A Comparative Study of Thermodynamic Properties 0f Pure Graphene and H-Bn Sheet Focusing on Contribution from Planar and Flexural Modes
Sarita Mann1*, Pooja Rani2and Ranjan Kumar1
1Department of Physics, Panjab university, India
2Dept. of Physics, MM Modi college, India
Submission: December 09, 2017; Published: January 12, 2018
*Corresponding author: Sarita Mann, Department of Physics, Panjab university, India, Email: saritaiitr2003@gmail. com
How to cite this article: Sarita M, Pooja R, Ranjan K. A Comparative Study of Thermodynamic Properties 0f Pure Graphene and H-Bn Sheet Focusing on Contribution from Planar and Flexural Modes. Glob J Nano. 2018; 3(4): 555618. DOI: 10.19080/GJN.2017.03.555618.
Abstract
Graphene, a 2D honeycomb lattice structure of carbon is widely studied due to its unique electronic and remarkable thermal properties such as negative thermal expansion and extremely high thermal conductivity. The h-BN sheet is another 2D material with same crystal structure having entirely different electronic properties but similar thermodynamic propertiesas compared to graphene. We have studied earlier that the flexural acoustic mode has a major contribution in negative behavior of thermal expansion in grapheneand h-BN sheet too. In view of further understanding the unique behavior of various thermodynamic properties like heat capacity, entropy and free energy of these 2D materials, we havecompared these properties for the two and also focused specifically on the individual contribution of planar and flexural modesin the temperature range of 0-1500K.
The study is performed using ab-initio density functional perturbation theory (DFPT) employed in VASP code. Then dynamical matrix and force constants are calculated in interface with phonopy software. The calculation of branch dependent various thermodynamic properties suggests that it is the transverse acoustic mode which has major contribution in dictating these properties at low temperatures (<300K) in graphene as well as h-BN sheet. The extensive study of comparison of specific heat, entropy and free energy for graphene and h-BN sheet and their branch dependence are reported here for the first time.
Introduction
Graphene is the name given to a monolayer of graphite which was discovered in year 2004 when few layered graphene sheet was mechanically exfoliated from pyrolytic graphite by KS Novoselov & AK Geim [1]. It was termed as a semiconductor with zero band gap having extremely high electron mobility and unusual thermal properties so it attracted great interest of scientific researchers with a number of research articles and reviews published every year signifying its various remarkable properties like mechanical, electronic, optical properties [2,311]. Although a lot of research is ongoing in the area of thermal expansion and unsually high thermal conductivity of graphene [12-17].
But in-depth study of thermal behaviour of various thermodynamic properties of graphene is still in growing state. The h-BN sheet is another interesting 2D material having same hexagonal honeycomb lattice structure as graphene although electronic band structure is quite different with h-BN having a large band gap of 5-6 eV compared to zero band gap of graphene [18] . Despite differences in electronic properties, thermal expansion behavior of these materials follow a similar trend [19] . Inthe present paper we have made an effort to investigate and compare various thermodynamic properties of these materials and emphasizing on contribution from fexural and planar modes.
Earlier we have studied phonon dispersion and various thermodynamic properties of pure graphene and thermal expansion behavior [12]. In an attempt to study the reason behind negative thermal expansion behavior of graphene, the out of plane flexural acoustic mode (ZA) mode contribution was separated from other modes contribution. ZA mode has largely negative Grüneisen parameters near r point which contributes to negative LTEC near room temperature [19]. It is the effect of negative Gruneisen parameter of ZA mode near r point which makes it to behave entirely different from other modes of vibration. Among all other modes, the in-plane modes (longitudinal and transverse acoustical and optical modes) have positive Gruneisen parameters while ZO mode (out of plane optical mode) also shows negative Gruneisen parameter but its value is very small as compared to the value for ZA mode. Thus it is only the ZA mode whose contribution dictates the negative thermal expansion behavior in pure graphene at low temperatures. Similar behavior was obtained for h-BN sheet having largely negative Gruneisen parameter corresponding to ZA mode which produces more negative thermal expansion in h-BN sheet compared to pure graphene.
In continuation of the above mentioned studies, in the present paper we have compared thermodynamic properties of two materials and focused on the role of ZA mode in contributing to various thermodynamic properties. There is only one reported theoretical work on mode dependence of specific heat [20] for graphene while no comparison or mode dependence on entropy and free energy has been reported till date.
Theory and Computational Details
The phonon frequencies and Gruneisenparameters are calculated using ab-initio density functional theory implemented in VASP code in combination with phonopy code [21-23]. A 4x4 supercell of graphene is used with a plane wave cut off energy of 750eV and a separation of 12A between layers is maintained. The generalized gradient approximation is used under Perdew- Burke-Ernzerhof (PBE) exchange correlational functional. The monkhorst pack gamma centered k-mesh of 7x7x1 is used for brilluion zone sampling. The structure is fully relaxed until the Hellmann-Feynman forces were less than about 0. 005 eV/A. The heat capacity per unit cell and entropy (S) is obtained by summing the contribution of all the phonon branches and separately for ZA branch alone as given inthe expressions below [24].
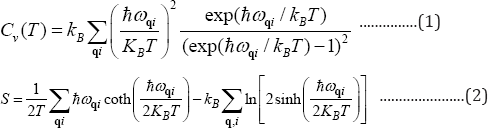
In these expressions, ωqi are the phonon frequencies for wave vector q and phonon branch index i.
The free energy expression [24] used for calculating total and ZA mode free energy is given as
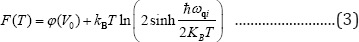
Where Φ(V0) is the ground state energy.
Results and Discussion
A comparative plot of total specific heat, entropy and free energy of pure graphene and h-BN sheet in the temperature range of 0-1500K using the expressions given in equations (1), (2) and (3) is shown in Figure 1. Since melting point of graphene is around 4000K and h-BN is around 3000K, for our study we have selected the range of temperature to 0-1500K in which contribution of anharmonic terms to phonon frequencies can be neglected. The specific heat of pure graphene has earlier been studied and found in good agreement with other theoretical and experimental studies [12]. Using the similar approach for h-BN sheet, we have found the variation in specific heat with temperature and compared with the pure graphene.

The specific heat curve is found to be linear in the low temperture region contrary to specific heat for pure graphene at low temperatures. The value of specific heat is higher for h-BN sheet at all temperatures and reaches a constant value of 23.3J/K/mol slightly higher than graphene in accordance with dulong-petit limit of 25J/K/mol. The comparison of entropy and free energy curves also shows that the values for h-BN sheet are higher as compared to pure graphene with same trend and same range of values. Thus thermodynamic properties of both the materials are closely related to each other and when compared with doped graphene, it follows naturally that increasing the doping of pure graphene sheet with B and N dopants increases the values of these thermoynamic properties [12] and further increases for h-BN sheet which can be obtained by replacing both the carbon atoms of unit cell of graphene with boron and nitrogen.
After compairing the thermodynamic properties of these materials, we studied the branch dependence of these properties for pure graphene and h-BN sheet. Earlier studies on in-plane and out of plane mode contribution to specific heat [20] of pure graphene shows that out of plane and in-plane contributions are nearly same at high temperatures(>1000K). Below 200K, only ZA mode contributed to specific heat while between 200-500K, both in-plane and out of plane contributions are dominant. We have attempted to separate the ZA mode contribution from total specific heat. Figure 2a clearly indicates that near room temperature ZA mode contribution is almost half of the total specific heat. Below 50K, the specific heat is contributed only by ZA mode and as temperature increases, the other modes also start contributing and overall specific heat value increases and starts deviating from ZA branch with increase in temperature.

While specific heat reaches towards a constant value at high tempertures, the contribution of ZA mode is almost one fifth part of the total value of specific heatat high temperatures. This implies that ZA mode contribution overpowersthat of other modesat low temperatures, since at low temperatures only acoustic modes are excited, of which ZA (flexural) mode dominates the other two modes (longitudinal and transverse acoustic). We have calculated the contribution of each branch separately and the overall specific heat matches well with theory [5,25] and experiment [26]. The specific contribution of various branches to specific heat is the least studied area.
The entropy of graphene is not studied precisely till date with a contribution from each phonon mode separately calculated so that the contribution of most dictating branch i.e. ZA branch stands out specifically at low temperatures. The specific heat and entropy curves show a similar trend with ZA mode dictating the low temperature and approaching towards almost one fifth contributions as temperature reaches the value of 1000K. The entropy curve shows a linear behaviour at high temperatures which is expected since entropy of any material increases with increase in temperature (Figure 2b).
The variation of total free energy with temperatureis compared with its ZA mode contribution in Figure 2c. It shows that the contribution of ZA mode to free energy is minimum at low temperatures and reduces to negative value as the temperature rises. At high temperatures beyond 1400K the contribution of ZA mode diminishes to largely negative value. This is unusual as ZA mode contribution becomes higher than total free energy above this point. This is due to negative contribution from other modes also lowering the total free energy of the system. As the temperature is increased, free energy of the system keeps on decreasing and the ZA mode has dominant effect in decreasing the overall free energy.
In continuation of this study, the branch dependence of all modes for h-BN sheet has also been studied and reported here. Figure 3 shows the contribution of each mode separately contributing to these thermodynamic properties. In Figure 3a, the variation of specific heat with temperature is shown. As can be seen in the Figure, ZA mode has highest contribution while optical modes have lowest contribution at low temperatures; whiles at high temperatures all the modes contribute almost equally to reach a constant value. Since at low temperatures only acoustic modes are excited, the contribution from optical modes is negligible.
Similarly Figure 3b shows the variation of entropy with temperature, where ZA mode contribution is highest at all temperatures which stands out from all other modes. Entropy does not reach a constant value and keeps on increasing with temperature. Figure 3c shows the variation of free energy with temperature. Similar to behaviour in pure graphene, here ZA mode contribution is lowest and continues to decrease with temperature reaching even negative values at high temperatures. This is interestingly different behaviour when the total free energy crosses all other modes contribution at high temperatures. Thus it becomes clear from the curve that ZA mode has highest contribution in decreasing the total free energy of the structure as its value is largely negative at high temperatures.
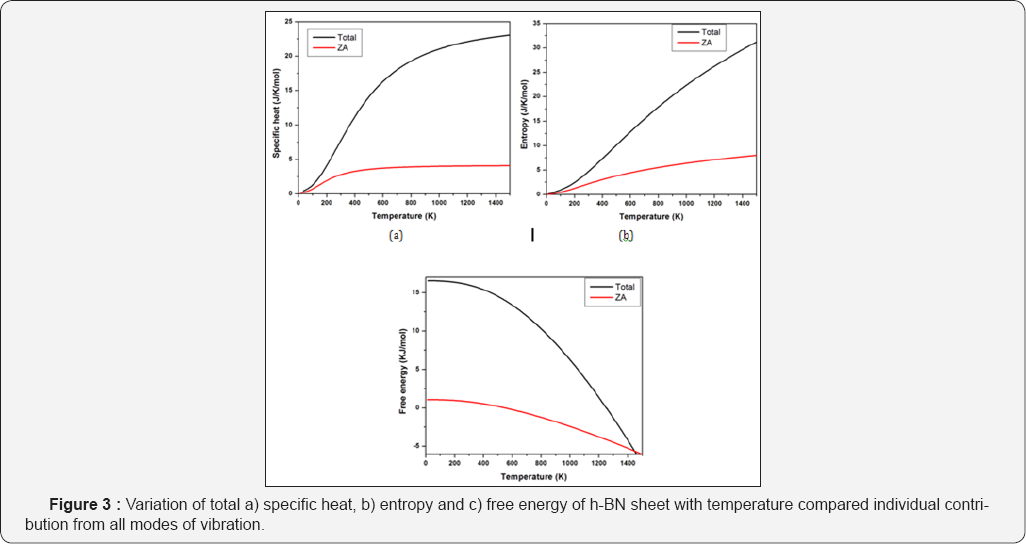
We have made an comparative study of thermodynamic properties of pure graphene and h-BN sheet. The behaviour of two materials are quite similar in thermal and thermodynamic properties with h-BN sheet having slightly higher values of specific heat and entropy while lower values of free energy as compared to pure graphene. The analysis of contribution of ZA mode separately to total thermodynamic properties in graphene shows that ZA mode has significant contribution to each property. Although all the modes of vibration contribute almost equally at high temperaturesas can be seen in the extensive study of all modes contribution in h-BN sheet towards various thermodynamic properties but the effect of ZA mode is highest among them dominantly at low temperatures. The ZA mode contribution is highest to entropy and specific heat while its contribution is minimum in free energy suggesting maximum contribution in reducing the free energy of the system. The ZA mode contribution to various thermal properties of graphene and h-BN sheet stands out from other modes contribution which can be utilised in temperature dependent applications by retstricting or allowing out of plane vibrations in these materials.
Acknowledgement
We are grateful to VASP and PHONOPY team for the code and IUAC for computational support and the computing facilities at Department of Physics, Panjab University, Chandigarh.
References
- Novoselov KS, Geim AK, Morozov SV, Jiang D, Zhang Y (2004) Electric field effect in atomically thin carbon films. Science 306(5696): 666669.
- Bolotin KI, Sikes KJ, Jiang Z, Klima M, Fudenberg G et al. (2008) Ultra- high electron mobility in suspended graphene. Solid State Commun 146: 351-355.?
- Ghosh S, Calizo I, Teweldebrhan D, Pokatilov EP, Nika DL, et al. (2008) Extremely high thermal conductivity of graphene: Prospects for thermal management applications in nanoelectronic circuits. Appl Phys Lett 92: 151911.
- Yoon D, Son YW, Cheong H (2011) Negative thermal expansion coefficient of graphene measured by Raman spectroscopy. Nano Lett 11(8): 3227-3231.
- Mounet N, Marzari N (2005) First-principles determination of the structural, vibrational and thermodynamic properties of diamond, graphite, and derivatives. Phys Rev B71: 205214.
- Reich S, Thomsen C (2002) Elastic properties of carbon nanotubes under hydrostatic pressure. Phys Rev B65: 153407.
- Kalosakas G, Lathiotakis NN, Galiotis C, Papagelis KJ (2013) In-plane focus field and elastic properties of graphence. Appl Phys 113: 134307.
- Geim AK (2009) Graphene: status and prospects. Science 324(5934): 1530-1534.
- Neto Castro AH, Guinea F, Peres NMR, Novoselov KS, Geim AK (2009) The electronic properties of graphene. Rev Mod Phys 81: 109-162.
- Rani P, Jindal VK (2013) Designing band gap of graphene by B and N dopant atoms J RSC Adv 3: 802.
- Rani P, Dubey GS, Jindal VK (2014) Physica E: Low-dimensional Systems and Nanostructures. Physica 62: 28.
- Sarita M, Rani P, Ranjan K, Dubey GS, Jindal VK (2016) Thermodynamic properties of pure and doped (B, N) graphene RSC Adv 6: 12158.
- Pan W, Xiao J, Zhu J, Yu C, Zhang G, et al. (2012) Biaxial compressive strain engineering in graphene/boron nitride heterostructures. Sci Rep 2: 893.
- Magnin Y, Forster GD, Rabilloud F, Calvo F, Zappelli A, et al. (2014) Thermal expansion of free-standing graphene: benchmarking semi-empirical potentials. J Phys Condens Matter 26: 185401.
- Calizo I, Balandin AA, Bao W, Miao F, Lau CN (2007) Temperature Dependence of the Raman Spectra of Graphene and Graphene Multilayers. Nano Lett 7 (9): 2645-2649.
- Nika DL, Pokatilov EP, Askerov AS, Balandin AA (2009) Phonon thermal conduction in graphene: Role of Umklapp and edge roughness scattering. Phys Rev B79: 155413.
- Lindsay L, Broido DA, Mingo N (2010) Flexural phonons and thermal transport in graphene. Phys Rev B82: 115427.
- Golberg D, Bando Y, Huang Y, Terao T, Mitome M, et al. (2010) Boron nitride nanotubes and nanosheets. ACS Nano 4(6): 2979-2993.
- Mann S, Kumar R, Jindal VK (2017) Negative thermal expansion of pure and doped graphene. RSC adv 7: 22378-22387.
- Cocemasov A, Nika DL, Balandin AA (2015) Engineering of the ther-modynamic properties of bilayer graphene by atomic plane rotations: the role of the out-of-plane phonons. Nanoscale 7(30): 1285112859.
- Hohenberg P, Kohn W (1964) Inhomogeneous Electron Gas. Phys Rev B136: 864.
- Kresse G, Hafner J (1993) ab initio molecular dynamics for liquid me-tals. Phys Rev B47: 558.
- Atsushi T, Fumiyasu O, Isao T (2008) First-principles calculations of the ferroelastic transition between rutile-type and CaCl2-type SiO2 at high pressures. Phys Rev B78: 134106.
- Jindal VK, Kalus J (1983) A calculation of the anharmonic phonon frequencies in solid deuterated naphthalene-d8. Phys J C Solid State Phys16: 3061-3080. 8
- Eric Pop, Vikas V, Ajit KR (2012) Thermal properties of graphene: Fundamentals and applications. MRS Bulletien 37(12): 1273-1281.
- D Gray, McCaughan A, Mookerji B (2009) Crystal structure of graphite, graphene and silicon. Physics For Solid State Applications 6: 730.






























