Bioorganism Combination Method for Platinium Nanoparticles
Imtiyaz Rasool Parrey1and Rayees Ahmad Zargar2*
1Department of Chemistry, Govt Degree College Doru, India
2Department of Physics, Islamic University of Science and Technology, India
Submission: September 20, 2017; Published: January 18, 2018
*Corresponding author: Rayees Ahmad Zargar, Department of Physics, Islamic University of Science and Technology, India, Email: rayeesphy12@gmail.com; imitiyazchemistry@gmail.com
How to cite this article: Imtiyaz R P, Rayees A Z. Bioorganism Combination Method for Platinium Nanoparticles. Glob J Nano. 2018; 3(4): 555619. DOI: 10.19080/GJN.2018.03.555619
Abstract
Platinum (Pt) nanoparticles have been combined from an antecedent arrangement of potassium tetrachloroplatinate (K2PtCl4) utilizing a network of bacterial cellulose (BC). The arrangement of Pt nanoparticles happens at the surface and within the BC film by diminishing the antecedent arrangement with a hydrogen gas reductant. The Pt nanoparticles got from the varieties of antecedent focus, in the vicinity of 3mM and 30mM, and the arrangement of Pt nanoparticles have been considered utilizing X-beam diffraction (XRD), checking electron microscopy- vitality dispersive X-beam spectroscopy (SEM-EDS), and thermogravimetry examination (TGA).
Keywords: X-beam spectroscopy; Bacterial cellulose; X-beam diffraction; Thermogravimetry examination; Platinum
Introduction
Platinum (Pt) has pulled in much consideration since Pt is the best impetus to use for an assortment of particular purposes. As a respectable metal, Pt has the best synergist movement among every single unadulterated metal, particularly in power devices [1] . Many components influence the electrocatalytic action of Pt particles, for example, the size, appropriation, and combination strategy [2,3]. The reactant movement of Pt is better when nanosized Pt particles are created. A nanosized Pt impetus that is dispersed legitimately will bring about high electrocatalytic movement [1,4,5]. Among these strategies, the combination of Pt nanoparticles utilizing the lessening technique is an exceptionally straightforward strategy. This strategy is finished by lessening the antecedent arrangement with a decreasing operator. The antecedents that are regularly utilized as a wellspring of Pt particles are H2PtCl6 and the decreasing specialists, that are likewise ordinarily utilized, are formaldehyde, Lithium triethylborohydride, sodium borohydride and ethylene glycol [6-9]. Yang et al. [10] have integrated Pt nanoparticles from Pt (IV) arrangement, to be specific, H2PtCl6, into BC framework utilizing sodium borohydride and a formaldehyde reductant. They revealed that the Pt molecule sizes of 3-4 nm are created and connected as impetuses for energy components. This exploration has motivated us to orchestrate Pt nanoparticles utilizing forerunner Pt (II) rather than Pt (IV). BC has been known as a grid that integrates a portion of the nanometals, for example, palladium and silver. BC has the capacity of a lattice in light of the fact that the fiber structure of BC has a cavity that is utilized as a minireactor for the amalgamation of nanoparticles [11].
Materials
Potassium tetrachloroplatinate (II) (K2PtCl4) was gotten from Sigma-Aldrich (99,9%), while ammonium sulfate, frigid acidic corrosive, hydrochloric corrosive, sodium hydroxide, and ethanol were acquired from Merck. Hydrogen gas (ultra- profoundly unadulterated) was acquired from a neighborhood provider. Coconut water, sucrose (sustenance review white sugar), and Acetobacterxylinum were gotten from a nearby customary market. All chemicals were utilized without encourage filtration.
Preparation of BC
Readiness of BC was completed as depicted already by Radiman & Yuliani. To acquire dry BC layers, the BC was first sliced to a size of 4cm x 4cm, at that point squeezed to evacuate water, lastly air dried at room temperature for 6 days. Dried BC film was utilized for examination by SEM-EDS.
Synthesis of Pt Nanoparticles Impregnated into BC
The squeezed BC hydrogels were absorbed fluid K2PtCl4 arrangements, with various focuses (3mM, 5mM, 10mM, 20mM, and 30mM), for 2hrs and sonicated at room temperature for another 2hrs. The BC gel was evacuated, flushed with deionized water, and absorbed again deionized water. The Pt (II) was then decreased with hydrogen gas at a connected steady weight of 0.5 psi and a mixing pace of 500rpm at room temperature for 1hr. Pt-BC gel was expelled from the arrangement and dried, as in the above technique, and created a dry composite layer that is meant by Pt-BCcm. The thickness of the subsequent Pt-BCcm film was 0.5-0.7mm
XRD Analysis
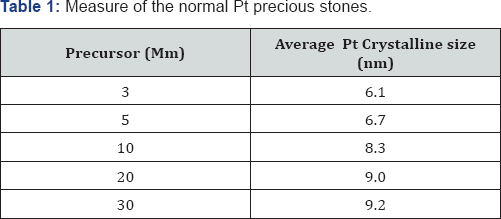
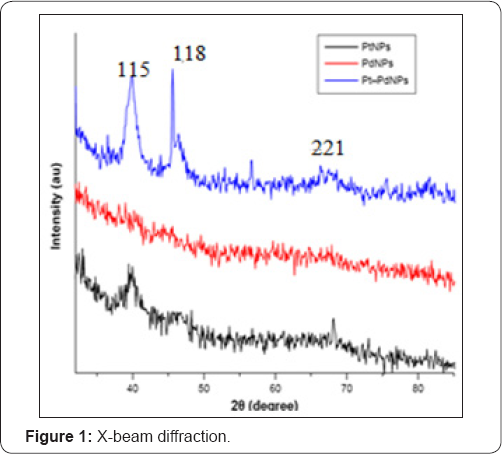
Pt nanoparticles have been effectively incorporated from fluid K2PtCl4 arrangement inside the BC layer utilizing hydrogen gas as a lessening specialist. The proof of Pt nanoparticles in Pt-BCcm layer is appeared by the X-beam diffraction range, in Figure 1. X-beam diffraction designs additionally show that, other than the trademark Pt tops, trademark pinnacles of BC in the zone 2θ=14.2°, 16.6°, and 22.4° likewise show up. A BC top showed up in every Pt-BCcm film. BC diffraction designs were like those done by past analysts [12-17]. Be that as it may, expanding the grouping of the Pt antecedent declines the pinnacle force of the BC crest zones, particularly the range at 2θ=14.2° and 16.6°. The Pt (111) top is utilized as a source of perspective in ascertaining the span of the normal Pt precious stone since its power is the most noteworthy among alternate pinnacles. Table 1 demonstrates the measure of the normal Pt precious stones which were figured from FWHM (111) crest utilizing the Scherrer recipe.
SEM Observation
The arrangement of Pt particles on the BC layer is outwardly seen through a shading change in the BC film. The BC layer was initially straightforward white however it changed to dark when Pt was embedded in the BC film. The difference in layer to dark shows that Pt (II) particles have been effectively lessened and kept on the filaments of BC. Be that as it may, there were no conspicuous shading contrasts among the composites acquired by various convergences of the antecedent.
Perception at first glance morphology of the Pt-BCcm layer demonstrated that white granules were installed in the BC film and that the grains are Pt particles. White grain shows that Pt particles have been framed and kept in the pit of the BC network. Testimony of Pt particles shaped agglomerations or groups at a specific level. At low antecedent focus, just a couple of Pt particles were obvious, yet at higher forerunner fixations, the measure of Pt particles was progressively and had a bigger size, demonstrating that framed agglomerations are extensive. SEM pictures of the surface morphology on the Pt-BCcm layer can be found in Figure 2.


In light of SEM pictures, the Pt molecule estimate appears to be expansive in view of the event of agglomeration. In the interim, an examination by utilizing X-beam diffraction designs demonstrates that the span of the Pt precious stones is nanoscale for all antecedent fixations. This distinction is because of the X-beam diffraction design examination that depicts the extent of the Pt precious stones contained in Pt particles, since Pt particles are shaped from accumulations of Pt gems. Thusly, the two examinations appear to be unique in dissecting the Pt molecule estimate. The arrangement of Pt particles shows up on the BC film surface as well as within the BC fiber. Pt particles are framed within the BC strands; this is known from the crosssectional SEM pictures of the Pt-BCcm film. The nearness of Pt particles within the BC filaments likewise demonstrates that the antecedent arrangement went into the internal BC strands and communicated with BC cellulose. Accordingly, when lessening happens, Pt particles are caught within (between the strands) of BC, as appeared in Figure 3.
Conclusion
Pt nanoparticles have been blended from the K2PtCl4 arrangement utilizing H2 as a lessening operator. The nearness of Pt particles has been exhibited through the examination of X-beam diffraction and BC film staining, from straightforward white to dark. The nearness of Pt nanoparticles is found on the film surface, as well as within the Pt-BCcm layer. In light of SEM pictures, the Pt molecule measure increments with an expanding antecedent fixation, from nanometers to micrometer estimate. Be that as it may, in light of the XRD investigation that utilizations Scherrer condition.
References
- Lim DH, Lee WD, Lee HI (2008) Highly dispersed and nano-sized Pt- based electrocatalysts for low-temperature fuel cells. Catalysis Surveys from Asia 12(4): 310-325.
- Takasu Y, Ohashi N, Zhang XG (1996) Size effects of platinum particles on the electroreduction of oxygen. ElectrochimicaActa 41(16): 25952600.
- Bergamaski K, Pinheiro ALN, Teixeira-Neto E, Nart FC (2006) Nanoparticle size effects on methanol electrochemical oxidation on carbon supported platinum catalysts. The Journal of Physical Chemistry B 110(39): 19271-19279.
- He Z, Chen J, Liu D, Tang H, Deng W, et al. (2004) Deposition and electrocatalytic properties of platinum nanoparticals on carbon nanotubes for methanol electrooxidation. Materials Chemistry and Physics 85(2-3): 396-401.
- Rao CV, Viswanathan B (2010) Monodispersed platinum nanoparticle supported carbon electrodes for hydrogen oxidation and oxygen reduction in proton exchange membrane fuel cells. Journal of Physical Chemistry C 114(18): 8661-8667.
- Zhou Z, Wang S, Zhou W, Weijiang Z, Luhua J, et al. (2003) Preparation of highly active Pt/C cathode electrocatalysts for DMFCs by an improved aqueous impregnation method. Physical Chemistry Chemical Physics 5(24): 5485-5488.
- Şen F, Gökağaf G (2007) Different sized platinum nanoparticles supported on carbon: an XPS study on these methanol oxidation catalysts. The Journal of Physical Chemistry C 111(15): 5715-5720.
- Maiyalagan T, Alaje TO, Scott K (2012) Highly stable Pt-Ru nanoparticles supported on three-dimensional cubic ordered mesoporous carbon (Pt-Ru/CMK-8) as promising electrocatalysts for methanol oxidation. The Journal of Physical Chemistry C 116(3): 26302638.
- Wu Y, Liao S, Wang K, Chen M, Birss V (2010) High pressure organic colloid method for the preparation of high performance carbon nanotube-supported Pt and PtRu catalysts for fuel cell applications. Science in China Series E: Technological Sciences, vol. 53(1): 264-271.
- Yang J, Sun D, Li Jet al. (2009) In situ deposition of platinum nanoparticles on bacterial cellulose membranes and evaluation of PEM fuel cell performance. Electrochimica Acta 54(26): 6300-6305.
- Evans BR, O'Neill HM, Malyvanh VP, Lee I, Woodward J (2003) Palladium-bacterial cellulose membranes for fuel cells. Biosensors and Bioelectronics 18(7): 917-923.
- Pileni MP (1997) Nanosized particles made in colloidal assemblies. Langmuir 13(13): 3266-3276.
- Wu SH, Chen DH (2003) Synthesis and characterization of nickel nanoparticles by hydrazine reduction in ethylene glycol. Journal of Colloid and Interface Science 259(2): 282-286.
- Sanchez-Dominguez M, Boutonnet M, Solans C (2009) A novel approach to metal and metal oxide nanoparticle synthesis: the oil- in-water microemulsion reaction method. Journal of Nanoparticle Research 11(7): 1823-1829.
- Suffredini HB, Salazar-Banda GR, Avaca LA (2009) Carbon supported electrocatalysts prepared by the sol-gel method and their utilization for the oxidation of methanol in acid media. Journal of Sol-Gel Science and Technology 49(2): 131-136.
- Angelucci CA, D'Villa SM, Nart FC (2007) Preparation of platinum- ruthenium alloys supported on carbon by a sonochemical method. Electrochimica Acta 52(25): 7293-7299.
- Sieben JM, Duarte MEE, Mayer CE (2008) Supported Pt and Pt-Ru catalysts prepared by potentiostatic electrodeposition for methanol electrooxidation. Journal of Applied Electrochemistry 38(4): 483-490.






























