Copper-Drug Based Complexes: Antimicrobial, Antioxidant and Pharmacological Study
Ouiza Hamrani1, Sultana Boutamine1, Safia Kellou-Tairi2 and Zakia Hank1*
1Université des Sciences et de la Technologie Houari Boumediene, Laboratoire d’Electrochimie-Corrosion, Algérie.
2Université des Sciences et de la Technologie Houari Boumediene, Laboratoire de Physico-Chimie Théorique et de Chimie Informatique, Algérie.
Submission: October 29, 2017; Published: November 30, 2017
*Corresponding author: Zakia Hank, Université des Sciences et de la Technologie Houari Boumediene, Laboratoire d’Electrochimie-Corrosion, Métallurgie et Chimie Minérale, Faculté de Chimie, Algérie ; Tel: 00213550627595; Email: zhank@usthb.dz
How to cite this article: Ardhendu K M. Silver Nanoparticles as Drug Delivery Vehicle against Infections. Glob J Nano. 2017; 3(2): 555610. DOI:10.19080/GJN.2017.03.555610
Introduction
The study on complexation of drug molecules with various metal ions is an important field of research in the chemical, biochemical, medicinal and pharmacological point of views [1]. Our body possesses a large number of metal ions for operating normal physiological activities and we also intake a number of metals as drug, dietary factor, drinks and we also come in close contact with different drugs. It’s also well established that many pathological situations involve deregulation in the metabolism of metals: therapeutic responses are then necessary and although most drugs or compounds used in medicine are purely organic. The challenge is to enhance the properties of these drugs by complexing them and to study their interaction with the trace elements present in the human organism. In fact, the complexation offers the metal ion a multitude of coordination possibilities and a wide range of geometries. As a result, the metal complexes, due to their thermodynamic and kinetic properties, and in some cases their redox activities, offer novel mechanisms of action that organic compounds do not exhibit themselves so it’s very important to control all these properties to obtain the desired therapeutic effect when a drug or a metal complex is introduced into the body or the cell [2-4].
The interest of this study is to examine the modifications that the metal can make to the properties of an organic molecule when the latter is coordinated to it. Copper complexes have attracted great deal of attention due to their therapeutic applications as antimicrobial and antioxidant so, we have been interested in the study of the complexation of this metal by some drug molecules such as Paracetamol, Indomethacin and Spiramycin. Few works report similar study [5-9].
Discussion
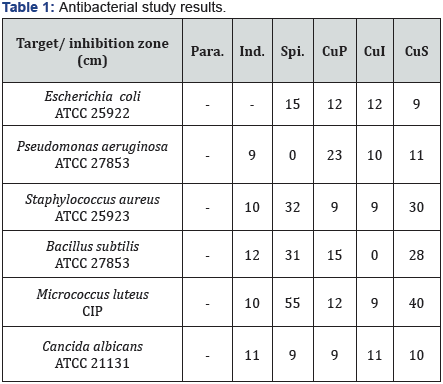
Antibacterial and antifungal activity (in vitro)
The results of our investigations (Table 1) show that some metal complexes are more toxic than their parent ligands against the same microorganism and under identical experimental conditions. This would suggest that chelation could facilitate the ability of the complex to cross a cell membrane. It’s well established that the mode of action of antimicrobials may involve various targets in the microorganisms [10]. These targets include the following: the higher activity of the metal complexes may be due to the different properties of the metal ions upon complexation [11]. According to Overtone’s concept [12] of cell permeability, the lipid membrane that surrounds the cell favors the passage of only lipid-soluble materials, so lipophilicity is an important factor controlling the antifungal activity. On the other hand and upon Tweedy’s chelation theory [13] the polarity of the metal ion will be reduced because of the partial sharing of the positive charge of the metal with the donor groups present in the ligand. Thus, chelation enhances the penetration of the complexes into lipid membranes. These results are corroborated by DFT calculations.
Acute toxicity
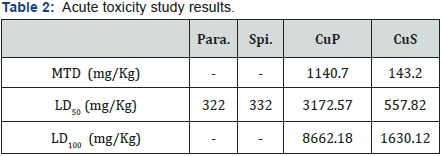
The comparison of the LD50 values of the complexes with that of the free ligands shows formally that the complexes are less toxic with a much higher tolerance to the synthetic product. Some results of this investigation are given in Table 2.
Subacute toxicity

As a result, we find that batches treated with CuP complex with doses lesser than 50% of determined lethal dose do not affect the weight evolution of the mice. Therefore, the results obtained suggest that CuP complex is fairly non-toxic (Table 3).
Anti-inflammatory assay
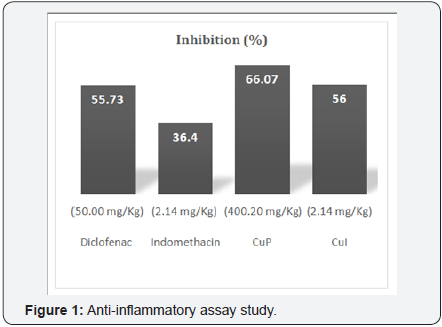
The injection of Carrageenan induces the liberation of bradykinin, which later induces the biosynthesis of prostaglandin and other autacoids, which are responsible for the formation of the inflammatory exudates [14-16]. The study of the acute antiinflammatory tests showed that copper complexes produced a significant (P=0.028) reduction at 6h in Carrageenan induced paw edema when compared to the positive control group (Diclofenac at 50mg/Kg-body weight). Some results are gathered in Figure 1. These results suggest that the complexation of indomethacin increases its anti-inflammatory effect and that of paracetamol confers anti-inflammatory activity.
Antioxidant assay
The antioxidant activity of Cu(II) complexes was measured in terms of their hydrogen donating or radical scavenging ability by UV–vis spectrophotometer using the stable 2,2-diphenyl- 1-picrylhydrazyl radical (DPPH) (DPPH: 18H12N5O6). For illustrating this study, we give here the results corresponding to CuP tests. Radical scavenging activity of paracetamol and CuP complex as well as the standard was increased in a dosedependent manner; nevertheless, paracetamol and CuP complex showed less good activities as a radical scavenger compared to that of ascorbic acid, which was used as a standard. However, they exhibit appreciable activity with 80% in percentage scavenging in from a concentration of 300μg/mL.
Electronic properties
The electronic structures of the studied compounds have been investigated by the quantum chemical parameters like energy of the highest occupied molecular orbital (EHOMO), energy of the lowest unoccupied molecular orbital (ELUMO), LUMOHOMO energy gap (ΔE) and natural atomic charges calculated by natural population analysis. The HOMO and the LUMO are important parameters in organ metallic chemistry especially in chemical reactivity. ΔE is an important stability index helping to characterize the chemical reactivity and kinetic stability of the compound [17].
In the copper complexes, the EHOMO and ELUMO are negative (Table 4) which indicates their stability. The complexes have the smallest energy gap (beta), so they are more reactive than the parent ligands since they easily offer electrons to an acceptor.
Conclusion
Microbial studies suggested that the copper complexes showed importantly raised antibacterial and antifungal activities and presented higher antimicrobial activity than the corresponding free ligand.
Based on the results of the toxicological study, the acute toxicity of the complexes tested on mice of NMRI strain revealed that the copper - drug complex is generally not toxic. The antiinflammatory assay study shows that the copper-ligand binding induced an anti-inflammatory effect to the drug molecule, which thus changes the therapeutic class. These experimental tests are corroborated by the DFT study which shows the higher reactivity of the complex with respect to the free ligand.
Acknowledgment
The authors would like to extend their grateful thanks to F. Benguergoura, A. Mokrani, K. Tahar Djebbar (Institut Pasteur de Dely Ibrahim, Alger-Algérie), A. Benabdelouahid (Département de Pharmacie, Université d’Alger-Algérie) and Dr. H. Henni (Laboratoire Nationale de Contrôle des Produits Pharmaceutiques, Alger-Algérie) for technical support.
References
- Moamen SR, Sharshar T, Khaled ME, Zein, K H, (2013) Journal of Molecular Structure. 1047: 37-4.
- Beril A (2012) Therapeutic Organometallic Compounds, Pharmacology, Dr. Luca Gallelli (Ed.), Istanbul University, Institute of Forensic Sciences, Turkiye, p. 652.
- Hartinger CG, Dyson PJ, (2009) Bioorganometallic chemistry- from teaching paradigms to medicinal applications. J Chem Soc Rev 38(2): 391–401.
- Kathryn LH, Katherine JF, (2009) Application of Metal Coordination Chemistry To Explore and Manipulate Cell Biology. Chem Revs 109(10): 4921–4960.
- Lawal A, Obaleye JA, (2007) Synthesis, characterization and antibacterial activity of aspirin and paracetamol metal complexes. Biokemistri 19: 9-15.
- El-Megharbel SM, Reham ZH, Moamen SR, (2014) Preparation, spectroscopic, thermal, antihepatotoxicity, hematological parameters and liver antioxidant capacity characterizations of Cd (II), Hg (II) and Pb (II) mononuclear complexes of paracetamol anti-inflammatory drug. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 131: 534–544.
- Ionut L, Georgeta S, Gabriela V, Germaine S, Titus V, et al. (2013) REV CHIM (Bucharest) 64: 1127-1130.
- Sangita PK, Farhad MA, Sania S, Zakir MdS, Mohammad AH et al. (2012) Study of Differential Scanning Calorimetry of complex of Magnesium Sulfate with Aspirin, Paracetamol and Naproxen. Bangladesh Pharmaceutical Journal 15(1): 7-12.
- El-Shahawy AS, Ahmed SM, Sayed NKh (2007) Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 66: 143–152.
- Al-Amiery AA, Abdul AHK, Abu Bakar M, (2012) Antifungal and Antioxidant Activities of Pyrrolidone Thiosemicarbazone Complexes. Bioinorganic Chemistry and Applications 795812: 1-6.
- Chohan ZH, Arif M, Akhtar MA, Supuran CT, (2006) Metal-Based Antibacterial and Antifungal Agents: Synthesis, Characterization, and In Vitro Biological Evaluation of Co(II), Cu(II), Ni(II), and Zn(II) Complexes With Amino Acid-Derived Compounds. Bioinorganic Chemistry and Applications 83131: 1–13.
- Parekh HM, Pansuriya B, Patel MN (2005) Characterization and Antifungal Study of Genuine Oxovanadium (IV) Mixed-Ligand Complexes with Schiff Bases. Polish J Chem 79(12): 1843-1851.
- 13. Tweedy BG (1964) Plant extracts with metal ions as potential antimicrobial agents. Phytopathology 55: 910–914.
- Farkhanda S, Amin B, Marcel G, Gianluca C, Ulrich FDV et al. (2010) In vitro assessment of cytotoxicity, anti-inflammatory, antifungal properties and crystal structure of metallacyclic palladium (II) complexes. Journal of Organometallic Chemistry 695: 315–322.
- Ueno A, Naraba H, Ikeda Y, Ushikubi F, Murata T, et al. (2000) Intrinsic prostacyclin contributes to exudation induced by bradikinin or carrageenan: A study on the paw edemainduced in ip-receptor-deficient mice. Life Sci 66(12): 155-160.
- Farkhanda S, Amin B, Marcel G, Christine G, Mariyam J et al. (2008) Synthesis, characterization, in vitro cytotoxicity and anti-inflammatory activity of palladium (II) complexes with tertiary phosphines and heterocyclic thiolates: Crystal structure of [PdC28H19N8PS2]. Journal of Organometallic Chemistry 693: 1117-1126.
- El-Gammal OA, Rakha TH, Metwally HM, Abu El-Reach GM, (2014) Synthesis, characterization, DFT and biological studies of isatinpicolinohydrazone and its Zn(II), Cd(II) and Hg(II) complexes. Spectrochim Acta A 127: 144-156.






























