Nongenomic Effect of T3 on the Na, K-ATPase Activity of Syncytiotrophoblast from Human Term Placenta
Ángel R Mota1, Delia I Chiarello1,2, Cilia Abad1, Teresa Proverbio1, Sandy Piñero1, Reinaldo Marín1, and Fulgencio Proverbio1*
1Laboratorio de Bioenergética Celular, Instituto Venezolano de Investigaciones Científicas (IVIC), USA
2Cellular and Molecular Physiology Laboratory (CMPL), Division of Obstetrics and Gynaecology, School of Medicine, USA
Submission: June 17, 2017; Published: August 22, 2017
*Corresponding author: Fulgencio Proverbio, Laboratorio de Bioenergética Celular, Centro de Biofísica y Bioquímica (CBB), Instituto Venezolano de Investigaciones Científicas (IVIC), USA, Tel: (+582) 504.1395; E-mail: fproverb@yahoo.com;fproverb@ivic.gob.ve
How to cite this article: Ángel R M, Delia I C, Cilia A, Teresa P, et al. Nongenomic Effect of T3 on the Na, K-ATPase Activity of Syncytiotrophoblast from Human Term Placenta. Glob J Reprod Med. 2017; 2(2): 555583. DOI: 10.19080/GJORM.2017.02.555583
Abstract
Several ion transport enzymes are positively regulated by genomic mechanisms, through the thyroid hormones T3 and T4. However, there is evidence indicating that these hormones might also exert non genomic effects on different transporting enzymes. A normal activity of the Na, K-ATPase of syncytiotrophoblast is essential for ion homeostasis in this tissue, as well as for the growth and development of the fetus. In the current work it was studied the non genomic effect of thyroid hormones on the Na, K-ATPase activity of syncytiotrophoblast. The study was carried-out by incubating human term placental explants in the absence and presence of 10nMT3 or 350nMT4 and then isolating the syncytiotrophoblast plasma membranes and assaying them for Na, K-ATPase activity. The enzyme activity was also assayed for freshly prepared plasma membranes in the presence of (0-15)nMT3 or (0-500)nMT4.
It was found that 10nMT3 inhibits the activity of the enzyme of the microvillous membranes, without exerting any effect on the basal membranes. This inhibitory effect is due to a direct binding of T3 to the plasma membrane.T4, on the other hand, does not show any effect on the Na, K-ATPase activity of the syncytiotrophoblast plasma membranes. The differential effect of T3 on the Na, K-ATPase activity of the syncytiotrophoblast plasma membranes, inhibiting the enzyme activity on the maternal side of the membranes, but not on their fetal side, could result in an important disorder in the transepithelial Na+ transport between the mother and the fetus. These results might explain, at least in part, the low weight developed by fetuses born from mothers with hyperthyroidism during their pregnancy.
Keywords: Placenta; Syncytiotrophoblast; Thyroid hormones; Na, K-ATPase; Nongenomic effects
Abbreviation: MVM: Micro Villous Membrane; BM: Basal Membrane; SE: Standard Error
Introduction
Several ion transporting mechanisms, such as the plasma and sarcoplasmic membrane Ca-ATPase, the Na,K-ATPase, the Na/Hex changer and the Na/Pico transporter can be modulated by thyroid hormones, i.e.3,5,3'-triiodo-L-thyronine (T3) and 3,5,3',5'-tetraiodothyronine (T4) [1]. Although these transporters are positively modulated by thyroid hormones through genomic mechanisms [2] some of them have been also found to be affected by non genomic mechanisms [2-5], with short time courses and probably mediated by specific receptors of the plasma membranes. Na,K-ATPase is an important primary ion transport system that is essential to keep the ion homeostasis and cell volume as well as the resting membrane potential. This enzyme is widely distributed in practically all animal tissues and it is regulated by different hormonal and non-hormonal factors. Among the hormonal factors, the thyroid hormones regulate the functionality of the enzyme by both genomic and non genomic mechanisms [6].
The syncytiotrophoblast of the human placenta is a polarized cell layer with a maternal-facing microvillous membrane (MVM) and a fetal-facing basal membrane (BM). This cell layer interfaces the maternal and fetal circulations. The Na,K-ATPase of the syncytiotrophoblast has been shown to be present in both MVM and BM, playing a key role in intracellular ion homeostasis as well as constituting the force generator for the growth of the developing fetus [7]. In fact, the Na+ gradient between the cell and its medium, maintained by the enzyme, activates Na+ dependent amino acid transporters, driving toward the fetus amino acids and nutrients essentials for his normal growth. Besides, the Na+ gradient is essential for the intracellular pH control and it is involved in the net transport of Na+ to the fetus [7-9]. Considering the fact that the Na, K-ATPase activity plays a key role in the growth and development of the human fetus, it was evaluated the non genomic effect of the thyroid hormones on the activity of this enzyme in MVM and BM from human term placenta.
Materials and Methods
Placenta collection
Term placentas obtained from uncomplicated normal women were collected immediately after delivery, from the Maternity Hospital "Concepción Palacios” in Caracas, and transported to the laboratory on ice. This study was carried-out in accordance with the ethical standards established by the Declaration of Helsinki. The study protocol was approved by the Institutional Review Board of the Maternity "Concepción Palacios” and the Bioethics Committee of IVIC; all women gave signed informed consent.
Explant preparation and culture
After removal of the chorionic plate and about 0.25cm of deciduas, explants were prepared using only tissue from the intermediate region of the placenta and following the procedure described elsewhere [10].
Preparation of syncytiotrophoblast plasma membranes
The membranes (MVM and BM) were isolated from placental explants following a method previously described [11,12].
Na, K-ATPase activity assay
The ATPase activity was determined by measuring the quantity of inorganic phosphate liberated from the hydrolysis of ATP, according to the method described elsewhere [13]. The ATPase activity is expressed as nmol Pi/mg protein. min. The protein concentration, in all the cases, was determined according to the micro assay method of Bio Rad [14]. In order to avoid presence of membrane vesicles, the membrane fractions were pretreated before the assays with SDS, as previously described [15].
Statistical analysis
Statistical analysis was performed by the Student's t-test. All results are expressed as means±standard error (S.E.) and n represents the number of experiments performed with different preparations. ATPase activities were calculated from paired data. A p-value <0.05 was accepted as statistically significant.
Results
Figure 1 shows the effect of different concentrations of T3 and T4 in the incubation medium, on the Na,K-ATPase activity of human placenta syncytiotrophoblast microvillous (MVM, panel A) and basal (BM, panel B) plasma membranes. Notice how T3, at concentrations over 2.5nM inhibits in a concentration dependent way the activity of the enzyme of MVM, but it does not inhibit the ATPase activity of BM, even at a concentration of 15nM. On the other hand, T4 even at a concentration of 500nM does not produce any effect on the Na,K-ATPase activity of both membranes. Considering the fact that T3 and T4 are both present in the blood plasma, there is a possibility that T4 could interfere with the effect of T3 on the Na,K-ATPase activity of MVM. To test this possibility, we determined the enzyme activity in the presence of both 10nM T3 and 350nM T4. The results of this experiment are shown in Figure 2. It can be seen that T4 does not interfere with the inhibitory effect of T3 on the Na,K-ATPase activity of MVM.
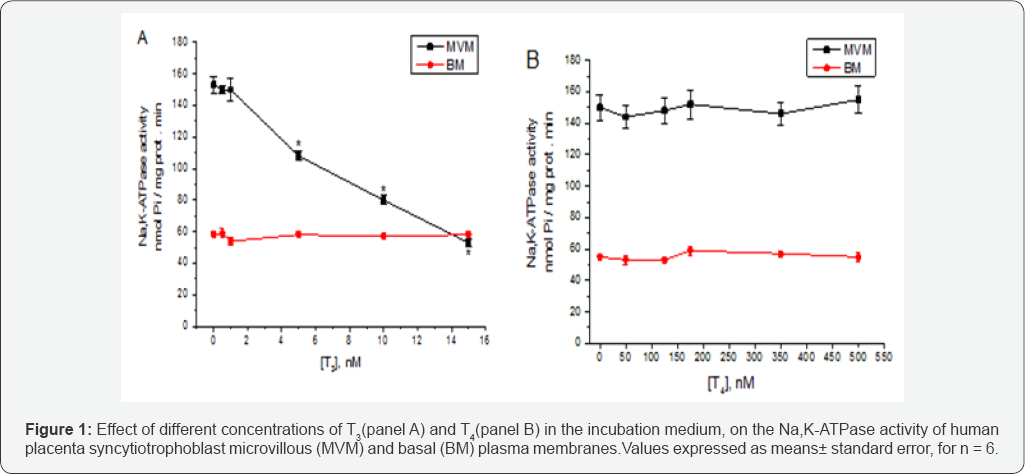
* p< 0.001 vs 0 T3.
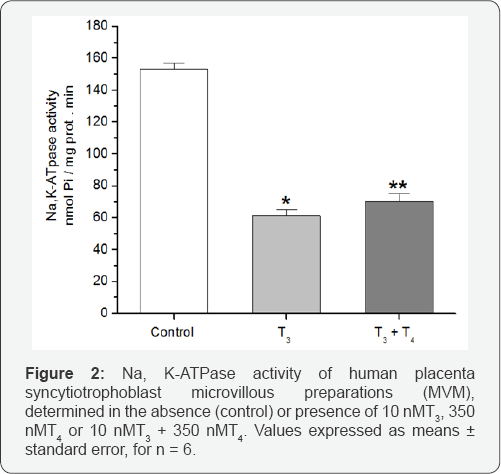
* p< 0.001 vs control
**p < 0.001 vs control and p =ns vs T3
The presented results indicate a nongenomic effect of T3 on the Na,K-ATPase activity of MVM. The possibility of T3 binding to receptors in MVM in order to exert its effect was tested, incubating placental explants for six hours at 37 °C under normoxia conditions (8% O2, 87% N2, 5% CO2), in the presence and absence of 10nMT3. The explants were then washed, homogenized and assayed for Na,K-ATPase activity. The results of this experiment are shown in Table 1. The Na,K-ATPase activity of explant homogenates preincubated in the presence of T3, shows an important inhibition, as compared to explants incubated under the same conditions but without T3. The Na,K- ATPase activity from MVM fractions isolated from the same homogenates does not show any inhibition (data not shown). This result may be taken as a clear indication that T3is wased away from the membernses during the MVM isolation procedure.

The tissue explants were incubated for 6 h at 37°C in4 mL medium (DMEM-F12 with 10% FCS) in a sterile CO2 incubator (Shel Lab Model IR2424) with a gas mixture composed of 8% O2, 87% N2, 5% CO2(normoxia), with constant gas pressure. Values expressed as means±standard error, for n = 6.
* p< 0.001 vs control.
To test this possibility, MVM fractions were separated in four groups:
I. of 10nMT3 (Condition A);
II. MVM fractions preincubated for 10min at 37 °C in the presence of 10nMT3, and then washed by centrifugation (Condition B);
III. MVM fractions preincubated for 10min at 37 °C in the presence of 10nMT3, washed by centrifugation and then preincubated Freshly prepared MVM (control),
IV. MVM fractions preincubated for 10min at 37 °C in the presence again with T3 under the same conditions as in Condition A.
The four groups were then assayed for Na,K-ATPase activity. The results of this experiment are shown in Figure 3.
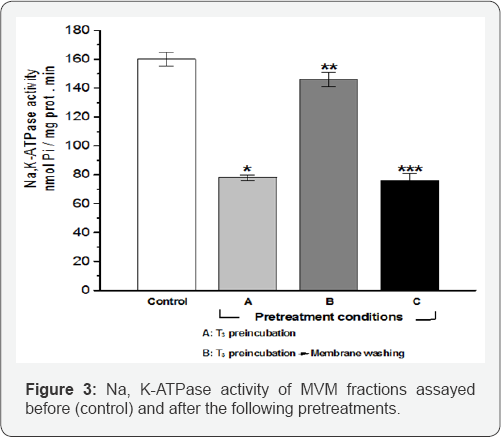
Condition A: Freshly prepared MVM fractions (2 mg protein/ml) were preincubated for 10 min at 37 °C in the presence of 10 nMT3. The membrane fraction was then diluted 10 fold (in order to dilute T3), and assayed for Na, K-ATPase activity.
Condition B: MVM fractions were preincubated with T3, washed by centrifugation at 100,000xg for 1 hour and then assayed for Na, K-ATPase activity.
Condition C: MVM fractions were preincubated with T3, washed by centrifugation, preincubated again with T3 and then assayed for Na, K-ATPase activity. Values expressed as means± standard error, for n = 6.
* p< 0.001 vs control
** p = ns vs control; p < 0.001 vs condition A
*** p< 0.001 vs control and vs condition B; p = ns vs condition A
Notice that:
o Preincubation of MVM with 10nMT3 leads to a significant reduction in the activity of their Na,K-ATPase activity;
o Centrifugation of the membranes preincubated with T3 returns the ATPase activity to its normal values and
o Second preincubation with 10nMT3 of the already preincubated and then centrifuged membranes, produces again a significant reduction of their ATPase activity.
Discussion
In general, the thyroid hormones effects have been explained in terms of the expression of specific genes, i.e. genomic effects, or direct effects through interactions at the plasma membrane, in cytoplasm or at intracellular organelles, such as mitochondria, i.e. non genomic effects. The genomic mechanism has been explained as a primary interaction of T3 with nuclear thyroid hormone receptor proteins. This primary interaction leads to the formation of intranuclear complexes formed by T3 and its nuclear receptor. These intranuclear complexes modulate transcription through binding to the promoter regions of thyroid hormone- responsive genes [16-18].
It has been suggested that the nongenomic effects of the thyroid hormones in intact cells may be initiated at the plasma membrane, in cytoplasm or at intracellular organelles. In the current study it was found an inhibitory effect of T3 on the Na,K-ATPase activity of microvillous membranes of syncytiotrophoblast from human term placenta (Figure 1). This inhibitory effect is due to a direct binding of T3 to the plasma membrane, as indicated by the fact that the Na,K-ATPase activity is diminished either in homogenates of placental explants preincubated with T3 (Table 1) or in MVM fractions preincubated with T3 (Figure 3). Even more, T3 bound to MVM can be washed away by centrifugation of the plasma membranes at 100,000x,g (Figure 3).
The Na, K-ATPase activity inhibition of the placental MVM produced by T3 could have important implications for the growing fetus. On this regard, different transport processes across the plasma membranes of eukaryotic cells are sodium coupled, and depend on the maintenance of a low intracellular Na+ concentration. This condition is mainly achieved by the Na, K-ATPase, exchanging intracellular Na+ ions for extracellular potassium K+ ions. Consequently, any alteration in the transport capacity and/or expression of the Na,K-ATPase, would lead to a dysfunction in the maintenance of the Na+ gradient across the cell plasma membrane, which will indirectly affect the driving forces of other membrane transport systems.
The differential effect of T3 on the Na,K-ATPase activity of the syncytiotrophoblast plasma membranes, inhibiting the enzyme activity on the maternal side of the membranes (MVM), but not on their fetal side (BM), could result in an important disorder in the transepithelial Na+ transport between the mother and the fetus. Consequently, pregnant mothers with hyperthyroidism might be facing this specific disorder. Even more, the resulting intracellular Na+ accumulation, which is accompanied by water, will affect some of the normal cell parameters and mechanisms, such as cell volume, intracellular pH and Ca2+ concentration, glucose and amino acid transepithelial transport from the maternal blood to the fetal blood [4,19]. The presented results may be taken as an indication that inhibition of the Na,K- ATPase of syncytiotrophoblast plasma membranes by T3could be contributing to the low weight gain of fetuses of pregnant women with hyperthyroidism.
Acknowledgment
The current study is part of the M.Sc. Thesis dissertation of ARM. The authors wish to thank the department of "Sala de Partos”, Maternity Hospital "Concepción Palacios” for its collaboration during this study. This research was supported in part by grant No. H9/181/R427, Project 96350, from the World Health Organization and by grant from FONACIT - Venezuela No. F-2005000222.
Declaration of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this paper.
References
- Davis PJ, Goglia F, Leonard JL (2016) Nongenomic actions of thyroid hormone. Nat Rev Endocrinol 12(2): 111-121.
- Incerpi S, De Vito P, Luly P, Spagnuolo S, Leoni S (2002) Short-term effects of thyroid hormones and 3,5-diiodothyronine on membrane transport systems in chick embryo hepatocytes. Endocrinology 143(5): 1660-1668.
- Davis PJ, Davis FB (1996) Nongenomic actions of thyroid hormone. Thyroid 6(5): 497-504.
- Segal J (1989) A rapid, extranuclear effect of 3,5,3'-triiodothyronine on sugar uptake by several tissues in the rat in-vivo. Evidence for a physiological role for the thyroid hormone action at the level of the plasma membrane. Endocrinology 124(6): 2755-2764.
- Silva FG, Giannocco G, Santos MF, Nunes MT (2006) Thyroid hormone induction of actin polymerization in somatotrophs of hypothyroid rats: potential repercussions in growth hormone synthesis and secretion. Endocrinology 147(12): 5777-5785.
- Lin HY, Tang HY, Davis FB, Mousa SA, Incerpi S, et al. (2012) Nongenomic regulation by thyroid hormone of plasma membrane ion and small molecule pumps. Discov Med 14(76): 199-206.
- Johansson M, Jansson T, Powell TL (2000) Na+-K+-ATPase is distributed to microvillous and basal membrane of the syncytiotrophoblast in human placenta. Am J Physiol 279(1): R287-294.
- Johansson M, Karlsson L, Wennergren M, Jansson T, Powell TL (2003) Activity and protein expression of Na+/K+ ATPase are reduced in microvillous syncytiotrophoblast plasma membranes isolated from pregnancies complicated by intrauterine growth restriction. J Clin Endocrinol Metab 88(6): 2831-2837.
- Whitsett JA, Wallick ET (1980) [3H]ouabain binding and Na+-K+-ATPase activity in human placenta. Am J Physiol 238: E38-45.
- Chiarello DI, Marín R, Proverbio F, Benzo Z, Pinero S, et al. (2017) Effect of hypoxia on the calcium and magnesium content, lipid peroxidation level, and Ca2+-ATPase activity of syncytiotrophoblast plasma membranes from placental explants. Biomed Res Int 2014: 597357.
- Illsley NP, Wang ZQ, Gray A, Sellers MC, Jacobs MM (1990) Simultaneous preparation of paired, syncytial, microvillous and basal membranes from human placenta. Biochim Biophys Acta 1029(2): 218-226.
- Jimenez V, Henriquez M, Llanos P, Riquelme G (2004) Isolation and purification of human placental plasma membranes from normal and pre-eclamptic pregnancies. a comparative study. Placenta 25(5): 422437.
- Marin R, Proverbio T, Proverbio F (1985) Characterization of the Na+, K+-ATPase activity of basolateral plasma membranes of kidney proximal tubular cells from young and old rats. Biochem Pharmacol 34: 4197-4201.
- Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein- dye binding. Analyt Biochem 72: 248-254.
- Proverbio F, Proverbio T, Marin R (1986) Na+-ATPase is a different entity from the Na++K+-ATPase in rat kidney basolateral plasma membranes. Biochim Biophys Acta 858: 202-205.
- Cheng SY, Leonard JL, Davis PJ (2010) Molecular aspects of thyroid hormone actions. Endocr Rev 31(2): 139-170.
- Yen PM (2001) Physiological and molecular basis of thyroid hormone action. Physiol Rev 81(3): 1097-1042.
- Zhang J, Lazar MA (2000) The mechanism of action of thyroid hormones. Annu Rev Physiol 62: 439-466.
- Sibley CP, Glazier JD, Greenwood SL, Lacey H, Mynett K, et al. (2002) Regulation of placental transfer: the Na+/H+ exchanger--a review. Placenta 23 Suppl A: S39-46.






























