Inhibitory Effect of Quercetin-Loaded Nanostructure Lipid Carrier on Compound 48/80-Induced Mast Cell Degranulation
Asit Kumar De, Rajkumar Shil, Santanu Ghosh, and Tanmoy Bera*
Division of Pharmaceutical Biotechnology, India
Submission: August 11, 2017; Published: October 25, 2017
*Corresponding author: Tanmoy Bera, Division of Pharmaceutical Biotechnology, India, Tel: +, E-mail: proftanmoybera@gmail.com
How to cite this article: Asit K D, Rajkumar S, Santanu G, Tanmoy B. Inhibitory Effect of Quercetin-Loaded Nanostructure Lipid Carrier on Compound 48/80-Induced Mast Cell Degranulation. Glob J Nano. 2017; 3(2): 555608. DOI:10.19080/GJN.2017.03.555608
Abstract
Quercetin is naturally occurring flavanoids, it is abundant in nature and the human beings are taken up it through their daily meals. The different pharmacological activity of quercetin has been evaluated since a long time. The scientific exploration of quercetin revealed that it is effective as anti-oxidant, anti-inflammatory agent; it can reduce the elevated levels of cholesterol, triglycerides, low-density-lipoproteins etc. However, solubility and bioavailability has been an issue for the quercetin. In this experimental work we not only evaluate the antihistaminic activity of quercetin but also developed a nano structure lipid carrier system for quercetin and explore its effectiveness as an antihistaminic agent.
It was found that the quercetin loaded nano structured lipid carrier system was successfully fabricated with adequate drug loading and entrapment efficiency. The intracellular uptake of formulated delivery system by isolated mice peritoneal mast cells has also been studied. The antihistaminic activity of quercetin and quercetin loaded lipid carrier system was compared against the standard antihistaminic agents like prednisolone, cromolyn sodium and it was found that the quercetin loaded lipid carrier system have showed superior activity than the free quercetin and the standard antihistaminic agents.
Keywords: Quercetin; NLC; Lipid based formulation; Compound 48/80; Mast cell; Antihistaminic
Abbreviations: Nano structured Lipid Carrier; qNLC: quercetin Loaded Nano Structured Lipid Carrier
Introduction
Allergic diseases and asthma have been prevalent since a long ago and still, now the incidence of these types of disease condition increased in such a manner that it has appeared as a major health concern. As per world health organization, more than 1.8lakhs of mortality is caused by asthma annually [1]. Ages are no bar for the occurrence of this type of disease; it may happen in any age group. The diseases like asthma and allergy are initiated by the process of inflammation. CD4+T helper cell responses are important for the initiation and propagation of the disease condition [2]. Interleukin-4, 5, 9 and 13 are the cytokines which are secreted by TH2 cells and they are the most important mediators of the process of inflammation. Mast-cell degranulation, increased IgE level is the characteristic phenomenon of asthma and allergy related diseases [3].
The treatment of this kind of disease condition falls into different categories like mast cell stabilizers, corticosteroids, antihistaminic agents. They may act by blocking the mediators responsible for inflammation or by preventing the process which initiates the process of inflammation. The present chemotherapy for this purpose is efficient however long term side effects still be a concern. Skin fragility, immunosuppressant is associated with the long term use of oral corticosteroids. It was also observed that the new generation antihistaminic agents are showed sedative effects [4,5]. Hence, there is always a need for a new antihistaminic agent which could effectively treat the disease condition as well as tolerable in patients in long term.
Searching for a new antihistaminic agent as well as a new drug delivery system for this type of diseases we have developed quercetin loaded nano structured lipid carrier system. Quercetin is a naturally occurring phenolic compound and it is flavonoid in class. Quercetin is abundant in nature and it is generally found in the food plants. The flavonoids commonly present as glycosides. Like, the aglycone quercetin linked to quercetin as 3-o-glycoside [6]. The beneficial pharmacological effects of quercetin always draw the attention of many scientists for several decades. According to Gupta et al, the quercetin has potent antioxidant activity [7]. Quercetin could scavenge the free radicals, chelatemetal ions and it shows synergistic activity with other antioxidants [8-12].
Quercetin could be effective in the treatment of inflammation and oxidative stress as well [13]. It was found in a study that regular intake of quercetin could decrease the elevated levels of cholesterol, triglyceride, and low-density-lipoproteins [14]. Tzankova et al in his recent study have found that quercetin possessed hepatoprotective activity [15]. It was reported that more than 90% of quercetin was metabolized after an hour when it was administered intraperitoneally. When Justino et al. [16] was treated Sprague–Dawley rats with a single intraperitoneal injection of quercetin at a dose of 50mg/kg body weight, it was found that the plasma contains very low amount of free quercetin [16]. Therefore, the development of a drug delivery system with quercetin which could effectively increase the solubility and bioavailability of it, have always been a challenge. In these circumstances, the nano structured lipid carrier system (NLC) for quercetin was developed to evaluate its antihistaminic property which would open up a new beneficial pharmacological effect of the naturally occurring flavonol.
Material and Methods
Materials
Gattefose India Pvt. Ltd. supplied Comparitol® 888 ATO. Other substances like soy lecithin, compound 48/80, triolein, o- phthalaldehyde, sorbitol, Pluronic F68, and quercetin were purchased from Sigma- Aldrich. Fetal bovine serum, RPMI1640 and medium 199 were supplied from HiMedia, India. All the solvents used in the experiment were of analytical grade.
Instruments and Equipment
Sartorius balance, 700 MW sonicator (Vibra cell VCX750; Sonics, CT, USA), Vortex (CM101 cyclomixer; Remi, India),high-speed homog¬enizer (IkaT10 basic, Ultra-Turrax), magnetically stirred hot plate (Tarsons, Spinot digital model MCO2), lyophilizer (LaboGene-ApS, 6-B-DR-3450 Lynge, Denmark),High performance liquid chromatography (HPLC) (Dionex ultimate3000; Thermo Fisher Scientific, MA, USA), UV spectrophotometer (Shimadzu UV-1800; Shimadzu, Tokyo, Japan), BOD incubator shaker (BOD Inc 1S, Kolkata, India), CO2incubator (MCO-15AC; Sanyo, Tokyo, Japan), Laminar Airflow (Stericlean; Deepak Meditech Pvt. Ltd, New Delhi, India), Cold Centrifuge (Rota 4R-V/FM; Plasto Crafts, Mumbai, India). Standard Borosil glass wares were used for experimental purposes. GraphPad Prism 5.0.1. was used for statistical analysis.
Ethics statements
BALB/c mice (20-25gm) of either sex or apparently same ages were used for the studies. Animals were housed in polypropylene cage. All of the experiments, where animals were used, were done according to the guidelines of Jadavpur University Animal Ethics Committee. Animals were fed with standard diet and water and they were exposed to normal day and night cycle.
Methods
Preparation of Cedrol-loaded NLCs: A combination of melt emulsification along with homogenization and ultrasonication method was employed to fabricate the quercetin loaded NLC (qNLC) [17]. Briefly, according to the drug and lipid ratio, the weighted amount of comparitol® 888 ATO (1.5mg) [(glycerol mono behenate 15%, glyceryl tribehenate 35% and glyceryl di behenate 50%)], triolein (0.31mg) and quercetin were melted together at about 85 ˚C. That melted mixture was stirred occasionally for 15mins to develop transparent and uniform oil phase. It was observed that there was no degradation of quercetin happened at that particular temperature condition.
The aqueous phase was prepared by dissolving Pluronic F68 and soya lecithin (0.31mg) at the same temperature condition and for the same period of time in a 60 ml double distilled water. Now, the aqueous phase was added drop-wise into the oil phase during homogenization at 20000rpm for 10mins. The formed emulsion was then sonicated for 10mins in an ultra sonicator at 85 ˚C. The formed NLC was cooled at 4 ˚C for overnight followed by membrane filtration through the 0.45μm membrane filter. After frozen at -80 ˚C NLC suspension was lyophilized to obtain a dry finished product.
Blank NLC was prepared by the same technique except adding the quercetin (Table 1).

Characterization
Particle size, zeta potential, and poly dispersity index: The average particle size, zeta potential, and polydispersity index (PDI) of formulated NLCs were determined by the dynamic light scattering (DLS) technique by the Zetasizer Nano ZS90 (Malvern Instrument ltd., UK) using the software Data transfer assistance (DTS). For these studies, 1 mg of the formulations were dispersed in 2ml of Milli Q water and then sonicated for half an hour before the analysis.
Quercetin loading and entrapment efficiency study: To carry out the drug loading and entrapment efficiency study, 5mg of qNLC was dispersed in 5 ml of 0.5% tween 80 in PBS buffer (pH 7.4). The dispersion was sonicated for 15mins. After that, it was centrifuged at 20000rpm for 15min. The supernatant was collected and the quantity of quercetin was determined by the liquid chromatographic method. For that purpose, C18 column (4.6 × 250 mm, 5μ) was used. Acetonitrile and phosphate buffer (pH 4.0 maintained by o- phosphoric acid) in a ratio of 50:50 was used as a mobile phase and the flow rate was 1ml/min. Quercetin was detected at a detecting wavelength of 292nm. Drug loading and the entrapment efficiency was calculated by the following equations.
Drug loading= (Wtotal-Wfree/Wlipids) × 100%
Entrapment efficiency= (Wtotal-Wfree/Wlipids) × 100%
Where Wtotal was the weight of drug added in the system, Wfree was the analyzed weight of drug in the supernatant and Wlipids weight of lipids added to the system.
Field emission scanning electron microscopic study: The surface morphology of the formulated NLC was evaluated by field emission scanning electron microscopy. The lyophilized product was used for taking the SEM images. Briefly, lyophilized NLC was spread on the carbon tape then subjected to gold coating. After completion of the coating, particles were observed under FESEM (Model-JSM-6700F; JEOL, Tokyo, Japan).
In vitro cumulative drug release study: In vitro cumulative drug release study of formulated qNLC was carried out for 21 days. Briefly, 5mg of qNLC was suspended in 5 ml PBS buffer pH 7.4 and kept it in the dialysis bag (MW cutoff 12000 kDa). That dialysis bag was further kept into a 100 ml 0.5% v/v tween 80 with PBS buffer (pH 7.4). That system was kept in a shaker incubator. 1 ml sample aliquots were collected and 1ml of fresh release medium was incorporated for a predetermined time period (45 mins, 90mins, 3h, 6h, 12h, 24h and up to 504h). The collected samples were analyzed by the similar chromatographic condition as previously described.
Mast cell isolation and culture: Peritoneal mast cells were isolated from BALB/c mice [18]. Briefly, after sacrificing the mice by cervical dislocation, 5mL of heparinized (10 IU/mL) calciumand magnesium-free Hank’s solution (HBSS) was injected into the mice peritoneal cavity. After 2mins, the peritoneal fluid was collected and centrifuged at 1,500rpm for 10minutes at 4 °C. The supernatant was discarded and the cells were resuspended in fresh HBSS containing 0.1% protease-free bovine serum albumin and then it was purified by continuous isotonic Percoll gradient (72%) for isolation of mast cell.
Mast cells thus purified were resuspended in fresh HBSS media containing calcium and magnesium. Toluidine blue and trypan blue exclusion staining were performed to evaluate the purity and the viability of mast cells, respectively.
After the tests for purification and viability, mast cells were transferred to RPMI-1640 medium containing 15% fetal bovine serum and 5μg/mL streptomycin and 50U penicillin. After every 3 days sub culturing was carried out, and the culture was maintained for 2 months.
Trypan blue exclusion assay of mast cell viability: Mast cell viability was assessed by trypan blue staining. For this, 0.2ml of mast cells were transferred aseptically into a 1.5ml eppendr of tube along with an equal amount of 0.4% (w/v) trypan blue solution prepared in 0.8% (w/v) sodium chloride and 0.06% (w/v) dibasic potassium phosphate and incubated it for 3min at room temperature. Cells were counted by using the hemocytometer under a light microscope. The count of viable and nonviable cells was recorded separately.
Mast cell sensitization by compound 48/80: HEPESTyrode’s buffer containing mast cell suspension was incubated with 4μg/ml of compound 48/80 for the period of 10mins at 37 °C. Mast cells smears were prepared, fixed and stained with 0.1% (w/v) toluidine blue and kept it for 10mins. The morphology of the mast cells was observed under a light microscope [19].
Cellular uptake study: To carry out the cellular uptake study, coumarin-6 loaded NLC was prepared it was used instead of qNLC. For this study, sterile cover slips were placed in each well of the 24 well plate was 4×105 cells/well were seeded with coumarin-6 loaded NLC (0.5mg/mL) and incubated in a 5% CO2 incubator at 37 °C for 20min. After the specific period of time, the cover slips were collected, washed for three times and then observed under confocal microscope (TCS SP8; Leica Microsystems, Wetzlar, Germany) using Leicasuitelas X software. The magnification of the objective was 63×.
Effects of anti-asthmatic drugs, quercetin, and qNLC on histamine release assay from mast cells: For the stabilization purpose, the isolated and purified mice peritoneal mast cells (2×105 cells/mL) were incubated at 37 °C in a 5% CO2 incubator for 10mins. Standard anti-asthmatic drugs, quercetin, and qNLC at variable concentrations were added to the pre-incubated media containing the mast cells and further incubated at 37 °C. After the incubation period of 24h, the cells were washed and further incubated with 4μg/ml of compound 48/80 for the period of 20minutes. After that, the test tubes were kept in ice water. The released histamines were separated from the mast cells by the process of centrifugation at 2,000rpm for 5minutes at 4 °C. 0.05% (v/v) Triton X-100 was added to liberate the residual histamine. Then, 0.02% opt-methanol solution was added and histamine content in the supernatant and in the pellets of cells was determined spectrofluorimetrically (excitation 360nm, emission 450nm). For the estimation of the spontaneous release of histamine (spontaneous), exactly the same procedure without adding drugs and samples was followed. The following equation was used to calculate the release percentage of histamine [20,21].
% Histamine release= {(Supernatant- Spontaneous)/ (Supernatant+ cell pellets)- Spontaneous]× 100%
Statistical analysis: Experimental data were expressed as a mean ± standard deviation. One way analysis of variance was used to calculate the statistical significance. P <0.05 was considered as significant and P >0.05 was considered as not significant.
Results
Average particle size, zeta potential, PDI
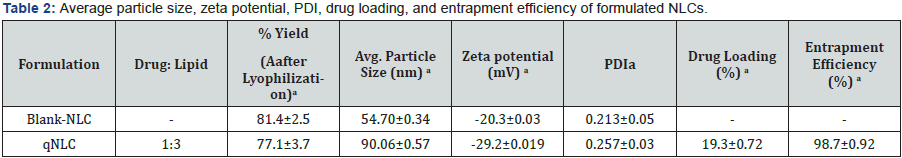
The average particle size, zeta potential, PDI values were calculated for blank-NLC and qNLC nanoparticles. According to the Table 2 the both types of formulated nanoparticles were in nano size range and their average particle sizes were less than 100nm. The percentage yields of the NLCs were more than adequate. The PDI values were found to be 0.213±0.05 and 0.257±0.03 for the Blank-NLC and qNLC, respectively. The NLCs have shown negative zeta potential values like -17.3±0.03 for Blank-NLC and -34.2±0.019 for qNLC.
Drug loading and entrapment efficiency
From the Table 2, it could be observed that the percentage drug loading for qNLC was 19.3±0.72% and the entrapment efficiency of the formulation was 98.7±0.92% (Figure 1).
Scanning electron microscopic study

The scanning electron microscopic study was carried out to evaluate the size and shape of the formulated NLC. It could be found from the Figure 2 that the formulated qNLC was in nano size range and particles were almost spherical in shape.
In vitro cumulative drug release study
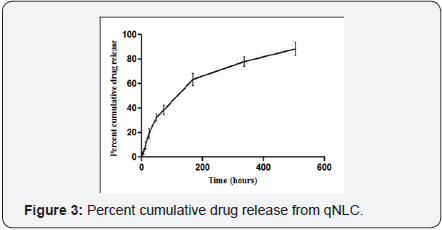
The in vitro cumulative release study was carried out for 21days. It was found that after 504h, 88.7±1.5% of quercetin was released from the NLC core (Figure 3).
Mast cell uptake study
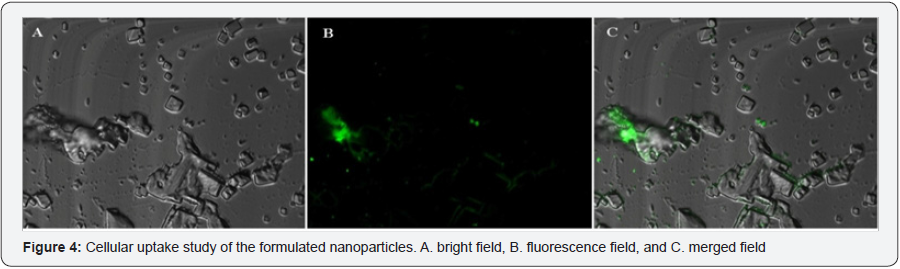
The confocal study was carried out to evaluate the formulated NLC uptake by the isolated and purified mast cell. From the Figure 4, it was observed that within the period of 20mins coumarin-6 loaded NLC was taken up by the mast cells. The green fluorescence appeared in the Figure 4B & 4C were the coumarin-6.
In vitro histamine release assay
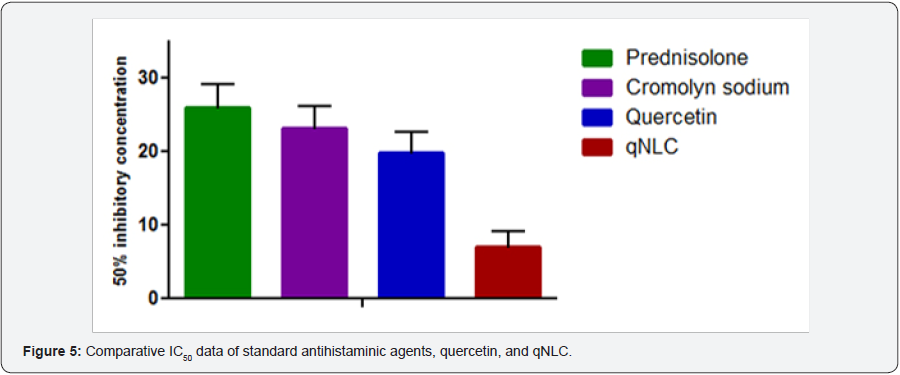
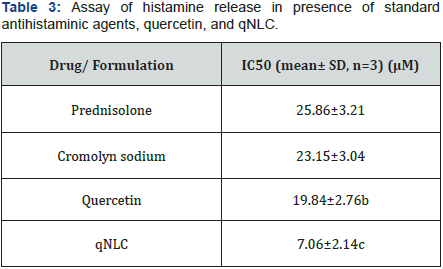
Table 3 represented the IC50 values of Prednisolone, cromolyn sodium, quercetin and qNLC which indicated that at the particular concentration of the drug or formulation the histamine release from the mast cells was 50% inhibited. It was observed from the Table 3 that the IC50 of free quercetin was 19.84±2.76μM which was less than the standard antihistaminic agents and the qNLC formulation of quercetin decreased the IC50value to 7.06±2.14μM (Figure 5).
Discussion
Fabrication of quercetin loaded NLC was carried out by a combination of melt emulsification along with homogenization and ultrasonication method. It was observed from the character studies that the formed drug loaded particles were in nano size range. From Table 2 and Figure 1A it could be observed that the average particle size of qNLC was 90.06±0.57nm. The FESEM analysis was also confirmed the formation of nano size ranged particles of NLC. From Table 2 it was also observed that the both the blank and qNLC possessed negative surface charge. Zeta potential is an important parameter which could depict the stability of the nanoparticles inside the formulation.
It was found from the Table 2 and Figure 1B that the zeta potential of qNLC was -29.2±0.019 mV which indicated that the particles in the formulation would not agglomerate in suspension if it will remain suspended for an extended time period [22]. When the quercetin loading into the NLC was evaluated it was found that the formulation of qNLC exerted superior drug loading of 19.3±0.72% with 98.7±0.92% entrapment efficiency.
In vitro percentage cumulative drug release study was carried out for an extended period of time. It was an important parameter as it will indicate the amount of drug release for the predetermined period of time as well as the pattern of release. It was observed from Figure 3 that the qNLC formulation released the 88.7±1.5% quercetin after 504h.
As the mast cell was our target so uptake of NLC by the mast cells was an essential parameter to evaluate and for that purpose confocal microscopy study was carried out. It was clearly observed from the Figure 4 that the coumarin-6 loaded NLC was well taken up by the isolated and purified mast cells within 20mins. Here the coumarin-6 was mimicking the quercetin which meant that quercetin will be taken up also when it will deliver through the NLC system.
Histamine released from the mast cells and inhibition of that phenomenon will be a property of an antihistaminic agent. For that purpose, 50% inhibitory concentrations of the free quercetin and qNLC formulation were determined and compared the data against the data generated from the standard antihistaminic drug like prednisolone and cromolyn sodium. From Table 3 and Figure 5 it was observed that the IC50 values of prednisolone, cromolyn sodium, and quercetin were 25.86±3.21μM, 23.15±3.04μM, and 19.84±2.76μM, respectively and it was also found that there were no significant differences among them which could indirectly indicate the similar antihistaminic property of quercetin like the standard drugs. Moreover, when the quercetin was loaded inside the lipid core of the NLC it was found that the qNLC exerted superior inhibition of histamine release from the mast cells with an IC50 value of 7.06±2.14μM. There was an extremely significant difference obtained among the IC50 value of qNLC and the other drugs.
Conclusion
From this experimental work, it could be concluded that the qNLC was successfully fabricated. The formulated NLCs were in nano size range. The uptake study revealed the adequate homing of the drug of interest inside the mast cells and the qNLC was efficiently inhibited the histamine release from the mast cells and the quercetin loaded NLC delivered significantly higher efficacy than the standard antihistaminic agents.
Acknowledgement
We want to acknowledge the Director of Indian Association of Cultivation of Science Kolkata, Director of Bose Institute, Kolkata for providing us the instrumental facilities.
References
- World Health Organization (2017) Media center: Bronchial asthma. Switzerland, Geneva.
- Georas SN, Guo J, De Fanis U, Casolaro V (2005) T-helper cell type-2 regulation in allergic disease. Eur Respir J 26(6): 1119-1137.
- Abbas AK, Murphy KM, Sher A (1996) Functional diversity of helper T lymphocytes. Nature 383(6603): 787-793.
- Baroody FM, Naclerio RM (2000) Antiallergic effects of H1-receptor antagonists. Allergy 55(Suppl 64): 17-27.
- Rizzo MC, Solé D (2006) Inhaled corticosteroids in the treatment of respiratory allergy: safety vs. efficacy. J Pediatr (Rio J) 82(5 Suppl): S198-205.
- Formica JV, Regelson W (1995) Review of the Biology of Quercetin and Related Bioflavonoid. Fd Chem Toxic 33(12): 1061-1080.
- Gupta C, Vikram A, Tripathi DN, Ramarao P, Jena GB (2010) Antioxidant and antimutagenic effect of quercetin against DEN induced hepatotoxicity in rat. Phytother Res 24(1): 119-128.
- Cos P, Ying L, Calomme M, Hu JP, Cimanga K et al. (1998) Structure-activity relationship and classification of flavonoids as inhibitors of xanthine oxidase and superoxide scavengers. J Nat Prod 61(1): 71-76.
- Mira L, Silva M, Rocha R, Manso CF (1999) Measurement of relative antioxidant activity of compounds: a methodological note, Redox Rep 4(1-2): 69-74.
- Morel I, Lescoat G, Cogrel P, Sergent O, Pasdeloup N et al. (1993) Antioxidant and iron-chelating activities of the flavonoids catechin, quercetin, and diosmetin on iron-loaded rat hepatocyte cultures. Biochem Pharmacol 45(1): 13-19.
- Miller NJ, Castelluccio C, Tijburg L, Rice-Evans C (1996) The antioxidant properties of the flavins and their gallate esters-radical scavengers or metal chelators? FEBS Lett 392(1): 40-44.
- Filipe P, Lança V, Silva JN, Morlière P, Santus R et al. (2001) Flavonoids and urate antioxidant interplay in plasma oxidative stress. Mol Cell Biochem 221(1-2): 79-87.
- Boots AW, Drent M, de Boer VC, Bast A, Haenen GR (2011) Quercetin reduces markers of oxidative stress and inflammation in sarcoidosis. Clin Nutr 30(4): 506-512.
- Talirevic E, Jelena S (2012) Quercetin in the treatment of dyslipidemia. Med Arh 66(2): 87-88.
- Tzankova V, Aluani D, Kondeva-Burdina M, Yordanov Y, Odzhakov F et al. (2017) Hepatoprotective and antioxidant activity of quercetin loaded chitosan/alginate particles in vitro and in vivo in a model of paracetamol- induced toxicity. Biomed Pharmacother 92: 569-579.
- Justino GC, Santos MR, Canário S, Borges C, Florêncio MH, et al. (2004) Plasma quercetin metabolites: structure-antioxidant activity relationships. Arch Biochem Biophys 432(1): 109-121.
- Chakraborty S, Kar N, Kumari L, De A, Bera T (2017) Inhibitory effect of a new orally active cedrol-loaded nanostructured lipid carrier on compound 48/80-induced mast cell degranulation and anaphylactic shock in mice. Int J Nanomedicine 12: 4849-4868.
- De Oliveira Barreto E, de Frias Carvalho V, Diaz BL, Balduino A, Cordeiro RS, et al. (2003) Adoptive transfer of mast cells abolishes the in flammatory refractoriness to allergen in diabetic rats. Int Arch Allergy Immunol 131(3): 212-220.
- Ikarashi Y, Yuzurihara M, Sakakibara I, Takahashi A, Ishimaru H, et al. (2001) Effects of an oriental herbal medicine,” sai Boku-to”, and its constituent herbs on compound 48/80-induced histamine release from peritoneal mast cells in rats. Phytomedicine 8: 8-15.
- Jung CH, Lee JY, Cho CH, Kim CJ (2007) Anti-asthmatic action of quercetin and rutin in conscious guinea-pigs challenged with aerosolized ovalbumin. Arch Pharm Res 30(12): 1599-1607.
- Inoue T, Sugimoto Y, Masuda H, Kamei C (2002) Antiallergic effect of flavonoid glycosides obtained from Mentha piperita L. Biol Pharm Bull 25(2): 256-259.
- Ghosh S, Kar N, Bera T (2016) Oleanolic acid loaded poly lactic co- glycolic acid- vitamin E TPGS nanoparticles for the treatment of Leishmania donovani infected visceral leishmaniasis. Int J Biol Macromol 93: 961-970.






























