Zinc Oxide Nanoparticles Ameliorate Oxidative Stress Induced by Dimethyl Nitrosamine in Rat Kidney
Varsha Rani, Yeshvandra Verma, Kavita Rana and SVS Rana*
Department of Toxicology, Chaudhary Charan Singh University, India
Submission:February 14, 2020; Published:February 28, 2020
*Corresponding author:SVS Rana, Department of Toxicology, Chaudhary Charan Singh University, India
How to cite this article:Varsha Rani, Yeshvandra Verma, Kavita Rana, SVS Rana. Zinc Oxide Nanoparticles Ameliorate Oxidative Stress Induced by Dimethyl Nitrosamine in Rat Kidney. Glob J Nano. 2020; 5(2): 555656. DOI: 10.19080/GJN.2020.05.555656
Abstract
Dimethyl nitrosamine (DMN) is an established carcinogen. It is toxic to several organs viz. liver, kidney, lungs and immune system. Several drugs have been used in the past to modulate its toxicity using experimental models. Present study was designed to investigate the effect of zinc oxide nanoparticles (ZnONPs) on renal toxicity caused by DMN in laboratory rat. Since oxidative mechanisms mainly contribute to its toxicity, the proposed study focuses on amelioration of oxidative stress manifested by ZnONPs, if any. Present results show that administration of ZnONPs (50 mg/kg) to DMN (2μl/100g body weight) treated rats diminuted the concentration of malonaldehyde, H2O2 and NO in the kidney. However, GSH concentration increased after ZnONPs treatment. Results on glutathione S-transferase, glutathione peroxidase suggested an antioxidative effect of ZnONPs. These results find support from diminished oxidative DNA damage manifested by ZnONPs in DMN treated rats. Histopathological results on kidney also indicate a protective effect of ZnONPs on renal toxicity of DMN. A comparison of results between control and ZnONPs treated rats suggests that ZnONPs might be toxic to renal tissue. Nevertheless, the study concludes that ZnONPs possess a strong therapeutic/antioxidative potential against DMN induced toxicity.
Keywords: Dimethyl Nitrosamine; Kidney; Zinc Oxide Nanoparticles; Oxidative Stress; Histopathology
Abbreviations: DMN: Dimethyl Nitrosamine; ZnONPs: Zinc Oxide Nanoparticles; NEDA: N-(1-Naphthyl) Ethylenediamine Dihydrochloride; Ip: Intraperitoneally; Zn-MT: Zinc Metallothionein; H2O2: Hydrogen Peroxide; NO: Nitric Oxide; MDA: Malondialdehyde; GSH: Reduced Glutathione; ROS: Reactive Oxygen Species; CDNB: 1-Choloro-2’, 4’ Dinitrobenzene; 8-OHdG: 8-Hydroxy-2’ -Deoxy Guanosine; AD: Adenocarcinoma; CO: Cortex; ND: Nuclear Degeneration; BR: Brush Border; ED: Epithelial Damage/Degeneration; GL: Glomerulus; MA: Mitotic Activity; NPL: Neoplasm; NPR: Nuclear Proliferation; BC: Binucleated Cells; EP: Epithelial Lining; PCT: Proximal Convoluted Tubule; GL: Glomeruli; TEM: Transmission Electron Microscope; SEM: Scanning Electron Microscope; XRD: X-Ray Diffraction; JSPDS: Joint Committee On Powder Diffraction Standards; EDAX: Energy Dispersive X- Ray
Introduction
Dimethyl nitrosamine (DMN) is an established carcinogen IARC [1], ATSDR [2]. It has been reported that preferential site of biotransformation of nitroso compounds is liver, but kidney, lungs and other organs may also participate in its degradation albeit to minor degree Lee et al. [3], Knecht [4]. Magee, Barnes for the first time showed that a single dose of DMN could induce renal tumors. Later studies made to decipher the etiology of renal cancer induced by DMN attributed it to the generation of reactive oxygen species (ROS) and resulting oxidative stress Bansal et al. [5], Mittal et al. [6]. Subsequently, efforts were made to protect DMN toxicity in suitable experimental models using several drugs and antioxidants. Hamza et al. [7] showed therapeutic effects of α-lipoic acid (ALA) against DMN induced renal toxicity in rats. Rana, Kumar [8]. demonstrated that cadmium and zinc induced metallothionein inhibits lipid peroxidation (LPO) in the kidney of DMN treated rats. Metallothionein is involved in the reduction of hydroxyl radicals (OH) and sequestration of ROS produced under stress conditions Ruz, Carrasco [9], Chasapis, Loutsidou [10]. Furthermore, zinc plays an important role as a transcription factor and antioxidant defense system in prevention of DMN toxicity. Zinc channels create a balance between cell survival and cell death via controlling the free and intracellular zinc movements Dhawan, Chadha [11]. Therefore, zinc has been considered as a suitable agent to prevent toxicity of several xenobiotics i.e. carbon tetrachloride Rana & Tayal [12], Saldeen [13] ethyl alcohol Yunice, Lindeman [14] and DDT Feaster et al. [15].
Recent advent of nanomedicine employs nanotechnology in the treatment and diagnosis of several diseases. In this context, several nanoparticles have been synthesized and tested for toxicity and health aspects Oberdorster et al. [16], Hulla et al. [17]. Zinc oxide nanoparticles (ZnONPs), amongst them, have potent therapeutic potential since they express bioavailability, biocompatibility and high solubility. They possess the capacity to regulate cell cycle and cellular homeostasis Vizirianakis. Recently, Food and Drug Administration (FDA) has approved zinc oxide nanoparticles for anticancer therapy Shen et al. [18]. They can produce selective toxicity towards cancer cells causing disequilibrium of zinc dependent protein activity Vinderall, Mitjans [19], Rasmussan et al. [20] hypothesized that ZnONPs can kill cancer cells through the induction of oxidative stress in the cancerous cells. Thus, ZnONPs emerged as nanotheranostic platform against several diseases specially those caused by oxidative stress. Nonetheless, several laboratories have published reports dealing with toxic effects of ZnONPs in specific organs and cell lines Li et al. [21], Fazilah [22], Soheili et al. [23], Esmaeillou et al. [23]. Therefore, there appears to be sufficient reason to examine further the antioxidative potential of ZnONPs manifested in a suitable experimental model and conditions. With this perspective, a study on the protective effects of ZnONPs against DMN induced hepatotoxicity in rat was recently made in our laboratory Rani et al. [24]. Present study was made with an objective to assess the protective effects of ZnONPs, if any, on DMN induced renal toxicity in rat. Further, Renal toxicity of ZnONPs, if any, has also been simultaneously assessed.
Materials and Methods
Chemicals and Reagents
Zinc oxide nanoparticles were procured from Sigma Chemical Co. Missouri (USA). According to the manufacturer, nanoparticles contained approximately 80% zinc basis, 100% purity and < 100 nm size with a surface area 15-252/g. Dimethyl nitrosamine, thiobarbituric acid, 5’-5’-dithiobis-2-nitrobenzoic acid, 1-2’-4’ dinitrobenzene, glutathione reductase, glutathione and N-(1- Naphthyl) ethylene- diamine dihydrochloride (NEDA) were also purchased from Sigma Chemical Co. (USA). All other reagents of highest purity were obtained from High Media (Mumbai) through local suppliers.
Characterization of Zinc Oxide Nanoparticles
ZnONPs were characterized using a battery of methods as described earlier Rani et al. [24]. Briefly, the size and shape of ZnONPs were observed through transmission electron microscope at Sophisticated Analytical Instrument Centre, Punjab University, Chandigarh (India). Scanning electron microscopic observations and energy dispersive X-ray analysis (EDAX) were made at Department of Physics, Choudhary Charan Singh University, Meerut (India). Analysis of size, distribution, zeta potential and XRD analysis of ZnONPs were performed at Indian Institute of Technology, Roorkee (India).
Maintenance of Animals and Experimental Protocol
Prior approval of Institutional Animal Ethical Committee was sought to make present investigations. Experiments were conducted on male Wistar rats (150±25 g), procured from the animal facility of Jamia Hamdard, Delhi. They were maintained under standard laboratory conditions (room temperature 25±5⁰C), relative humidity (50±10%) and a 12 h dark/light cycle. Each rat was individually housed in polypropylene cage, offered food pellets (Golden Feeds, Delhi) and tap water ad libitum. After acclimatization to laboratory conditions for two weeks, rats were separated into four groups, each containing five rats. Rats of group A were injected DMN (2μl/100g body weight) in saline intraperitoneally (ip) on each alternate day for 15 days as described earlier Rani et al. [24]. Rats of group B were treated as the rats of group A and subsequently administered a pre-determined NOEL of ZnONPs (50 mg/kg) on each alternate day for 30 days. Rats of group C were treated with ZnONPs only as the rats of group B. Rats of group D were injected (ip) saline (2μl/100g body weight) only on each alternate day for 45 days and treated as controls. After 45 days, rats were starved overnight, and their urine samples were collected next morning through metabolic cages. Thereafter, they were sacrificed after euthanasia. Kidneys were carefully removed and processed for the estimation of reactive species viz. malondialdehyde, nitric oxide and hydrogen peroxide. Oxidative stress was determined through standard parameters viz. reduced glutathione (GSH), glutathione S- transferase and glutathione peroxidase estimations as described below.
Kidney Function Test
Creatinine in the urine samples was determined following the method of Toro, Ackerman [25] using a commercial kit procured from M/S Span Diagnostics, Surat (Gujarat, India).
Determination of Oxidative Stress
Malondialdehyde (Mda): MDA in the kidney samples was determined using thiobarbituric acid following the method of Jordan, Schenkman [26]. Absorbance was recorded at 532 nm using a spectrophotometer (Systronics, India). 1, 1, 3’, 3’, tetramethoxypropane (Sigma) was used as the standard. Protein contents in the kidney samples were determined following the method of Lowry et al. [27]. Bovine serum albumin (Sigma) was used as the standard.
Hydrogen Peroxide (H2O2): H2O2 in the kidney homogenates prepared in 0.25 M sucrose was measured employing Ferrithiocyanate method as described by Thurman et al. [28]. Absorbance was recorded at 480nm using a spectrophotometer (Systronics, India).
Nitric Oxide (NO): NO in the kidney samples was estimated through Greiss reagent following the method suggested by Cortas, Wakid [29]. Absorbance was recorded at 550 nm using a spectrophotometer (Systronics, India).
Reduced Lutathione (GSH): Ellman’s reagent was used to determine reduced glutathione in kidney samples Ellman [30]. Absorbance was recorded at 412nm using a spectrophotometer (Systronics, India).
Glutathione S-transferase: Glutathione S-transferase was assayed using 1-choloro-2’, 4’ dinitrobenzene (CDNB) that conjugated with glutathione. Absorbance was recorded at 340nm Habig et al.
Glutathione Peroxidase: The enzyme was assayed following the method of Paglia, Valentine [31]. Glutathione di sulphide (GSSG) produced as a result of glutathione peroxidase is reduced by an excess of glutathione reductase. Conversion of GSSG to GSH was monitored at 340nm using a spectrophotometer (Systronics, India).
Metallothionein: Metallothionein in kidney samples was analyzed following the cadmium saturation method Onaska et al. [32]. using atomic absorption spectrophotometer (EC, Hyderabad, India).
8-Hydroxy-2’-Deoxy Guanosine (8-OHdG)
Urine sample of each rat was collected in a sterilized vial through metabolic cage. These samples were stored at -80⁰C till further analyses. Competitive ELISA technique was used for the estimation of 8-OHdG (biomarker of oxidative DNA damage) using a commercial kit procured from Bioassay Technology Laboratory (China). Absorbance was recorded at 450nm using a microplate reader (EC, Hyderabad, India).
Histopathology
Small pieces of kidney were fixed in 10% neutral formaldehyde, dehydrated and embedded in paraffin. 5 μm thick sections were stained with hematoxylin and eosin and examined under a research microscope (Nikon, Japan).
Statistical Analysis
Students’ “t” test was employed to make intergroup comparisons amongst different groups. Differences amongst groups with p value <0.05 were considered significant. SPSS software version 2.0 was used for Inter group comparisons.
Results
Characterization of ZnONPs
Shape, size, structure and electric composition of ZnONPs was determined applying standard methods. Results show that average diameter of ZnONPs was <100nm (Figure 1). SEM observations showed that the nanoparticles formed agglomerates (Figure 2). Electrical composition of the ZnONPs was determined as zeta potential (Figure 3). through EDAX. The XRD pattern of ZnONPs showed a hexagonal structure when compared with the standard data (JSPDS: 00-001-1136) published elsewhere Rani et al. [24]. Intensity-weighed particle size distribution of ZnONPs has been exhibited by (Figure 4).


Renal Function
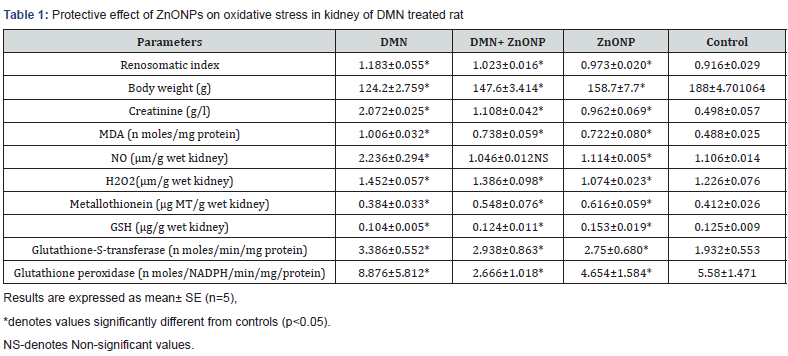
Higher concentration of creatinine indicated renal impairment in DMN treated rats. Subsequent treatments of DMN treated rats with ZnONPs improved renal function. ZnONPs treated rats also showed higher values for creatinine than control rats (Table 1).
Lipid Peroxidation
Lipid peroxidation is considered to be an important mechanism involved in DMN toxicity. Malondialdehyde, the product of LPO increased in the kidney of DMN treated rats. Administration of ZnONPs to DMN treated rats diminuted malondialdehyde concentration in renal tissue. However, its concentration was higher in the kidney of ZnONPs treated rats than control rats (Table 1). Higher values for NO in the renal tissue of DMN treated rats supported the results on malondialdehyde. ZnONPs therapy offered to DMN treated rats decreased NO concentration in the kidney. A comparison of NO values obtained in the kidney of ZnONPs and control rats showed higher values though insignificant in the kidney of ZnONPs treated rats (Table 1). Results on hydrogen peroxide also showed higher values in the kidney of DMN treated rats. Average values of H2O2 in the kidney of DMN+ ZnONPs treated rats were lower than DMN treated rats (Table 1). Taken together, all these results suggest an antiperoxidative and anti nitrosative role of ZnONPs.
GSH
In the kidney of DMN treated rats, a significant decline in GSH values was observed. GSH status improved after administration of ZnONPs to DMN treated rats. These observations also show that ZnONPs offer antioxidative protection against DMN induced renal toxicity (Table 1).
Glutathione S-transferase and Glutathione Peroxidase
Results on GSH were supported by the observations on glutathione-S-transferase. Enzyme activity decreased in the kidney of DMN treated rats. Supplementation of ZnONPs to DMN treated rats restored its activity near to control values (Table 1). Glutathione peroxidase activity also decreased in the kidney of DMN treated rats. However, it increased in the kidney of DMN and ZnONPs treated rats (Table 1).
Oxidative DNA Damage
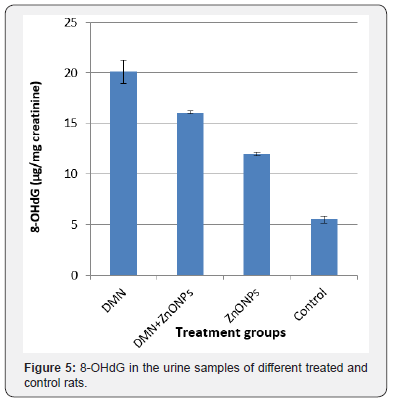
Present results showed greater oxidative DNA damage in the kidney of DMN treated rats as measured by 8-OHdG. ZnONPs supplementation to DMN treated rats inhibited this damage (79.97 % inhibition) to some extent. However, treatment with ZnONPs alone also induced DNA damage in kidney of rats (Figure 5).
Metallothionein
Results on renal metallothionein (MT) suggest that concentration of MT decreased in the kidney of DMN treated rats. However, 63.50 % increase in MT was recorded in the kidney of DMN and ZnONPs treated rats. ZnONPs, alone were also found to be a potent inducer of MT in rat kidney (Table 1).
Histopathology
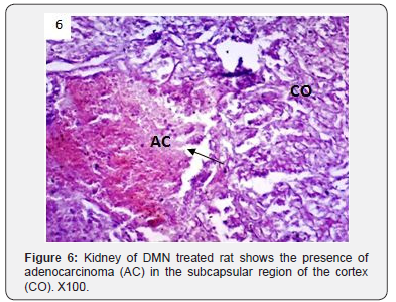
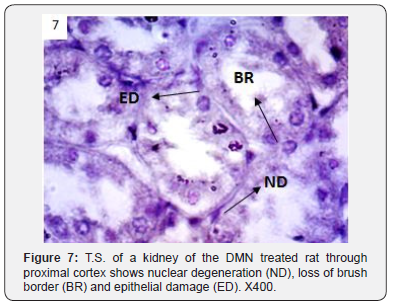
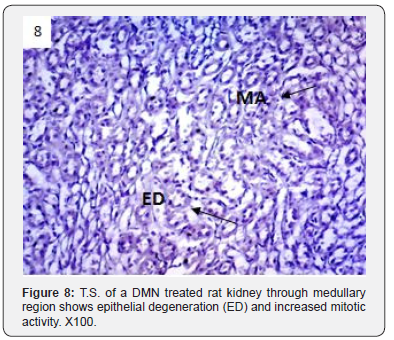
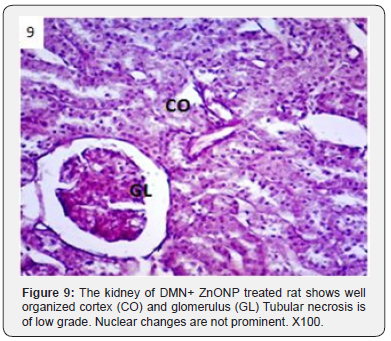
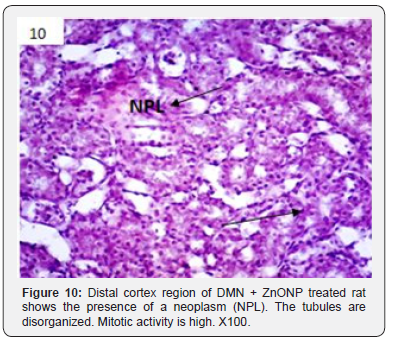
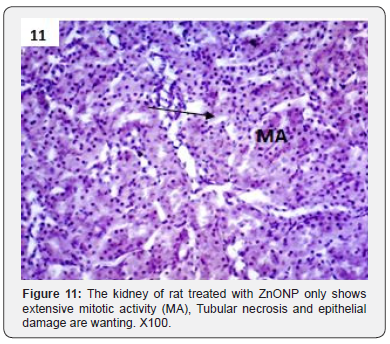
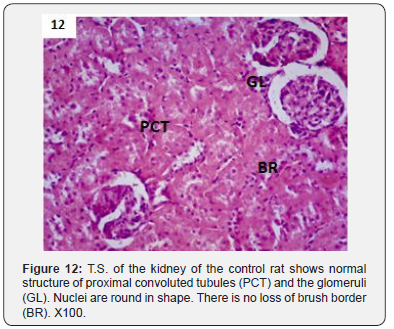
In addition to glomerulonephritis and proximal tubular necrosis, adenocarcinoma formation in subcapsular cortex was recorded in the kidney of DMN treated rats (Figure 6). Epithelial degeneration was conspicuous in proximal as well as distal tubules (Figure 7). Nuclei of different shapes and sizes were noticed throughout cortex and medulla (Figure 8). Histopathological observations in the kidney of DMN and ZnONPs treated rats indicated mild glomerulonephritis and tubular necrosis. Adenocarcinoma was wanting. However, neoplasia was noticed a few locations in the distal cortex (Figure 9). Large number of binucleated tubular epithelium was found to be intact. Cells indicated high mitotic activity (Figure 10). Histopathological observations in the kidney of ZnONPs treated rats showed no nephritis. Proximal and distal tubules were well formed showing no sign of epithelial degeneration. Brush border was found to be intact. However, increased mitotic activity was noticed in distal cortex (Figure 11). All the pathological changes described above were wanting in the kidney of control rats. Renal tubular cortex and medulla exhibited no sign of injury. Normal nuclei in cortex as well as medulla were observed (Figure 12).
Discussion
Present study demonstrates that DMN is equally injurious to kidney as it is to liver and lungs. Mechanism of its toxicity has been discussed by a few workers in the past. It has been established by now that dimethyl nitrosamine and other nitroso compounds are preferentially metabolized in the liver, however, kidney also participates in their biodegradation. DMN is metabolized by CYP2E1 which hydroxylates one methyl group. The resulting hydroxyl methyl nitrosamine is unstable and decomposes to formaldehyde that methylate’s DNA and protein or reacts with water to form methanol Guengerich et al. [33], Frei et al. [34]. The formation of reactive oxygen species (ROS) like hydrogen peroxide and hydroxyl radicals leads to oxidative stress which is the key factor in induction of pathological changes, carcinogenicity and neoplastic changes not only in the liver but kidney and lungs also Pradeep et al. [35], Wills et al. [ 36]. Restoration of renal function remains a challenging issue in experimental toxic renal injury. In a recent study from our laboratory, ZnONPs were found to be protective against DMN induced hepatic injury in rats Rani et al. [24]. Therefore, a similar study on kidney was considered essential to establish the therapeutic potential of ZnONPs. Very first indication of a beneficial effect of ZnONPs against DMN toxicity was exhibited by observations on creatinine. It was elevated in the urine samples of DMN treated rats but decreased in DMN and ZnONPs treated rats. ZnONPs treatment alone also increased creatinine concentration. Elevated urinary/serum creatinine is a reliable biomarker of renal function Bennett [37]. It is associated with abnormal glomerular function Bishop et al. [38], Ali Noori et al. [39] also reported that treatment of Balb/c mice with ZnONPs (50-300 mg/kg) increased serum creatinine concentration. They correlated it with glomerular and tubular degeneration. During present study also we found a correlation between creatinine concentration and renal morphological changes. Improved renal glomerular and tubular morphology in DMN and ZnONPs treated rats corresponded with a decline in urine creatinine concentration. However, ZnONPs at present concentration and dose regimen exhibited moderate renal toxicity.
Several studies have documented that metabolism of DMN generates ROS in the liver of experimental animals that lead to oxidative stress Guengerich et al. [40]. However, very few workers have shown that ROS are responsible for its renal toxicity also Usunomena et al. [41]. Present results confirm that DMN could induce LPO in the kidney as well. Subsequent treatment with ZnONPs inhibited the generation of ROS. Dawei et al. [42]. postulated that zinc oxide nanoparticles possess the ability to decrease malondialdehyde and increase the activity of antioxidant enzymes. Contrarily, malondialdehyde increased in the kidney of ZnONPs treated rats in comparison to control rats. Findings of other experiments conducted on the toxicity of ZnONPs have also revealed that its elevated MDA concentration in zebra fish Zhao et al. [43] human epidermal cells Sharma et al. [44] and human liver cells Sharma et al. [45]. Nitric oxides also showed elevated values in DMN treated rats. It declined in the kidney of DMN and ZnONPs treated rats. Earlier studies have shown that nitric oxide donors like sodium nitrite partially prevented the chronic hepatitis induced by dimethyl nitrosamine Lukivskaya et al. [46]. ZnONPs might have affected DMN induced renal toxicity by modulating nitric oxide synthase. Nitric oxide synthase inhibitors like N (ω) nitro. L- arginine (L-NNA) might attenuate the protective effects over DMN toxicity expressed by nitric oxide donors Fukawa et al. [7].
H2O2 is a major metabolic product of DMN Pradeep et al. [35]. Elevated values were registered for H2O2 in the kidney of DMN treated rats. However, a decline was recorded in DMN and ZnONPs treated rats. This observation suggests that ZnONPs influence the metabolism of DMN. This influence might be expressed through CYP2E1. However, further studies are needed to confirm this presumption. Significant increase in renal concentration of MDA, H2O2 and NO reciprocated with depression of GSH in kidney of DMN treated rats. Subsequent administration of ZnONPs to DMN treated rats restored GSH status. ZnONPs treatment to normal rats also elevated GSH level. GSH, a nonenzymatic antioxidant is known to counteract damaging effects of ROS Rana [47], Rana [48], Dorval, Hontela [49]. ZnONPs express antioxidant effect which may be attributed to its anti- inflammatory potential mediated by down regulation of iMOS, cyclo-oxygenase-2 and various cytokines Nagajyothi et al. [50]. Other workers attribute the beneficial effects of ZnONPs to metallothionein Mo et al. [51], Jing et al. [52].
Present results show that DMN inhibits MT in kidney as compared to its concentration in normal rat kidney. MT concentration increased in the kidney of DMN and ZnONPs treated rats. Administration of ZnONPs alone significantly increased MT concentration in renal tissue. These results suggest that ZnONPs too, are strong inducers of MT. Earlier reports show that zinc is the potential inducer of MT Rana, Kumar [8], Maret [53]. MT exchanges zinc relatively quickly in intramolecular and intermolecular reactions with other zinc/sulfur clusters despite relatively high thermodynamic stability Maret et al. [54]. DMN is known to affect glutathione S-transferase (GST) activity in the liver Aniya, Anders [55], Armato et al. [56], Sheweita et al. [57]. However, its effects on renal glutathione S-transferases are not known. Present investigations showed that DMN increased the activity of GST in kidney. Aniya, Anders [55] reported that DMN administration decreased hepatic GST but increased it in serum. This elevation is accompanied by increase in serum GPT (SGPT) activity and serum bilirubin concentrations. Previous study from our laboratory has also confirmed elevation of serum transaminases in DMN treated rats Rani et al. [24]. Treatment of rats with ZnONPs to normal rats increased GST activity but decreased it in the kidney of DMN and ZnONPs treated rats. However, no increase in renal GSH concentration was recorded. GST and GSH play an important role in the detoxification of mutagens and carcinogens Sheweita, Tilmisany [58]. Further, GST can reduce covalent binding of epoxides of carcinogens like DMN Gopalan et al. [59].
Many workers agree that protective effects of ZnONPs against chemically induced damage in liver/ kidney are manifested through its anti-oxidative potential and prevention of ROS mediated mutagenicity and carcinogenicity Taccola et al. [60]. DMN treatment to rats affects an array of antioxidant enzymes viz: superoxide dismutase, catalase and glutathione peroxidase. Post treatment of ZnONPs to DMN treated rats increased glutathione peroxidase activity in comparison to control rats indicating its enhanced capacity to scavenge H2O2 and lipid hydroperoxides Zhao et al. [43]. Morphological improvement in the kidney of DMN treated rats, manifested by ZnONPs, support above observations. Zak et al. [61] and Magee, Barnes confirmed that DMN could induce renal tumors in rats. Hard, Butler [62] studied the morphogenesis of epithelial neoplasms induced in rat kidney by DMN. Riopelle, Jasmine [63] further classified renal tumors induced by DMN. They named them as dysplastic epithelial islands. However, subsequent administration of ZnONPs abolished these tumors and suppressed other morphological lesions. Functional changes, diminished oxidative stress and improvement in antioxidative enzymes, significantly contribute to morphological repair in the kidney. One of the critical features of ZnONPs is their selective toxicity towards cancerous cells in comparison to normal cells Premanathan et al. [64]. ZnONPs express cytotoxicity owing to their specific composition and surface properties. ZnONPs are chemically more active, lead to spontaneous formation of ROS at their surface and cause oxidative stress Yang et al. [65].
Formation of ROS contributes to cellular toxicity and the release of Zn+ ions from the ZnONPs due to their instability in the acidic compartment of lysosomes. Yu et al. [66], Fukui et al. [67] also concluded that ZnONPs toxicity arise from Zn2+ ions released from ZnONPs in vitro and in vivo. Wiseman et al. [68,69]. revealed that excess free Zn2+ (dissolved from ZnONPs) resulted in depletion of sulfhydryl groups in metallothionein and reduction of mitochondrial function leading to apoptotic or necrotic cell death. In principle, ZnONPs toxicity may be manifested through several mechanisms viz. oxidative stress, inhibition of antioxidative enzymes, mitochondrial dysfunction and apoptosis. Interestingly, type of the cell system treated with ZnONPs, the strength of oxidative stress and existing intercellular/ intracellular environment are important factors that will determine ZnONPs toxicity. On the basis of present investigations, following conclusions can be drawn. Firstly, DMN is a potent renal toxin. Even sub chronic exposure to DMN can cause renal tubular damage and induce tumorigenesis. Secondly, ZnONPs possess antitumor properties that are manifested through their selective toxicity against cancer cells. ZnONPs offer protection against DMN induced renal toxicity by inhibiting oxidative stress. This function is attributed to significant induction of metallothionein, antioxidant enzymes and reduction in oxidative DNA damage by ZnONPs. Finally, present study indicates that ZnONPs with the structural, physical and chemical properties can cause a moderate renal tissue damage in kidney of rat. Further studies are needed to establish antitumorigenic behavior of ZnONPs [70-75].
Acknowledgement
Authors thank Director of Sophisticated Analytical Instrumentation Facility, Punjab University, Chandigarh for TEM analysis of nanoparticles. We are grateful to Dr. B. P. Singh, Head, Department of Physics, C.C.S. University, Meerut for providing SEM facility. Technical assistance received from Mr. Ashwani Kumar of Indian Institute of technology, Roorkee, in characterization of nanoparticles by XRD, EDAX, size distribution and zeta potential are gratefully acknowledged.
References
- IARC (1978) Monographs on the evaluation of carcinogenic risk of chemicals to humans. 17 Some n-nitroso compounds, Lyon.
- ATSDR (1989) Toxicological profiles for n-nitrosomethylamine. Agency for Toxic Substances and Disease registry Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service. CAS 9: 62-75.
- Lee KY, Lijinsky W, Magee PN (1964) Methylation of ribonucleic acids of liver and other organs in different species treated with c14- and h3-dimethylnitrosamines in vivo. J Natl Cancer Inst 32: 65-76.
- Knecht M (1966) On the localization of microsomal N-demethylase in the organs of the rat. Naturwissenschaften 53(3): 85.
- Bansal AK, Bansal M, Soni G, Bhatnagar D (2005) Modulation of N-nitrosodiethylamine (NDEA) induced oxidative stress by vitamin E in rat erythrocytes. Hum Exp Toxicol 24(6): 297-302.
- Mittal G, Brar AP, Soni G (2006) Impact of hypercholesterolemia on toxicity of N-nitrosodiethylamine: Biochemical and histopathological effects. Pharmacol Rep 58 (3) 413-419.
- Fukawa A, Kabayashi O, Yamaguchi M, Uchida M, Hosono A (2017) Bovine milk derived α-lactalbumin prevents hepatic fibrosis induced by dimethylnitrosamine via nitric oxide pathway in rats. Biosc Biotechnol Biochem 81(10): 1941-1947.
- Rana SVS, Kumar A (2000) Metallothionein induced by cadmium or zinc inhibits lipid peroxidation in rats exposed to dimethylnitrosamine. Arh Hig Rada Toksikol 51(3): 279-286.
- Ruz M, Carrasco F, Rojas P, Codoceo J, Inostroza J et al. (2013) Zinc as a potential coadjuvant in Therapy for Type 2 Diabetes. Food Nutrition Bulletin 34(2): 215-221.
- Chasapis CT, Loutsidou AC, Spiliopoulou CA, Stefanidou ME (2012) Zinc and human health: an update. Arch Toxicol 86(4): 521-534.
- Dhawan DK, Chadha VD (2010) Zinc: a promising agent in dietary chemoprevention of cancer. Indian J Med Res 132: 676-682.
- Rana SVS, Tayal MK (1981) Influence of zinc, vitamin B12 and glutathione on the liver of rats exposed to carbon tetrachloride. Ind Health 19(2): 65-69.
- Saldeen T (1969) On the Protective Action of Zinc against Experimental Liver Damage due to Choline Free Diet or Carbon Tetrachloride. Z Ges Exp Med 150(3): 251-259.
- Yunice AA, Lindeman RD (1977) Effect of ascorbic acid and zinc sulphate on ethanol toxicity and metabolism. Proc Soc Exp Biol Med 154(1): 146-150.
- Feaster JP, Van Middelem CH, Davis GK (1972) Zinc DDT interrelationship in growth and reproduction in the rat. J Nutr 102(4): 523-528.
- Oberdorster G, Stone V, Donaldson K, (2007) Toxicity of nanoparticles: A historical perspective. Nanotoxicology 1(1): 2-25.
- Hulla JE, Sahu SC, Hayes AW (2015) Nanotechnology: history and future. Human and Experimental Toxicology 34(12): 1318-1321.
- Shen C, James SA, De Jonge MD, Turney TW, Wright PF, et al. (2013) Relating cytotoxicity, zinc ions, and reactive oxygen in ZnO nanoparticle-exposed human immune cells. Toxicological Sciences 136(1): 120-130.
- Vinardell MP, Mitjans M (2015) Antitumor Activities of Metal Oxide Nanoparticles. Nanomaterials (Basel) 5(2): 1004-1021.
- Rasmussen JW, Martinez E, Louka P, Wingett DG (2010) Zinc oxide nanoparticles for selective destruction of tumor cells and potential for drug delivery applications. Expert Opinion on Drug Deliv 7(9): 1063-1077.
- Li CH, Shen CC, Cheng YW, Huang SH, Wu CC, et al. (2012) Organ biodistribution, clearance, and genotoxicity of orally administered zinc oxide nanoparticles in mice. Nanotoxicology 6(7): 746-756.
- Fazilati M (2013) Investigation toxicity properties of zinc oxide nanoparticles on liver enzymes in male rat. Eur J Exp Biol 3(1): 97-103.
- Esmaeillou M, Moharamnejad M, Hsankhani R, Tehrani AA, Maadi H (2013) Toxicity of ZnO nanoparticles in healthy adult mice. Environ Toxicol Pharmacol 35(1): 67-71.
- Rani V, Verma Y, Rana K, Rana SVS (2018) Zinc oxide nanoparticles inhibit dimethylnitrosamine induced liver injury in rat. Chem Biol Interact 295: 84-92.
- Toro G, Ackermann P (1975) Practical clinical chemistry. first eds, Little Brown and Company, Boston, United States pp. 154.
- Jordan RA, Schenkman JB (1982) Relationship between malondialdehyde production and arachidonate consumption during NADPH-supported microsomal lipid peroxidation. Biochem Pharmacol 31(7): 1393-1400.
- Lowry OH, Rosenbrough NJ, Forr AL, Randall RJ (1951) Protein measurement with the Follin phenol reagent. J Biol Chem 193(1): 265-275.
- Thurman RG, Ley HG, Scholz R (1972) Hepatic microsomal ethanol oxidation. Hydrogen peroxide formation and the role of catalase. Eur J Biochem 25(3): 420-430.
- Cortas NK, Wakid NW (1990) Determination of inorganic nitrate in serum and urine samples by a kinetic cadmium reduction method. CLIN CHEM 36(8Pt1): 1440-1443.
- Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82(1): 70-77.
- Paglia DP, Valentine VM (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70(1): 158-169.
- Onosaka S, Tanaka K, Doi M, Okahara KA (1978) Simplified procedure for determination of metallothionein in animal tissues. Eisei Kagaku 24: 128-133.
- Guengerich FP, Kim DH, Iwasaki M (1991) Role of human cytochrome P-450 IIE1 in the oxidation of many low molecular weight cancer suspects. Chem Res Toxicol 4 (2): 168–179.
- Frei E, Kuchenmeister F, Gliniorz R, Breuer A, Schmezer P (2001) N-nitrosodimethylamine is activated in microsomes from hepatocytes to reactive metabolites which damage DNA of non-parenchymal cells in rat liver. Toxicol Lett 123(2-3): 227-234.
- Pradeep K, Mohan CV, Gopichand K, Karthikeyan S (2007) Effect of Cassia Fistula Linn. Leaf extract on diethylnitrosamine induced hepatic injury in rats. Chem Biol Interact 167(1): 12-18.
- Wills PJ, Suresh V, Arun M, Asha VV (2006) Antiangiogenic effect of Lygodium flexuosum against N-nitrosodimethylamine induced hepatotoxicity in rats. Chem Biol Int 164(1-2): 25-38.
- Bennett WM (1996) Mechanisms of acute and chronic nephrotoxicity from immunosuppressive drugs. Ren Failure 18(3): 453-460.
- Bishop LM, Fody PE, Schoe HL (2005) Clinical Chemistry: Principles, procedures, correlations, 5th Lippincott Williams & Wilkins, Philadelphia, Pennsylvania pp: 730.
- Noori A, Karimi F, Fatahian S, Yazdani F (2014) Effect of zinc oxide nanoparticles on renal function in mice. Int J Biosc 5(9): 140-146.
- Guengerich FP, Johnson WW, Ueng YF, Yamazaki H, Shimada T (1996) Involvement of cytochrome P450, glutathione S-transferase, and epoxide hydrolase in the metabolism of aflatoxin B1 and relevance to risk of human liver cancer. Environ. Health Perspect 104(3): 557-562.
- Usunomena U, Ademuyiwa AJ, Tinuade OO, Uduenevwo FE, Martin O, et al. (2012) N-nitrosodimethylamine (NDMA), Liver Function Enzymes, Renal Function Parameters and Oxidative Stress Parameters: A Review. British Journal of Pharmacology and Toxicology 3(4): 165-176.
- Dawei AI, Zhisheng W, Angu Z (2009) Protective effects of Nano-ZnO on the primary culture mice intestinal epithetlial cells in in vitro against oxidative injury. Int J Nanotechnol 3: 1-6.
- Zhao X, Wang S, Wu Y, You H, Lv L (2013) Acute ZnO nanoparticles exposure induces developmental toxicity, oxidative stress and DNA damage in embryo-larval Zebrafish. Aquat Toxicol 136-137: 49-59.
- Sharma V, Shukla RA, Saxena N, Parmar D, Das M, et al. (2009) DNA damaging potential of zinc oxide nanoparticles in human epidermal cells. Toxicology Letters 185(3): 211-218.
- Sharma V, Anderson D, Dhawan A (2012) Zinc oxide nanoparticles induce oxidative DNA damage and ROS-triggered mitochondria mediated apoptosis in human liver cells (HepG2). Apoptosis 17(8): 852-870.
- Lukivskaya O, Lis R, Zwierz K, Buko V (2004) Effect of nitric oxide doner and nitric oxide synthase inhibitor on the liver of rats with chronic hepatitis induced by dimethylnitrosamine. Pol J Pharmacol 56(5): 599-604.
- Rana SVS, Singh R, Verma S (1997) Protective effects of GSH, vitamin E and selenium on lipid peroxidation in cadmium-fed rats. Biological Trace Element Research 51(2): 161-168.
- Rana SVS, Kumar A (2001) Effect of cadmium and zinc metallothionein on methemoglobin and nitric oxide in dimethylnitrosamine treated rats. Indian J Exp Biol 39(5): 487-489.
- Dorval J, Hontela A (2003) Role of glutathione redox cycle and catalase in defense against oxidative stress induced by endosulfan in adrenocortical cells of rainbow trout (Oncorhynchus mykiss). Toxicol Appl Pharmacol 192(2): 191-200.
- Nagajyothi PC, Cha SJ, Yang IJ, Sreekanth TV, Kim, KJ, et al. (2015) Antioxidant and anti-inflammatrory activities of zinc oxide nanoparticles synthesized using Polygala tenuifolia root extract. Journal of Photochemistry and Photobiology B: Biology 146: 10-17.
- Mo R, Jiang T, Gu Z (2014) Recent progress in multidrug delivery to cancer cells by liposomes. Nanomedicine 9(8): 1117-1120.
- Jing L, Li L, Zhao J, Zhao J, Sun Z, Peng S (2015) Zinc induced metallothionein overexpression prevents doxorubicin toxicity in cardiomyocytes by regulating the peroxiredoxins. Xenobiotica 46(8), 715-725.
- Maret W (2000) The function of zinc metallothionein: a link between cellular zinc and redox state. J Nutr 130(Suppl 5): 1455-1458.
- Maret W, Larsen KS, Vallee BL (1997) Coordination dynamics of biological zinc "clusters" in metallothioneins and in the DNA-binding domain of the transcription factor Gal4. Proc Natl Acad Sci U S A 94(6): 2233-2237.
- Aniya Y, Anders MW (1985) Alteration of hepatic glutathione S-transferases and release into serum after treatment with bromobenzene, carbon tetrachloride, or N-nitrosodimethylamine. Biochem Pharmacol 34(24): 4239-4244.
- Armato U, Wu J, Menegazzi M, Menapace L, Ribecco M, et al. (1993) The in-utero initiation with DMN alters the complement of cytosolic glutathione S-transferases and the phenobarbital-induced expression of c-jun and c-myc oncogenes in primary neonatal rat hepatocytes. Cytotechnology 11(1): 18-20.
- Sheweita SA, Mousa N, Al-Masry HM (2008) N-Nitrosodimethylamine changes the expression of glutathione S-transferase in the liver of male mice: The role of antioxidants. J Biochem Mol Toxicol 22(6): 389-395.
- Sheweita SA, Tilmisany AK (2003) Cancer and phase II drug-metabolizing enzymes. Curr Drug Metab 4(1): 45-58.
- Gopalan P, Jensen DE, Lotlikar PD (1992) Glutathione conjugation of microsome-mediated and synthetic aflatoxin B1-8, 9-oxide by purified glutathione S-transferases from rats. Cancer Lett 64(3): 225-233.
- Taccola L, Raffa V, Riggio C, Vittorio O, Iorio MC, et al. (2011) Zinc oxide nanoparticles as selective killers of proliferating cells. Int J Nanomedicine 6: 1129-1140.
- Zak FG, Holzner JH, Singer EJ, Popper H (1960) Renal and pulmonary tumors in rats fed dimethylnitrosamine. Cancer Res 20: 96-99.
- Hard GC, Butler WH (1971) Morphogenesis of epithelial neoplasms induced in the rat kidney by dimethylnitrosamine. Cancer Res 31(10): 1496-1505.
- Riopelle JL, Jasmin G (1969) Nature, classification, and nomenclature of kidney tumors induced in the rat by dimethylnitrosamine. J Natl Cancer Inst 42(4): 643-662.
- Premanathan M, Karthikeyan K, Jeyasubramanian K, Manivannan G (2011) Selective toxicity of ZnO nanoparticles toward Gram-positive bacteria and cancer cells by apoptosis through lipid peroxidation. Nanomedicine 7(2): 184-192.
- Yang H, Liu C, Yang D, Zhang H, Xi Z (2009) Comparative study of cytotoxicity, oxidative stress and genotoxicity induced by four typical nanomaterials: the role of particle size, shape and composition. J Appl Toxicol 29(1): 69-78.
- Yu L, Lu Y, Man N, Yu SH, Wen LP (2009) Rare earth oxide nanocrystals induce autophagy in HeLa cells. Small 5(24): 2784-2787.
- Fukui H, Horie M, Endoh S, Kato H, Fujita K, et al. (2012) Association of zinc ion release and oxidative stress induced by intratracheal instillation of ZnO nanoparticles to rat lung. Chem Biol Interact 198(1-3): 29-37.
- Wiseman DA, Wells SM, Wilham J, Hubbard M, Welker JE, et al. (2006) Endothelial response to stress from exogenous Zn2+ resembles that of NO-mediated nitrosative stress and is protected by MT-1 overexpression. Am J Physiol Cell Physiol 291(3): 555-568.
- Wiseman DA, Wells SM, Hubbard M, Welker JE, Black SM (2007) Alterations in zinc homeostasis underlie endothelial cell death induced by oxidative stress from acute exposure to hydrogen peroxide. Am J Physiol Lung Cell Mol Physiol 292(1): 165-177.
- Durnan DM, Palmiter RD (1981) Transcriptional regulation of the metallothionein- I gene by heavy metals. J Biol Chem 256(11): 5712-5716.
- Gong Q, Hart BA (1997) Effect of thiols on cadmium-induced expression on metallothionein and other oxidant stress genes in rat lungs epithelial cells. Toxicology 119(3): 179-191.
- Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249(22): 7130-7139.
- Hamza RZ, Ismail HA, El-Shenawy NS (2017) Oxidative stress, histopathological and electron microscopic alterations induced by dimethylnitrosamine in renal male mice and the protective effect of α-lipoic acid. J Basic Clin Physiol Pharmacol 28(2): 149-158.
- Soheili S, Moradhaseli S, Shokouhian A, Ghorbani M (2013) Histopathological effects of ZnO nanoparticles on liver and heart tissues in wistar rats. Adv Biores 4(2): 83-88.
- Vizirianakis IS (2011) Nanomedicine and personalized medicine toward the application of pharmacotyping in clinical practice to improve drug-delivery outcomes. Nanomedicine: Nanotechnology, Biology and Medicine (Lond) 7(1): 11-17.






























