Variations in the Phase Composition of Calcium-Phosphate Ce-Ramics by Addition of Sodium Ions
MV Tkachenko1, MS Barabashko2* and М Yu Vovsianiker1
1Physics of Solids Department, VN Karazin Kharkiv National University, Ukraine
2B Verkin Institute for Low Temperature Physics and Engineering of NAS of Ukraine, Ukraine
Submission:July 15, 2021; Published:August 31, 2021
*Corresponding author: MS Barabashko, B Verkin Institute for Low Temperature Physics and Engineering of NAS of Ukraine,47 Nauky Ave., Kharkiv, 61103, Ukraine
How to cite this article: MV Tkachenko, MS Barabashko, М Yu Vovsianiker. Variations in the Phase Composition of Calcium-Phosphate Ce-Ramics by Addition of Sodium Ions. Glob J Nano. 2021; 5(4): 555668. DOI: 10.19080/GJN.2021.05.555668
Abstract
Ceramics hydroxyapatite / β-tricalcium phosphate (HA/β-TCP) with a phase ratio 100/0, 60/40, 40/60 and 0/100, respectively, have been obtained by sintering at 1100°C. Four series of calcium-phosphate powders with the ratio of Ca/ P in the range from ~1.5 to 1.67 have been used. The amount of the Na additives in the HA/β-TCP powder was varied in the range from 1 to 1.5 wt.%. The resulting HA/β-TCP powder was calcinated at a temperature of 900°C for 1 h. It was shown in X-ray and IR studies that the composition of the powders significantly influence on the phase composition of the ceramics. Na additives lead for the increase of the content of the carbonated sodium-substituted hydroxyapatite phase (CHA) and decrease of the β-TCP phase. The β-TCP is stabilized, and the HA phase is transforming to CHA in the presence of sodium is calcinated powder.
Keywords: Synthesis; Hydroxyapatite; β-tricalcium phosphate; Ceramics; Na additives
Introduction
Ceramic materials based on calcium phosphates are widely used as bone replace materials [1,2]. Typically, biphasic calcium phosphates ceramics (BCP) that consisting of 60 wt.% hydroxyapatite (HA) and 40 wt.% β-tricalcium phosphate are used [3]. A dissolution rate in the biological environment of this biphasic mixture is comparable to the rate of growth of new bone tissue, due to the presence of β-TCP. Excellent osteoconductive is realized due to the content of HA [1]. X-ray amorphous apatite structure for the carbonized HA is similar to the structure of the mineral component of the bone tissue [4]. Two structurally different phases of components (the rhombohedral β-form of TCP and the hexagonal apatite HA phase) are usually present in the highly crystalline BPC. In this regard, the human body has to spend a lot of work both to reduce the crystallinity of the implant and to transform it into an appetite structure. Some microelements are incorporated into the structure of the synthetic apatite, and some elements are adsorbed by its surface. It leads for the additional differs between the biological apatite and synthetic. The presence of microelements in bioapatite contributes to giving it unique biological, physical, and chemical properties [5]. Ions such as Na+ and СO32- are presented in significant amounts in biological apatite [6]. Sodium and carbonate ions distributions in the HA lattice were studied in ref [7]. It was found that for the formation of Na-substituted carbonized HA (B-type), the replacement of calcium by sodium ions is accompanied by the formation of vacancies in both positions of M1 (nine-fold oxygen environment of calcium) and M2 (sevenfold oxy-gen environment of calcium).
PO43- are replacement by groups of carbonate ones. The formation of a Na-substituted carbonized HA with the mixed type of A- and B-sublattices is a result of the sintering of the synthesized sodium-substituted HA due to the co-substitution of СO32- carbonates n both sublattices [8,9]. There is little information in the literature about the interaction of the β-TCP with sodium additives. Two sodium ions can replace a calcium ion and a vacancy in the Ca (4) positions of the β-TCP lattice [10]. At the same time, no structural changes occur with β-TCP. β-TCP is converted to Na substituted carbonated HA in the melted Na-containing flux [11]. The sodium additives led to the formation of an appetite structure in this case. At the same time, no significant change in the phase behavior was found for the incorporation of sodium into the apatite structure. It was shown that a two-phase mixture with change of the ratio of sodium-substituted HA and β-TCP is formed at high temperatures with varies of calcium deficiency [12]. In the ref. [13], it was discussed that the presence of sodium in the two-phase mixture stabilizes β-TCP that is contradicted with the result on the phase transformation of Na-doped β-TCP [11].
It was shown in refs. [8,14,15] that a single-phase sodiumsubstituted carbonized ceramic is formed from the initial calciumphosphate powder with introduced sodium ions during sintering. The content of carbonate groups in ceramics correlates with the number of additives of Na+ ions. The step on the temperature dependence of linear shrinkage at a temperature near 700°C is associated with the replacement of calcium ions by sodium ions in the apatite structure and the formation of carbonated hydroxyapatite as a result of the substitution of carbonate СО32‒ ions for phosphate (РО43‒) and hydroxyl (ОН‒) positions. Such ceramics have a higher dissolution rate than pure hydroxyapatite ceramics in the point of view of biomedical application. It is seeming possible to achieve equalization of dissolution rate with the growth rate of new bone by the varying of the concentration of sodium ions. It is usually achieved for a two-phase mixture of HA/ β-TCP by adjusting the ratio between components. The aim of this work was to determine the conditions of stabilizing the apatite and β-TCP phase at presence of additives of sodium additives in sintered ceramics with a Ca/P ratio from 1.5 to 1.67.
Materials and Methods
A synthesis reaction between calcium carbonate (Merck, Darmstadt, Germany, analytical grade) and orthophosphoric acid (Merck, Darmstadt, Germany, analytical purity) with a Ca/P ratio = 1.67 was used to obtain the initial powders. Calcium nonstoichiometric was created by using an extra amount of phosphoric acid [14]. As a result, four series of powder samples (HA, β-TCP and two-phase HA/β-TCP with the ratio 60/40 and 40/60, respectively) were obtained. Ceramics were sintered from these powders by annealing at temperatures above 900°C. Some of the powders were calcinated at 900°C for an hour. Thus, two series of samples were formed. The so-called “green” nonstoichiometric calcium-phosphate powder was formed without annealing. A single-phase or two-phase composition was formed in this powder, in which, as a result of calcination. Sodium additives were added to both types of powders from sodium bicarbonate solution. Concentration 1 and 1.5 wt. % of Na+ ions in the powders was used in this study. Compacts were prepared under a pressure of 120 MPa from the powders and then were annealed in air at 1100°C for 1 hour.
X-ray studies were carried out on DRON-2 diffractometer in copper Kα radiation (λ = 0.154 nm) with a nickel filter in the interval of diffraction angles 2θ from 20 ÷ 55, with a scan step 0.5 deg/min. IR spectra were recorded using a BIO-RAD FFS 175 spectrometers (Germany) with the KBr pellet method with a resolution of 0.05 cm-1 and averaging over 200 scans. Dynamic dilatometric measurements were performed in the temperature range from room temperature up to 1200 °C. The heating rate of the samples was 4 K/min.
Results
It was shown by X-ray studies (Figure 1a) that obtained ceramics with additives of sodium ions in HA does not have structural changes from initial powder of HA. In ceramic samples without additives, a formed texture is manifested by the redistribution of the intensities for (112) and (300) lines. Intensity of these lines is slightly enhanced for ceramics with the additives of Na+ ions. Significant changes of the phase composition occur in the case of two-phase HA/β-TCP and almost single-phase β-TCP (with HA impurity less than 5 wt.%). A two-phase mixture of HA/ β-TCP with a phase ratio 60/40 is formed at sintering from the powder without sodium additives (Figure 1b).
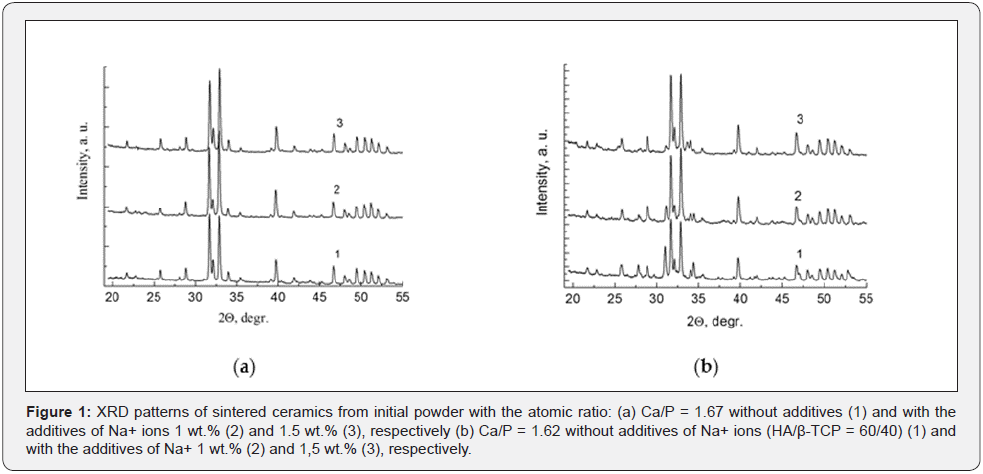
The intensity of the HA lines increases, and the intensity of β-TCP lines decreases for the case of mixture with added sodium. A similar situation is observed in the case of sintering the β-TCP (Figure 2a). Thus, the sodium additives in the powders with an atomic ratio of Ca/P ˂ 1.67 lead for the formation of β-TCP ceramics or two-phase calcium-phosphate ceramics with an increase of the concentration of apatite phase during sintering. A carbonized hydroxyapatite is a characteristic feature of this resulting apatite phase.
It can be seen from the (Figure 2b) that for ceramics from initial powder with Ca/P = 1.52 and without sodium additives is observed the typical IR spectrum for β-TCP. The phase composition of the sintered ceramics with added sodium in an amount of 1 - 1.5 wt.% is changes. A carbonized apatite phase appears. The degree of carbonization increases with the increasing of sodium content. The IR spectra in the range of wavenumbers 1400 - 1600 cm-1 and 870 - 880 cm-1 show appear and corresponding enhancement of the transmission bands of carbonate ions.
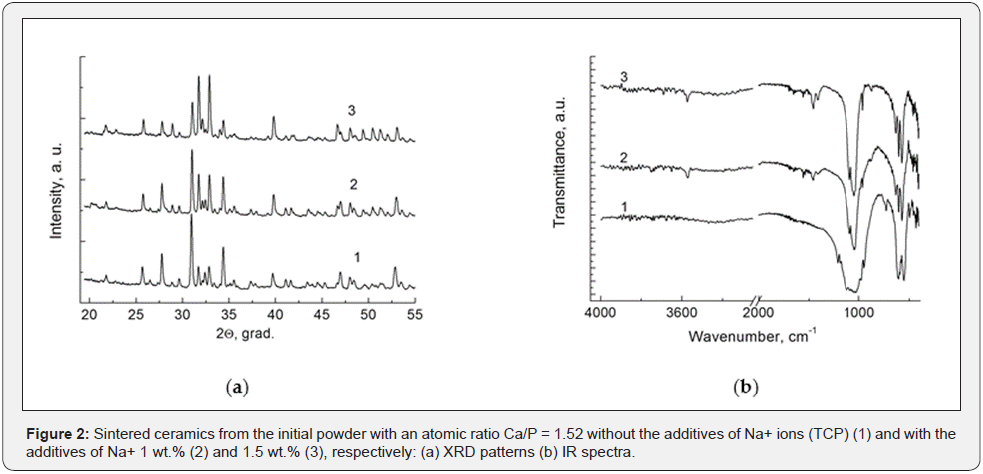
This occurs in the case when single-phase HA is sintered from the initial powder (Ca/P = 1.67) and when carbonated sodiumsubstituted HA (CHA) is sintered. Carbonated apatite predominates in the final ceramic that sintered from the initial powder of HA/β- TCP with the ratios 40/60 and 60/40, respectively, Ca/P in the range 1.5 - 1.67 and sodium concentration 1 - 1.5 wt.%. β-TCP is only a small additive.
The process of replacing calcium by sodium and the formation of sodium-substituted HA is reflected in the temperature dependence of the linear shrinkage of compacts with different initial Ca/P ratio. The linear shrinkage curves with the formation of a purely apa-tite phase in the region of 700°C show a step associated with reaction sintering [15,16], which can be an indicator of the entry of sodium ions into the apatite lattice (Figure 3).
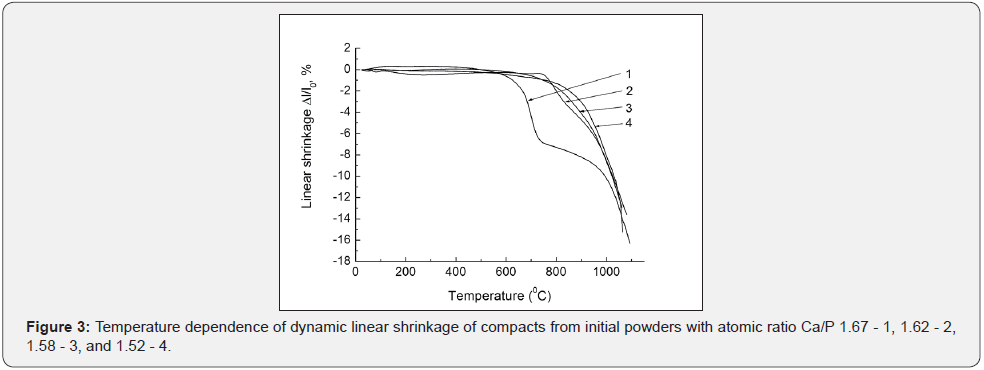
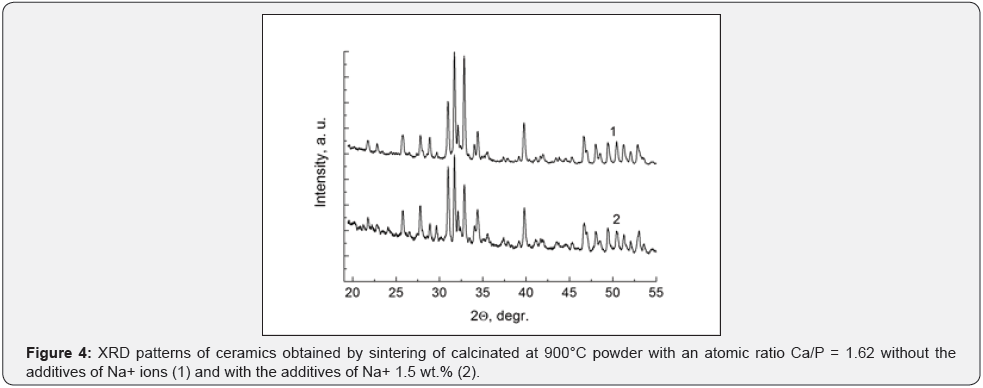
Another picture is observed when the initial calciumphosphate powder with sodium additives previously calcinated at 900°C. The final phase composition has already formed at this temperature, in this case in the powder. The additives of sodium ions to the calcinated HA powder does not effect on the crystal structure and phase composition of the final ceramic like in the case of the initial powder without sodium additives. Weak qualitative differences are also observed in the structure of twophase mixtures, in comparison with the use of uncalcined initial powder (Figure 4). The stabilizing of the β-TCP phase is observed, that is some contradiction. A small relative increase of the β-TCP amount can be due to the partial decomposition of HA during sintering.
The significant effect of calcination on the shrinkage processes of pure and sodium doped compacts was studied for all powder compositions. This is the most clearly reflected for the HA sintering compacts. Curve (1) in (Figure 5) refers to the original powder, and curve (2) - to the calcinated one. The inset in (Figure 5) shows the temperature dependences of the shrinkage rate for both samples. The characteristic step is present in both cases on the curves and reflects the formation of sodium-substituted CHA.
For the initial powder, this process occurs in a rather narrow temperature range and more intensively (the shrinkage rate is much higher). In the case of calcinated powder, this occurs at higher temperatures and less intensively. This is not surprising. The powder is finely dispersed, which pro-motes diffusion processes in the first case. More time and higher temperatures are re-quired to replace calcium with sodium in the second case of the powder that coarse-crystalline.
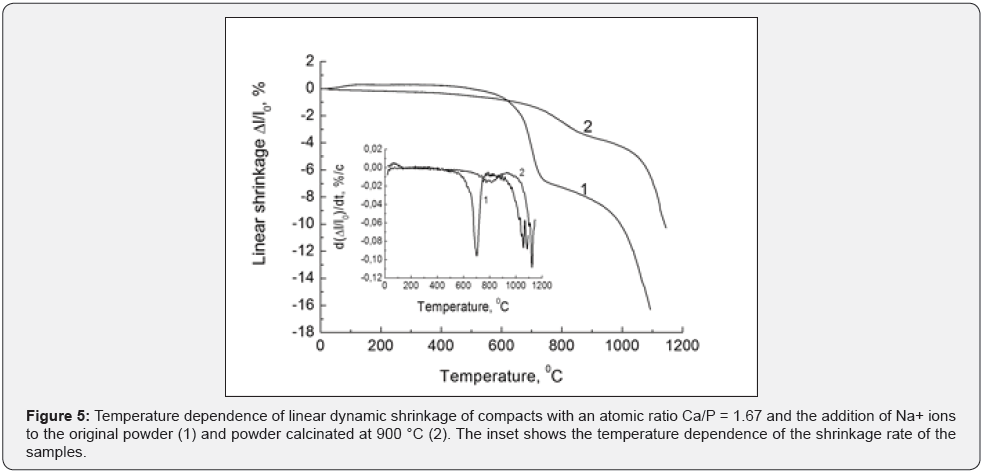
As a result, the additives of sodium in calcinated powder have very little effect on the formation of the final phase composition of two-phase ceramics during sintering. It is determined by the structure of the initial powder. Calcium is replaced by sodium in the cat-ionic sublattice during sintering. In addition, the ОН‒ and РО43‒ ions are replaced by carbonate groups in the anionic sublattice, which leads to the transformation of HA into sodiumsubstituted CHA.
Discussion
In the case of single-phase HA ceramics, the initial powder is always non-stoichiometric due to the presence of HPO42‒ ions in it at the applied method. The re-quired stoichiometry is achieved only when it is annealed at temperatures above 750°C. Two-phase samples and β-TCP are not synthesized directly by the “wet method”. The obtained calcium-deficient hydroxyapatite Ca9(HPO4) (PO4)5(OH) have different Ca/P ratios. Calciumdeficient HA decomposes into β-TCP and stoichiometric HA depending on the atomic ratio Ca/P, at temperatures above 750°C [14,17].
The additives of sodium to leads to the formation of two types of clusters during annealing, from which two crystallization centers are formed: HA and β-TCP. The corresponding phases are formed on their basis. The increase of the sodium concentration leads for the improving of the stoichiometry of HA. Sodium is incorporated into the apatite lattice together with the carbonate ion, and as a result, sodium-substituted carbonized HA is formed for procuring of the electroneutrality of the entire compound. The amount of the β-TCP phase decreases in this case.
New phases already exist in the case of pre-calcinated powder, and the additives of sodium does not play any role in the subsequent nucleation. The insignificant increase of the β-TCP amount during sintering of the compact may be due to the partial dehydration of the apatite phase.
Conclusion
The effect of influence of sodium additives is studied on two types of calcium-phosphate powders, from which HA and β-TCP ceramics are synthesized during sintering, as well as two-phase mixtures with ratio of 60/40 and 40/60 of HA/β-TCP. The initial powders were calcinated by annealing at 900 °C for 1 hour. It was shown that the phase composition of the ceramics can be effectively varies by change of concentration of additives of sodium ions, as well as the Ca/P atomic ratio of the initial powder that gives the HA/β-TCP mixture in sintered ceramics. The phase ratio in ceramics strongly depends on the degree of crystallinity of the powder in this case and it is differing for the as pre-pared and calcinated powders. Calcium-phosphate ceramics with ratio 60/40 of HA/β-TCP is easily transformed into sodium-substituted CHA in the case of concentration in the initial powder of sodium ions is slightly more than 1.5 wt.%. It was found that the calcinated powder is effective to use as the initial one for the case of preserve the initial ratio of HA/β-TCP and replace the apatite phase by sodium-substituted CHA.
Acknowledgment
The authors thank to Professor ZZ Zyman (V.N.Karazin Kharkiv National University, Ukraine) for the useful discussion, as well as Professor M. Epple (Duisburg-Essen University, Germany) and Dr. O. Prymak (Duisburg-Essen University, Germany) for the opportunity to carry out verification measurement.
Refrences
- SV Dorozhkin (2011) Application of Calcium Orthophosphate Bioceramics. BIO 1: 1-51.
- E Champion (2013) Sintering of calcium phosphate bioceramics. Acta Biomaterialia 9(4): 5855-5875.
- RZ Legeros, S Lin, R Rohanizaden, D Mijares, JP Legeros (2003) Biphasic Calcium Phosphate Bioceramics: Preparation, Properties and Applications. J. Mat. Sci.: Mat. Med 14: 201-209.
- JM Bouler, P Pilet, O Gauthier, E Verron (2017) Biphasic calcium phosphate ceramics for bone reconstruction: A review of bio-logical response. Acta Biomaterialia 53: 1-12.
- M Dermience, G Lognay, FO Mathieu, P Goyens (2015) Effects of thirty elements on bone metabolism. J. Trace Elem. Med. Biol 32: 86-106.
- Sergey V Dorozhkin (2007) Calcium orthophosphates. J. Mat. Sci 42: 1061-1095.
- Hafed El Feki, Jean Michel Savariault, Abdelhamid Ben Salah, Mohamed Jemal (2002) Sodium and carbonate distribution in sub-stituted calcium hydroxyapatite, Sol. Stat. Sci 2(5): 577-586.
- Zoltan Z Zyman, Mykola V Tkachenko (2013) Sodium-carbonate co-substituted hydroxyapatite ceramics. Processing and Application of Ceramics 7(4), 153-157.
- MV Tkachenko, AS Kamzin (2015) Formation of a Sodium Bicarbonate Cluster in the Structure of Sodium Substituted Hydroxyapatite. Physics of the Solid State 57(2): 407-411.
- K Yoshida, H Hyuga, N Kondo, H Kita, M Sasaki, et al. (2006) Substitution Model of Monovalent (Li, Na, and K), Divalent (Mg), and Trivalent (Al) Metal Ions for β-Tricalcium Phosphate. J. Am. Ceram. Soc 89(2): 688-690.
- G Buvaneswari, K Valsalan (2014) Single step conversion of β-TCP to Na-modified hydroxyapatite and its application in green chemical synthesis of α, β-unsaturated compounds. Materials Letters 134(1): 252-255.
- S Kannan, JMG Ventura, AF Lemos, A Barba, JMF Ferreira (2008) Effect of sodium addition on the preparation of hydroxyapatites and biphasic ceramics. Ceramics International 34(1): 7-13.
- S Kannan, F Goetz Neunhoeffer, J Neubauer, JMF Ferreiraw (2008) Ionic Substitutions in Biphasic Hydroxyapatite and b-Tricalcium Phosphate Mixtures: Structural Analysis by Rietveld Refinement. J. Am. Ceram. Soc 91(1): 1-12.
- ZZ Zyman, MV Tkachenko, DV Polevodin (2008) Preparation and characterization of biphasic calcium phosphate ceramics of desired composition. J. Mater. Sci.: Mater. Med 19: 2819-2825.
- MV Tkachenko, AS Kamzin (2015) Formation of a Sodium Bicarbonate Cluster in the Structure of Sodium_Substituted Hydroxyapatite. Physics of the Solid State 57(2): 407–411.
- NK Orlov, AK Kiseleva, PA Milkin, PV Evdokimov, VI Putlyaev (2020) Reaction Sintering of Bioceramic Based on Substituted Calcium Phosphates CaMPO4 (M = K, Na). Inorganic Materials: Applied Research 11(2): 394-402.
- A Massit, A El Yacoubi, A Kholtei, BC El Idrissi (2021) XRD and FTIR Analysis of Magnesium Substituted Tricalcium Calcium Phosphate Using a Wet Precipitation Method. Biointerface Research in Applied Chemistry 11(1): 8034-8042.






























