Rectal Indomethacin is an Effective Analgesic During Copper T380A Intrauterine Device Insertion: A Randomized Double-Blind Placebo-Controlled Trial
Ahmed M. Abbas1*, Abdelrahman M. Abdelkader1, Ahmed H. Elsayed2, Shaimaa Adel 3 >and Mohamed S. Fahmy 2
1Department of Obstetrics & Gynecology, Faculty of Medicine. Assiut University, Assiut, Egypt
2Department of Obstetrics & Gynecology, Faculty of Medicine. Aswan University, Aswan, Egypt
3Department of Obstetrics & Gynecology, Sohag General Hospital, Sohag, Egypt
Submission: May 09, 2017; Published: July 27, 2017
*Corresponding author: Ahmed M Abbas, Department of Obstetrics and Gynecology, Faculty of Medicine, Assiut University, Egypt, Tel: +20-88-2414616; Fax: +20-88-9202503; Email: bmr90@hotmail.com
How to cite this article: Ahmed M. A, Abdelrahman M. A, Ahmed H E, Shaimaa A, Mohamed S. F. Rectal Indomethacin is an Effective Analgesic During Copper T380A Intrauterine Device Insertion: A Randomized Double-Blind Placebo-Controlled Trial. Glob J Reprod Med. 2017; 2(1): 555576. DOI: 10.19080/GJORM.2017.02.555576
Abstract
Objectives: To investigate the analgesic effect of rectal indomethacin during copper T380A intrauterine device (IUD) insertion.
Materials and methods: A randomized, double-blind, placebo-controlled trial, was conducted in Aswan University Hospital, Egypt between March and July 2016. Parous women eligible for Copper IUD insertion were recruited and randomized in a 1:1 ratio to indomethacin or placebo. The participants were asked to self administer the rectal suppositories 30 minutes before IUD insertion. The primary outcome was the participant's self-rated pain perception using a 10cm Visual Analogue Scale (VAS) during IUD insertion. We considered a 1.5cm difference in VAS scores between study groups as clinically significant.
Results: Ninety six women were enrolled (n=48 in each group). Indomethacin significantly reduce the median VAS pain scores during speculum placement (1 vs. 2.5) tenaculum placement (2 vs. 4), sound insertion (2 vs. 4.25), IUD insertion (2.25 vs. 5) and 5 minutes postinsertion (1 vs. 2) with p<0.001. A higher satisfaction score was also present in indomethacin group (7.18±2.45 vs. 4.95±2.7, p<0.001).
Conclusion: The use of rectal indomethacin 30 minutes prior to copper T380A IUD insertion significantly reduces the insertion pain with no adverse effects.
Keywords: Intrauterine device; Indomethacin; Contraception; Pain relief
Introduction
Intrauterine devices (IUDs) are highly effective contraceptive methods that are associated with lower discontinuation rates if compared to other methods [1]. Worldwide, IUD is the most popular reversible method of contraception being used by more than 180 million women [2]. They are convenient to use for most of women since they do not require frequent user or clinician actions to maintain contraceptive protection [3].
Their convenience and high efficacy make them an excellent option that women should consider to reduce the unplanned pregnancy rate worldwide [4]. However, fear of pain is a major concern and disincentive to use the IUD [5,6] besides the health care provider worry of difficult insertion, especially in nullipara [7] . This limits its use to approximately 8% of reproductive-aged women in developed countries and 15% in developing countries [8] .
Pain perception during IUD insertion procedure is related to application of the cervical tenaculum to grasp the cervix or transcervical actions as inserting the uterine sound, inserting the IUD in the inserter tube through the cervix, and irritation of the endometrial cavity with the device itself [9]. So, it is important to identify a safe, effective and easy to apply analgesic method during IUD insertion that can reduce women's pain and increase IUD uptake.
Numerous systematic reviews have evaluated different medications through various routes for pain relief with IUD insertion [10,11]. Pre-insertion oral, intravenous, intramuscular, intrauterine and topical analgesics have been included; however, interpretation of the current data is limited and there is no consensus regarding administration of the optimal analgesic, timing or route. A recent Cochrane systematic review (2015) included seven trials on the efficacy of non-steroidal antiinflammatory drugs (NSAIDs) in reducing pain during IUD insertion [10]. The efficacy of oral naproxen, ibuprofen and intramuscular ketorolac were tested with variable degrees of success [10]. The effect of rectal NSAIDs for pain relief during IUD insertion has not been evaluated.
Indomethacin is a NSAID with potent anti-inflammatory, analgesic action. Previous studies attempted to use rectal indomethacin for pain relief during office gynecological procedures as endometrial biopsy and hysterosalpingography (HSG) reported contradictory results regarding its analgesic effect [12,13].
To the best of our knowledge, no study has examined the efficacy of rectal indomethacin on reducing IUD insertional pain. So, the current study aims to evaluate the analgesic effect of rectal indomethacin in pain relief during IUD insertion.
Materials and Methods
We performed a randomized, double-blind, placebo- controlled trial (NCT02 711358), in Aswan University Hospital, Egypt between March and July 2016. The Institutional Ethical Review Board approved the study, and we obtained a written informed consent from all participants before enrollment. Women 18 years of age or older who presented to the Family Planning Clinic requesting a copper IUD insertion were enrolled in the study. We excluded women received any analgesics or misoprostol within 24 hours prior to insertion. Those with a Category 3 or 4 conditions for IUD insertion according to the WHO Medical Eligibility Criteria for contraceptive use [14] were also excluded. Moreover, we excluded women declined participation in the study.
A statistician, not otherwise involved in the study, prepared a computer generated random table and placed the allocation data in sequentially numbered opaque sealed envelopes. Each envelope had a card noting the group identifier inside. A single pharmacist was responsible for the manufacturing of the placebo cream and packaging of both preparations into sterile boxes with labeling them as A and B. Only the Pharmacist knew what the medication in box A and B was, so neither the clinician nor the women knew the type of the preparation.
Once enrolled, the clinic nurse opened the envelopes according to the sequence of attendance of women. We randomized participants in a 1:1 ratio into one of two groups:
A. Indomethacin group: Received a single rectal dose of 50mg indomethacin suppository (KAHIRA PHARMA & CHEM IND, CO. CAIRO-EGYPT).
B. Placebo group: Received a single rectal placebo suppository similar in consistency, appearance, and color to indomethacin. All women were instructed to insert the suppository 30 minutes before the IUD insertion. Before insertion, one of the study researchers collected the baseline data. Then, he explained the standard 10cm visual analog scale (VAS) to the participants for pain scoring [15]. The severity of pain was assessed with VAS (with 0= no pain and 10= worst imaginable pain). Each woman received a copper T380A IUD (Pregna®T380A; Pregna International Ltd USA, Mumbai, India) for insertion. During IUD insertion, all women were menstruating. The day of the menstrual cycle ranged from the second to the fifth.
The IUD was inserted by an experienced study researcher, using the standard manufacturer approved technique. A research assistant blinded to the group assignment asked the women to rate the intensity of pain at five consecutive steps; at speculum placement, at tenaculum placement, at sound insertion, at IUD insertion and 5 minutes after the end of insertion using the same 10-point VAS with a different sheet of paper at every point.
After the end of procedure, the clinician assessed the ease of IUD insertion using the ease of insertion score (ES). The ES is a graduated VAS-like scale from zero to 10; in which 10 means terribly difficult insertion and zero means very easy insertion. This score was validated for use in previous similar studies about IUD insertion [16,17]. Satisfaction scores was also evaluated using a 10cm VAS at 15 minutes post insertion with 0 being not at all satisfied and 10 being highly satisfied.
All women were asked about the need for any additional analgesics at 15 minutes after completing the procedure. Ibuprofen 400mg was offered as an additional analgesic if needed. Also, they were asked to report any side effects occurred during and within 15 minutes after insertion.
The primary outcome was the difference in pain VAS scores during IUD insertion. The secondary outcomes included the difference in pain scores during speculum, tenaculum placement, sound insertion and 5 minutes post-insertion, the difference in ease of insertion score, the difference in women's satisfaction score, and the side effects of the used medication.
The required sample size was calculated based on previous studies assessing pain control with IUD insertion using a 10cm (100mm) VAS. In the study of Bednarek et al, the mean VAS pain score with IUD insertion in the control group was 4.3cm [18]. A clinically significant difference in VAS pain score has been defined as 1.3-2.0cm [19,20]. Using a standard deviation of 2.6 cm to obtain 80% power with a error of 0.05, a sample size of 48 women was needed in each group to detect 1.5cm difference in the VAS pain score with the use of indomethacin (Open Epi, Version 3, open source calculator-SS Mean).
All data were analyzed using SPSS software Chicago, IL, USA, version 21. Comparison between categorical variables in both groups was done by Chi-square test and continuous variables were compared using Student t-test. A Shapiro-Wilkes test was performed to test normality of the different pain scores. Data showing skewed distribution, so they were presented as median (interquartile range), and Wilcoxon rank-sum test was used for comparing intergroup differences. Univariate and multivariate regression analysis were performed with the dependent variable (VAS score during IUD Insertion) to determine the independent predictors of IUD insertion pain. We considered P value < 0.05 as a significant value.
Results
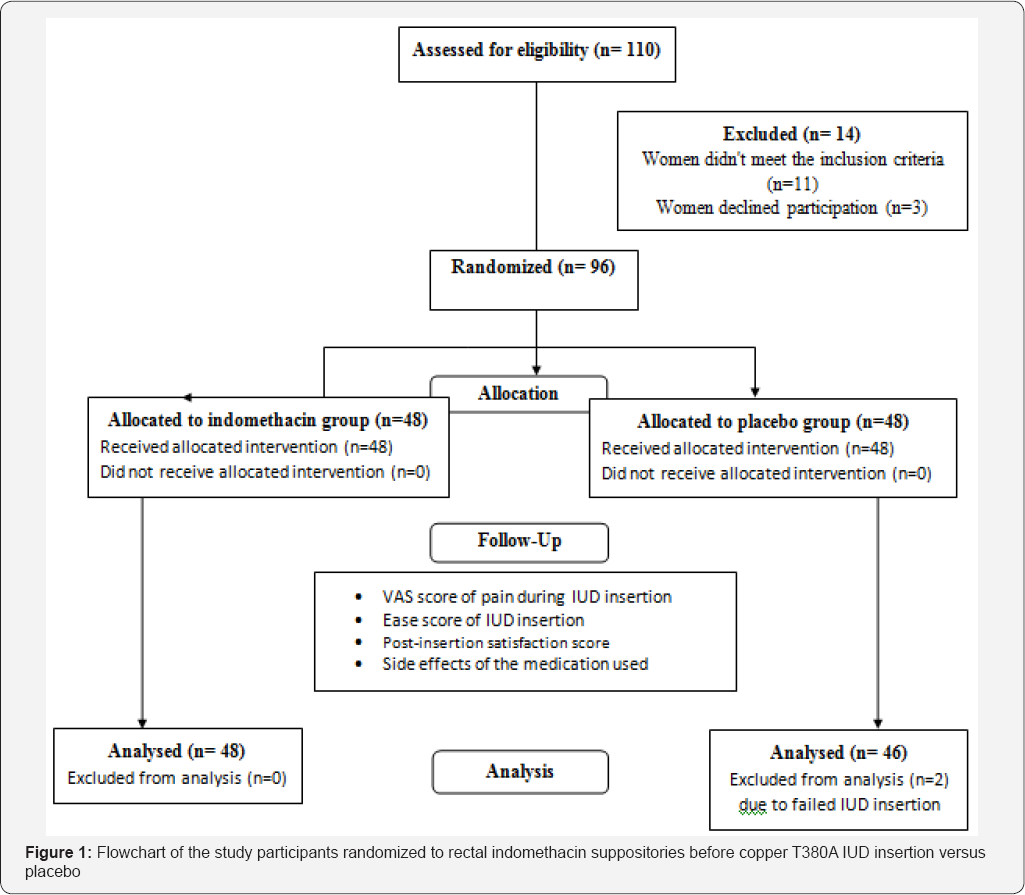
One-hundred ten women were approached to participate in the study. Fourteen women have been excluded: four women had uterine abnormalities; four women had received analgesics prior to insertion and three women had undiagnosed vaginal bleeding at the time of insertion. Moreover, three women declined participation in the study. We randomly assigned the remaining 96 women into both groups. Two cases were excluded from the final analysis in the placebo group as IUD insertion failed due to severe cervical stenosis (Figure 1, the study flowchart).
Table 1 show that both groups were comparable in the baseline characteristics. Table 2 shows the median pain scores for both groups. Women in indomethacin group were more likely to report lower pain scores during IUD insertion (2.25 (2-3) vs. 5 (3-7), p<0.001). The same observed with pain scores during speculum placement, tenaculum placement and sound insertion (p<0.001). There was also a decrease in pain scores at 5 minutes after IUD insertion (1 (1-1.5) vs. 2 (1-2), p<0.001).
There was a statistically significant difference in satisfaction score between the study groups. The mean satisfaction scores were 7.18±2.45 and 4.95±2.7 in the indomethacin and placebo groups respectively (p<0.001). A lower ease of insertion score was observed among indomethacin group (3.13±2.2 vs. 3.26±1.77) but the difference did not reach statistical significant (p=0.742). The duration of IUD insertion was significantly lower in the indomethacin group (4.9±1.28 vs. 5.78±1.6 minutes, p=0.004). Ten women asked for additional analgesics in the placebo group versus four in the indomethacin group (p=0.086). No cases of uterine perforation or vasovagal reactions were observed in both groups. Also, no women reported side effects from indomethacin use (Table 2).
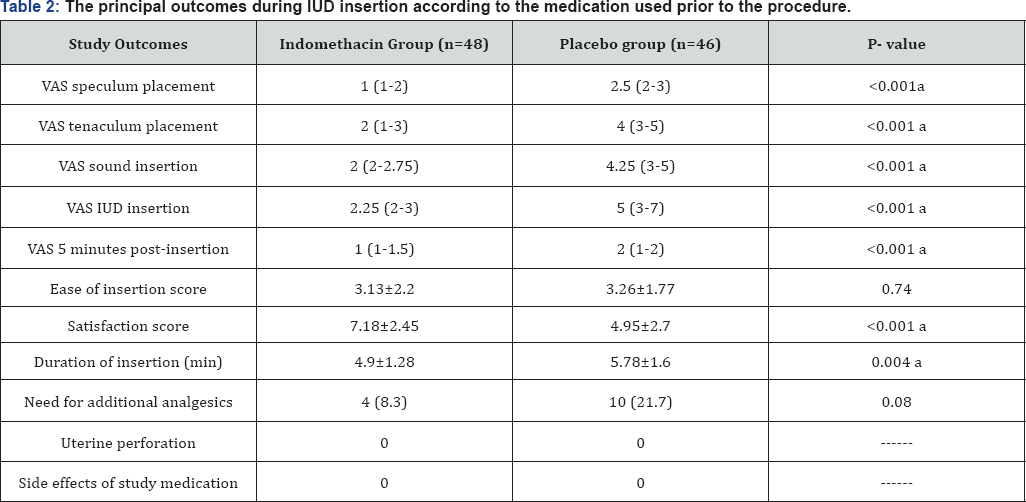
VAS: Visual Analog Scale; IUD; Intrauterine Device, SD: Standard Deviation; IQR; Inter-Quartile Range, (a) statistically significant difference.
All data are presented as n (%), median [inter-quartile range] or mean ± standard deviation.
Both history of dysmenorrhea and prior IUD insertion were associated independently with IUD insertional pain in univariate analysis (p=0.016, 0.041 respectively). Higher pain scores at IUD insertion were more likely in presence of history of dysmenorrhea (odds ratio of 0.25), while prior IUD insertion was independent predictor for decreased pain with IUD insertion (odds ratio of 0.26). In multivariate analysis, only history of dysmenorrhea remained a strong predictor of pain (p=0.012). Other variables tested include age (p=0.38), parity (p=0.19), prior cesarean deliveries (p=0.79), prior vaginal deliveries (p=0.20), prior office gynecologic procedures (p=0.79), and uterine position (p=0.86) were not predictors for pain experienced during IUD insertion.
Discussion
In the present study, we found that a single dose of rectal indomethacin 30 minutes prior to copper IUD insertion effectively reduces pain at all steps of insertion with no adverse effects. This is the first study to evaluate the efficacy of rectal indomethacin as an analgesic during IUD insertion. Pain in office gynecological procedures has always been a dilemma in daily practice. Fear of pain during IUD insertion can be a barrier to its use. Thus, better pain control during insertion could lead to more widespread IUD use and decrease the rate of unplanned pregnancies as it is safe, highly effective, long-term and reversible contraceptive method.
There is no consensus in the literature as regard to the optimal method of pain relief for IUD insertion. In a Cochrane review by Lopez et al. [10] that examined 7 randomized control trials included both oral and intramuscular NSAIDs [10] it was reported that six studies showed no difference in pain relief between women given NSAIDs or placebo [9,16,21-24] while only one study showed a beneficial effect of oral naproxen sodium [25]. More recently, Fouda et al. [26] reported a beneficial effect of oral diclofenac combined with topical lidocaine in reducing pain with IUD insertion [26].
Indomethacin is a potent NSAID which act as a prostaglandin synthetase inhibitor leading to reduce prostaglandin release. The locally increased secreted prostaglandins induce uterine cramps and pain during office gynecological procedures. Indomethacin was evaluated before for pain relief in office gynecological procedures, such as endometrial sampling [12] and HSG [13] as well as in non-gynecological conditions like after repair of episiotomy and perineal tears [27].
The main advantage of the rectal route is the avoidance of the first-pass effect on the metabolism of orally administered drugs from the enterohepatic circulation. This may affect the optimum dosage of the drug creating the need to use higher doses to obtain similar analgesic effect. Also, it avoids the significant adverse effects of oral NSAIDs as epigastric pain, gastritis or peptic ulcer. In our study, no reported local or systemic adverse effects in women received rectal indomethacin. This coincides with the study of Karaman et al. [13] in which 42 women received rectal indomethacin before HSG with no reported adverse effects.
The current study shows a significant difference in the pain intensity scores with all steps of IUD insertion in the indomethacin group being lower than placebo group (p<0.001). Moreover, fewer women in the indomethacin group requested additional analgesics than placebo group, however that difference did not reach statistical significance (4 vs 10, p=0.086).
Our results are not consistent with the literature that used oral or intramuscular NSAIDs for pain relief during IUD insertion. However; Karabayirli et al. [25] found that naproxen sodium 550mg can be recommended for pain relief during IUD insertion. Women who received naproxen had a lower mean pain scores during insertion compared with the placebo group (Mean difference; -1.94, 95%CI -2.35 to -1.53). Inserting the IUD tube was the most painful step and those who received indomethacin had less pain as compared with placebo group (median VAS; 2.25 (2-3) vs 5(3.7), respectively, p=0.0001).
A literature search revealed two studies evaluating the analgesic effect of rectal indomethacin during other gynecological procedures with contradictory results [12,13]. In agreement with our results, Karaman et al. showed that rectal indomethacin significantly decreases perceived pain during HSG in all steps apart from speculum placement [13]. On the contrary, Telly et al. [12] showed no reduction in pain scores with rectal indomethacin compared to placebo (median VAS; 4(2-5.1) vs 4.8(2-6.3), respectively, p=0.502) during Pipelle endometrial biopsies [12]. This discrepancy may be attributed to the difference in age between the study participants (mean age <30 years in Karaman et al and our study vs >45 years in Telly et al study) that is definitely affect the pain perception. Also, the difference in the procedure itself, as almost no application of cervical tenaculum in endometrial biopsy, beside the smaller diameter of the pipelle device compared with IUD insertion tube and HSG cannula. This can lead to less pain perception during the procedure.
The main strength of our study is the randomized, placebo- controlled design. We were able to recruit our calculated sample size for achieving adequate power to detect a clinically significant difference in our primary outcome. Additionally, the study was conducted at the same clinic, with a single provider to avoid any inter-assessor variation in VAS scores estimation. Also, we used validated pain scores and assessed women satisfaction, which was not assessed in many other trials. Finally, we evaluated the educational status of women, as this could affect the ability to identify perceived pain on VAS, so our results could be generalizable.
The main limitation of our study is the subjectivity in reporting pain through VAS score, even though it is the widely accepted method and there are currently no other objective parameters to evaluate pain. Also, the medication was selfadministered, so pain relief could be affected by improper drug insertion. In conclusion, rectal indomethacin 30 minutes prior to copper IUD insertion seems to effectively alleviate pain during insertion. It is easily administered and relatively inexpensive with no side effects. Thus, we recommend its use before IUD insertion for pain relief.
Conflict of Interest
The authors declare that they have no conflict of interest.
References
- Peipert JF, Zhao Q, Allsworth JE, Petrosky E, Madden T, et al. (2011) Continuation and satisfaction of reversible contraception. Obstet Gynecol 117: 1105-1113.
- Darney P, Speroff L (2010) A clinical guide for contraception. Lippincott Williams & Wilkins, Philadelphia, USA, pp. 242-243.
- ACOG Committee Opinion no. 539 (2012) Adolescents and long acting reversible contraception: implants and intrauterine devices. Obstet Gynecol 120: 983-988.
- Winner B, Peipert JF, Zhao Q, Buckel C, Madden T, et al. (2012) Effectiveness of long-acting reversible contraception. N Engl J Med 366: 1998-2007.
- Potter J, Rubin SE, Sherman P (2014) Fear of intrauterine contraception among adolescents in New York City. Contraception 89: 446-450.
- Hubacher D, Spector H, Monteith C, Chen PL, Hart C (2015) Rationale and enrollment results for a partially randomized patient preference trial to compare continuation rates of short-acting and long-acting reversible contraception. Contraception 91(3): 185-192.
- Buhling KJ, Hauck B, Dermout S, Ardaens K, Marions L (2014) Understanding the barriers and myths limiting the use of intrauterine contraception in nulliparous women: results of a survey of European/ Canadian healthcare providers. Eur J Obstet Gynecol Reprod Biol 183: 146-154.
- d'Arcangues C (2007) Worldwide use of intrauterine devices for contraception. Contraception 75: 2-7.
- Hubacher D, Reyes V, Lillo S, Zepeda A, Chen PL, et al. (2006) Pain from copper intrauterine device insertion: randomized trial of prophylactic ibuprofen. Am J Obstet Gynecol 195(5): 1272-1277.
- Lopez LM, Bernholc A, Zeng Y, Allen RH, Bartz D, et al. (2015) Interventions for pain with intrauterine device insertion. Cochrane Database Syst Rev 29(7): CD007373.
- Zapata LB, Jatlaoui T, Marchbanks PA, Curtis KM (2016) Medications to ease intrauterine device insertion: a systematic review. Contraception 94(6): 739-759.
- Telli E, Aydin Y, Oge T, Yalcin OT.. (2014) Vaginal misoprostol versus a rectal non steroidal anti-inflammatory drug to reduce pain during Pipelle endometrial biopsies: A prospective, randomized, placebo- controlled trial. Gynecol Obstet Invest 78(4): 230-234.
- Karaman E, pm N, Alkijt, Yalcin OT (2016) Rectal indomethacin use in pain relief during hysterosalpingography: A randomized placebo controlled trial. J Obstet Gynecol Res 42(2): 195-201.
- World Health Organization (2010) Medical Eligibility Criteria for Contraceptive Use. WHO, Geneva, Switzerland.
- Bouhassira D, Attal N, Alchaar H, Boureau F, Brochet B, et al. (2005) Comparison of pain syndromes associated with nervous or somatic lesions and development of a new neuropathic pain diagnostic questionnaire (DN4). Pain 114 (1-2): 29-36.
- Abdellah MS, Abbas AM, Hegazy AM, El-Nashar IM (2017) Vaginal misoprostol prior to intrauterine device insertion in women delivered only by elective cesarean section: a randomized double-blind clinical trial. Contraception 95(6): 538-543.
- Abbas AM, Abdellah MS, Khalaf M, Bahloul M, Abdellah NH, et al. (2017) Effect of cervical lidocaine-prilocaine cream on pain perception during copper T380A intrauterine device insertion among parous women: A randomized double-blind controlled trial. Contraception 95(3): 251256.
- Bednarek PH, Creinin MD, Reeves MF, Cwiak C, Espey E, et al. (2015) Prophylactic ibuprofen does not improve pain with IUD insertion: a randomized trial. Contraception 91(3): 193-197.
- Todd KH, Funk KG, Funk JP, Bonacci R (1996) Clinical significance of reported changes in pain severity. Ann Emerg Med 27(4): 485-489.
- Rowbotham MC (2001) What is a 'clinically meaningful' reduction in pain? Pain 94(2): 131-132.
- Massey SE, Varady JC, Henzl MR (1974) Pain relief with naproxen following insertion of an intrauterine device. J Reprod Med 13(6): 226231.
- Jensen HH, Blaabjerg J, Lyndrup J (1998) Prophylactic use of prostaglandin synthesis inhibitors in connection with IUD insertion. Ugeskrift for Laeger160 (48): 6958-6961.
- Chor J, Bregand-White J, Golobof A, Harwood B, Cowett A (2012) Ibuprofen prophylaxis for levonorgestrel-releasing intrauterine system insertion: a randomized controlled trial. Contraception 85(6): 558-562.
- Ngo LL, Ward KK, Mody SK (2012) Ketorolac for pain control with intrauterine device placement. Obstet Gynecol 126: 29-36.
- Karabayirli S, Ayrim AA, Muslu B (2012) Comparison of the analgesic effects of oral tramadol and naproxen sodium on pain relief during IUD insertion. J Minim Invasive Gynecol 19(5): 581-584.
- Fouda UM, Salah Eldin NM, Elsetohy KA, Tolba HA, Shaban MM, et al. (2016) Diclofenac plus lidocaine gel for pain relief during intrauterine device insertion. A randomized double blinded placebo-controlled study. Contraception 93(6): 513-518.
- Yildizhan R, Yildizhan B, Sahin S, Suer N (2009) Comparison of the efficacy of diclofenac and indomethacin suppositories in treating perineal pain after episiotomy or laceration: A prospective, randomized, double-blind clinical trial. Arch Gynecol Obstet 280(5): 735-738.






























