Contemplating Significance of Zingiber Officinale and Antibiotics Coated Silver Nanoparticles against Multi-Drug Resistant Bacterial Strains
Mahnoor Bakhtiar1, Asad Ullah1, Maria Shoukat*2,3, Amna Younus3, Umar Saeed3, Umair Hassan3, Omera Naseer1, Manal Rauf4, Abdul Rehman2, Ghazala Parveen3, Rahim Shah3 and Memoona Rauf5
1Department of Microbiology & Biotechnology, Abasyn University, Pakistan
2Dept of Microbiology Quaid-i-Azam University, Pakistan
3Biological Production Division, National Institute of Health, Pakistan
4Department of Pathology, Pakistan Institute of Medical Sciences, Pakistan
5Department of Biological Sciences, International Islamic University, Pakistan
Submission:September 13, 2021; Published:October 27, 2021
*Corresponding author: Maria Shoukat, Department of Microbiology Quaid-i-Azam University, Biological Production Division, National Institute of Health, Islamabad, Pakistan
How to cite this article: Mahnoor B, Asad U, Maria S, Amna Y, Umar S, et al. Contemplating Significance of Zingiber Officinale and Antibiotics Coated Silver Nanoparticles against Multi-Drug Resistant Bacterial Strains. Glob J Nano. 2021; 5(5): 555672. DOI: 10.19080/GJN.2021.05.555672
Abstract
Zingiber Officinale has been extensively used as therapeutic option against several infectious diseases due to presence of bioactive compounds. Current study demonstrates biosynthesis of silver nanoparticles (AgNPs) from the Zingiber Officinale extract to investigate augmented effect of several antibiotics including ampicillin, amikacin, meropenem, ciprofloxacin, gentamycin, ceftazidime and piperacillin against multi-drug resistant (MDR) bacterial strains such as K. pneumoniae, P. aeruginosa, Yersinia enterocolitica and Proteus. The UV-Visible spectroscopy absorption peak was observed at 425 nm which was in the prescribed range and confirmed the synthesis of AgNPs. The Scanning electron microscopy (SEM) demonstrated the morphology of AgNPs while Energy Dispersive Analysis of X-rays (EDAX) expressed the extent of silver, carbon and oxygen in the synthesis of AgNPs. While, Fourier Transform Infrared Spectroscopy (FTIR) gave confirmation about capping and reduction of AgNPs due to presence of phenolic, carboxyl and hydroxyl groups. Moreover, it was also observed that AgNPs significantly enhanced the antibacterial activities of test antibiotics against MDR bacterial strains (p = 0.01) and it was confirmed by comparing the zones produced by non-coated antibiotics with the zones produced by AgNPs coated antibiotics. AgNPs solution displayed maximum zone of inhibition against Yersinia followed by K. pneumoniae, P.aeruginosa and Proteus while the activity of AgNPs coated ampicillin, amikacin, meropenem, ciprofloxacin, gentamycin, ceftazidime and piperacillin antibiotics increased significantly against Yersinia, K. pneumoniae, P.aeruginosa and Proteus. AgNPs synthesized from Zingiber Officinale extract might be critical in pharmaceutical and medical science.
Keywords:Zingiber officinale extract; Biosynthesis; Silver nanoparticle; MDR bacterial strains; Antibiotics
Abbreviations: AgNPs: Silver Nanoparticles; MDR: Multi-drug Resistant; UV: Ultra Violet; SEM: Scanning Electron Microscopy; EDAX: Energy Dispersive Analysis of X-rays; FTIR: Fourier transform Infrared Spectroscopy; Ag+: Silver Ions; WHO: World Health Organization; XRD: X-Ray Diffraction; HFMR: Hydroxyl-Footed Methyl Resorcinarene; ROS: Reactive Oxygen Species
Introduction
Nanotechnology is the field of material science which deals with the synthesis of minute particles having less than 100 nm in size. Furthermore, these particles are having multiple chemical and biological properties due to which they have been used in several types of industries like pharmaceutical industries, medical sciences, mechanics, chemical industry, catalysis, photoelectrochemical sciences [1]. Silver (Ag) is an inorganic compound, which is present in white powder form and insoluble in water under normal conditions. It is widely used in the formulation of many types of products such as pigments ceramics, lubricants, adhesives tapes and ointment [2]. Silver is a harmless and biocompatible semiconductor material which is mainly used in biological science such as in Nano medicine due to their high level of antimicrobial properties [3].
There are different types of methods for the synthesis of nanoparticles such as chemical, physical and biological methods. Among all these methods, biological methods are considered to be safe and cost-effective as compared to other methods [3]. Throughout the biological synthesis of nanoparticles, different factors are involved which are needed to be considered most important and these are maintenance of cell culture, intracellular synthesis and multi-purification steps. Plants are an eco-friendly and beneficial approach for the synthesis of nanoparticles as they are easily accessible for production of nanoparticles at a large scale as well the rate of production is faster in contrast to other biological models such as algae, fungi and bacteria [5-7].
Zingiber Officinale belongs to a family of Zingiberaceae and widely used as an alternate therapy of medicine for the treatment of several infectious diseases like colds, cough, inflammation, tumors, cardiac disorders, and several others [8-10]. It contains several bioactive compounds, such as shogaols, alkaloids, gingerols, zingiberene and flavonoids [11]. Biomolecules present in Zingiber Officinale extract play an important role in the reduction of silver ions (Ag+) to metallic AgNPs (Ag0) because Zingiber Officinale has high antioxidants activity [1].
Silver nanoparticles (AgNPs) synthesized from the leaves extract of plants which is used as an antimicrobial agent has proven very high level of antimicrobial properties against several bacterial strains as they are small in size so they can easily penetrate the target site l and destroy synthesis machinery within the cell and caused cell death of the target organism [12]. In addition to this, AgNPs are also used for the synergetic effect with antibiotics to increase their activity against multidrug-resistant bacterial species [13].
World health organization (WHO) has a responsibility to implement different actions to overcome infection caused by multidrug-resistant (MDR) bacteria because Antimicrobial resistance (AMR) has become a serious issue to the entire world [14]. The main reason for the spread of bacterial MDR is the use of appropriate broad-spectrum antibiotics for the treatment of infectious diseases whose specific causative agent is not diagnosed [15]. To overcome this problem, researchers are trying their best to develop new and alternative drugs. Nanotechnology is expecting the field to help us in fighting against such pathogens at the atomic level. The silver nanoparticles among all are well known for their inhibitory activity and antimicrobial effects and can be utilized for treating numerous infections [16]. The effect of nanoparticles against MDR strains was found to be more significant [1].
The present research study was designed to mainly focus on to synthesis of AgNPs from Zingiber Officinale (ginger) extract and then determine its bio-catalytic activity with other commercially available antibiotics against multidrug-resistant bacterial species. This study will help to improve the quality of commercially available antibiotics against multidrug-resistant bacterial strains. As the emergence of multidrug-resistance phenomenon increases day by day, therefore by utilizing AgNPs in combination with commercially available antibiotics can provide an alternate therapy for the treatment of bacterial infections.
Results
Characterization of AgNPs synthesized from Zingiber officinale
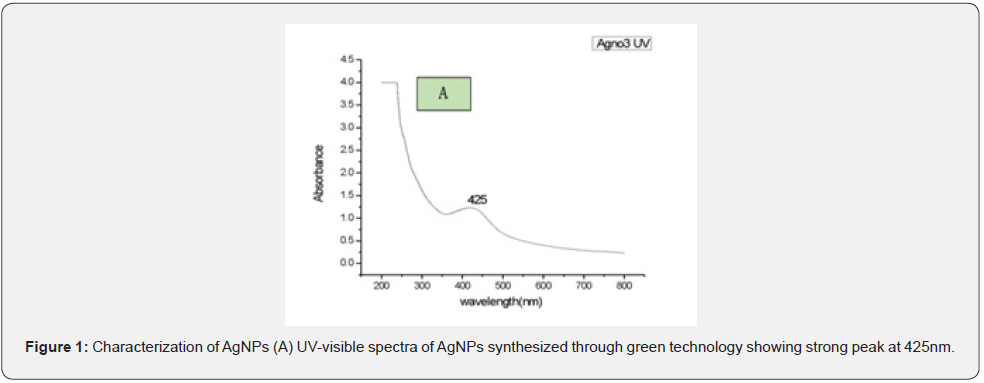
In the current study, AgNPs were prepared from the Zingiber Officinale extract by utilizing a green synthesis approach and then different analytical techniques such as UV-visible spectroscopy, SEM, EDAX, X-Ray Diffraction (XRD) and FTIR were used for the characterization of AgNPs. PerkinElmer UV-Visible spectroscopy lambda 25 with a wavelength range from 300-800 nm was used in the current study to measure the absorbance of synthesized AgNPs. From UV-visible spectroscopy, it was observed that the sample had absorbed energy at 425 nm which was a characteristic peak value of AgNPs. Besides this, the absorption peak at 425 nm with no other peak displayed high purity of the nanoparticles as shown in (Figure 1).
In addition, SEM technique was used in the current study for the overall appearance of the AgNPs. The SEM micrograph of the synthesized AgNPs from Zingiber Officinale confirmed the monodispersed and irregular morphology of the AgNPs showing particle size in the range of 100 nm at a magnification of 30,000X as shown in (Figure 2). The SEM results indicated that Zingiber Officinale extract acts as a strong reducing agent during the synthesis of irregular shaped AgNPs. In addition to this, EDAX was used to measure the elemental composition of the samples.
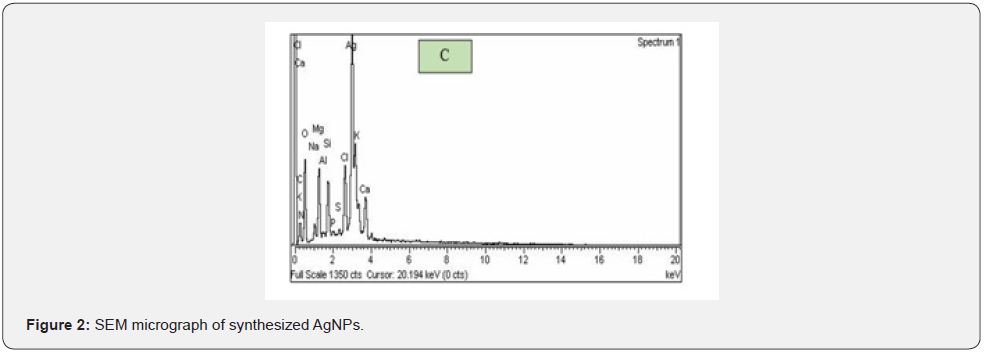
The EDAX spectra of AgNPs presented from a simple precipitation method using Zingiber Officinale extract and AgNO3 were the starting materials for the synthesis of nanoparticles. The results of EDAX spectrum showed a high intensive peak of silver which confirmed the synthesis of AgNPs. The spectra indicated the AgNPs peak at 34 kV, whereas calcium, chlorine, magnesium, oxygen and potassium peaks were also present indicating the mixed precipitation in the Zingiber Officinale extract as shown in (Figures 3-5).

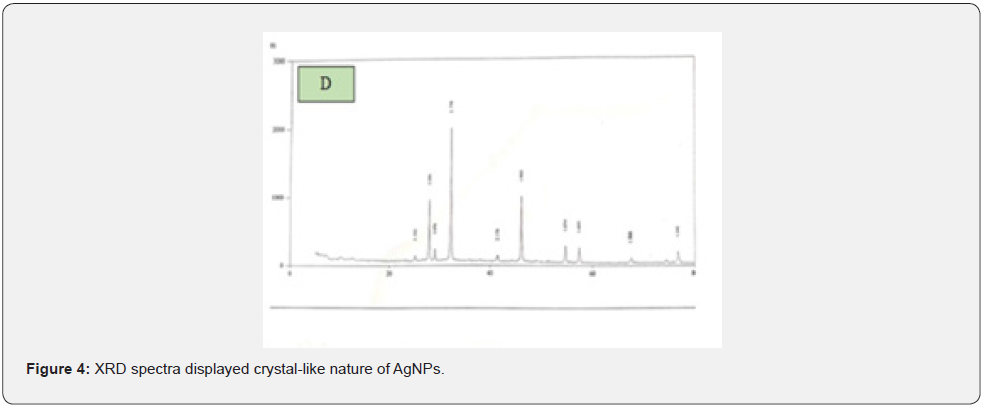
Moreover, XRD was a rapid analytical technique primarily used for phase identification of crystalline material. Besides this, it was also used to provide information on unit cell dimensions. It was observed from XRD spectrum that the sample was finely grounded and homogenized material. The crystal-like nature of AgNPs was established by the analysis of XRD (Figure 1D). The three district diffraction peaks at 2θ values of 27.8, 32.2, and 46.2 could be indexed to the (1054), (2223), and (1064) reflection planes of cubic structure. In addition to the Bragg peaks, additional peaks were also observed at 25.050, 29.000, 41.500, 54.800, 57.450, 67.400 and 76.650 θ which were due to the presence of organic contaminants.
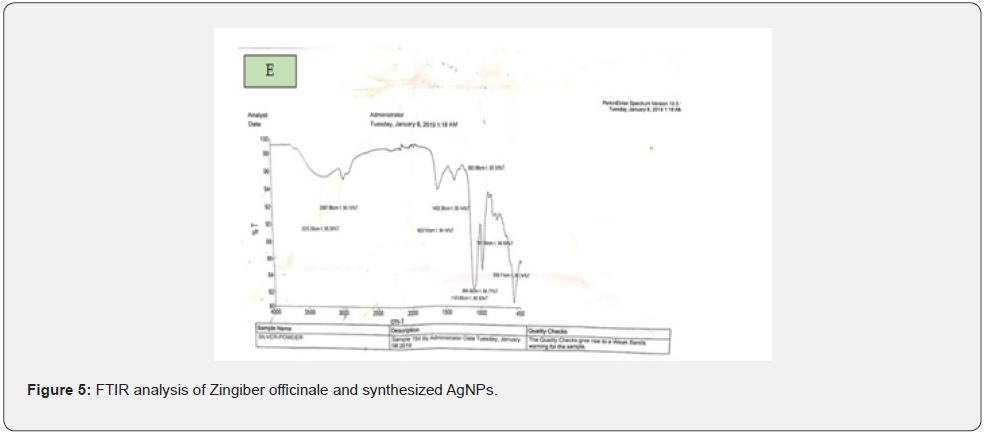
For further characterization, FTIR analysis of the Zingiber Officinale and synthesized AgNPs were performed and different peaks were observed in the FTIR spectrum representing amines, carboxylic acids and alkanes, which played an important role in capping, stabilization and synthesis of AgNPs. In the current study, the peaks in the FTIR spectrum of Zingiber Officinale extract at 3255.56, 2987.96, 1637.51, 1405.38, 994.56, 1103.85, 903.88, 791.80 and 535.71cm showed interrelation of different functional groups such as carboxylic acid, amine salt or alkane, conjugated alkene or alkene, carboxylic acid, alkenes, secondary alcohol or floro-compound, 1,2,4-trisubstituted, alkene and halo compound respectively as shown in Figure 1E were compared with standard IR absorption table retrieved at 8.8.2019.
Confirmation of previously identified bacterial isolates
As discussed earlier in the methodology section that bacterial strains such as K. Pneumonia, P.aeruginosa, Yersinia enterocolitica and Proteus were obtained from the Hayatabad Medical Complex, Peshawar, which was previously identified by 16S RNA sequencing. In the current study, these bacterial strains were determined by colony morphology using the standard guidelines. On nutrient agar milky white, translucent, convex mucoid colonies of K. pneumoniae, off white, moist and healthy growth of Proteus, slimy white, transparent, small and large round shape colonies of P. aeruginosa and white, transparent, cocci shaped of Yersinia were observed. Besides, all the colonies were observed as pink color Gram-negative rods except Yersinia which was gram-negative coccobacilli after microscopy. Furthermore, it was confirmed after the accomplishment of different biochemical tests as shown in (Table 1).
coated antibiotics against test organisms
The complete profile of antibacterial activity of AgNPs, noncoated and AgNPs coated antibiotics against test organisms is depicted in (Table 2). In the current study, it was observed AgNPs solution displayed a maximum zone of inhibition against Yersinia (22.6±3.05mm) followed by K. pneumoniae (22±2mm), P. aeruginosa (19±4mm) and Proteus (17.6 ±2.5mm). However, it was perceived, K. pneumoniae, P. aeruginosa and Proteus all of them displayed resistance patterns against non-coated Ampicillin, Amikacin, Meropenem, Ciprofloxacin, Gentamycin, Ceftazidime and Piperacillin antibiotics.
On the other hand, the activity of antibiotics increased significantly (p = 0.01) against these strains after coating with AgNPs. It was observed that the activity of AgNPs coated Ampicillin, Amikacin, Meropenem, Ciprofloxacin, Gentamycin, Ceftazidime and Piperacillin antibiotics increased up to ¬72, 69.8, 64.6, 72.2, 66.6, 78.8 and 78.8% against Proteus, 136, 80, 60.3, 113.8, 93.5, 75.6 and 42.8% against K. pneumoniae, 114.4, 80.5, 60.3, 55.7, 56.2, 41.9 and 53.1% against P.aeruginosa and 116.5, 116.9, 50.7, 51.2, 62.5, 56.1 and 112% against Yersinia respectively.
Discussion
Green technology used for the synthesis of silver nanoparticles (AgNPs) gained significant importance in the recent year within the research community due to their simple, nontoxic, less time consumption, cost-effective nature and also due to their effectiveness in large scale production [17]. Besides this, AgNPs produced through green synthesis also possessed strong antimicrobial and anti-inflammatory activity that’s why extensively used in pharmaceutical and cosmetic products [18,19]. In the current study, AgNPs were prepared from the extract of Zingiber Officinale and then different analytical techniques such as UV-visible spectroscopy, SEM, EDAX, XRD and FTIR were used for the characterization of AgNPs. UV-visible spectroscopy revealed sample absorbance energy of 425 nm which was a characteristic peak value of AgNPs. Besides this, absorption peak at 425 nm with no other peak displayed high purity of the nanoparticles and also confirmed that maximum absorbance of AgNPs depends on Ag+ ions concentration concerning peel extract of Zingiber Officinale [20,21]. Different researchers observed variation in the absorption peak of AgNPs in the range of 390-455 nm and possible reasons for this might be differences in the manufacturing process (chemical, physical and biological method) as a result variation occurs in the morphology of AgNPs [22].
In the current study, SEM and EDAX techniques were used for the overall topology of the AgNPs. The SEM micrograph of the synthesized AgNPs using Zingiber Officinale extracts confirmed the monodispersed and irregular topology of the AgNPs showing particle size in the range of 100 nm at a magnification of 30,000X. The SEM results indicated that chemical constituents of extract Zingiber officinalea act as strong reducing agent resulting in the formation of irregular shaped AgNPs. Rout and his co-workers [23]. Utilized hydroxyl-footed methyl resorcinarene (HFMR) in SEM technique for the investigation of AgNPs size. They observed that as the amount of HFMR increased, the size of the AgNPs also increased. Furthermore, they observed irregular or hexagonal-shaped AgNPs in SEM spectrum which was in agreement with our results. But the size of the nanoparticle in the current study was greater as compared to their results which might be due to different manufacturing conditions like temperature, time of incubation, nature of plant extract and handling applications. Also, EDAX was used to measure the elemental composition of the samples. The EDAX spectra gave a sharp peak in the silver region which confirmed the synthesis of AgNPs. The spectra indicated the AgNPs peak at 34 kV, whereas calcium, chlorine, magnesium, oxygen and potassium peaks were also present indicating the mixed precipitation in the extract of Zingiber officinale. Similar peaks of silver, carbon and oxygen were observed in EDAX spectrum of AgNPs by different researchers in their studies [24].
Moreover, XRD was a rapid analytical technique primarily used for phase identification of crystalline material. Besides, it was also used to provide information on unit cell dimensions. It was observed from XRD spectrum that the sample was finely grounded and homogenized material. Moreover, the three district diffraction peaks at 2θ values of 27.8, 32.2 and 46.2 could be indexed to the (1054), (2223) and (1064) reflection planes of cubic structure which coincide with the results of Dang et al., [25]. Begumet and his co-worker’s [26]. synthesized silver (Ag) nanoparticles by chemical as well as green technology and used sodium hydroxide plus silver nitrate as a reagent and found that the size of AgNPs was in the range of 830 nm whereas, those produced by chemical method had size in the range of 813 nm. In the present research study, only AgNO3 was used as a reagent in the synthesis of AgNPs that’s why the size of nanoparticles didn’t affect. In addition to this, FTIR spectroscopy was actually used to measure the absorption of infrared radiations by AgNPs as a function of wavelength and different peaks were observed in FTIR spectrum representing amines, carboxylic acids and alkanes, which facilitate the capping, stabilization and synthesis of AgNPs. Moreover, after reaction with AgNO3, new peaks in FTIR spectrum were observed which clearly indicated that carboxylic, hydroxyl and amide groups in the Zingiber Officinale extract of Zingiber Officinale played role in the synthesis of AgNPs. Similar results were also reported by Singh et al. [27]. utilizing Dillenia indica fruit extract and found the same band’s position in FTIR spectrum.
In the present study, it was observed that K. pneumonia, P.aeruginosa, Yersinia enterocolitica and Proteus showed resistance to non-coated ampicillin, amikacin, meropenem, ciprofloxacin, gentamycin, ceftazidime and piperacillin antibiotics and this might be due to alteration in the target sites, enzymatic modification, genetic mutation and efflux mechanism [28]. On the other hand, it was observed that AgNPs solution displayed maximum zone of inhibition against Yersinia (22.6±3.05mm) followed by K. pneumoniae (22±2mm), P.aeruginosa (19±4mm) and Proteus (17.6±2.5mm) while the activity of AgNPs coated Ampicillin, Amikacin, Meropenem, Ciprofloxacin, Gentamycin, Ceftazidime and Piperacillin antibiotics increased significantly (p = 0.01) against Yersinia (50.7-116.9%), K. pneumoniae (42.8-136%), P.aeruginosa (41.9-114.4%) and Proteus (64.6- 78.8%). It was reported that AgNPs carried out modification in the membrane permeability of bacterial strains and also act as a carrier to transport antibiotics to the inner portion of K. pneumoniae, P.aeruginosa, Yersinia enterocolitica and Proteus and in this way they enhance the activity of antibiotics against these resistant strains [29]. In other words, Ag+ ions coated on the surface of antibiotics bind with negatively charged structural components i.e. proteins and nucleic acid and cause alteration in the basic structure of cell wall, cell membrane and nucleic acids of the target organisms [30,31]. In addition to this, Ag+ ions also interact with other electron-donating groups such as thiols, PO4+2 and OH- of the cell wall, cell membrane and nucleic acids of the target organisms. As a result, the damaged membranes induced the release of reactive oxygen species (ROS) which forming free radicals with a powerful bactericidal action [31]. also conducted a similar study by synthesizing plant-mediated silver nanoparticles and then applied them in combination with ofloxacin, norfloxacin and cephalexin antibiotics against E. coli, S. aureus and P. aeroginosa and observed synergistic effects i.e. the antibacterial activity of antibiotics increased after coating with nanoparticles. According to their observations, the conjugate of nanomaterial and antibiotics showed enhanced antimicrobial activity against all the test organisms. Sulochanaet and his coworkers [32] also conducted similar study by utilizing different antibiotics in combination with silver nanoparticles against pathogenic bacterial species and found almost similar results.
Conclusion
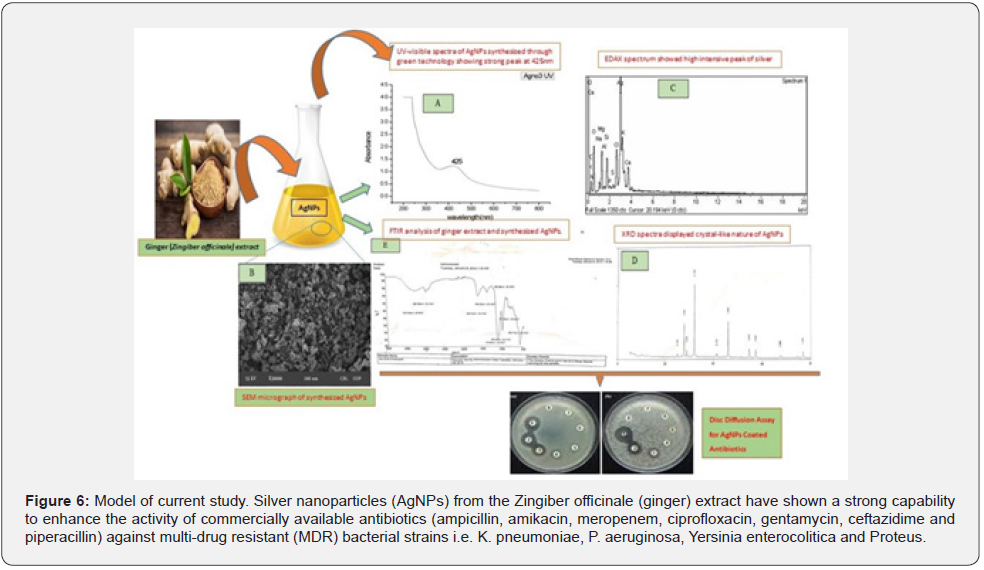
Model of current study is shown in (Figure 6). AgNPs solution displayed maximum zone of inhibition against Yersinia followed by K. pneumoniae, P.aeruginosa and Proteus while the activity of AgNPs coated Ampicillin, Amikacin, Meropenem, Ciprofloxacin, Gentamycin, Ceftazidime and Piperacillin antibiotics increased significantly against Yersinia, K. pneumoniae, P.aeruginosa and Proteus. AgNPs synthesized from Zingiber Officinale extract could be of great importance in pharmaceutical and medical science for their biocatalytic activity as they enhanced the activity of existing antibiotics by modifying their pharmacokinetic as well as pharmacodynamics properties.
Methods
Collection of test Organisms
In the current research study, a total of four previously identified antibiotics resistant bacterial strains such as Klebsiella pneumoniae, Pseudomonas aeruginosa, Yersinia enterocolitica and Proteus vulgaris were collected from the Hayatabad Medical Complex Peshawar, Pakistan which was preliminarily confirmed before performing bioassay.
Preparation of plant extract
Zingiber Officinale was collected from the local grain market at District Peshawar. Took a vegetative part of the Zingiber Officinale plant. Washed it 2 to 3 times with tap water to eliminate impurities, then treated it with de-ionized water to remove the remaining contaminants. The selected part was dried at room temperature in shade for 10-15 days, and finely grinded into small pieces. After that, the pieces were crushed in grinder mortar pestle (Muller) to form a fine powder. Then 60 g of that powder was stirred in 600 ml distilled water and boiled at 80°C for 30 minutes to form a complete solution. After that, the extract was cool down at room temperature and then filtered through Whatman filter paper no 1 and also centrifuged that solution at 2000rpm for 10 minutes to get fine and pure filtrate. The filtrate was stored at 4°C.
Preparation of 1mM silver nitrate solution
For 1mM silver nitrate solution preparation, 0.51 silver nitrate (AgNO3) powder was dissolved in 3000ml distilled water and mixed well.
Synthesis of silver nanoparticles
The solution was prepared by mixing 300ml of pure extract to 2700ml of 1 mM aqueous solution of silver nitrate (AgNO3) in three distinct 1000ml Erlenmeyer flask. Covered the flasks with newspaper or aluminum foil to minimize photoactivation of silver nitrate. The mixture was left at room temperature for 2days to complete the reaction. Reduction from Ag+ to Ag0 was confirmed by the color change of the solution from colorless to milky white and bright yellow (the color change shows the presence of silver nanoparticles). Its formation can also be confirmed by using UVVIS spectrophotometer.
Purification of nanoparticles
The silver nanoparticle thus obtained was purified by repeated centrifugation at 2500 rpm for 10 min. The purified pellet was dried by putting it into the oven at 60°C for two days. The dried pellet is then scratched from the Petri plate and later on collected into the Eppendorf tube. The total amount of nanoparticles produced from 3000ml solution is 0.95 which is followed by redispersion of the pellet into 10 ml de-ionized water. Then silver nanoparticles were kept in the refrigerator till further analysis.
Characterization of synthesized silver nanoparticles
Characterization of AgNPs was made through UV-visible spectroscopy, Scanning electron microscopy (SEM), Energy dispersive analysis of X-rays (EDAX), Fourier transform infrared spectroscopy (FTIR) and X-ray diffraction (XRD) in Centralized Resource Laboratory (CRL), University of Peshawar, Pakistan.
Antimicrobial activity of silver nanoparticles solution by well diffusion method
The antimicrobial activity of silver nanoparticles was then tested by the standard well diffusion method. The strains were cultured through previously used methods i.e. on Muller–Hinton Media. Wells of 6mm was made using a sterile cork-borer. The silver nanoparticle solution was prepared by mixing 0.58gram silver nanoparticles in 5ml de-ionized water. About 150μl solution was then poured to already prepared culture media of known strains marked under the sterile condition and incubated at 37ºC for 24h. The zone of inhibition was measured and expressed in millimeters.
Disc diffusion assay of non-coated antibiotics
Total seven antibiotics such as Gentamycin (CN), Ciprofloxacin (CIP), Amikacin (AK), Ceftazidime (CAZ), Piperacillin (TZP), Meropenum (MEM) and Ampicillin (AMP) were used against four different strains i.e. Klebsiella pneumoniae, Pseudomonas aeruginosa, Yersinia Entero colitica and Proteus vulgaris. Four plates of Muller-Hinton Media were prepared. Then the known strains were streaked on media by using a sterile cotton swab. The swab was first dipped in known strain then dipped in the 5ml water of injection to get the clean colonies. After that, the swab was streaked on the whole media plate. Then antibiotic discs were put on the cultured media. After 24hour of incubation at 37 ºC, the result obtained showed that all the strains were sensitive towards the above-used antibiotics.
Disc diffusion assay for AgNPs coated antibiotics
To check the synergistic effect of antibiotics, the antibiotics were coated with the silver nanoparticle solution. About 50μl AgNPs solution was poured on an antibiotic within petri plates then placed in an oven at 80ºC for 15 min for drying and this method of the coating was repeated for each antibiotics. Then applied the coated antibiotics on the prepared cultured media. All the strains showed sensitivity to this synergistic effect which indicates that silver nanoparticles could increase the activity of antibiotics. The zone of inhibition was measured and expressed in millimetres [33].
Statistical analysis
The mean values of AgNPs coated and non-coated antibiotics activity against test organisms were calculated using the Microsoft Excel program. Furthermore, the t-test was determined using the Microsoft Excel program and p-value of less than 0.05 was measured as a statistical impact value.
Acknowledgement
The authors would like to acknowledge the Department of Microbiology & Biotechnology, Abasyn University, Peshawar for research guidance and financial assistance. Also, the author would like to thanks the co-authors from the Department of Pathology, Pakistan Institute of Medical Sciences, Islamabad; Biological Production Division of National Institute of Health, Islamabad; Department of Microbiology Quaid-i-Azam University, Islamabad; Department of Biological Sciences, International Islamic University, Islamabad; and Department of Pharmacy, University of Peshawar, Pakistan for their technical assistance and guidance in writing and reviewing the manuscript. The work presented in this review article is a collaborative effort.
Author’s contributions
MB and AR conceived the idea and designed the study. MB and AR collected and prepared the samples.AU, MR and MR assist in sampling. US, MS, AY and UH drafted the manuscript. US, GP, RS and AR critically reviewed the manuscript. All authors read and approved the final manuscript.
Refrences
- Heidary M, Goudarzi H, Hashemi A, Eslami G, Goudarzi M, et al. (2013) Deploy vaccines to fight superbugs. Antimicrob Agents Chemother 4(1): 1446-1449.
- Song JY, Kim BS (2009) Rapid biological synthesis of silver nanoparticles using plant leaf extracts. Bioprocess Biosyst Eng 32(1): 79.
- Zheng X, Wang J, Xu W, Xu J, Hu J (2017) Clinical and molecular characteristics, risk factors and outcomes of Carbapenem-resistant Klebsiella pneumoniae bloodstream infections in the intensive care unit. Antimicrob Resist Infect Control 6(1): 102.
- Ingale AG, Chaudhari AN (2013) Biogenic synthesis of nanoparticles and potential applications: an eco-friendly approach. J Nanomed Nanotechol 4(165): 1-7.
- Nair R, Varghese SH, Nair BG, Maekawa T, Yoshida Y, et al. (2010) Nanoparticulate material delivery to plants. Plant Sci 179(3): 154-163.
- Borase HP, Patil CD, Suryawanshi RK, Patil S V (2013) Ficus carica latex-mediated synthesis of silver nanoparticles and its application as a chemophotoprotective agent. Appl Biochem Biotechnol 171(3): 676-688.
- Nahar MK, Zakaria Z, Hashim U, Bari MF (2015) Biological synthesis of nanosilver by using plants. In: Advanced Materials Research. Trans Tech Publ, pp 30-34.
- Leach MJ, Kumar S (2008) The clinical effectiveness of Ginger (Zingiber officinale) in adults with osteoarthritis. Int J Evidence‐Based Healthc 6(3): 311-320.
- Ding M, Leach MJ, Bradley H (2013) A systematic review of the evidence for topical use of ginger. Explore 9(6): 361-364.
- Drummond N, Murphy BP, Ringwood T, Prentice MB, Buckley JF, et al. (2012) Yersinia enterocolitica: a brief review of the issues relating to the zoonotic pathogen, public health challenges, and the pork production chain. Foodborne Pathog Dis 9(3): 179-189.
- Pages J-M, Lavigne J-P, Leflon-Guibout V, Marcon E, Bert F, et al. (2009) Efflux pump, the masked side of ß-Lactam resistance in Klebsiella pneumoniae clinical isolates. PLoS One 4(3): e4817.
- Ahmed S, Ahmad M, Swami BL, Ikram S (2016) A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: a green expertise. J Adv Res 7(1):17-28
- Bankar A, Joshi B, Kumar AR, Zinjarde S (2010) Banana peel extract mediated novel route for the synthesis of silver nanoparticles. Colloids Surfaces A Physicochem Eng Asp 368(1-3): 58-63
- Jansen KU, Knirsch C, Anderson AS (2018) The role of vaccines in preventing bacterial antimicrobial resistance. Nat Med 24(1): 10-19.
- Rappuoli R, Bloom DE, Black S (2017) Deploy vaccines to fight superbugs. National library of medicine 552(7684): 165-167.
- Mudshinge SR, Deore AB, Patil S, Bhalgat CM (2011) Nanoparticles: emerging carriers for drug delivery. Saudi Pharm J 19(3): 129-141.
- Nagarajan S, Kuppusamy KA (2013) Extracellular synthesis of zinc oxide nanoparticle using seaweeds of gulf of Mannar, India. J Nanobiotechnology 11(1): 39.
- Otunola GA, Afolayan AJ, Ajayi EO, Odeyemi SW (2017) Characterization, antibacterial and antioxidant properties of silver nanoparticles synthesized from aqueous extracts of Allium sativum, Zingiber officinale, and Capsicum frutescens. Pharmacogn Mag 13(Suppl 2): S201.
- Mathew S, Prakash A, Radhakrishnan EK (2018) Sunlight mediated rapid synthesis of small size range silver nanoparticles using Zingiber officinale rhizome extract and its antibacterial activity analysis. Inorg Nano-Metal Chem 48(2): 139-145.
- Shukla VK, Pandey S, Pandey AC (2010) Green synthesis of silver nanoparticles using neem leaf (Azadirachta indica) extract. In: AIP Conference Proceedings. American Institute of Physics pp. 43-49.
- Mahamudin M, Samah NA, Muhamad S, Ibrahim AL (2019) Green synthesis of silver nanoparticles from polygonum minus extract and its antimicrobial properties. J Acad 7(2): 46-55.
- Sharma VK, Sayes CM, Guo B, Pillai S, Parsons JG, et al. (2019) Interactions between silver nanoparticles and other metal nanoparticles under environmentally relevant conditions: A review. Sci Total Environ 653: 1042-1051.
- Rout Y, Behera S, Ojha AK, Nayak PL (2012) Green synthesis of silver nanoparticles using Ocimum sanctum (Tulashi) and study of their antibacterial and antifungal activities. J Microbiol Antimicrob 4(6): 103-109.
- Nguyen T-D, Dang C-H, Mai D-T (2018) Biosynthesized AgNP capped on novel nanocomposite 2-hydroxypropyl-β-cyclodextrin/alginate as a catalyst for degradation of pollutants. Carbohydr Polym 197:29–37.
- Shankar SS, Ahmad A, Pasricha R, Sastry M (2003) Bioreduction of chloroaurate ions by geranium leaves and its endophytic fungus yields gold nanoparticles of different shapes. J Mater Chem 13(7): 1822-1826.
- Agarwal H, Kumar SV, Rajeshkumar S (2020) Antidiabetic effect of silver nanoparticles synthesized using lemongrass (Cymbopogon Citratus) through conventional heating and microwave irradiation approach. J Microbiol Biotechnol Food Sci 9(6): 371-376.
- Singh S, Saikia JP, Buragohain AK (2013) A novel ‘green’synthesis of colloidal silver nanoparticles (SNP) using Dillenia indica fruit extract. Colloids Surfaces B Biointerfaces 102: 83-85.
- Zaki SS, Kheiralla ZH, Rushdy AA, Betiha MA, Abousittash HB (2017) Embedded Mesoporous Silica Silver Nanoparticles as potential antibacterial agent againstMultidrug-Resistant Bacteria. J Sci Res Sci 34(part1): 158-178.
- Hussain A, Alajmi MF, Khan MA, Pervez SA, Ahmed F, et al. (2019) Biosynthesized silver nanoparticle (AgNP) from Pandanus odorifer leaf extract exhibits anti-metastasis and anti-biofilm potentials. Front Microbiol 10: 8.
- Choi O, Clevenger TE, Deng B, Surampalli RY, Ross Jr L (2009) Role of sulfide and ligand strength in controlling nanosilver toxicity. Water Res 43(7): 1879-1886.
- Kim S, Choi JE, Choi J, Chung K-H, Park K, et al. (2009) Oxidative stress-dependent toxicity of silver nanoparticles in human hepatoma cells. Toxicol Vitr 23(6): 1076-1084.
- Roy S, Das TK (2015) Plant mediated green synthesis of silver nanoparticles. A Rev Int J Plant Biol Res 3(3): 1044-1055.
- Hocquet D, Bertrand X, Köhler T, Talon D, Plésiat P (2003) Genetic and phenotypic variations of a resistant Pseudomonas aeruginosa epidemic clone. Antimicrob Agents Chemother 47(6): 1887-1894.






























