Analytical Method Development and Validation of UV-Visible Spectrophotometric Method for the Estimation of Linagliptin
Deepika Joshi1*, Bhavana Singh1, Archana Rautela2 and Nidhi Semwal1
1School of Pharmaceutical Sciences, SGRR University, Dehradun, 248001, India
2Gyani Inder Singh Institute of Professional Studies, Dehradun, 248001, India
Submission:July 21, 2021; Published:September 13, 2021
*Corresponding author: Deepika Joshi, School of Pharmaceutical Sciences, SGRR University, Dehradun, 248001, India
How to cite this article: Deepika J, Bhavana S, Archana R, Nidhi S. Analytical Method Development and Validation of UV-Visible Spectrophotometric Method for the Estimation of Linagliptin. Glob J Nano. 2021; 5(5): 555671. DOI: 10.19080/GJN.2021.05.555671
Abstract
Aim: Simple, precise, accurate, cost effective, rapid, and sensitive UV/visible spectrophotometric method was developed at the wavelength of maximum absorption, λmax (290 nm). The developed UV/visible spectrophotometric method was validated as per ICH guidelines for the estimation of Linagliptin in active pharmaceutical dosage form and bulk form.
Method: Linagliptin was characterized for reformulation studies by drug solubility profile, Functional groups (Fourier Transform Infra-Red) and melting point detection. The drug was analyzed using UV/visible spectrophotometric method that was further validated in terms of linearity, accuracy, precision, specificity, Limit of detection and Limit of quantitation. The solvent used was methanol: water (15:85, v/v) and the wavelength corresponding to maximum absorbance of the drug were found at 290 nm.
Result: Melting point of drug was found 201°C nearly corresponds to its actual melting range. The linear response for concentration range of 6-16 μg/ml with a regression equation; y = 0.1147x + 0.0184, with a correlation coefficient, r2 = 0. 98074.The accuracy was found between 98.51- 102.06%. Precision for intra-day and inter-day was found to be within the limits. To establish the sensitivity of the method, limit of detection (LOD) and limit of quantification (LOQ) were determined which were found to be 5.03 μg /mL and 21.22 μg /mL respectively.
Conclusion: The drug was confirmed by interpretation of UV spectra. Hence proposed method stands out validated and thus may be used for routine analysis of Linagliptin in pharmaceutical dosage forms.
Keywords: Linagliptin; UV/visible spectrophotometric method; Lambert-Beer’s Law; ICH Guidelines; Linear; Validate; Accuracy
Introduction
Linagliptin, an oral hypoglycemic drug is a highly potent, selective, and long-acting dipeptidyl peptidase-4 (DPP-4) inhibitor chemically known as 8-[(3R)-3-aminopiperidin-1-yl]-7-but-2-ynyl-3-methyl-1-[(4-methylquinazolin-2-yl) methyl] purine-2,6-dione as shown in (Figure 1). The empirical formula is C25H28N8O2, and the molecular weight is 472.54 g/mol [1,2]. It act by competitive and reversible inhibition of dipeptidyl peptidase (DPP)-4 enzyme that degrades the incretin hormones glucagon- likepeptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). Incretin hormones inhibits glucagon production from pancreatic alpha cells with resultant increase in insulin production from pancreatic beta cells [3-5]. On 2nd May 2011 US FDA approved linagliptin (BI-1356 on 2 May 2011 for treatment of Type-2 diabetes mellitus as a monotherapy or in combination with other medications (metformin etc.), in combination with exercise and dietary variation. It is being marketed by Boehringer Ingelheim and Lilly under the trade names are Tradjenta and Trajenta [6-11].
Reported literature survey shows that the drug can only be determined by reversed-phase liquid chromatography [12,13]. As per literature survey there is no stability indicating simultaneous RP- HPLC and UV method with degradation studies for linagliptin with simple solvent system (distilled water) [14-16]. Thus the aim of the present work was to develop and validate an accurate, simple, sensitive and precise spectrophotometric method (UV/Visible) for the determination of linagliptin in its pure form and pharmaceutical formulation.
Materials and Methods
Chemical and Reagents
All the chemicals used were of high analytical grade. All the solutions were freshly prepared in Methanol and 0.1N HCl (15:85, v/v). Authentic of linagliptin were obtained as gift samples from Mylan Laboratories Limited, Hyderabad
Methods
A sufficient amount of linagliptin sample was dissolved in 100 mL of distilled water and analyzed in the range 200 to 400 nm using UV spectrophotometer (Shimadzu model 1800 double beam) and the wavelength of maximum absorption, λmax was determined. Further drug was characterized by solubility, melting point, and Fourier Transform Infra-Red (FTIR) techniques. Drug was analyzed using UV-Visible method and was further validated for analytical parameters like linearity, precision, and accuracy as per guidelines laid down by International Conference on Harmonization (ICH).
Physical characterization of linagliptin
The physical characterization of procured drug sample of linagliptin was determined on the basis of following parameters.
a) Organoleptic properties: The pure sample of linagliptin was characterized for organoleptic properties such as nature, colour, taste and odour.
b) Solubility: Excess amount of linagliptin was dissolved in 10 ml distilled water and this saturated solution was stirred for 48hrs on magnetic stirrer at 100 rpm and 25 ± 1°C. Then sample was centrifuged for 10 min at 10,000 rpm using ultracentrifuge (Remi International, Mumbai, India). The clear supernant was collected using 0.22μm syringe filter and analyzed using UV spectrophotometer at 290 nm. The results were analysed and noted [17].
c) Melting point: Melting point of linagliptin was determined using capillary melting point apparatus, where a small quantity of drug was filled in a capillary tube open both the ends and it was placed along with thermometer in melting point apparatus.
d) Fourier Transform Infrared Spectroscopy (FTIR): FTIR spectrum was used as an analytical technique for identification of pure drug sample by KBr method using FTIR spectrometer (Agilent Technologies, USA). Peaks of individual pure drug were compared with reference FTIR peaks. Samples were previously prepared with KBr at 1:5 (sample: KBr, w/w). KBr disks were prepared by compressing the powders at a pressure of 5 tons for 3 min in a hydraulic press and scanned against a blank KBr disk at wave numbers ranging from 500 to 4000 cm-1 with a resolution of 1.0 cm-1 [18].
Analytical method for drug concentration measurements (UV/Visible method)
The ultraviolet spectrophotometric method was selected for the determination of linagliptin in the range of 200 to 400 nm and the λmax was determined.
Diluent Preparation
Methanol, Distilled water, and other reagents were of analytical grade.
Preparation of Linagliptin stock solution (20μg/ml) in 0.1N HCl
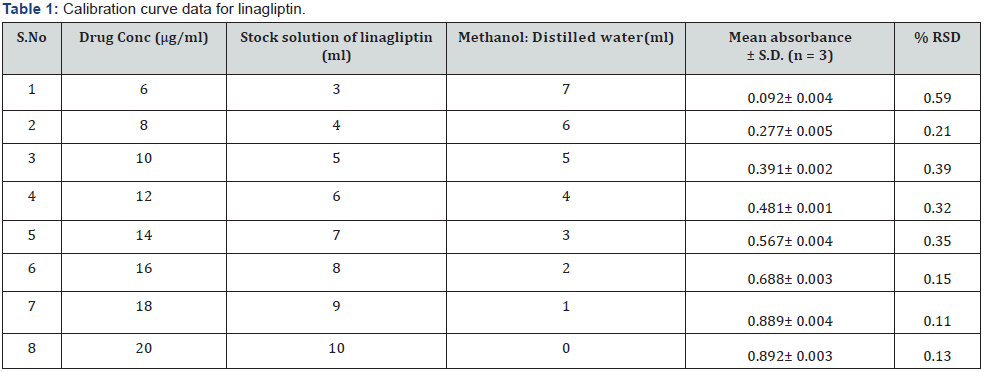
Linagliptin equivalent to 100mg was weighed and transferred into 100ml volumetric flask then 15ml of methanol was added and shaken well to dissolve it after that the volume was made up to 100ml with distilled water. From that 2ml of solution was withdrawn and taken in 100ml volumetric flask. The volume was adjusted with diluent up to up to 100ml with distilled water [19] as shown in (Table 1).
Selection of detection wavelength
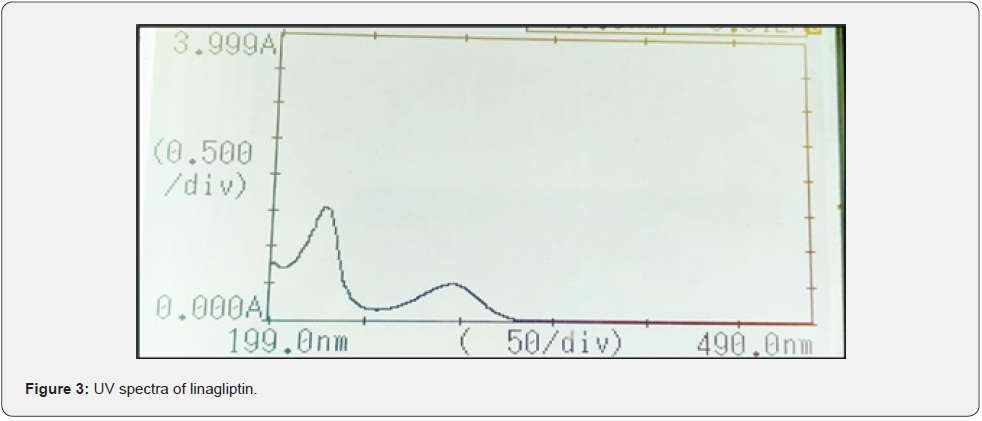
Drug solution was scanned over the range of 200- 400 nm. The wavelength of linagliptin was determined to be 290 nm.
Preparation of working standard
Prepare a series of dilute solution from the above stock solution according to (Table 2) in 10 ml volumetric flask. Measure the absorbance of the solutions at 290 nm using distilled water as a blank. Plot a graph by taking concentration (μg/ml) on X-axis and absorbance on Y-axis. This plot gives a straight line, and the linearity can be determined using y = mx + c formula. From the calibration curve calculate the Coefficient of determination r2 value, slope m and intercept c by using excel sheet.
Construction of calibration curve
Pipette out 1,2,3,4,5,6,7, and 8 ml of working solution and transfer into separate 10 ml volumetric flasks. Dilute all of them to 10 ml with water to get solution of concentrations to 2, 4, 6,8,10,12,14,16 μg/ml respectively.
UV Method Validation
The ultraviolet spectrophotometric method was validated for accuracy, precision, linearity, detection limit, quantitation limit and robustness [18-23].
Linearity
Appropriate aliquots of linagliptin working standard solutions were taken in various 10 ml volumetric flasks and diluted with distilled water up to the mark to obtain final concentrations of 6, 8, 10, 12, 14 μg/ml. Calibration curves were constructed by plotting absorbance versus concentrations and regression equations were calculated for linagliptin.
Range
The Range of the analytical method was decided from the interval between the upper and lower level of the calibration curve by plotting curve.
Precision
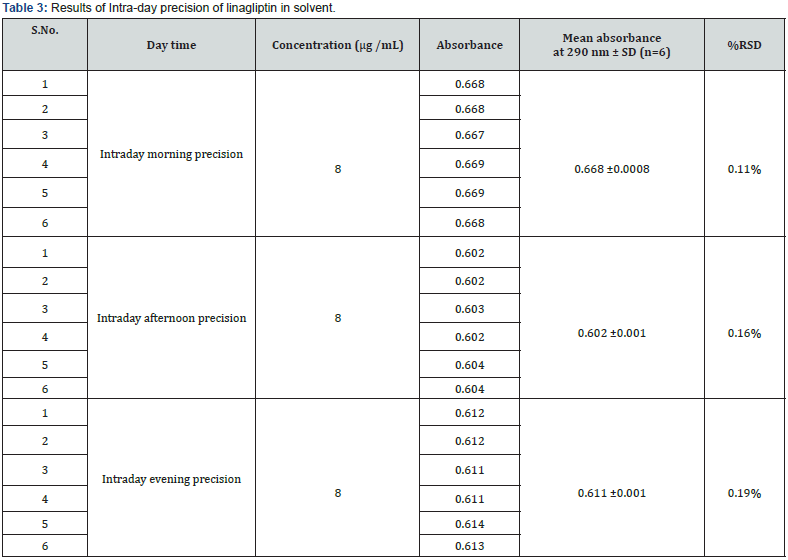
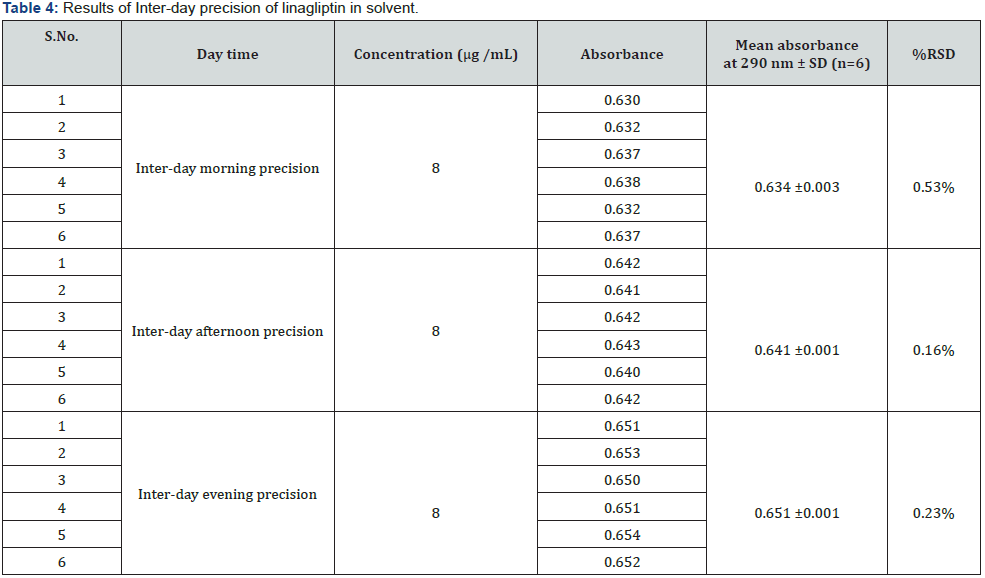
Intraday precision was determined by analyzing the drug at concentration (8μg/mL) and each concentration for three times, on the same day. Similarly, inter-day precision was determined daily, for two consecutive days. Repeatability (intraday) of the method was determined by analyzing six samples of the same concentrations of the drug (8μg/mL). The absorbance of each was measured and reported in terms of relative standard deviation (RSD) to detect the variation.
Accuracy
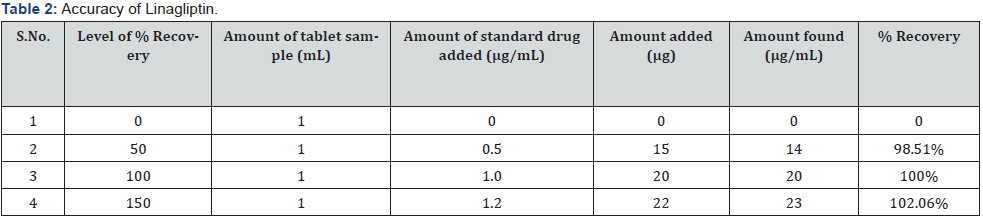
The accuracy of the method was determined by calculating recoveries of linagliptin by method of standard additions at three different levels 60, 100 and 140 %. Mean percentage recovery was determined. Recovery values were calculated and shown in (Table 1).
Detection Limit
The Detection Limit of an individual analytical procedure is the lowest amount of analyte in a sample, which can be detected but not necessarily quantitated as an exact value [24]. The detection limit (LOD) may be expressed as: LOD= 3.3σ/S
Where σ = Relative standard deviation of the response. S = the slope of the calibration curve (of the analyte).
Quantitation limit
The Quantitation limit of an analytical procedure is the lowest amount of analyte in a sample, which can be quantitatively determined with suitable precision and accuracy.
Quantitation Limit (LOQ) may be expressed as: LOQ = 10σ/S
Where, σ = Relative standard deviation of the response.
S = the slope of the calibration curve (of the analyte).
Results & Discussion
a) Organoleptic properties: Linagliptin was found to be white coloured, slightly hygroscopic, crystalline powder.
b) Solubility: The solubility of linagliptin in water was found to be 2.98 ± 0.68 mg/L corresponding to reference value of 3.4 mg/L at 25 °C.
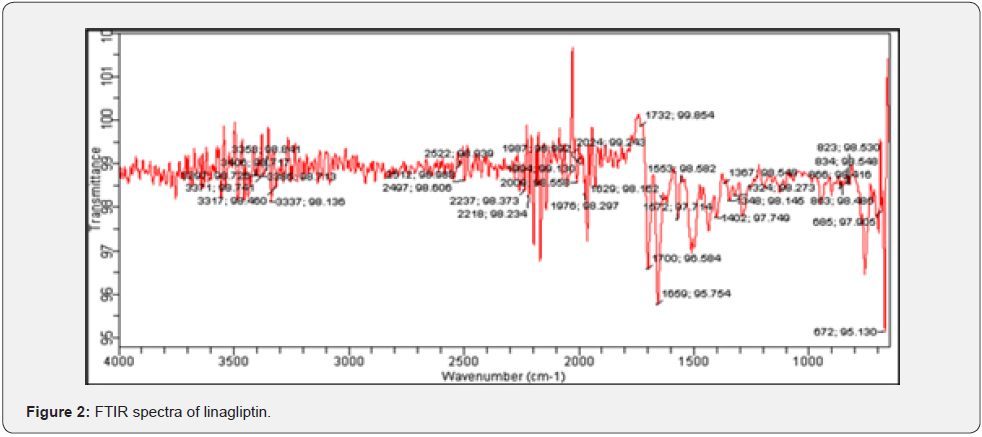
c) Fourier Transform Infrared Spectroscopy (FTIR): FTIR spectrum for identification of pure drug sample was done by FTIR spectrometer. The characteristic peaks of linagliptin at 3358 cm-1 (N-H stretching), 2237 cm-1(–C≡C–), 1348 cm-1 (C-N stretching), 1659 cm-1 (C=O stretching) as shown in (Figure 2) which corresponds to the literature peaks confirming the purity of drug sample.
d) Melting point: Capillary melting point apparatus determined the melting point. The observed melting point of linagliptin is 201 °C
Analytical method for drug concentration measurements (UV/VIS Method)
a) Selection of detection wavelength: The wavelength of linagliptin was determined to be 290 nm as shown in (Figure 3).
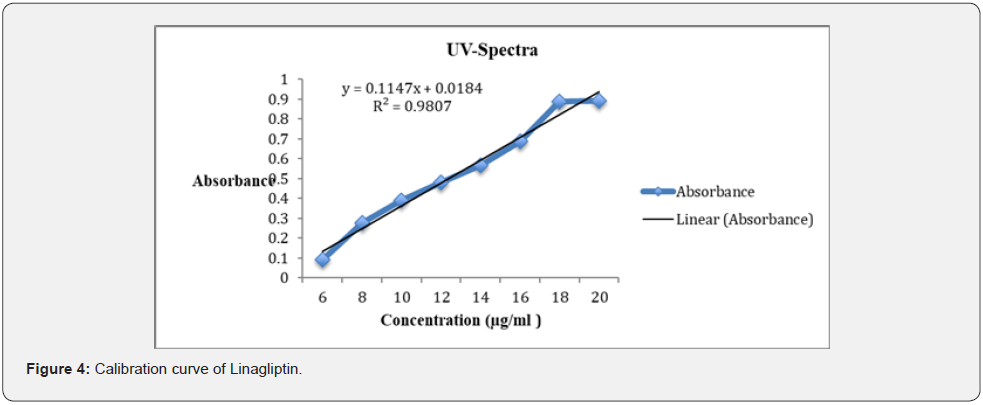
b) Preparation of standard plot for linagliptin: Absorbance of the resultant solution was measured at 290 nm using blank. A graph was plotted between the concentrations and their respective absorbance. The response of the drug was found linear in the entire investigational range of 6 to 16 μg/ml as shown in (Table 1). The calibration curve showed the linear equation as, y = 0.1147x + 0.0184, with a correlation coefficient, r2 = 0.98074, where y represents absorbance (optical density) and x represents the concentration (μg /ml) as shown in (Figure 4).
Method validation
As per ICH guidelines the developed UV-VIS spectroscopic method was validated for the following parameters
a) Linearity: The linearity for linagliptin was found to be linear in the range of 6-16 μg/ml. The regression equation was found to be y = 0.1147x + 0.0184, with a correlation coefficient, r2 = 0.98074.
b) Range: The observed range of linagliptin in test solution was observed from 0.092 ± 0.004 to 0.688 ± 0.003.
c) Accuracy: The accuracy of the analytical method for Linagliptin was determined at 60%, 100% and 150% levels of standard solution. Absorbance was measured at 290 nm and results were expressed in terms of % recoveries in (Table 2).
d) Precision: The Intra-day and Inter-day precision were carried out using similar optimized conditions. The precision (measurement of inter-day, intra-day repeatability) results showed good reproducibility with the relative standard deviation (% RSD) below 2.0 % as shown in (Tables 3 & 4) respectively. This indicated that the method was highly precise.
e) Limit of Detection (LOD) and Limit of Quantification (LOQ): LOD and LOQ of method were determined to be 5.03 μg /mL and 21.22 μg /mL respectively. LOD and LOQ indicate that method was accurate, fast, and highly sensitive.
Conclusion
The method was validated and showed high sensitivity, acceptable linearity, and accuracy as per ICH guidelines. In both inter-day and intra-day precision study, shows %RSD were not more than (NMT) 2.0% that indicated good repeatability and intermediate precision. Hence proposed method may be used for routine analysis of linagliptin in bulk and pharmaceutical dosage forms. The method for linagliptin estimation was found to be linear in the range of 6-16 μg/ml with regression equation; y = 0.1147x + 0.0184 and correlation coefficient, r2 = 0.98074. Accuracy of proposed method was confirmed by performing accuracy studies that showed the results within the range. Precision of proposed UV method was confirmed by performing intra-day and interday precision. Results were well within acceptance criteria that indicate excellent scope of the method for the determination of linagliptin in pharmaceutical dosage forms and bulk.
Acknowledgment
All authors have contributed significantly to the preparation of the manuscript and agree with the content of the manuscript and agree to submission to Global Journal of Nanomedicine. Authors are grateful to Mylan Laboratories Limited, Hyderabad for providing the gift sample. The authors express gratitude to School of Pharmaceutical Sciences, SGRR University, Dehradun.
Conflict of Interest
The authors declare that there is no conflict of interest that could be prescribed as prejudging the impartiality of the review. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Refrences
- Kirby M, Yu D, Conor S, Gorrell MD (2010) Inhibitor selectivity in the clinical application of dipeptidyl peptidase-4 inhibition. Clinical Sci 118(1): 31-41.
- Konari SN, Jacob JT, Prakash V (2017) Stability Indicating UV Spectrophotometric Method For Lina- gliptin and Metformin in Pharmaceutical Dosage Form. Pharm Methods 8(2): 121-126.
- Nirogi B, Kandikere V, Mudigonda K (2008) Sensitive liquid chromatography tandem mass spectrometry method for the quantification of sitagliptin, a DPP-4 inhibitor, in human plasma using liquid-liquid extraction. Biomed Chromatogr 22(2): 214-222.
- Salsali A, Pratley R (2007) Does addition of sitagliptin to metformin monotherapy improve glycemic control in patients with type 2 diabetes mellitus. Nat. Rev. Endocrin 3(6): 450-451.
- Mello S, Gregorio B, Fernando S, Carvalho L, Aguila MB, et al. (2010) Comparative effects of telmisartan, sitagliptin and metformin alone or in combination on obesity, insulin resistance, and liver and pancreas remodelling in C57BL/6 mice fed on a very high-fat diet. Clin Sci 119(6): 239-250.
- Ramzia I El Bagary, Ehab F Elkady, Bassam M Ayoub (2013) Spectrophotometric Methods for the Determination of Linagliptin in Binary Mixture with Metformin Hydrochloride and Simultaneous Determination of Linagliptin and Metformin Hydrochloride using High Performance Liquid Chromatography. Int J Biomed Sci 9(1): 41-47.
- Australian Drug Evaluation Committee Recommendations (2010) Australian Government Department of Health and Aging. .
- Amar Gangadhar Zalte, Ravindranath Bhanudas Saudagar, Pramod Nanasaheb Katkade (2016) Validated UV- Spectroscopic estimation of Linagliptin Concentration in Bulk and Dosage form. Research J. Pharm. and Tech 9(5): 490-492.
- Singh S, Sethi S, Khanna V, Benjamin B, Kant R (2011) A potent selective and slow-binding inhibitor of dipeptidyl peptidase-IV for the treatment of type 2 diabetes. Eur J Pharmaco 652: 157-163.
- Sherwyn L, Schwartz MD (2010) Treatment of Elderly Patients with type 2 diabetes mellitus: A Systematic Review of the Benefits and Risks of Dipeptidy l Peptidase-4 Inhibitors. Amer J Geria Pharmacothera, 8: 405-418.
- Hess C, Musshoff F, Madea B (2011) Simultaneous identi cation and validated quanti cation of 11 oral hypoglycaemic drugs in plasma by electrospray ionisation liquid chromatography–mass spectrometry . Anal Bioanal Chem, 400: 33-41.
- Lakshmi B, Reddy T (2012) A novel RP-HPLC method for the quantification of linagliptin in formulations. Journal of atoms and molecules 2: 155.
- El-bagary RI, Elkady EF, Ayoub B (2012) Liquid chromatographic determination of linagliptin in bulk, in plasma and in its pharmaceutical preparation. Int. J. Biomed. Sci 8(3): 209-214.
- Vemulla P, Dodda D, Ballekari U, Panga S, Veeresham C (2015) Simultaneous determination of linagliptin and metformin by reverse phase- high performance liquid chromatography method: An application in quantitative analysis of pharmaceutical dosage forms. J Adv Pharm Technol Res 6(1): 25-28.
- Bagary R, Elkady E, Ayoub B (2013) Spectrophotometric methods for the determination of linagliptin in binary mixture with metformin hydrochloride and simultaneous determination of lina- gliptin and metformin hydrochloride using high performance liquid chromatog- raphy. Int J Biomed Sci 9(1): 41-47.
- Varaprasad C, Asif M, Ramakrishna K (2015) RP-HPLC method for simultaneous estimation of metformin and linagliptin in tablet dosage form. Rasayan J Chem 8(4): 426-432
- Thadkala K, Nanam PK, Rambabu B, Sailu C, Aukunuru J (2014) Preparation and characterization of amorphous ezetimibe nanosuspensions intended for enhancement of oral bioavailability. International Journal of Pharmaceutical Investigation 4(3): 131-137.
- Sharma D, Bhavna (2019) Formulation and Evaluation of Polymeric Nanomicelles of Gliptin for Controlled Drug Delivery. Drug Delivery Letters 9(4): 1-11.
- Beckett AH, Stenlake JB (2004) Pharmaceutical Chemistry part 2. 4thed, CBS publisher and distributor. New Delhi. pp. 284-286.
- ICH Q1A (R2) (2003) Stability Testing of New Drug Substances and Products.
- Konari SN, Jacob JT (2015) Application of Analytical Validated High-Performance Thin-Layer Chromato- graphic Technique for the Multicomponent Analysis of Cardiovascular Drug Combos in Pharmaceutical Dosage Form. J P C 28(5): 354-361.
- ICH Q2 (R1) (2005) Validation of Analytical Procedures: Text and Methodology.
- ICH Harmonised Tripartite Guideline (1994) Text on Validation of Analytical Procedures, International Conference on Harmonization. Geneva. pp. 1-5.
- Aktar F, Kuddus MR, Hossen A, Hossain MK, Rashid MA (2012) Development and validation of a simple and rapid UV Spectrophotometric Method for assay of Nitazoxanide in Pharmaceutical Dosage Forms. Bangladesh Pharmaceutical Journal 15(1): 47-51.






























