Largest Obstacle of Drug Delivery to the Blood Brain Barrier and Current Approach to Solve this Problem: Recent Comprehensive Review
Md. Didaruzzaman Sohel1*, Tania Sultana1 and Md. Hassan Kawsar2
1 Incepta Pharmaceuticals Ltd, Bangladesh
2 Department of Pharmacy, State University of Bangladesh, Bangladesh
Submission: August 17, 2018; Published: September 10, 2018
*Corresponding author: Md. Didaruzzaman Sohel, Incepta Pharmaceuticals Ltd, Dewan Idris Road, Zirabo, Savar, Dhaka, Bangladesh
How to cite this article: Didaruzzaman S, Tania S, Hassan K. Largest Obstacle of Drug Delivery to the Blood Brain Barrier and Current Approach to Solve this Problem: Recent Comprehensive Review. Glob J Nano. 2018; 4(2): 555633. DOI: 10.19080/GJN.2018.04.555633
Abstract
Present review highlights the largest obstacle of Drug Delivery to the Blood Brain Barrier and current approach to solve this problem. The blood–brain barrier acts as a physical barrier that prevents free entry of blood-derived substances, including those intended for therapeutic applications. Medical treatment options for central nervous system (CNS) diseases are limited due to the inability of most therapeutic agents to penetrate the blood–brain barrier (BBB). Notable examples of potential drugs where the intact blood–brain barrier (BBB) precludes their use are neuropeptides, proteins, and chemotherapeutic agents. Indeed, all large-molecule products of biotechnology such as monoclonal antibodies, recombinant proteins, antisense, or gene therapeutics do not cross the BBB. Barrier mechanisms in the brain are important for its normal functioning and development.
Keywords: Blood brain barrier; Drug development; Current approach; Disease
Abbrevations: BCSFB: Blood–Cerebrospinal Fluid Barrier; TEER: Trans Endothelial Electrical Resistance; HBEC: Human Brain Endothelial Cells; CSF: Cerebrospinal Fluid; BBB: Blood Brain Barrier
Introduction
Brain is the most sensitive organ of human body. A few diseases like encephalitis, neurological disorders, various sclerosis, stroke, epilepsy, Alzheimer disease, Parkinson’s disease, different scleroses, headache and tumor prompt decline of brain function. The blood - brain barriers excess the body to stay pathogens and potentially harmful chemicals circulating in the blood from entering the brain and spinal cord. The semi-permeable blockade, composed of tightly packed cells, is important for maintaining a controlled environment to excess the central nervous system (CNS) to function accurately. However, the barrier also makes it nearly impossible for many drugs and other drug molecules to be delivered to the brain via the bloodstream [1]. Neurological diseases are an important reason of mortality and constitute 12% of total deaths globally. Among the neurological disorders, Alzheimer and other dementias are probable to constitute 2.84% of the total deaths, while cerebrovascular disease constitutes about 8% of the total deaths in high income countries in 2005 [2]. development of new therapeutic approaches for these diseases is a difficult challenge, and there is no effective treatment for almost all the brain diseases. The blood-brain barrier (BBB) efficiently restricts penetration of therapeutic agents to the brain from the periphery [3].
The blood–brain barrier, which is formed by the endothelial cells that line cerebral micro vessels, has a significant role in maintaining an accurately regulated microenvironment for reliable neuronal signaling [4]. The blood– brain barrier (BBB) is a dynamic hindrance ensuring the brain against attacking life forms and undesirable substances. It is also noted that the most important barrier impeding drug transport into the brain via the blood circulation. This is not because there is a lack of candidate drugs but due to the inability of many therapeutic molecules to cross the BBB or BCSFB other specialized CNS barriers to reach the specific areas of brain [5]. The blood brain barrier (BBB) keeps up homeostasis by organizing the vehicle of synthetic substances at the brain interface [6,7].
The endothelial cells in the BBB are appended to each other by tight intersection protein edifices. Tight intersections are found in regions in the body other than the cerebrum, for example, the small intestine. Estimations of electrical obstruction over the endothelia, named as TEER, are utilized to analyze the penetrability of tight intersections inside the body. TEER of the cerebrum endothelium is more important than 1000 ohm/ cm2, which shows that the intersections at the BBB are tight bringing about more limitations for the paracellular transport of materials crosswise over BBB [8]. In a large portion of the cases, the major reason for the distress in the development of medications to treat brain diseases is the nearness of BBB. Out of the several brain disorders, brain tumors commonly have poor prognosis, which varies according to the type and grade of the tumor. Due to the presence of BBB, drug delivery to brain tumors has long been a problematic issue [9]. The BBB is additionally a diffusion barrier obstruction that permits just water and little lipophilic molecules to easily permit the brain in accordance with their concentration gradients. Experimental animal studies have revealed differences in the barrier tightening time-course during development.
The BBB ends up impervious to bigger particles previously it stops littler ones. Thus, its maturation has a progressive scheme. A similar scheme is true for BBB transporters. Due to all these facts, the BBB is the most significant element responsible for the preservation of CNS homeostasis. Under both physiological and obsessive conditions, the BBB segregates and protects nervous tissue of the brain and spinal line from fluctuations in nutrients, hormones, metabolites, and other blood constituents [10]. Therefore, it is essential to find strategies to be capable to transiently open the BBB for drug delivery, and repair BBB in the case of neurodegenerative diseases like Alzheimer’s disease, Parkinson’s, Amyotrophic lateral sclerosis (ALS), and multiple sclerosis [11].
Discovery of Blood-Brain Barrier
The revelation of the BBB goes back over 100 years when, in the 1880s, Paul Ehrlich watched that intravenous administration of specific colors (e.g. trypan blue) recolored all organs except for the cerebrum and the spinal cord. He presumed that the colors had a lower affinity for binding to the sensory system when contrasted with different tissues. In 1913, Edwin Goldman, a partner of Ehrlich, showed the simple same colors, when specifically infused into the cerebrospinal liquid (CSF), promptly recolored sensory tissue however not different tissues. The expression “blood-brain barrier” was instituted, in any case, by Lewandowsky in 1898, after he and his partners had performed investigations to exhibit that neuro toxic agents influenced cerebrum work just when straight forwardly infused into the mind yet not when infused into the vascular framework It took an extra 70 years until the point when Reese and partners confined the obstacle to the narrow endothelial cells inside the mind by electron-microscopic studies [12].
Physiology and biology of the blood–brain barrier
The human blood brain barrier (BBB) is a barrier shaped by human brain endothelial cells (hBECs), which is essential ensure the adequate neuronal function and ensure the central nervous system (CNS) from disease [13]. It is well protected and dynamically regulated to provide a sanctuary for the central nervous system (CNS). There are several pathways to enter brain parenchyma; the most important two are blood circulation and cerebrospinal fluid (CSF) circulation. In the human cerebrum, there are around 100 billion vessels altogether, giving a joined length of brain capillary endothelium of roughly 650 km and an aggregate surface region of around 20 m2 [14]. Any molecules’ entry into the brain via parenteral administration is strictly controlled by the BBB and the BCSFB. As the surface of BCSFB faces the ventricle that is full of CSF not the blood. This, in combination with the high turnover rate of CSF, leads to continuously flushing the injected drug (i.e. those injected into the ventricle) back to the blood.
The BBB, therefore, is universally considered as the most essential barrier in preventing molecules from reaching the brain parenchyma via extensive branches of blood capillary networks. The chief anatomical and functional site of the BBB is the brain endothelium. Physiologically, in addition to brain capillary endothelial cells, extracellular base membrane, adjoining pericytes, astrocytes, and microglia are all integral parts of the BBB supporting system. Together with surrounding neurons, these components form a complex and functional “neurovascular unit” [15,16]. In addition, efflux transporters such as p-glycoprotein contribute to the barrier properties by returning small lipophilic molecules capable of diffusing into BMECs back to the bloodstream [17] (Figure 1).

Molecular components of tight junctions
The tight junctions between endothelial cells are liable for the barrier function. Occludin was the first integral membrane protein found to be exclusively localized within the tight junctions. In contrast to occludin, claudins have been shown to be required for the formation of tight junctions. The integral membrane proteins of tight junctions are linked to the cytoskeleton via zone occluding-1 (ZO-1), ZO-2 and ZO- 3. In addition, non-occludin adherens junctions are found intermingled with tight junctions. In adherence junctions, the endothelial specific, integral membrane proteins VE-cadherins are found. Furthermore, a family of proteins called junctional adhesion molecules (JAM), and recently discovered endothelial cell-selective adhesion molecules (ESAM), are localized in the tight junctions of the BBB. Their precise function in BBB integrity remains to be determined. All areas of the brain do not have a blood-brain barrier. The structures located at strategic positions in the midline of the ventricular system and lack the BBB are collectively referred to as circumventricular organs (CVOs). In these non-barrier regions, the tight junctions between endothelial cells are discontinuous thus allowing entry of molecules. Many of these areas participate in hormonal control [16,17] (Figure 2).

Biological functions of the blood brain barrier
The BBB restrictions the passage of large and hydrophilic solutes, while allowing tiny lipophilic molecules (O2, CO2, and hormones) to freely diffuse concentration gradients. The BBB possesses specific transporters which are used to move complex nutrients such as glucose and amino acids into the brain. The BBB can also use receptor-mediated endocytosis to transport certain proteins such as insulin, leptin, and iron transferrin into the brain [20,21]. Approximately 98% of small molecule drugs and all large molecule neurotherapeutics (e.g., recombinant peptides, proteins, anti-sense-agents and genetic vector) are normally expelled from the brain. When confronted with the BBB, they behave essentially like trypan blue.
Passive and active mechanisms of transport across the BBB
Preventing random influx of molecular compounds into the brain, the BBB does allow selective entrance of nutrients and provide of regulatory factors into the brain. Thus, the endothelial cells enable transport of molecules in a passive, gradient-driven manner or active, energy-dependent mode. Physiological conditions, passive movement of water solutes along the aqueous route of the intercellular cleft between endothelial cells via small pores in the tight junctions, i.e., the paracellular pathway. The exact molecular nature of paracellular transport is still largely obscure, and neurodegenerative and neuro-oncological diseases at the early stages of development are not necessarily accompanied by defects in BBB integrity, general exploitation of this pathway for transport of drug-loaded nanoparticles into the brain might be virtually negligible. Thus, hydrophobic molecules, like many psychoactive drugs, with a size less than 500 Da, may diffuse transcellularly-provided they escape from MDR-mediated extrusion-to reach the brain parenchyma from the systemic circulation. However, convey of nutrients and building blocks are more tightly synchronized and facilitated by specialized transporters. For example, glucose uses the glucose transporter (GLUT1), while amino acids are transported by the large neutral amino acid transporter (LAT1). The latter transporter is also involved in transport of drugs like L-dopa, gabapentin, and baclofen.
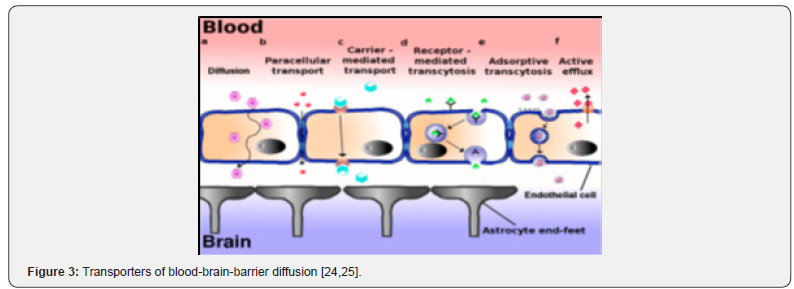
The blood brain barrier consists of a polarized layer of vascular endothelial cells, tightly attached to each other by means of tight junctions, and lined up by astrocytes. A variety of transcellular transport processes can be distinguished
a. Diffusion, driven by a concentration gradient, mainly involving small hydrophobic molecules. This pathway represents the main entry route into the brain of current therapeutics;
b. Paracellular transport–limited to small water-soluble molecules;
c. Carrier-mediated transport, as occurs for e.g., glucose, amino acids, nucleosides, and therapeutics such as vinca alkaloids, azidothymidine etc.;
d. Receptor-mediated transcytosis for peptidic signaling and regulatory molecules (insulin, leptin, interleukins), nutrients (iron, LDL);
e. Adsorptive transcytosis, presumably relying on transport of positively charged cargo (serum proteins) in a non-specific manner and
f. Proton pump efflux transporters [22] (Figure 3).
Challenge of drug delivery to BBB
The BBB confines drug delivery by generally allowing only those molecules that are lipophilic and have low molecular weight (less than 400−500Da) to enter the brain from the blood stream through the transcellular route. In this context, it has been reported that approximately 98% of small molecules and nearly all large therapeutic molecules, such as MABs, antisense oligonucleotides, or viral vectors, cannot pass through this barrier. For these reasons, delivery of drugs to the brain is still a major challenge, and recent reports designate that less than 10% of therapeutic agents for neurological diseases enter clinical trials because of poor brain penetration. Attempts to overcome this barrier involve increasing drug delivery of intravascularly administered drugs by manipulating either the drug or capillary permeability, and/or by local administration into brain fluids, such as the cerebrospinal fluid of brain ventricles or the interstitial fluid of brain tissue [6,16].

Lipid soluble small molecule drugs may cross the BBB if they are capable of diffusing through the endothelial cell membrane itself [23], but few drugs fall into this category. The BBB’s remarkable exclusionary capability is attributed to tight junctions that join the endothelial cells lining the vasculature throughout the brain. Tight junctions are comprised of several proteins, including various claudins, occludins, junctional adhesion molecules, and cadherins, which function to prevent molecules from passively diffusing between cells and out of the vasculature. Small molecules must pass through the endothelial cells themselves, either through diffusion, for lipid soluble molecules, or active transport, for most nutrients and other substances necessary for normal brain function. Furthermore, if a certain molecule does manage to pass through the endothelial cell layer, the basement membrane provides an additional barrier to diffusion [24,25] (Figure 4).
Delivery of therapeutics to the brain is demanding because many organic molecules have inadequate aqueous solubility and limited bioavailability. Aqueous solubility of haloperidol was increased by more than 100-fold in the developed formulation. Formulation was assessed via different routes of administration for behavioral (cataleptic and locomotor) responses, and for haloperidol distribution in plasma and brain tissues. Dendrimerbased formulation showed extensively higher distribution of haloperidol in the brain and plasma compared to a control formulation of haloperidol administered via intra peritoneal injection. Additionally, 6.7 times lower doses of the dendrimer− haloperidol formulation administered via the intranasal route produced behavioral responses that were comparable to those induced by haloperidol formulations administered via intraperitoneal injection. This study demonstrates the potential of dendrimer in improving the delivery of water insoluble drugs to brain [26].
Factors affecting permeation rate and distribution of drug across BBB
In healthy conditions, the passage of solutes and cells from the blood to the CNS may be controlled by BBB. There are various factors that affect permeation of drug through BBB, partition coefficient, molecular size, ionization constant of drug. The transport of drug across the BBB has a relationship with drug structure and physicochemical properties. But the main crucial factor is lipophilicity of drug. Increasing lipophilicity of a drug may cause rise in its volume of distribution. Pharmacokinetic parameters are also be affected by lipophilicity of some drug. As lipophilicity of drug increases, the rate of its oxidative metabolism by enzyme cytochromes P450 may increase. Therefore, to improve bioavailability of a drug, there is a balanced effect of lipophilicity on first pass metabolism and membrane permeability. Most of the drugs used for treatment of CNS diseases are lipoidal in nature. They may have a log octanol/ water partition coefficient between 0.5 and 6.0. The molecular weight of these drugs is between 150 and 500 Da. Because lipid soluble small drug molecules possess molecular weight between the ranges 400-600Da transported quickly through BBB by lipid mediated transport mechanism. Charged, highly branched, hydrophilic molecules and molecules having routable bonds show a poor BBB penetration [27].
New technologies for drug delivery across the blood brain barrier: current approach
a) Artificial hydrophobization of peptides and proteins for delivery to CNS
b) Receptor-mediated delivery of nanoparticles to the Brain
c) Inhibition of drug efflux systems in BBB by Pluronic® block copolymers [28]
d) Drug and gene delivery across the blood–brain barrier with focused ultrasound [29].
Blood brain barrier can be temporarily and locally opened by focused ultrasound in the presence of circulating micro bubbles. Micro bubbles may be defined as the “bubbles” of mono lipids that are capable to pass through the blood brain barrier. They form a lipophilic bubble that can easily move through the barrier. The ultrasound increases the permeability of the blood brain barrier by causing interference in the tight junctions in localized areas. Thus, combined effect of micro-bubbles and ultrasonic sound allows drug into a very specific area with the diffusion of micro-bubbles. More often, micro bubbles diffuse only where the ultrasound disrupts the barrier. Focused ultrasound is also used to deliver targeted NK-92 cells to the brain using a model of metastatic breasts cancer. Thus, loading a micro-bubble with an active drug to diffuse through the barrier and target a specific area increases the usefulness and action of drug. It was also found to be more reasonable for targeted gene transfer into central nervous system by MRI guided focused ultrasound induced blood brain barrier disruption. Similarly, doxorubicinloaded micro bubble technology has been developed that destroys tumors with focused ultrasound and makes fragments. Further nano-shards formed are capable of escaping through the leaking tumor vasculature, promoting accumulation of drug within the interstitial [30].
Significance of Nanoparticles for Delivery of Drug to BBB
Nanoparticles (NP) have been used for drug delivery to the brain [31,32]. NPs are solid colloidal particles ranging in size from (1 to 1000) nm that are utilized as drug delivery agents. The use of NPs to deliver drugs to the brain across the blood-brain barrier (BBB) may provide a significant advantage to current strategies. The primary advantage of NP carrier technology is that NPs mask the blood-brain barrier limiting characteristics of the therapeutic drug molecule. Furthermore, this system may slow drug release in the brain, decreasing peripheral toxicity [33]. Consequently, drugs are transported across the BBB at very low efficiency, and this currently limits the treatment of e.g. schizophrenia, depression, brain tumors, HIV and epilepsy [34]. The precise mechanism of transcytosis has not been clarified and is still debated. A possible mechanism is that poly sorbate 80 (tween-80) coated polybutyl-cyanoacrylate nanoparticles adsorb apolipoprotein E and B from the bloodstream after IV injection and therefore use LRP for transcytosis across the BBB [35]. There are two main categories of nanoparticles, inorganic and organic. Inorganic nanoparticles are mainly magnetic, metallic, nano shells, and ceramic. Magnetic nanoparticles are super paramagnetic iron oxide particles that display large magnetic moments in a magnetic field. These are bio competitive, noncompatible chemically stable, and easy to manufacture. These are mostly used for targeted delivery of drugs/genes and are used in thermotherapy. Next category of nanoparticles is metallic nano particle which comprises gold or silver or copper andiron nanoparticles.
These are smaller in size (<50nm) having large surface area, carry high drug doses, but these show poor biocompatibility and have no decided function when used in vivo. These are used for controlled release of drugs, proteins, and DNA encapsulated in hollow cores of metal shells at desired sites. These are widely used in catalysis, sensing, imaging, and drug delivery. Silica nanoparticles are nano shells that possess similar imaging/ therapeutic potential as quantum. These are less toxic and are relatively large compared with quantum dots. These are used for photothermal tumor ablation. These form immune conjugates which are highly applicable for immunoglobulin bioassay. Ceramic nanoparticles are made up of nonmetallic materials that are cheap and stable. These can be formed by inorganic biocompatible materials, silica, Titania, and alumina. These are of smaller size (<100). These are relatively flexible, easy to manufacture, water soluble, and biologically stable. These can form coatings and make bulk materials at low temperatures. Many types of organic nanoparticles such as carbon nano tubes, quantum dots (semiconductors), dendrimers, liposomes, and polymeric nanoparticles. These are crystalline form of pure carbon. Carbon nanotubes are graphite sheets rolled into single or multi walled tubes. Carbon nanotubes are used in electromagnetic shielding of polymers composite for hydrogen, storage, and its batteries. These are used for targeted delivery of drugs, genes, and vaccines and are widely used in thermotherapy of tumors. Quantum dots are semiconductor crystals formed by combination of chemical elements from groups II, III, and V of the periodic table.
These are made up of cadmium core and metal shell and have similar size <10 nm. These are used in vitro labeling of live cells and for gene expression studies, fluorescent imaging assays to detect antigens or cells. These are used for in vivo cancer diagnosis [36]. Delivery of pharmacologically active molecules, especially macromolecules, to the brain is challenged by specific properties of the cells forming the interfaces between the blood and the central nervous system (CNS). By restricting paracellular and transcellular diffusion of hydrophilic and lipophilic substances, these cellular interfaces provide the strictly controlled environment required for CNS development and functions [37]. Moreover, NPs can be functionalized by covalent conjugation to various ligands (such as antibodies, proteins, or aptamers) to target specific tissues. When designing NPs for clinical applications, it should be remembered that their systemic administration generates important modifications. The nonspecific interaction between the shell of NPs and many classes of proteins circulating in the bloodstream leads to the adsorption of opsonin’s on their surface, forming the so-called “corona” [38] (Figure 5).

Advantages of nanoparticles [39]
Nanoparticles are having several potential advantages over other carrier mediated drug delivery system.
i. Targeting ability of drugs to organ or tissue.
ii. Increase in bioavailability.
iii. Development of new formulation, which are safer.
iv. Ability to sustained release of drugs.
v. High carrier capacity.
vi. Prolonged circulation time.
vii. Stable in blood.
viii. Acquiescent to small molecules, peptides, proteins, or nucleic acids.
Disadvantages of nanoparticles
a) Increase in cost of formulation due to high manufacturing costs, which can be optimized property
b) May cause allergic reactions.
c) Over use of polyvinyl alcohol as a stabilizer have toxic reaction.
2B-Trans™
Drug delivery across the blood–brain barrier (BBB) is a major obstacle for the development of effective treatments of many central nervous system disorders. Sophisticated cell culture models of the BBB have helped us to identify, characterize, and validate a novel targeted drug delivery technology, designated 2B-Trans™, for the receptor-mediated uptake and transport of drugs across the BBB. This paper describes in great detail how such a BBB cell culture model should be prepared and handled and applied for the use of targeted drug delivery across the BBB [39].
Future Prospective to Solve this Obstacle to Delivery of Drug to BBB
It may be feasible to develop several systemically effective neuro pharmaceuticals that will be effective following systemic administration. Novel strategies based mainly on exploitation of specific transport systems at the BBB are being planned and developed. The advancement for delivering drug or peptide across BBB requires the integration of antibody engineering, pharmacokinetics, and receptor-based drug design. The development of a successful BBB drug delivery system seems possible. Thus, there is need of development of CNS drug delivery.
Conclusion
Brain targeting drug delivery system has essential in management of CNS disorders. It can be concluded from this review that by means of the nanotechnology, nasal routes, disruption of BBB, prodrugs, etc. the drug can be delivered across the BBB efficiently. Additional drug exposure to brain can be improved by utilizing modified colloidal particles and liposomes. Because it is assumed that they have prolong blood circulation, which helps in more interaction and penetration into brain endothelial cells. Thus, these approaches can be useful in the brain targeting offers an improved clinical efficiency but still there is need of most reliable techniques or methods which high clinical significance and cost effective.
Acknowledgment
The authors wish to thank, Department of Pharmacy, State University of Bangladesh for providing excellent idea to carry out the study.
Availability of Data and Materials
All data are presented in the main paper
Authors’ Contributions
MDS and MHK designed the concept of the study. MDS and TS collect the information. MDS drafted the manuscript. MHK critically reviewed and revised the manuscript. All authors read and approved the final manuscript.
References
- Neuwelt E, Abbott NJ, Abrey L, Banks WA (2008) Strategies to advance translational research into brain barriers. Lancet Neurol 7(1): 84-96.
- Gidwani M, Singh AV (2013) Nanoparticle Enabled Drug Delivery Across the Blood Brain Barrier: in vivo and in vitro Models, Opportunities and Challenges. Curr Pharm Biotechnol 14(14): 1201-1212.
- Groothuis DR (2000) The blood-brain and blood-tumor barriers: a review of strategies for increasing drug delivery. Neuro Oncol 2(1): 45-59.
- Butt AM, Jones HC, Abbott NJ (1990) Electrical resistance across the blood-brain barrier in anaesthetized rats: a developmental study. J Physiol 429: 47-62.
- Pardridge WM (2003) Blood–brain barrier drug targeting: the future of brain drug development. Mol Interv 3(2): 90-105.
- Rip J, Schenk GJ, de Boer AG (2009) Differential receptor-mediated drug targeting to the diseased brain. Expert Opin Drug Deliv 6(3): 227- 237.
- Chen Y, Liu L (2012) Modern methods for delivery of drugs across the blood–brain barrier. Advanced Drug Delivery Reviews 64(7): 640-665.
- Bhowmik A, Khan R, Ghosh MK (2015) Blood Brain Barrier: A Challenge for Effectual Therapy of Brain Tumors. Biomed Res Int 320941.
- Bernacki J, Dobrowolska A, Nierwiñska K, Malecki A (2008) Physiology and pharmacological role of the blood-brain barrier. Pharmacol Rep 60(5): 600-622.
- Rönnbäck L, Hansson E (2006) Astrocyte–endothelial interactions at the blood–brain barrier. Nat Rev Neurosci 7(1): 41-53.
- Strazielle N, Ghersi-Egea JF (2013) Physiology of Blood−Brain Interfaces in Relation to Brain Disposition of Small Compounds and Macromolecules. Mol Pharm 10(5): 1473-1491.
- Pardridge WM (2005) Molecular biology of the blood-brain barrier. Mol Biotechnol 30(1): 57-69.
- Pardridge WM, Eisenberg J, Yang J (1987) Human blood-brain barrier transferrin receptor. Metabolism 36(9): 892-895.
- Lippmann ES, Azarin SM, Kay JE, Nessler RA (2012) Human Blood- Brain Barrier Endothelial Cells Derived from Pluripotent Stem Cells. Nat Biotechnol 30(8): 783-791
- Naik P, Cucullo L (2012) In Vitro Blood–Brain Barrier Models: Current and Perspective Technologies. Pharm Sci 101(4): 1337-1354.
- Khawli LA, Prabhu S (2014) Drug Delivery across the Blood-Brain Barrier. Curr Drug Deliv 10(5): 1471-1472.
- Kabanov AV, Batrakova EV (2004) New Technologies for Drug Delivery across the Blood Brain Barrier. Curr Pharm Des 10(12): 1355-1363.
- Lockman PR, Mumper RJ, Khan MA, Allen DD (2002) Nanoparticle Technology for Drug Delivery across the Blood-Brain Barrier. Drug DevInd Pharm 28(1): 1-13.
- Gabathuler R (2010) Approaches to transport therapeutic drugs across the blood-brain barrier to treat brain diseases. Neurobiology of Disease 37(1): 48-57.
- Gaillard P, de Boer AG (2008) 2B-Trans™ Technology: Targeted Drug Delivery Across the Blood-Brain Barrier. Drug Delivery Systems. 437: 161-175.
- Pardridge WM (2005) The blood brain barrier: bottleneck in brain drug development. Neuro Rx 2(3): 3-14.
- Bauer HC, Krizbai IA, Bauer H, Traweger A (2014) You Shall Not Passtight junctions of the blood brain barrier. Front Neurosci 8: 392.
- Timbie KF, Mead BP, Price RJ (2015) Drug and gene delivery across the blood–brain barrier with focused ultrasound. J Control Release 219: 61-75.
- Katare YK, Daya RP, Gray CS, Luckham RE, Bhandari J, et al. (2015) Brain Targeting of a Water Insoluble Antipsychotic Drug Haloperidol via the Intranasal Route Using PAMAM Dendrimer. Mol Pharm 12(9): 3380-3388.
- Kumar S, Gupta SK, Sharma PK (2012) Recent Developments in Targeted Drug Delivery System for Crossing Blood brain barrier: A Review. Int J Pharm Sci 4(2): 36-41.
- Gabathuler R (2010) Approaches to transport therapeutic drugs across the blood–brain barrier to treat brain diseases: A REVIEW. Neurobiol Dis 37(1): 48-57.
- Upadhyay RK (2014) Drug Delivery Systems, CNS Protection and the Blood Brain Barrier. Biomed Res Int 869269.
- Kreuter J (2014) Drug delivery to the central nervous system by polymeric nanoparticles: what do we know?” Adv Drug Deliv Rev 71: 2-14.
- Vauthier C, Dubernet C, Fattal E, Pinto-Alphandary H, Couvreur P (2003) Poly(alkylcyanoacrylates) as biodegradable materials for biomedical applications. Advanced Drug Delivery Reviews 55(4): 519- 548.
- Georgieva JV, Hoekstra D, Zuhorn IS (2014) Smuggling Drugs into the Brain: An Overview of Ligands Targeting Transcytosis for Drug Delivery across the Blood–Brain Barrier. Pharmaceutics 6(4): 557-583.
- Kopecek J (2003) Smart and genetically engineered biomaterials and drug delivery systems. Eur J Pharm Biopharm 20(1): 1-16.
- Torchilin VP (2001) Structure and design of polymeric surfactantbased drug delivery systems. Control Release 73(2-3): 137-172.
- Muller-Goymann CC (2004) Physicochemical characterization of colloidal drug delivery systems such as reverse micelles, vesicles, liquid crystals and nanoparticles for topical administration. Eur J Pharm Biopharm 58(2): 343-356.
- Kabanov AV, Batrakova EV (2014) New technologies for drug delivery across the blood brain barrier. Curr Pharm Des 10(12): 1355-1363.
- Kong SD, Lee J, Ramachandran S, Eliceiri BP, Shubayev VI, et al. (2012) Magnetic targeting of nanoparticles across the intact blood–brain barrier. J Control Release 164(1): 49-57
- Sarkar A, Fatima I, Jamal QM, Sayeed U, Khan MKA, et al. (2017) Nanoparticles as a Carrier System for Drug Delivery Across Blood Brain Barrier. Curr Drug Metab 18(12): 129-137.
- Masserini M (2013) Nanoparticles for Brain Drug Delivery.
- Honjo K, Black SE, Verhoeff NP (2012) Alzheimer’s disease, cerebrovascular disease, and the β -amyloid cascade. Can J Neurol Sci 39(6): 712-728.
- Montet X, Funovics M, Montet-Abou K, Weissleder R, et al. (2006) Multivalent effects of RGD peptides obtained by nanoparticle display. J Med Chem 49(20): 6087-6093.






























