Applicationof Pulsed Arc Electrohydraulic Discharges for the Synthesis of PVA and Dextran coated Magnetic nanoparticles
Mikelashvili V*, Sh Kekutia, Markhulia J and Saneblidze L
Vladimir Chavchanidze Institute of Cybernetics of the Georgian Technical University, Georgia
Submission: October 30, 2017; Published: December 12, 2017
*Corresponding author: Mikelashvili V, Vladimir Chavchanidze Institute of Cybernetics of the Georgian Technical University, Georgia, Tel: +995593675586; Email: vmikelashvili@gtu.ge
How to cite this article: Mikelashvili V, Sh Kekutia, Markhulia J, Saneblidze L. Applicationof Pulsed Arc Electrohydraulic Discharges for the Synthesis of PVA and Dextran coated Magnetic nanoparticles. Glob J Nano. 2017; 3(3): 555613. DOI: 10.19080/GJN.2017.03.555613.
Abstract
The electrohydraulic processing after chemical co-precipitation technique of magnetite (Fe3O4) nanofluids, for better dispersity, stabilization and homogenous size distribution is proposed. Biocompatible polyvinyl alcohol (PVA) and Dextran coated 10-20nm sized superparamagnetic iron oxide nanoparticles (SPIONs) have been synthesized via co-precipitation method in the vacuum environment with pH«7.4 and solid phase content ranging from 0.02-0.75 % w/v. Scanning electron microscopy (SEM) and Transmission Electron Microscopy (TEM), also UV-VIS spectroscopy, Dynamic light scattering (DLS) and Vibrating Sample Magnetometer (VSM) was used to characterize obtained samples.
Keywords: Magnetic nanofluids; Homogenization; Dispersity; Electrohydraulic discharge
Abbreviations: SPIONs: Superparamagnetic Iron Oxide Nanoparticles; SEM: Scanning Electron Microscopy; VSM: Vibrating Sample Magnetometer; UV-VIS: Ultraviolet-Visible; PVA: Polyvinyl Alcohol; MRI: Magnetic Resonance Imaging; NP: Nanoparticle; MNPs: Magnetic Nanoparticles
Introduction
The union of scientific achievements in Nanotechnology and biotechnology led to a new discipline-Origin of nano biotechnology, the development of which promises to create nano constructions able to manage biosystems in a molecular level. The revealing unique properties (changes in their physical and chemical properties) of the substance in a nanoscale provide a broad perspective of using them in different directions. Energetically, the reduction of particle size leads to incrisement of surface energy share in the chemical potential of small particles under different conditions (Coordination number, local surrounding symmetry and so on) than atoms in a volume crystal [1], which allows them to operate effectively with any chemical bonds. In recent years, much attention is paid to study nanoscale magnetic particles, such asFe3O4, γ-Fe2O3, CoFe2O4, ZnFe2O4, BaFe12O19. Among which magnetite (Fe2O3) and maghemite (γ-Fe2O3) are very popular candidates with their biocompatibility in "in-vivo" applications such as contrast agents for Magnetic Resonance Imaging (MRI), Tissue repair, immune analysis, biological fluids detoxification, targeted delivery of therapeutic agents, hyperthermia and cell separation. As well as in the research of specific bio objects, such as bacteria, white blood cells and proteins [2,3].
To construct such nanostructures it is actual the technological task that involves controlling the nanoparticle (NP) size, shape, stability and dispersity in the desired solvents. Magnetic nanoparticles (MNPs) have a big surface / volume ratio that is why they are characterizing by high surface energy; as a result, they exhibit aggregation ability, in order to minimize the surface energy. In addition, bare iron oxide NPs characterized by high chemical activity and easily oxidize in the air (especially magnetite), which generally leads to the deterioration of magnetism and dispersity. Thus, it is important to cover the NP surface with appropriate coating and evaluate an effective strategy to improve fluid stability.
The most widely known method to synthesize Fe3O4 and γ-Fe2O3NPs is a chemical co-precipitation [4]. In this method, the metal oxide NP size and shape is depending on the type of salts used (namely, Chlorides, sulfates, nitrates, etc.), the trivalent and divalent metal ions ratio, reaction temperature, pH value, environment ionic strength and other parameters of the reaction (for example, mixing speed, the sedimentation rate of basic solution). However, this method of improving the dispersion requires refinement, which is essential for biomedical use. These strategies generally include grafting or modification with organic molecules, including small organic molecules or surface- active substances, polymers and biomolecules, or covering by inorganic layer, such as silicon dioxide, metal or non-metals, metal oxides, metal sulfides [5]. Should be noted that protective coat not only stabilize iron oxide NPs, but they can be used for further functionalization.
Different types of synthesis techniques are used for the synthesis of magnetite nanomaterials such as bottom- up approach, chemical precipitation technique , thermal decomposition of organic iron precursor in organic solvents, polyol process, sol-gel method, sonochemical synthesis, solvothermal synthesis, hydrothermal synthesis, and emulsion technique and in top-down, ball milling, etc. Among them the most widely known method for synthesize SPIONs is a chemical co-precipitation. In this method, the synthesized size and shape of nanoparticles (NPs) is depending on the type of salts used, their molar ratio, reaction temperature, pH value, environment ionic strength, mixing speed and other parameters [6-19].
While these batch processes have the advantage of simplicity, cheapness and mass production, the growing need for emerging nanotechnology is the monodisperse nanoparticles. The problem lies in the imperfections of the batch mixing technology [20-23]. This technique is known by its difficulties in controlling the mixing and the temperature synthesis, which result in a broad distribution of particle sizes and polydispersivity, due to the instability of the growing process of NPs. Besides, in order to functionalize synthesized MNPs, additional procedures are needed, (washing, reducing pH, change the vessel, de- agglomerating before covering the surfaces, etc.) which drives to the oxidization of the particles and lowering adsorbtion ability for surfactation (stabilization).
Problem solving lies in improving the existing method of the synthesis of stabilized magnetite nanoparticles using in situ synthesis with pulsed electrohydraulic discharges during chemical co-precipitation in order to better disperse the formed nanoparticles at the initial stage, process their surface (static stabilization, H and OH radical addition for better adsorbance of the surface) by pulsed discharges and gain to the fluid the bactericidal properties [24,25] that is necessary in the case of biomedical applications. After that, the covering (stabilizing) of the monodisperse nanoparticles with surfactant is relatively easy to follow, with bioactive molecules, followed by washing from chemical reaction residuals, additional ultrasound homogenization and centrifugation.
During recent years plasma systems in liquid such as waste water treatment and purification, or for simple low cost production of nanoparticles have become of topical interest. Arc discharge methods in liquids have been used for direct production of metal particles, metal nitrides/carbides/oxides and carbon nanotubes etc., but, electrohydraulic discharges after or during co-precipitation of iron oxide nanoparticles has not ever been reported [26-34].
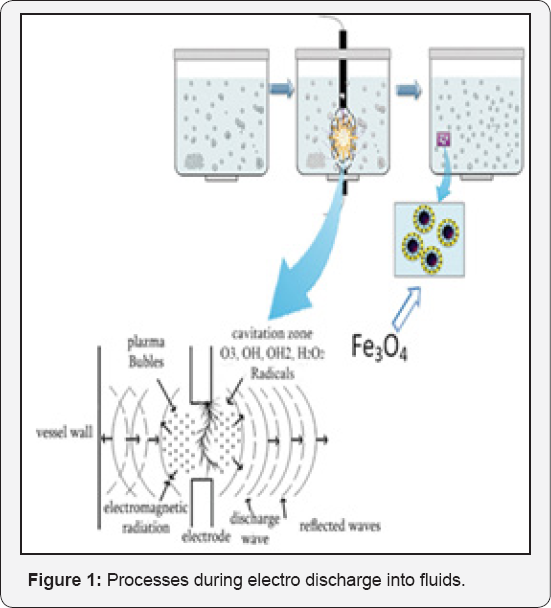
The essence of this method lies in performing high voltage discharges between electrodes in the open open or closed vessel/ reactor with special electric impulse. Complex physical and chemical phenomena, which progress instantly leads to various physicochemical changes (The process generates UV radiation, hydrogen peroxide, hydrogen, oxygen and other radicals and ions) in the liquid and objects placed on the processed fluid (Figure 1) [35].
The schematic picture of plasma discharging device presented in (Figure 2) consists of power transformer T2 (U = 3kV), rectifier diodes D1-D4, condenser C, air gap - Gap1 and discharge gap - Gap 2. The energy emission characterized to the plasma discharge method realized by active resistance of the contour:
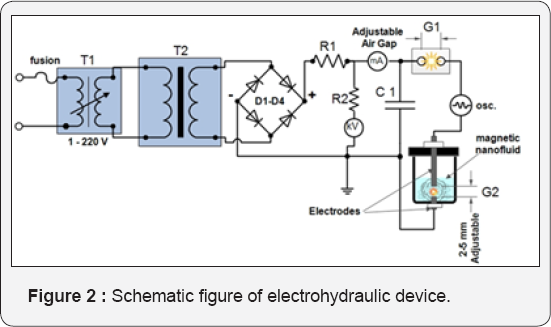

Where C is the capacitance of the condenser, R and L-active resistance and active inductance, respectively.
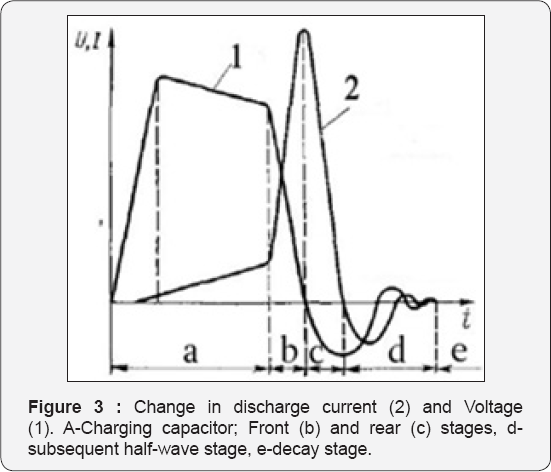
GAP1 allows collecting a certain amount of energy from the condenser C to avoid a bridged discharge and create a sharp front of the impulse, (Figure 3, curve 2). By adjustment of the length of the GAP 1 and GAP 2, we can change the shape of the impulse and nature of the discharge in the main processing area and thus control this device. As shown in current coltage diagram (Figure 3) the sharp front of current (curve 2) determines the power of discharge and effectiveness. Discharging rods must be non metal (for example carbon) in order to avoid contamination info fluid. Non metals can easily separate from magnetite by permanent magnet, OR should be made from pure iron.
Materials and Methods
All ofanalytic reagents were used without further purification. For the synthesis was used Iron (III) chloride hexahydrate (FeCl3•6H2O) (≥98%-Sigma-Aldrich-Germany), Iron (II) sulfate heptahydrate (FeSO4•7H2O) (≥99 %-Carl Roth Gmbh+Co. KG- Germany), Ammonium hydroxide solution (NH4OH) (≥25%-Carl Roth Gmbh+Co. KG-Germany), Poly (vinyl alcohol)-PVA (Carl Roth GMBH+Co. KG) and dextran (macrochem.ua).
Bare Fe3O4 nanoparticles
3.78g of FeSO4•7H2O and 7g of FeCl•6H2O (1:1.9 molar ratio of FeCl2/FeCl3) was diluted into 42ml distilled water at 400C with mechanical stirring (speed 650rpm) within 30min. When the iron salts were well dissolved, 15mL of a 0.75M NH4OH solution was added under magnetic stirrer (700rpm) during 30min at 400C in a vacuum (-0.15Mpa) environment. After that cooled down to room temperature within additional 30 minutes. The ferrofluid washed several times with water by decanting the supernatant in order to remove excess of chemical reaction residues and polymer, since reversible flocculation and rapid sedimentation observed before washing. This caused by the depletion attraction created by unabsorbed polyelectrolytes in solution [36]. Finally, we got pH=7.5. After that fluid was processed with electrohydraulic discharges and compared some properties with the same fluid before processing.
PVA-coated Fe3O4 nanoparticles
The chemical co-precipitation method was used for obtaining PVA-coated Fe3O4 nanoparticles. Firstly, 1gr PVA was diluted into 6ml distilled water by mechanical stirring at 600C during 30min. 3.78g of FeSO4•7H2O and 7g of FeCl3•6H2O (1:1.9 molar ratio of FeCl2/FeCl3) was diluted into 42ml distilled water at 400C with mechanical stirring (speed 650rpm) within 30 min. When the iron salts were well dissolved, 15mL of a 0.75M NH4OH solution was added under electrohydraulic discharges during 5 minute at 600C in a vacuum (-0.15Mpa) environment. After the addition of NH4OH, the suspension continued stirring on magnetic stirrer (700rpm) further 30min at 400C and cooled down to room temperature.
After synthesis fluid was placed into ultrasonic chamber and was processed 12min by 60% (total power 500wt) with regime (on 2sec, off 1sec) ultrasonic waves. In this period the firstly prepared 6.5ml PVA/water solution was added dropwise and after was additionally sonicated 30min with 50% of power, 2sec-on, lsec-off. During all ultrasound processing the fluid temperature did not exceeded 400C. After that, the ferrofluid poured into a beaker and placed on the permanent magnet. The ferrofluid washed several times with water by decanting the supernatant in order to remove excess of chemical reaction residues and polymer. Finally, we got pH=7.0. The final fluid was sonicated again and centrifuged at 3000rpm speed during 10min.
Dextran-coated Fe3O4 nanoparticles
0.16g of FeCl2•4H2O and 0.435g of FeCl3.6H2O (1:2 molar ratio) was diluted into 5ml distilled water by magnetic stirring in vacuum environment at 400C and added dropwise to 10mL of a 0.75M NH4OH solution in the electrohydraulic device chumber during 10 minutes following additional stirring on magnetic stirrer (speed 650rpm) within 30min. 3gr dextran was diluted into 3ml distilled water and added dropwise to magnetic fluid during Ultrasound processing (50%-power, 2sec-on, 1sec- off, temperature did not exceeded400C.) in 60 minutes. After synthesis, the ferrofluid washed several times, sonicated again and centrifuged at 3000rpm speed during 10min.
Results and Discussion
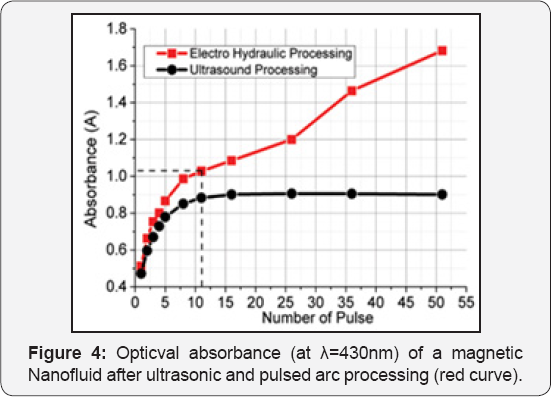
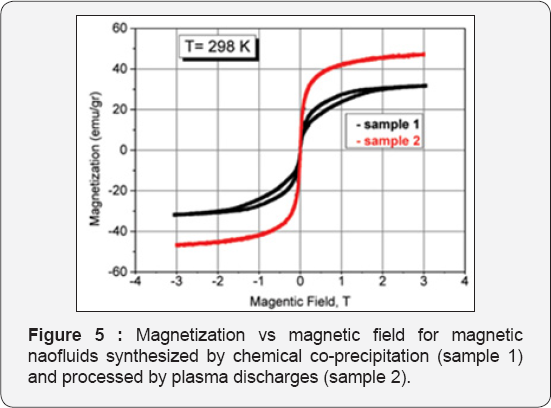
For the visibility of photo spectroscopy measurements the fluids with low concentration bare magnetite nanofluid were prepared, 0.1ml 4.6% magnetic nanofluid dissolved in 250ml. distilled water. The concentration of solution consisted 0.00184%. An Absorption in 430nm wavelength measured for 100ml samples in each case-after processing with ultrasound and electrohydraulic discharge (Figure 4)-with respect to processing pulse number/time. Concentration changes drastically after processing of sedimented nanoparticles in the liquid. In case of ultrasonic processing after certain pulse number absorbance goes through saturation. The change in particle concentration in the electrohydraulic processing relatively large, but do not saturates and continues to grow. We think it is associated with an increase free radicals and gas bubbles in the liquid, also particles ejected from rods during discharges. Figure 5 represents magnetic characterization of bare magnetite nanoparticles (sample 1) and after processing of pulsed arc discharges (sample 2).

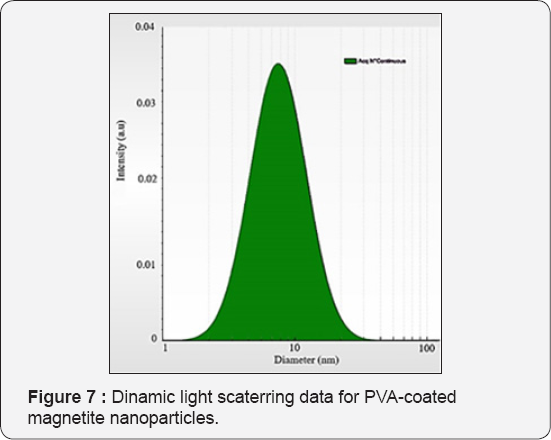
Scanning electron microscopy (SEM)used For powders (Figure 6a) of PVA-coated nanoparticles (Nivas Babu Selvaraj) and Transmission Electron Microscopy (TEM) of PVA stabilized magnetite nanoparticles analysis (Figure 6b) have been carried out with Hitachi High-Tech HT7700 instrument, operated in high resolution mode at 100kV accelerating voltage. Samples have been prepared by drop casting from diluted dispersions of nanoparticles on 300mesh holey carbon coated copper grids (Ted Pella) and vacuum dried (LaszloAlmasy). By the program ImajeJ we evaluated nanoparticles distribustion by size (Figure 6c) with comparision of Dinamic light Scaterring analysis nanoparticles mean diameter is 9-10nm (Figure 7).
UV-visible spectrometry can be used as a characterization technique that provides information on whether the nanoparticle solution has destabilized over time. The absorption of light, reflection, scattering and radiation of any material are highly dependent on the electrons of material, electrons' energy spectrum, and collective motion of the electrons into material. The last, the light induced collective vibration of electrons in the electrical field of lattice positive ions, described by quasiparticles called as Plasmon - a quantum of plasma oscillation that arises from the quantization of plasma oscillations.
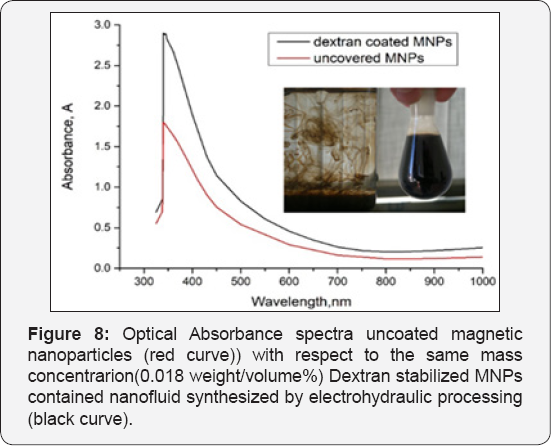
In the optical properties of materials, especially for metals plasmon plays an important role. The Incident light with lower frequencies than Plasmon frequency will not pass the material as the incident electromagnetic wave the faster moving electrons shielded. In addition, opposite, if the plasma frequency is lower than incident light, the electrons can no longer respond to the wave of change in the field and light passes through the material. The optical properties of magnetite nanoparticles change when particles aggregate and the conduction electrons near each particle surface become delocalized and shared amongst neighboring particles. When this occurs, the surface Plasmon resonance shifts to lower energies, causing the absorption and scattering peaks to red-shift to longer wavelengths (Figure 8).
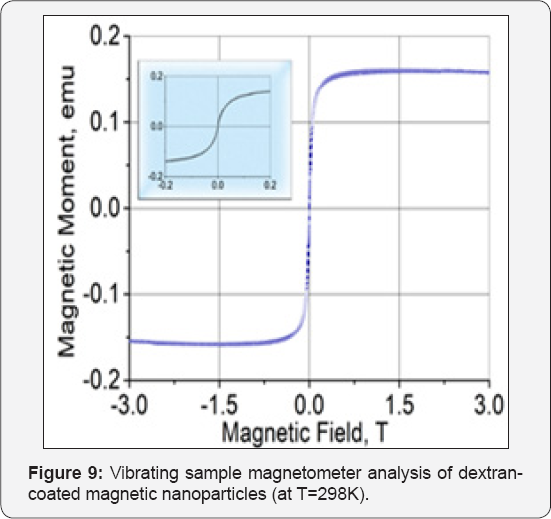
Vibrating sample magnetometer (VSM) is used to evaluate magnetization of the magnetic nanoparticles (MNPs) as a function of an applied external magnetic field (H) between-3 and+3 T. Based on the obtained VSM curve at room temperatures, magnetic behavior of the MNPs can be identified. For example at room temperature, the zero magnetic remanence (when H is zero), and the hysteresis loop feature indicates that the MNPs are superparamagnetic (Figure 9).
Conclusion
Experimental studies have shown the effectiveness of electrohydraulic processing on the high dispersive magnetite nanofluid synthesis. The concentration enhancement of Magnetite nanoparticle was caused by an instant increase of pressure and other physical phenomena influence, which accompanied during electrohydraulic effect. However, the effective use of this effect closely related to the control of some physical parameters. PVA and Dextran coated nanofluids did not sedimented long time, revealed strong magnetic and mono dispersal properties. TEM shows narrow size distribustion for PVA-coated magnetic nanoparticls with mean diameter of 10 nm. Finally, the recorded hysteresis loops shows that the particles are superparamagnetic at room temperature, which is characteristic of a soft ferromagnetic material such as magnetite.
Acknowledgement
This work was supported by Shota Rustaveli National Science Foundation of Georgia (Grant No.AR/96/3-250/13 and PG/54/3-250/13).
References
- Wei W, Quanguo H, Changzhong J (2008) Magnetic Iron Oxide Nanoparticles: Synthesis and Surface Functionalization Strategies, Nanoscale Res Lett 3(11): 397-415.
- Gupta AK, Gupta M (2005) Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications Biomaterials 26(18): 3995-4021.
- Ba nobre-Lopez M, Teijeiro A, Rivas J (2013) Magnetic nanoparticle- based hyperthermia for cancer treatment. reports of practical oncology and radiotherapy 18: 397-400.
- Garcia-Jimeno S, Estelrich J (2013) Ferrofluid based on polyethylene glycol-coated iron oxide nanoparticles: Characterization and properties, Colloids and Surfaces A: Physicochem. Eng. Aspects 420: 74- 81.
- Fedele L, Colla L, Bobbo S, Barison S, Agresti F (2011) Experimental stability analysis of different water based Nanofluids Nanoscale Research Letters 6(1): 300.
- Pang SC, Khoh WH, Chin SF (2010) Nanoparticulate magnetite thin films as electrode materials for the fabrication of electrochemical capacitors. J Mater Sci 45(20): 5598-5604.
- Pang SC, Chin SF, Anderson MA J (2007) Redox equilibria of iron oxides in aqueous-based magnetite dispersions: Effect of pH and redox potential. Colloid Interface Sci 311(1): 94-101.
- Yu WW, Falkner JC, Yavuz CT, Colvin VL (2004) Synthesis of monodisperse iron oxide nanocrystals by thermal decomposition of iron carboxylate salts. Chem Commun 20: 2306-2307.
- Li Z, Sun Q, Gao M (2005) Preparation of water-soluble magnetite nanocrystals from hydrated ferric salts in 2-pyrrolidone: mechanism leading to Fe3O4. Int Ed 44: 123-126.
- Liu Z L, Wang X, Yao KL, Du GH, Lu QH, et al. (2004) Synthesis of magnatic nanoparticles in W/O micromulcious. J Mater Sci 39: 2633.
- Xuan S, Hao L, Jiang W, Gong X, Hu Y, et al. (2007) Preparation of water- soluble magnetite nanocrystals through hydrothermal approach. J Magn Magn Mater 308: 210-213.
- Chin SF, Iyer KS, Raston CL, Saunders M (2008) Size Selective Synthesis of Superparamagnetic Nanoparticles in Thin Fluids under Continuous Flow Conditions. Adv Funct Mater 18(6): 922-927.
- Park J, Lee E, Hwang NM, Kang M, Kim SC, et al. (2005) One-Nanometer- Scale Size-Controlled Synthesis of Monodisperse Magnetic Iron Oxide Nanoparticles. Chem Int Ed 44(19): 2872-2877.
- Sun S, Zeng H (2002) Size-controlled synthesis of magnetite nanoparticles. J Am Chem Soc 124(28): 8204-8005.
- Smith B, Raston CL, Sobolev AN (2005) Poly (ethyleneglycol) (PEG): a versatile reaction medium in gaining access to 4'-(pyridyl)- terpyridines. Green Chem 7: 650-654.
- Kidwai M, Bhatnagar D, Mishra NK (2010) Polyethylene glycol PEG mediated green synthesis of 2,5-disubstituted 1,3,4-oxadiazoles catalyzed by ceric ammonium nitrate CAN. Green Chem 3(1): 55.
- Smith NM, Raston CL, Smith CB, Sobole AN (2007) PEG mediated synthesis of amino-functionalised 2, 4, 6-triarylpyridines. Green Chem. 9: 1185-1190.
- Yu W, Zhang T, Zhang J, Qiao X, Yang L, et al. (2006) The synthesis of octahedral nanoparticles of magnetite, Materials Letters 60: 29983001.
- Zhu Y, Wu Q (1999) Synthesis of magnetite nanoparticles by precipitation with forced mixing, Journal of Nanoparticle Research 1(3): 393-396.
- Anne-emmanuellestricker, michelbéland (2006) sequencing batch reactor versus continuous flow process for pilot plant research on activated sludge. Water environment foundation, Burlington, Canada, p. 1-11.
- Alexandre D, Costin B (2014) Integrated design and Simulation of chemical processes, book,elsevierpubl. (2nd edn), Elciver, Germany, p. 886.
- Lahure P, Jain P (2015) Preparation and Characterization of Magnetite Nanoparticle using Green Synthesis, Int J Res Chem Environ 5 (4): 3843.
- Baranov DA, Gubins P (2009) Magnetic nanoparticles: achievements and problems of chemical synthesis, Solid State Chemistry: monocrystals, nanomaterials, nanotechnologies. IX International Scientific Conference.
- Patrick V, Anton Y, NikiChristophe L (2016) Electrical Discharge in Water Treatment Technology for Micropollutant Decomposition. Intech, London Bridge street, UK, p. 574.
- Grabowski LR (2006) Pulsed corona in air for water treatment Technische Universiteit Eindhoven.
- Potocký Š, Saito N, Takai O, Potocký Š, Saito N, Takai O (2009) Needle electrode erosion in water plasma discharge. Thin Solid Films 518: 918-923.
- Emil O, Jasnakunov J, Mairykova N, Abdykerimova A, Maatkasymova A, et al. (2007) Synthesis Method of Nanomaterials by Pulsed Plasma in Liquid, Journal of Nanoscience and Nanotechnology 7(9): 3157-3159.
- Hassan K, Fariba G, Juliet O (2016) Synthesis of Magnetite/Hematite/ Iron Nanocomposites by the Low Voltage Arc Discharge in Water in the Presence of External Magnetic Field. J Electrochem Sci 11: 3074-3085.
- Helena Oi Lun LI, Jun K, Kuniko U, Nagahiro S (2013) Comparison between the Mechanism of Liquid Plasma Discharge Process in Water and Organic Solution. J Inst Electrostat Jpn 37(1): 22-27.
- Liangliang L, Wang Q (2015) Microplasma: A New Generation of Technology for Functional Nanomaterial Synthesis. Plasma Chem Plasma Process 35(6): 925-962.
- Jaroslav H, Vit K, Ondrej J (2013) Electrode-less plasma jet synthesis of core-shell iron/iron oxide nanoparticles. Czech Republic, EU, pp 16-18.
- Burakov VS, Savastenko NA, Tarasenko NV, Nevar EA (2008) Laser-induced modification of composite Cu-C nanosized particles synthesized using a pulsed electrical discharge in a liquid. Jpn Inst Metal 75(3): 394-401.
- Sato T, Usuki K, Okuwaki A, Goto Y (1992) Synthesis of metal nitrides and carbide powders by a spark discharge method in liquid mediaJ Mater Sci 27(14): 3879-3882.
- Xie SY, Ma ZJ, Wang CF, Lin SC, Jiang ZY, et al. (2004) Preparation and self-assembly of copper nanoparticles via discharge of copper rod electrodes in a surfactant solution: a combination of physical and chemical. J Solid State Chem 177: 3743.
- Yutkin LA (1986) Electrohydraulic effect and its application in industry.
- Markhulia J, Kekutia S, Jabua Z, Mikelashvili V, Saneblidze L (2017) Chemical co-precipitation synthesis and characterization of polyethylene glycol coated iron oxide nanoparticles for biomedical applications, SGEM proceedings 7(61): 51-58.






























