posterior Vaginal Prolapse, Department of Obstetrics, Gynaecology and Reproductive Medicine, Flinders Medical Centre and Flinders University, Adelaide, SA 5042, Obstetrics and Gynecology International Volume.
- Peter Rosenblatt, Emily Von Bargen (2017) Use of biologic grafts in pelvic organ prolapse surgery, Contemporary OB/ GYN.
- Silva WA, Karram MM (2005) Use of biologic grafts in pelvic organ prolapse surgery, Scientific basis for use of grafts during vaginal reconstructive procedures. Curr Opin Obstet Gynecol 17: 519-529.
- Karlovsky ME, Thakre AA, Rastinehad A, Kushner L, Badlani GH (2005) Biomaterial for pelvic floor reconstruction. Urology 66(3): 469-475.
- Zerris V, James K, Roberts J, Bell E, Heilman CB (2007) Repair of the dura mater with processed collagen devices. J Biomed Mater Res B Appl Biomater 83(2): 580-588.






























