Synthesis of Nanoparticles of Titanium Dioxide TiO2 for Toxicological Studies: A Short Review
Rouxel D1, Fleutot S1, Cherni N1, Darne C2, Gate L2 and Binet S2
1Université de Lorraine, France
2Department of Toxicol & Biometrol, Institute of National Research & Secure, France
Submission: February 23, 2017; Published: October 11, 2017
*Corresponding author: Rouxel D, Université de Lorraine, IJL– Institut Jean Lamour, CNRS UL 7198, F-54000 Nancy, France, Email: didier.rouxel@unil-lorraine.fr
How to cite this article: Rouxel D, Fleutot S, Cherni N, Darne, Gate, et al. Synthesis of Nanoparticles of Titanium Dioxide TiO2 for Toxicological Studies: A Short Review. Glob J Nano. 2017; 3(1): 555604. DOI:10.19080/GJN.2017.03.555604.
Abstract
With advancement in medicines, Nanomaterials have tremendous potential in the drug delivery system. The usage of polymeric and non-polymeric nanomaterials has attracted much attention because of its different compositions and extensive use for the gene and drug delivery. The drugs face several barriers during their movement inside the human body to reach their target site. The biological environments need to understand and get tuned to the interactions of nanomaterials with the human body such as targeting cell-surface receptors and drug release mechanisms. The polymeric nanomaterials such as hydrogels, micelles, liposomes and polymeric nanoparticles and non-polymeric nanomaterials such as quantum dots, carbon nanotubes, metallic nanoparticles etc are used for the formulations of several drug deliveries. In this review, we have reviewed on the prospective of polymeric and non-polymeric nanomaterials in drug delivery systems.
Keywords: Polymers; Nanomaterials; Nanoparticles; Drug delivery system
Abbreviations: FDA: Food and Drug Administration; PLA: Polylactide; PGA: Polyglycolide; PEGs: Polyethylene Glycol; PNIPAM: (Poly(N-isopropylacrylamide); CNT: carbon nanotubes; AFM: Atomic Force Microscopy; SEM: Scanning Electron Microscopy; TEM: Transmission Electron Microscopy; PMs: Polymeric Micelles; NPs: Polymeric Nanoparticles; DDS: Drug Delivery Systems; QDs: Quantum dots
Introduction
Nanoparticles have unique properties relative to the large particles, allowing their use in many nanomaterials. For example, TiO2 nanoparticles are widely used in cosmetics, paints and foodstuffs for their various properties, including colloidal stability, high reflection index and photo catalytic properties [1]. However, several toxicological studies have shown that nanoparticles can be potentially hazardous because of these unique physicochemical properties [2-4]. In particular, the very large specific surface area of nanomaterials greatly increases their surface reactivity with biological media.
In recent years, synthesis methods have been intensively developed to produce nanoparticles with high homogeneity and uniform microstructure [5-7]. These methods include hydrothermal synthesis [8], solvothermal synthesis [9], sol-gel method [10], ultrasonic irradiation [9] and microwave synthesis [11]. To synthesize TiO2 nanoparticles, solvothermal method appears to be particularly relevant for the preparation of nanometric-sized TiO2, because it allow a good control of size and crystallinity while limiting the agglomeration of nanoparticles [12]. Moreover, several toxicological studies show that the size, shape and degree of agglomeration can significantly affect toxicity [13-15]. Finally, studies have shown that mixing nanopowders with or without surfactants, such as water-soluble citric acid, provides better stability [16,17].
In this paper, after giving some general information about titanium dioxide TiO2, we will present the main methods of synthesis of nanopowders of TiO2, and discuss some important aspects of their preparation for toxicological studies.
General Information on Titanium Dioxide TiO2
Titanium dioxide (TiO2), also called titanium (IV) or titanium oxide, was initially discovered in 1821 but marketed in 1916 as a white pigment [18]. It is found in nature in three crystallographic forms: rutile, anatase and brookite. Only the anatase and rutile crystalline structures are used commercially, and are found in manufactured products for which the risk of exposure is of interest for toxicological studies. The anatase phase is metastable whereas the rutile phase is thermodynamically stable. For nanoparticles less than 15 nm, the anatase phase becomes the most stable because stability depends on the size of the particles [19].
The use of TiO2 nanoparticles makes it possible, for example, to improve the process of photo degradation and complete mineralization of toxic products and organic pollutants. In fact, TiO2 nanoparticles are successfully used in environmental technology for the treatment of wastewater and groundwater, and degradation of pollutants in air, in particular nitrogen and volatile organic compounds [20]. TiO2 nanoparticles are also often used in certain materials (sunscreens, glasses, paints) due to the size corresponding to wavelengths below the visible (which gives them transparency) and their ultraviolet absorbance [20].
Methods of Synthesis of Nanopowders of Titanium Dioxide Tio2
The synthesis of the nanoparticles of titanium dioxide TiO2 can be carried out by several methods using different precursors such as the titanium isopropoxide of the linear formula Ti[OCH(CH3)2]4 and generally reported in the literature under the abbreviation Ti(OiPr)4, the tetrachloride or other titanium salts. In this part, we will present various synthetic processes which make it possible to obtain monodisperse TiO2 nanoparticles.
Solvothermal method
It is a synthetic method that involves chemical reactions with a titanium precursor in organic solvents, at controlled temperatures and pressures. This synthesis method is used, for example, by Guo et al. [9]. They obtained non-agglomerated titanium dioxide particles of anatase crystalline structure by thermal hydrolysis of titanium isopropoxide precursor in the presence of a mixture of water and ethanol under ultrasonic irradiation of 100 W/cm2 at a temperature of 90 °C and for 3 hours.
Indeed, the addition of the solvent in the reaction system acts not only as an oxidizing agent, but also contributes to the improvement of the morphology [12]. The application of ultrasonic irradiation in this synthesis method has advantages over conventional TiO2 preparation methods such as uniform distribution, dispersion of nanoparticles, higher surface area and better thermal stability [21,22]. In this method of synthesis, the crystal structure and the size of the nanoparticles depend on the temperature of the reaction, the acidity of the medium and the reaction time.
Hydrothermal method
It is a synthetic method that involves the chemical reactivity of a titanium precursor in aqueous solvents at controlled temperatures and pressures. This method makes it possible to obtain nanoparticles of small size, small size distribution and controlled crystallinity while adjusting the experimental conditions [12]. This method was used by Katsumata et al. [8] to obtain nanoparticles of monodisperse TiO2 of brookite crystalline form, between 20 and 50 nm in size. To obtain the nanoparticles, a titanium powder is introduced into an NH3/H2O2solution. After 2 hours, glycolic acid is added. The synthesized product is dried at 65 °C.
The mixture thus obtained undergoes a hydrothermal treatment at 200 ° C for 6 hours. In this study, the authors showed that the addition of sodium oleate as surfactant allows the formation of cubic nanoparticles. This addition has above all a significant effect on the zeta potential: the synthesis made by adding sodium oleate results in a zeta potential of -50.6 and -55.2 mV for pH 6 and 7 whereas for synthesis without the sodium oleate, the zeta potential is -24,6 et -31,4 mV. The addition of sodium oleate in this study favors the formation of brookite at pH = 8.0 as demonstrated by Kobayashi et al. [23].
Method (NAC-FAS): Formation of crystals of nanometric size in an alcoholic solution
It is a method developed by Ito et al. [24] which makes it
possible to synthesize nanocrystals at low temperature. This
method involves mixing (water - alcohol) in a solution containing
complexes of metal chelates or metal salts. This method was
used for the synthesis of crystalline TiO
Microwave method
In most studies, microwave treatment is used to obtain TiO2 nanopowders in colloidal suspension. Heating by microwaves allows homogeneity of the temperature in the solution and consequently an acceleration of the hydrolysis in a short time, which leads to promoting the nucleation of the nanoparticles.
Hu et al. [11] prepared spherical and monodisperse nanoparticles from the titanium sulfate precursor by heating using a microwave. This study showed that the power of microwaves has an effect on particle size. Indeed, the small size particles have been obtained by a high power while a low power leads to particles of large size. Microwave synthesis may allow better control of particle size and morphology [25, 26].
Sol-gel method
The sol-gel process was also used to obtain the TiO
Discussion
Generally, studies have shown that the difficulty of synthesis is not related to obtaining the crystallinity of the nanoparticles itself but rather to their irreversible aggregation. The crystallinity could be controlled during the synthesis or by a post-synthesis treatment as mentioned above. For example, Wang & Ying [31] obtained an amorphous white precipitate while mixing the titanium isopropoxide with water at room temperature. The amorphous nanoparticles formed were then hydrothermally treated to give an anatase crystal phase. In this study, several hydrothermal treatments were carried out between 80 °C and 240 °C in order to define a minimum and optimum treatment temperature.
Over the last decade, several teams have carried out studies to improve the previous synthesis processes while optimizing the dispersion of non-agglomerated nanopowders. The addition of multi-charged ions and polymers coating the NP surface favor respectively the repulsive electrostatic forces and the steric repulsive forces, which reduces the agglomeration [32-34]. Let’s remember that the nanoparticles can be present in the form of free primary particles, agglomerates or aggregates. The agglomerates correspond to a set of particles bound by weak forces (eg Van der Waals force) whereas aggregates are particles maintained by strong chemical bonds. The state of aggregated or agglomerated nanoparticles can be monitored during synthesis [35,36]. The agglomerates in a solution can be dispersed using several methods such as sonication by vibrating rod or even by ultrasonic bath unlike the aggregates.
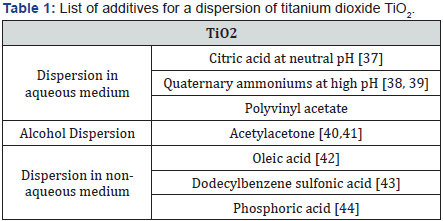
To allow a better dispersion of the nanoparticles in the liquid medium, there are two parameters to be taken into account: the pH and the isoelectric point. The isoelectric point is the pH for which the nanoparticle exists under a neutral electrical potential. If the pH is equal to the isoelectric point, there is no electrostatic repulsion and therefore the particles agglomerate. If the pH moves away from the isoelectric point, the particles repel each other as the electrostatic repulsive forces become dominant. Below, in Table 1, we describe the two most common methods which allow the stabilization of nanoparticles by adsorption of polymers or surfactants on their surface.
Barringer & Bowen [45] have shown that at the isoelectric point of TiO2 5.5±0.1, surface charges on particles in an acid medium help to maintain well dispersed particles in the solution. Similarly, Wang & Ying [31] have shown that the addition of hydrochloric acid HCl allows a change in the size of the nanoparticles from 20 nm to 14 nm, which leads to the conclusion that the acid has the effect of preventing the growth of the particles and the agglomeration while ensuring an electrostatic repulsion.
As said, the various studies stress the fact that the difficulty in syntheses is not related to the crystallinity of the nanopowders but rather to the spontaneous agglomeration of these nanoparticles which generates a production of particles with large sizes. The presence of water, inherent in the hydrolysis medium, seems to be at the origin of this agglomeration. Consequently the addition of water-substituting organic solvent or the addition of strong acid such as HCl in order to keep the solution at a very low pH may be an effective solution to reduce the agglomeration phenomenon, but this solution is opposed to the pH of the physiological medium (close to neutrality) required for biological application or toxicological studies.
The coating of nanoparticles is another way to avoid this phenomenon of agglomeration, just like washing the precipitate with ethanol may also cause a slowing down of the agglomeration. In order to avoid the aggregation phenomena of synthesized TiO2 nanoparticles, it is possible to functionalize them at the surface. One way is to work with citric acid in order to maintain the stability of the nanoparticles in an aqueous medium by controlling the rate of surface coating of the nanoparticles. In order to ensure colloidal stability over time, the adsorbed layer (polymers, surfactants and surfactant) must be sufficient to limit the attractive interactions between nanoparticles.
Conclusion
The various synthetic pathways lead to physico-chemical parameters (size, shape factor, aggregation and agglomeration state, porosity, impurities, surface state, surface chemistry, etc.) which can be very different. All these parameters can drastically affect the reactivity of the nanoparticles and therefore their toxicity. This highlights the crucial importance of precise physico-chemical characterization of nanoparticles for any nanotoxicology study. To conclude, with the precise definition of the nanoparticle, the challenge is to synthetize nanoparticles of various sizes while retaining constant the other parameters, if we want to truly highlight an effect of the size parameter.
References
- Znaidi JL, Seraphimova R, Bocquet J, Colbeau-Justin C, Pommier C (2001) A semi-continuous process for the synthesis of nanosize TiO2 powders and their use as photocatalysts. Materials Research Bulletin 36: 811-825.
- Oberdorster E (2004) Manufactured nanomaterials (Fullerenes, C-60) induce oxidative stress in the brain of juvenile largemouth bass. Environ Health Perspect 112(10): 1058-1062.
- Hoshino A, Fujioka K, Oku T, Suga M, Sasaki YF, et al. (2004) Physicochemical properties and cellular toxicity of nanocrystal quantum dots depend on their surface modification. Nano Letters 4(11): 2163-2169.
- Magrez A, Kasas S, Salicio V, Pasquier N, Seo JW, et al. (2006) Cellular toxicity of carbon-based nanomaterials. Nano Lett 6(6): 1121-1125.
- Aufray M, Menuel S, Fort Y, Eschbach J, Rouxel D, et al. (2009) New synthesis of nano-sized niobium oxides and lithium niobate particles and their characterization by XPS analysis. Journal of Nanoscience and Nanotechnology 9: 4780-4785.
- Mugnier Y, Houf L, El-Kass M, Le Dantec R, Djanta G, et al. (2011) In situ crystallization and growth dynamics of acentric iron iodate nanocrystals in w/o microemulsions probed by Hyper-Rayleigh Scattering measurements. J Phys Chem Chemistry 115(1): 23-30.
- Mohan S, Oluwafemi OS, Songca SP, Jayachandran VP, Rouxel D, (2016) Synthesis, antibacterial, cytotoxicity and sensing properties of starchcapped silver nanoparticles. Journal of Molecular Liquids 213: 75-81.
- Katsumata K, Ohno Y, Tomita K, Taniguchi T, Matsushita N (2012) Synthesis of Amphiphilic Brookite Nanoparticles with High Photocatalytic Performance for Wide Range of Application. ACS Appl Mater Interfaces 4(9): 4846-4852.
- Guo W, Lin Z, Wang X, Song G (2003) Sonochemical synthesis of nanocrystallineTiO2 by hydrolysis of titanium alkoxides. Microelectronic Engineering 66: 95-101.
- Massard C, Bourdeaux D, Raspal V, Feschet-Chassot E, Sibaud Y, (2012) One-pot Synthesis of TiO2 Nanoparticles in Suspensions for Quantification of Titanium Debris Release in Biological Liquids. Advances in Nanoparticles 1: 86-94.
- Hu M, Bai C, Song M, Lv X, Zhang S, (2012) Preparation of Spherical Monodispersed Titanium. International Journal of Remote Sensing Applications 2(1): 31-33.
- Cassaignon S, Koelsch M, Jolivet J (2007) Selective synthesis of brookite, anatase and rutile nanoparticles: thermolysis of TiCl4 in aqueous nitric acid. Journal of Materials Science 42(16): 6689-6695.
- Chithrani BD, Ghazani AA, Chan WCW (2006) Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Letter 6(4): 662-668.
- Wick P, Manser P, Limbach LK, Dettlaff-Weglikowska U, Krumeich F, et al. (2007) The degree and kind of agglomeration affect carbon nanotube cytotoxicity. Toxicol Lett 168(2): 121-131.
- Passagne I, Morille M, Rousset M, Pujalté I, L’azou B (2012). Implication of oxidative stress in size-dependent toxicity of silica nanoparticles in kidney cells. Toxicology 299(2-3): 112-124.
- GülYavuz A, Gök A (2007) Preparation of TiO2/PANI composites in the presence of surfactants and investigation of electrical properties. Synthetic Metals 157: 235-242.
- Rashad MM , Shalan AE, Lira-Cantu´ M, Abdel-Mottaleb M (2013) Synthesis and characterization of mesoporous anatase TiO2 nanostructures via organic acid precursor process for dye-sensitized solar cells applications. Journal of Industrial and Engineering Chemistry 19(6): 2052-2059.
- Chen X, Mao S (2006) Synthesis of Titanium Dioxide (TiO2) Nanomaterials. Journal of Nanoscience and Nanotechnology 6(4): 906- 925.
- H Zhang, Banfield J (1988) Thermodynamic analysis of phase stability of nanocrystalline titania. Journal of Materials Chemistry 9: 2073- 2076.
- FU, Jian-feng, JI, Min, An D (2005) Fulvic acid degradation using nanoparticle TiO2 in a submerged membrane photocatalysis reactor. Journal of Environmental Sciences (IOS Press) 17(6): 942-945.
- Zhang Y, Han K, Cheng T, Fang Z (2007) Synthesis, characterization, and photoluminescence property of LaCO3OH microspheres. Inorg Chem 46(11): 4713-4717.
- Wang X, Wang M, Song H, Ding B (2006) A Simple Sol- Gel Technique for Preparing Lanthanum Oxide Nanopowders, Mater Letter 60: 2261.
- Kobayashi M, Tomita K, Petrykin V, Yoshimura M, Kakihana M (2008) Direct synthesis of brookite-type titanium oxide by hydrothermal method using water-soluble titanium complexes. J Mater Sci 43(7): 2158-2162.
- Ito S, Inoue S, Kawada H, Hara M, Iwasaki M (1999) Low-Temperature Synthesis of Nanometer-Sized Crystalline TiO2 Particles and Their Photo induced Decomposition of Formic Acid. Journal of Colloid and Interface Science 216(1): 59-64.
- Bilecka I, Niederberger M (2009) Microwave Chemistry: Towards Predictable Nanoparticle Synthesis. Chimia 63(9): 581.
- Bilecka I, Niederberger M (2010) Microwave chemistry for inorganic nanomaterials synthesis. Nanoscale 2: 1358-1374.
- Su C, Hong B, Tseng C (2004) Sol-gel preparation and photocatalysis of titanium dioxide. Catal Today 96: 119-126.
- Ping Y, Cheng L, Nanping H (2002) Titanium Dioxide Nanoparticles Co-Doped with Fe3+ and Eu3+ Ions for Photo catalysis .Mater Lett 57: 794-801.
- Corradi A, Bondioli F, Focher B, Ferrari A, Grippo C, et al. (2005) Conventional and Microwave-Hydrothermal Synthesis of TiO2 Nanopowders Soc 88: 2639-264.
- Dai Y, Cobley CM, Zeng J, Sun Y, Xia Y (2009) Synthesis of Anatase TiO2 Nanocrystals with Exposed {001} Facets. Nano Lett 9(6): 2455-2459.
- Wang C, Ying J (1999) Sol-Gel Synthesis and Hydrothermal Processing of Anatase and Rutile Titania Nanocrystals. Chem Mater 11(11): 3113- 3120.
- Jiang J, Oberdosrster G, Biswas P (2009) Characterization of size, surface charge, and agglomeration state of nanoparticle dispersions for toxicological studies. J Nanoparticle Res 11(1): 77-89.
- Nguyen VS, Rouxel D, Hadji R, Vincent B, Fort Y (2011) Effect of ultrasonication and dispersion stability on the cluster size of alumina nanoscale particles in aqueous solutions. Ultrasonic Sonochemistry 18(1): 382-388.
- Nguyen VS, Rouxel D, Vincent B (2014) Dispersion of nanoparticles: from solvents to polymer solutions. Ultrason Sonochemistry 21 (1): 149-153.
- Jiang J, Chen D, Biswas P (2007) Synthesis of nanoparticles in a flame aerosol reactor (FLAR) with independent and strict control of their size, crystal phase and morphology. Nanotechnology 18: 285-603.
- Tsantilis S, Pratsinis SE (2004) Soft- and hard-agglomerate aerosols made at high Temperatures. Langmuir 20(14): 5933-5939.
- Mudunkotuwa IA, Grassian VH (2010) Citric Acid Adsorption on TiO2 Nanoparticles in Aqueous Suspensions at Acidic and Circumneutral pH: Surface Coverage, Surface Speciation, and Its Impact on Nanoparticle- Nanoparticle Interactions. J Am Chem Soc 132(42): 14986-14994.
- Chemseddine A, Moritz T (1999) Nanostructuring Titania: control over nanocrystal structure, size, shape and organization. Eur J Inorg Chem 2: 235-245.
- Yang J, Mei S, Ferreira J (2001) Hydrothermal synthesis of TiO2 nano powders from tetra alkyl ammonium hydroxide peptized sols. J Am Ceram Soc 84: 1696.
- Paz Y, Luo Z, Rabenberg L, Heller A (1995) Photo oxidative self-cleaning transparent titanium dioxide films on glass. J Mater Res 10(11): 2842- 2848.
- Scolan E, Sanchez C (1998) Synthesis and characterization of surfaceprotected nanocrystalline titania particles. Chem Mater 10 (10): 3217- 3223.
- Comparelli R, Fanizza E, Curri M, Cozzoli P, Mascolo G (2005) Photocatalytic degradation of azo dyes by organic-capped anatase TiO2 nanocrystals immobilized onto substrates. Appl Catal B 55: 81-91.
- Nussbaumer R, Caseri W, Tervoort T, Smith P (2002) Synthesis and characterization of surface-modified rutile nanoparticles and transparent polymer composites thereof. J Nanopart Res 4: 319-323.
- Demir M, Koynov K, Akbey U, Bubeck C, Park I, et al. (2007) Optical properties of composites of PMMA and surface-modified zincite nanoparticles. Macromolecules 40(4): 1089-1100.
- Barringer E, Bowen H (1985) Monodisperse TiO2 Powders by Hydrolysis of Titanium Tetraethoxide Aqueous Interfacial Electrochemistry and Dispersion Stability. Langmuir 1: 420-428.






























