Noxeno-Research & Development of an Affordable Nasal Foreign Body Removal Device in India
Ravi Jangir1, Jagdish Chaturvedi2*, Pooja Kadambi3, Vishal Prasad4, Abu Saquib Tauheed1, Rohan Dsouza1, Darien Rodrigues1, Rishabh Sirdesai1 and Parul Chachra1
1Affordable Invention in MedTech (AIM) Fellow, InnAccel Technologies Pvt. Ltd, India
2Department of Otorhinolaryngology, Fortis Hospital, India
3Lead System Designer, InnAccel Technologies Pvt Ltd, India
4Design and Validation Engineer, InnAccel Technology, India
Submission: March 21, 2018; Published: April 06, 2018
*Corresponding author: Jagdish Chaturvedi, Department of Otorhinolaryngology, Fortis Hospital, Bangalore, Tel: 91-9650928582; Email: dr.jagdishc@ymail.com
How to cite this article: Ravi J, Jagdish C, Pooja K, Vishal P, Abu S T, et al . Noxeno-Research & Development of an Affordable Nasal Foreign Body Removal Device in India. Glob J Oto 2018; 14(2): 555890. DOI: 10.19080/GJO.2018.14.555890.
Abstract
Introduction: Removal of Nasal Foreign Bodies (NFB) in the pediatric population is a difficult procedure for General Physicians (GP's) and pediatricians without adequate expertise in carrying out highly skilled nasal procedures. Given the distribution of ENT specialists in cities and urban regions, most children with impacted nasal foreign bodies are taken to GP's and Pediatricians who are available in semi-urban and rural regions. They however, are not experts in carrying out skill intensive nasal maneuvers that are required for a successful first attempt. With repeated unsuccessful attempts, extraction becomes complex, dangerous and often warrants removal under sedation or general anesthesia. This paper focuses on the research and development of a dedicated nasal foreign body removal device that is designed for easy removal of NFB's at the first point of care.
Methods: The methodology involved the use of the Stanford BioDesign process for unmet need assessment to identify this need, followed by a rigorous phase of product design & development. Features of the device were validated and incorporated based on an extensive clinical survey on cost, acceptability and feasibility assessment of the device.
Results: An affordable nasal foreign body removal device was specifically designed and developed for non-otorhinolaryngologists, especially General Physicians and Pediatricians, while meeting cost, skill requirement and usability criteria's gathered from the clinical survey.
Conclusion: A robust methodology similar to the one followed during the development of Noxeno is needed to be followed for developing clinically effective medical devices which meet the requirements of the user, patient and provides greater market acceptability.
Keywords: Ear Nose Throat; Nasal Foreign Body; Noxeno; Stanford Biodesign; Inventing Medical devices; Children
Abbreviations: ENT: Ear Nose And Throat; DBT: Department of Biotechnology; SIB: Stanford India BioDesign; NFB: Nasal Foreign Bodies; GP: General Physicians; AIIMS: All India Institute of Medical Sciences; BIRAC: Biotechnology Industry Research Assistance Council; BCIL: Biotech Consortium India Limited; BIG: Biotechnology Ignition Grant; SBIRI: Small Business Innovation Research Initiative
Introduction
There is an estimated incidence of 25-30 million nasal foreign body cases in India on an annual basis [1-3]. "In medical literature, a foreign object is one that is in the body but doesn't belong there" [4]. The frequency of cases of foreign objects in the ear, the nose & the throat is significantly higher in the pediatric population. This skew in frequency has been attributed to higher levels curiosity when the developmental milestone, pincer grip, has been achieved [5,6]. Anatomically, in the nasal passage the frequency of foreign bodies is higher in the region anterior to the middle turbinate and below the inferior turbinate within the nose [7]. Food, dirt, pebbles, clay, tissue, small toys, erasers pieces, paired disc magnets and button batteries are common foreign bodies found in the nasal cavity [4]. The first point of contact in the healthcare system for parents of these patients is typically a general physician or pediatrician. Due to lack of resources, especially in the rural areas, an ear nose and throat (ENT) referral is sort for but a specialist is seldom available in these geographies. Physicians & Pediatricians due to lack of resources, tend to use makeshift tools like bent needles, tweezers and glue in an to attempt to remove the foreign body. Such methods are ineffective in almost 70% of cases and can be a cause of injury to the child [3]. Failure to access the nasal foreign body in the initial attempts could push it deeper and block the airway which can lead to the patient going into respiratory distress (Figures 1
-3).
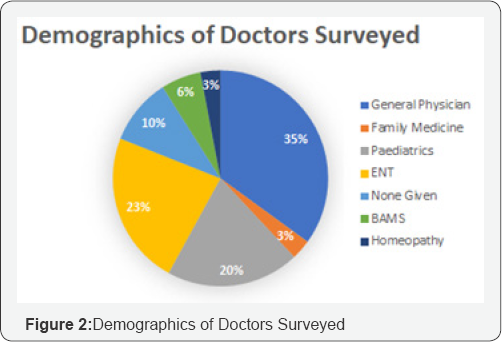
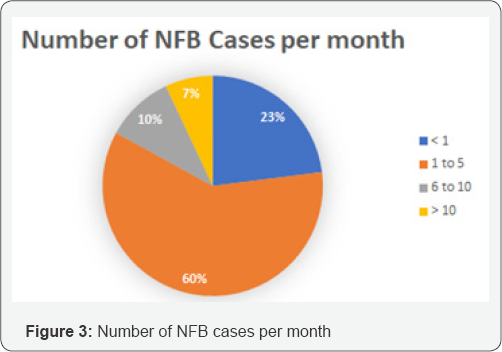
The eventual end users of a Medical Device should be involved in the entire cycle of product development from initial research and development to evaluation of the device. The end users are among the key stakeholders and play a vital role in the innovation, development, evaluation and implementation of the technology in the system [1]. Conducting ethnographic research will help comprehend the intricacies in the skill levels, the working environment and the unmet needs among the end users and this in-depth consideration of all functional capabilities and limitations goes a long way in the development of an effective product [2]. Apart from innovation of the device, holistic understanding of the end user and the ecosystem plays a pivotal role in developing business models, revenue stream, pricing strategies, sales and distribution models which will eventually define the success of the product. In keeping with the dictums mentioned above, this paper covers the product development cycle of Noxeno-A Nasal Foreign Body Removal Device-for non otorhinolaryngologists in India (Tables 1 & 2).

Methods and Materials
The need to develop a dedicated device for nasal foreign body extraction was identified through a modified research- based approach designed to identify need specifically for Indian healthcare system based on Stanford Bio-Design process which is a process template dedicated to identifying unmet need in various fields of medical science [8]. While conducting this research in the field of Otorhinolaryngology, nasal foreign body removal appeared as one of the top needs, since most cases were found to occur among children in rural settings where access to ENT specialists and dedicated resources were out of reach. More often than not, the physician at the primary health care center used makeshift tools to attempt extraction. This often led to complications which required referral and interventions at higher centers thus further increasing not only the risk associated with the foreign body but also the financial burden on the family. Clinician feedback, survey and responses on usability, ergonomics, cost, problems faced, current solutions available, prototype feedback and open-ended feedback were taken to build the must have and nice to have in the device in addition to filtering out the unwanted features (Figures 4-6)

Results
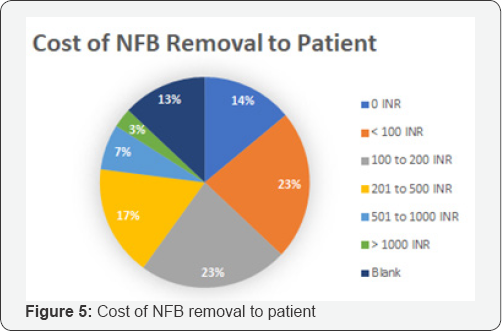
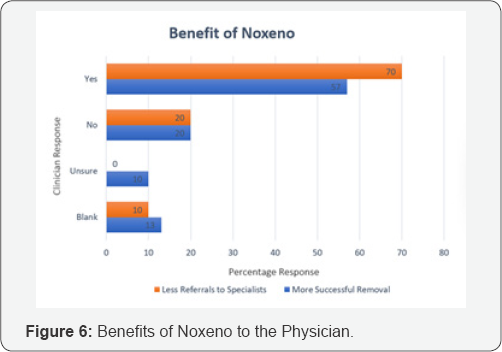
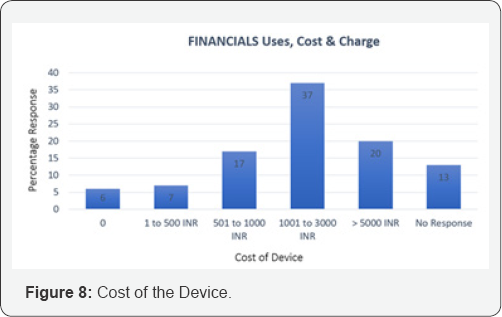
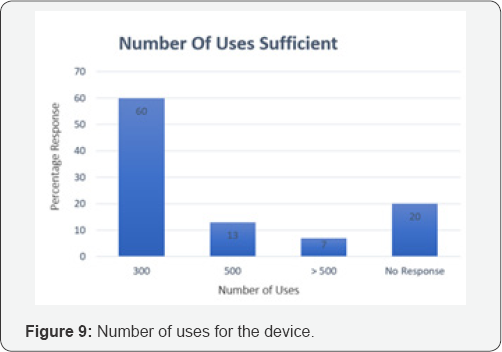
An exhaustive literature review and rigorous brainstorming was done to draw up 5 initial concepts. The concepts were classified based on their approach toward NFB extraction. Dr. Jagdish Chaturvedi, an ENT surgeon, oversaw the whole process of prototype development and testing. A silicon model- based testing was conducted for all prototypes to evaluate their efficacy. The most promising concept was found to be a hinge-based prototype. A first generation of prototype was then developed and demonstrated to clinicians to retrieve feedback. A clinician survey was designed as a three-step process. First, the working prototype was demonstrated followed by the delivery of a detailed survey questionnaire. Post survey a response analysis was conducted to evaluate the solution of the basis of efficiency, cost and acceptability. A total of 31 physicians from different clinical specialties, from varied locations in India, were surveyed with a 21-question questionnaire. The questions focused on demographics, the clinical problem, current solutions available, prototype feedback and finally open-ended feedback. 60% of the surveyed clinicians found to attend 1 to 5 patients per month with nasal foreign bodies. Almost all the cases were performed with makeshift instruments. In 46% of cases the NFB extraction was unsuccessful, though the exact reason was seldom known. The cost for nasal foreign body removal was found to have tremendous variations. The range from Rs.100 to Rs.1000 and above, was primarily associated was the complications that occurred during extraction. 70% percent of the surveyed clinician felt that the device demonstrated, Noxeno (NFB removal prototype), would help improve success rates and thus cut down referrals to ENT specialists. 87% responded that ergonomics was to their satisfaction. From a cost perspective, over 50% of those surveyed were prepared to purchase the product at price of Rs.5000. 60% responded that they should ideally be able to use the device at least 300 times before disposal. Based on the feedback and survey results, further development of the prototype was carried out. The criteria for a potential solution was set and further divided in must have and nice to have factors bas

Discussion
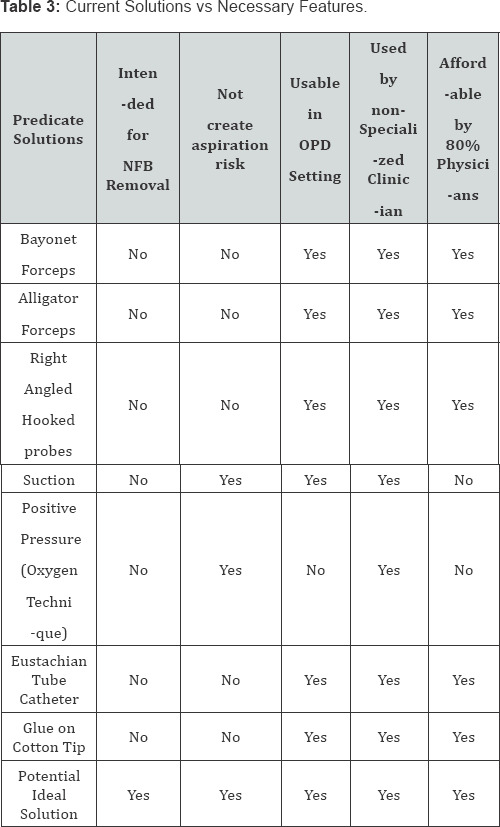
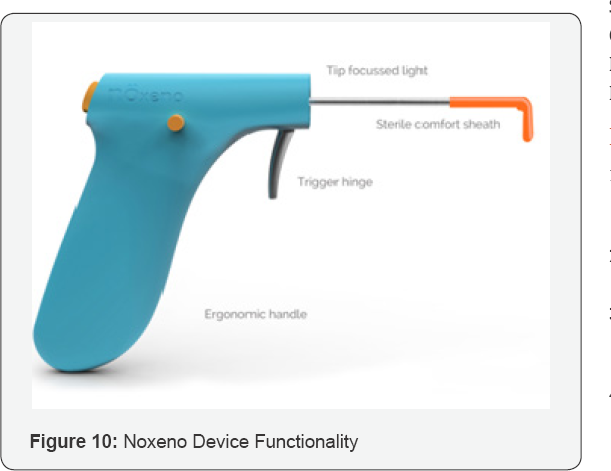
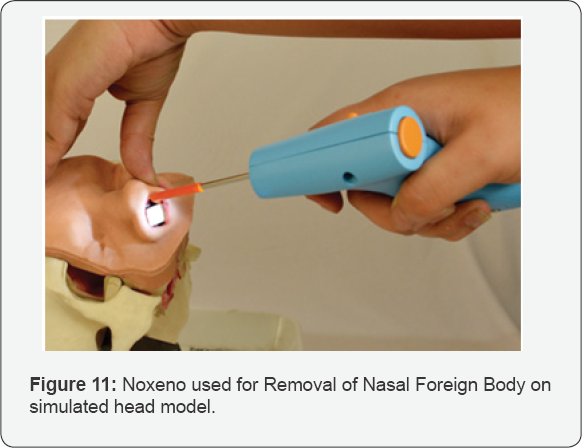
Foreign bodies in ENT have been found to account for approximately 11% of the cases seen in a clinical setting.Nasal foreign body removal in literature is said to be not only traumatic to the child but also for the on looking parents as well as embarrassing for the surgeon [9-11]. Nasal foreign bodies can lead ulceration, congestion, epistaxis, swelling and tissue destruction. There have also been reports of speech alteration, nasal discharge and halitosis [10]. The removal has been associated with various complications such as lodging in the bronchi, septal perforation etc [12]. Despite the risk of these co morbidities various techniques are attempted in the removal of an NFB. In literature these include, but are not limited to, forceps, curved hooks, ceriman loops, suction catheters, lubricated balloon-tip catheter [13]. Certain features were determined by a focus group of clinicians who gave their inputs on essential features in a potential solution. A potential solution should not create aspiration risk to the patient by further pushing the foreign body into the nasal cavity which may further cause it to enter the lungs. It should be usable in a low resource primary care setting without an operation room setup. It should be usable by a general physician and must not require a specialized clinician. Should be considered affordable by 80% of target physicians. A few features which if added, would prove beneficial are that it should remove NFB within 30 seconds. A potential solution should create a record of NFB removal and document the event of foreign body removal. A solution to bridge this unmet need is abundantly clear. Using the BioDesign process a solution was designed which would be a much-needed dedicated tool for nasal foreign body removal for non-ENT clinicians. Noxeno was launched in December 2017 powered by InnAccel Technologies Pvt. Ltd. and supported by the Department of Biotechnology and is now available in the Indian market. Indian Patent application for Noxeno is filed under name of removing foreign objects from the nasal cavity. Application Number- 3838/DEL/2012 (Figures 10&11) (Table 3).
Conclusion
The development of Noxeno was based on a robust innovation process extending from identifying the epidemiology and criticality of the unmet need to the eventual development and role of out of a nasal foreign body removal device. The clinician survey established the effectiveness, acceptability, price points, usability in various specialists beyond otolaryngologists. The development of this device represents a significant step forward in managing a condition in which makeshift devices were used with poor results and often leading to catastrophic consequences.
Acknowledgement
The authors would like to thank InnAccel Private Limited for supporting in writing this article. We are thankful to the Inventors of the Device: Dr. Jagdish Chaturvedi, Dr. Jonathan Pillai, Siddhartha Joshi, Siraj Bagwan, Dr. Suresh C Sharma, and Dr. Kapil Sikka. Most importantly, we would like to thank Department of Biotechnology (DBT) Government of India, Stanford India BioDesign (SIB), All India Institute of Medical Sciences (AIIMS), Biotechnology Industry Research Assistance Council (BIRAC), Biotech Consortium India Limited (BCIL), IIT Delhi, Biotechnology Ignition Grant (BIG) and Small Business Innovation Research Initiative (SBIRI) Grant for their support.
References
- Bairstow PJ, Mendelson R, Dhillon R, Valton F (2006) Diagnostic imaging pathways: development, dissemination, implementation, and evaluation. International Journal for Quality in Health Care 18(1): 51-57.
- Ostrander LE (1984) Attention to the medical equipment user in biomedical engineering. Medical Instrumentation 20(1): 3-5.
- (2018) Minister of S & T, Dr. Harsh Vardhan launches Noxeno, A Novel-Nasal Foreign Body Removal Device, developed under DBT supported SIB program, managed by BCIL.
- Surapaneni H, Sisodia SS (2016) Incidence of ear, nose and throat disorders in children: a study in a teaching hospital in Telangana. International Journal of Otorhinolaryngology and Head and Neck Surgery 2(1): 26-29.
- Shunyu NB, Akhtar H, Karim HM, Lyngdoh NM, Yunus M, et al. (2017) Ear, Nose and Throat Foreign Bodies Removed under General Anaesthesia: A Retrospective Study. Journal of clinical and diagnostic research JCDR 11(2): MC01.
- Davies PH, Benger JR (2000) Foreign bodies in the nose and ear: A review of techniques for removal in the emergency department. Emergency Medicine Journal 1:17(2): 91-94.
- Oreh AC, Folorunsho D, Ibekwe TS (2015) Actualities of management of aural, nasal, and throat foreign bodies. Annals of medical and health sciences research 5(2): 108-114.
- Biodesign (2015) The Process of Innovating Medical Technologies. (2nd edn.); Cambridge university press, USA.
- Balentine Jerry R, Davis Charles P Foreign Body in the Nose.
- Nath S (2016) "Visual Hook” for the removal of nasal foreign body-Case report. Romanian Journal of Rhinology 6(24).
- Giourgos G, Matti E, Pagella F (2009) Endoscopic removal of a nasal foreign body with the "hook-scope” technique. Eur Arch Otorhinolar- yngol 266(10): 1663-1665.
- Fox JR (1980) Fogarty catheter removal of nasal foreign bodies. Ann Emerg Med 9(1): 37-38.
- Chan TC, Ufberg J, Harrigan RA, Vilke GM (2004) Nasal foreign body removal. J Emerg Med 26(4): 441-445.





























