Comparison of Executive Functioning in Current and Former Methamphetamine Users, and a Control Group
Farzane Jahan bakhsh, Matin Afshari Poor, Ladan Shahba and Marjan Shamspour*
Department of psychiatry, Shahid Beheshti hospital, Afzalipuor faculty of medicine, Kerman University of Medical Sciences, Kerman, Iran
Submission: November 11, 2025;Published: November 24, 2025
*Corresponding author:Marjan Shamspour, Department of psychiatry, Shahid Beheshti hospital, Afzalipuor faculty of medicine, Kerman University of Medical Sciences, Kerman, Iran
How to cite this article:Farzane Jahan b, Matin Afshari P, Ladan S, Marjan S. Comparison of Executive Functioning in Current and Former Methamphetamine Users, and a Control Group. Glob J Addict Rehabil Med. 2025; 7(5): 555723.DOI: 10.19080/GJARM.2025.07.555723.
Abstract
Objectives: Methamphetamine use is a global health issue associated with cognitive and neurological disorders, particularly in executive function. This study compared executive function among current users, former users, and a control group. It also examined the relations between executive function with duration of use, psychosis history, simultaneous substance use, and mood disorders.
Methods: This cross-sectional study included 162 individuals through convenience sampling, divided into three groups of 54: current users, former users, and control group. We assessed the executive function with Wisconsin Card Sorting Test (WCST), mood disorders with DASS-21 questionnaire, and demographic data and substance use with a data collection form. Data were analyzed with SPSS 25.
Results: Current and former users performed worse than the control group on the WCST (p<0.001). Former users performed better than current users but lagged behind the control group. A negative correlation was found between the duration of methamphetamine use and WCST performance (r=-0.42, p<0.01). Simultaneous substance use, psychosis history and mood disorders were associated with poorer WCST performance (p<0.05). Longer abstinence in former users was correlated with improved executive function (r=0.38, p<0.01).
Conclusion: Methamphetamine use impairs executive function, persisting partially after abstinence. Cognitive intervention and long-term follow-ups are essential for recovery.
Keywords: Methamphetamine; Executive Function; Psychosis; Abstinence; Mood Disorders; Cognitive Rehabilitation
Introduction
Substance use disorders have become a widespread issue globally, leading to numerous serious problems across physical, psychological, and social domains. Despite increased effects of substance use, substance use disorders continue to rise worldwide [1]. Methamphetamineis one of the most widely used illicit stimulant around the word, with significant prevalence reported across various countries [2]. In Iran, methamphetamine, locally referred to as “Shisheh,” has recently emerged as a critical public health concern. This stimulant raises levels of monoamines, especially dopamine, norepinephrine, and serotonin, in the central nervous system. Chronic use of methamphetamine is linked to structural and functional brain changes, resulting in various psychiatric and behavioral issues such as mood disorders, hallucinations, delusions, and disruptions in executive function. Executive functions, which are high-level cognitive processes responsible for behavior regulation, are closely associated with key psychological processes involved in awareness, thinking, and action [3,4]. Long-term methamphetamine use is associated with cognitive impairments [5]. potentially due to disruption in several neurotransmitter systems in the cortex [6], including the dopaminergic [7,8], serotonergic, noradrenergic, and glutamatergic systems [9]. Studies indicate that high doses of methamphetamine lead to reduced dopamine [10] and serotonin in the striatum, cortex, olfactory bulb [11], and prefrontal cortex [8,12,13], resulting in deficits in executive functioning [14].
Executive function broadly refers to the cognitive processes that guide and regulate behaviork [15]. Although definitions vary among researchers, executive functions generally encompass higher-order organizational and integrative skills necessary for goal-setting, planning, self-regulation, task execution, and feedback processing. These processes are controlled by neural pathways in the prefrontal cortex [16]. Executive functions help individuals organize, plan, inhibit inappropriate responses, adapt to new tasks, and manage emotions, memory, attention, and task transitions, making them crucial for adaptive behavior [17]. Executive dysfunction, especially in response inhibition and impulse control, has been linked to increased relapse risk [18]. Research has shown that strong executive control prevents immediate pursuit of pleasurable stimuli and supports balanced behavioral patterns, both of which are essential for preventing problematic substance use disorders [19]. Impairment in these functions can lead to difficulties with inhibition, response accuracy, and control over environmental stimuli [20]. Emerging neurocognitive models highlight executive control deficits as central mechanisms in the development and maintenance of substance dependence [21]. Extensive research underscores methamphetamine’s detrimental impact on executive and cognitive functioning [22]. Given the high prevalence of methamphetamine use and the significant functional impairments it causes, assessing executive dysfunction and its contributing factors is critical for designing effective interventions. This study aims to compare executive functioning across current methamphetamine users, abstinent individuals, and a control group, and to examine factors such as anxiety, depression, duration of methamphetamine use, age of onset, methamphetamine-induced psychosis, and cosubstance use. Understanding these factors can aid in developing targeted rehabilitation programs for methamphetaminedependent individuals. It is important to clarify that throughout this manuscript, the term “drug” refers to illicit psychoactive substances unless otherwise stated. Substances such as alcohol, tobacco, and caffeine are not the focus of this study.
Material and Methods
Research Setting, Population, and Sample Size
This cross-sectional, descriptive-analytical study
was conducted on three groups of participants: active
methamphetamine users, former users who had quit
methamphetamine, and a control group without a history of
methamphetamine use. Participants were recruited from two
methadone maintenance treatment (MMT) clinic in Kerman,
Iran from March to October 2024. Sampling was performed via
convenience sampling, and 54 individuals meeting the study’s
inclusion criteria were selected for the first group. The sample
size was calculated using the formula for comparing means across
three independent groups:
n = (Z₁₋α/2k + Z₁₋β)² × k × σ² / Δ²
In this formula, n is the sample size per group, k is the number of groups (3 in this study) Z₁₋α/2k is the Z-value for a 95% confidence level with Bonferroni correction for 3 groups (2.394), Z₁₋β is the Z-value for 80% power (0.84), σ² is the variance of the primary outcome variable, and Δ is the minimum meaningful difference between groups.
Based on similar studies evaluating executive function in
methamphetamine users, we estimated the variance (σ²) to be
100 and the minimum expected difference (Δ) to be 8. Substituting
these values into the formula:
n = (2.394 + 0.84)² × 3 × 100 / 8² = 10.459² × 300 / 64 ≈ 49
Considering a 10% potential dropout rate, the sample size was increased to 54 participants per group. Accordingly, the total sample size was determined to be 162 participants across the three groups. The second group included 54 individuals with a history of methamphetamine use that based on self-report did not use methamphetamine for at least four months and had negative urine toxicology test for methamphetamine at the time of the study. All individuals in both current and former user groups, methamphetamine use was via inhalation, specifically through glass pipes. The control group consisted of 54 students and staff from Kerman University of Medical Sciences. Each group was matched for age (±5 years) and gender to ensure comparability.
Inclusion and Exclusion Criteria and Ethical Considerations
Inclusion Criteria: For the methamphetamine user group,
participants had to be actively using methamphetamine, verified
by a positive urine test, and be between 18-50 years old with
basic literacy. For former users, criteria included negative urine
test results for methamphetamine at the time of the study,
abstinence for at least four months, age 18-50, and minimum
literacy. The control group participants, recruited from university
staff and students, were aged 18-50 and without a history of
methamphetamine use.
Ethical Considerations: Ethical considerations were
carefully followed, and informed consent was obtained from
all participants after explaining the study’s purpose. Data
confidentiality was maintained, with anonymous responses,
after approval by the university. Participation was voluntary,
with the option to withdraw at any time. The ethics committee
reviewed and approved this study and its ethics code is IR.KMU.
AH.REC.1403.141
Method and Tools of Data Collection
Data were collected using three primary instruments: a demographic questionnaire, the Depression Anxiety Stress Scale-21 (DASS-21), and the Wisconsin Card Sorting Test (WCST). The demographic questionnaire developed by the researcher, covering age, gender, marital status, education level, and methamphetamine use history, such as age at initiation, duration of use, and any concurrent substance use.
Psychological assessment was conducted using the Depression Anxiety Stress Scale-21 (DASS-21). The DASS-21 is a 21-item tool assessing levels of stress, anxiety, and depression, with each category containing seven items rated on a four-point Likert scale ranging from 0 “Never” to 3 “Always.” The DASS-21 is a short form of the original DASS-42, so each subscale score was doubled to obtain final scores. The Persian version of DASS-21 has high internal consistency, with reported Cronbach’s alphas of %95 for depression, %90 for anxiety, and %93 for stress [23].
To evaluate executive function, the Wisconsin Card Sorting Test (WCST) was used. The WCST measures cognitive processes like abstract reasoning and cognitive flexibility. It consists of 64 cards that participants must sort based on feedback provided by the examiner, challenging the participant’s ability to form, maintain, and shift mental sets. Two primary WCST scores were calculated: Categories Completed and Perseverative Errors. Categories Completed indicates a participant’s ability to understand and apply new sorting rules based on feedback, reflecting abstract reasoning. Perseverative Errors occur when the participant continues to respond according to an outdated rule despite new feedback, indicating cognitive rigidity. The WCST is widely regarded as the “gold standard” for assessing executive function related to prefrontal cortex activity, with high reliability for identifying cognitive deficits [24,25]. To ensure participant understanding, the researcher provided detailed instructions on how to complete the online WCST. Research assistants were available during questionnaire completion to respond to participant queries.
Data Analysis Tools and Methods
Data were analyzed using SPSS software, version 25. Descriptive statistics (frequency distributions, means, and standard deviations) were reported to summarize participant characteristics. Inferential statistics included chi-square tests to compare categorical variables across groups and oneway ANOVA for continuous variable comparisons among the methamphetamine user, former user, and control groups. For significant ANOVA results, post-hoc tests (Tukey or Bonferroni) were applied to identify specific group differences. Pearson or Spearman correlations, based on data normality, examined relationships between independent variables (e.g., age of onset, duration of use, anxiety, stress, depression) and executive function scores. To adjust for potential confounders, ANCOVA and multiple regression analyses were employed as necessary. The significance level was set at P < 0.05.
Results
This study was conducted on 162 participants, divided into three groups of 54: current methamphetamine users, former users, and a control group. The participants from current and former users were recruited from Shahid Beheshti Treatment Center and Dr. Ghasemi Addiction Treatment Center. In contrast, the control group was composed of students and staff from University of Medical Sciences in Kerman, Iran, recruited via voluntary participation announcements from March to October 2024. There was no statically significant difference in gender and age between these three groups. Although, significant differences were observed in education level and marital status among the groups. The control group had a notably higher education level, with 37.1% holding university degrees compared to about 20% in the other two groups. Regarding marital status, the control group included a higher percentage of married individuals (63.0%) compared to 46.3% in the former user group and 33.3% in the current user group. Conversely, the current user group had the highest percentage of single individuals (53.7%), compared to 40.7% and 27.8% in the former user and control groups, respectively (Table 1). These differences, which were statically significant, may be attributed in part to the different recruitment strategies used for the control group and should be considered as potential confounding variables in interpreting the results.
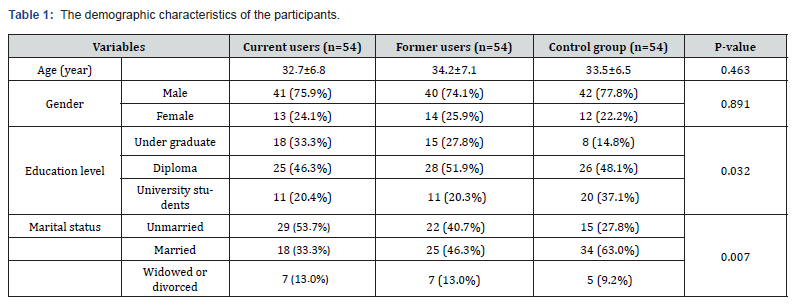
Turning to methamphetamine use characteristics in the current and former user groups, both groups had a similar age of onset, averaging around 24-25 years, and a similar duration of use. Additionally, current users were more likely to use other substances, including opioid, alcohol, heroin, and methadone, concurrently (70.4%) compared to former users (61.1%), though this difference was not statistically significant. A history of methamphetamine-induced psychosis was slightly more common among current users (35.2%) than former users (29.6%), although this difference was not statically significant (Table 2).
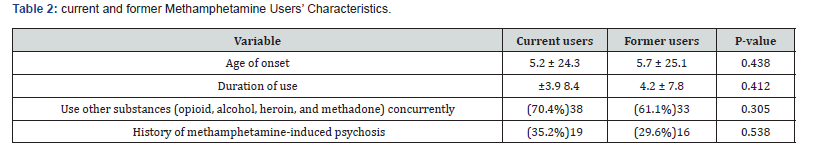
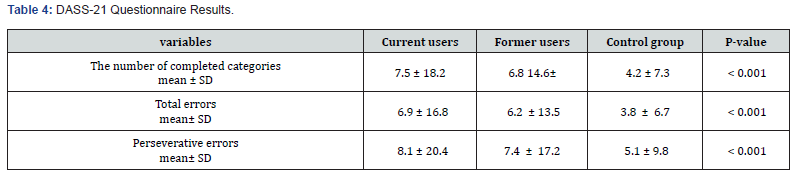
A one-way ANOVA showed statical significant differences in all WCST variables between the three groups. Tukey’s post hoc test revealed that the control group completed significantly more categories than the former (P < 0.001) and current users (P < 0.001), also the former users performed better than current users (P = 0.002). Besides that, the control group made significantly fewer total errors compared to both former (P < 0.001) and current users (P < 0.001), and former users made fewer errors than current users (P = 0.007). Additionally, the control group had significantly fewer perseverative errors compared to the former (P < 0.001) and current users (P < 0.001), with former users also showing fewer perseverative errors than current users (P = 0.003) (Table 3). A one-way ANOVA in the DASS-21 questionnaire across the three groups indicated significant differences in all subscales of the DASS-21. Tukey’s post hoc test showed that the control group had significantly lower scores in depression, anxiety, and stress compared to both former (P < 0.001) and current users (p < 0.001). Also, it revealed that former users had lower scores across all subscales compared to current users (depression: P = 0.012; anxiety: P = 0.018; stress: P = 0.031) (Table 4).
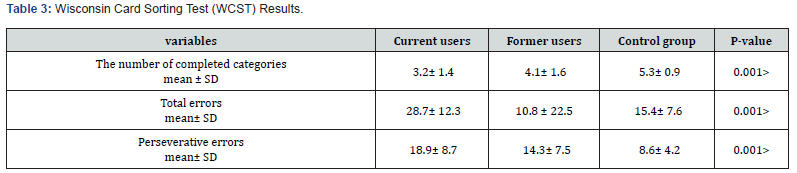
The result of pearson correlation coefficients for variables in the current user group, showed that the age of onset had a significant positive correlation with the number of categories completed in the WCST (r = 0.28, P < 0.05). Also, the duration of use positively correlated with both total errors (r = 0.39, P < 0.01) and perseverative errors (r = 0.37, P < 0.01). Additionally, performance on the WCST (number of completed categories, number of errors, and perseverative errors) shows a significant correlation with depression, anxiety, and stress scores (Table 5). To assess the impact of concurrent substance, use on executive function, an independent t-test was conducted. Results indicated that among current user group individuals who simultaneously use other substances (38 individuals) performed worse on the WCST. 16 individuals from current users’ group did not use another substance with methamphetamine..
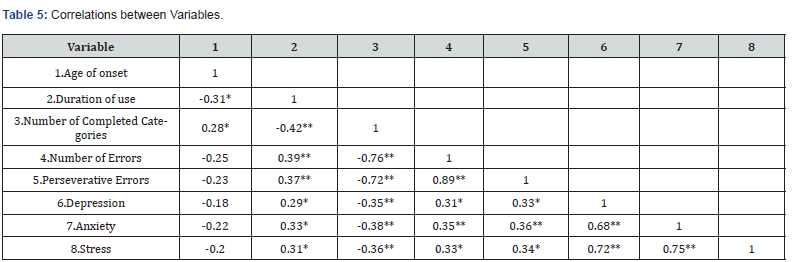
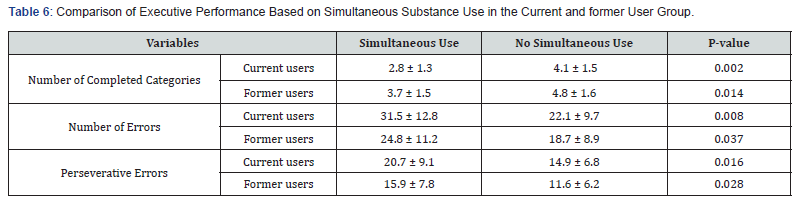
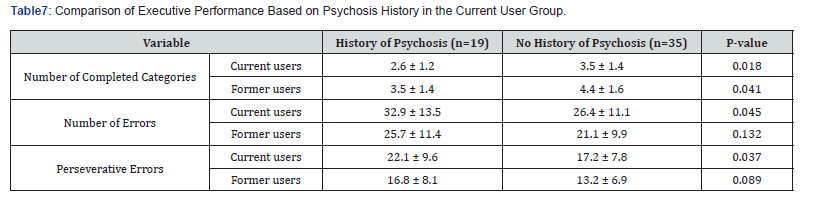
A similar trend was observed in the former user group, where those with concurrent substance use (21 individuals) showed poorer WCST performance. 33 individuals from this group did not use other substances simultaneously (Table 6). For finding the effect of Methamphetamine-Induced Psychosis on Executive Performance, an independent t-test was conducted to assess the effect of methamphetamine-induced psychosis on executive performance. 19 individuals had a history of psychosis and 35 of them did not have such experience. Besides that, results showed that in the current user group, individuals with a history of psychosis had poorer performance on the WCST. Similarly, significant differences in executive performance were observed in the former user group. 16 people from former users had a history of psychosis and 38 of them did not (Table 7). These results indicate that concurrent substance use and a history of methamphetamine-induced psychosis are associated with poorer performance on the Wisconsin Card Sorting Test (WCST).
Discussion
The present study aimed to compare executive functioning in current methamphetamine users, former users, and a control group. Our findings revealed that current methamphetamine users exhibited poorer performance on the Wisconsin Card Sorting Test (WCST) compared to the control group, aligning with [26], which showed significant impairments in executive functioning among methamphetamine users compared to control group [26]. Also, Kim et al. in 2006 reported that methamphetamine use is associated with deficits in cognitive flexibility and inhibitory control [27]. The study of Salas-González et al. in 2024, support these findings as well. They found significant deficits in WCST performance among methamphetamine users, emphasizing impairments in cognitive flexibility and control [28]. Moreover, our study demonstrated that the former user group also performed worse than the control group on the WCST but showed better results than the current methamphetamine users. This finding consistent with [29], which found that some cognitive deficits persist even after methamphetamine cessation [29]. However, the relative improvement in executive functioning among the former users group suggests the potential for recovery in executive function following abstinence. This aligns with Karabulut in 2023, which found that impairments in executive function could both predict and be impacted by ongoing methamphetamine use and abstinence [30].
One of the important findings of this study was the significant association between the duration of methamphetamine use and the severity of executive dysfunction. This result consistent with Nordahl et al. study in 2003, which indicated that both the duration and intensity of methamphetamine use are directly linked to cognitive impairment [31,32], similarly found that longer use durations were significantly associated with reduced attention and working memory performance among methamphetamine users [32]. Besides that, Kim and colleagues in 2020 conducted a longitudinal study tracking 50 methamphetamine users for two years. They found that improvements in executive functioning were directly related to the length of abstinence; however, even after two years, users’ performance did not reach the level of the control group [33]. Our results also indicated that simultaneous use of other substances was associated with poorer WCST performance, a finding consistent with Gonzalez et al. study in 2004. This study reported that poly-drug use exacerbates negative effects on cognitive function [34]. This may be due to the synergistic impacts of multiple substances on the nervous system [35]. confirm in his study that co-occurring substance use further impairs executive function and increases cognitive control deficits among methamphetamine users [35].
Another notable finding was the association between a history of methamphetamine-induced psychosis and poorer performance on the WCST. This result aligns with Glasner-Edwards et al., who showed that individuals with a history of methamphetamineinduced psychosis exhibited more severe cognitive impairments than those without psychosis history [36]. This might indicate broader neuronal damage in individuals predisposed to psychosis [32]. also demonstrated that methamphetamine users with psychotic symptoms had worse executive performance compared to those without psychosis [32]. In the present study, a significant relationship was observed between levels of anxiety, stress, and depression and executive functioning among methamphetamine users. This finding is in line with London et al. in 2004, which reported that mood disorders in methamphetamine users are associated with more pronounced cognitive deficits [37]. This relationship could be bidirectional, suggesting that mood disorders may act as both a cause and a consequence of cognitive impairments..
Conclusion
In conclusion, these findings emphasize the complex interplay between methamphetamine use, executive dysfunction, mental health, and poly-drug use. The data suggest that cognitive impairments are both a consequence and a potential perpetuating factor in substance use patterns. Interventions focused on early cessation, mental health support, and polysubstance abuse treatment are likely crucial to improving executive function and overall cognitive health in this population. Future studies should incorporate longitudinal designs and neurobiological assessments to further understand these associations.
References
- Gruber, S ST (2007) Neuropsychological Consequences of Opiate Use. Neuropsychic Rev 17(3): 299-315.
- TM Durell, L K C (2008) Prevalence of nonmedical methamphetamine use in the United States. Substance abuse treatment, prevention and policy 25(3): 19.
- Galbraith N (2018) The methamphetamine problem. BJPsych Bull 39(5): 218-220.
- Kray J K (2016) Executive Functions. cognative training.
- Simon S L, Andy C D, Xochitl C, John R M, Edythe D L (2010) Methamphetamine dependence and neuropsychological functioning: evaluating change during early abstinence. Journal of studies on alcohol and drugs 71(3): 335-344.
- Quinton M S Bryan K Y (2006) Causes and consequences of methamphetamine and MDMA toxicity. the AAPS journal 8(2): E337-47.
- Krasnova I N, Jean L C (2009) Methamphetamine toxicity and messengers of death. Brain research reviews 60(2): 379-407.
- Pm T, Kiralee M H, Sara L S, Jennifer A G, Michael S H, et al. (2004) Structural abnormalities in the brains of human subjects who use methamphetamine. J Neurosci 24(26): 6028-36.
- Meredith C W, Craig J, Kathleen A L, Andrew J S (2005) Implications of chronic methamphetamine use: a literature review. Harvard review of psychiatry 3(3): 141-154.
- Miyazaki M Y, Yukihiro Noda, Akihiro Mouri, Kazuto Kobayashi, Masayoshi Mishina, et al. (2013) Role of convergent activation of glutamatergic and dopaminergic systems in the nucleus accumbens in the development of methamphetamine psychosis and dependence. International Journal of Neuropsychopharmacology 16(6):1341-50.
- Deng X, Bruce Ladenheim, Subramaniam Jayanthi, Jean Lud Cadet (2017) Methamphetamine administration causes death of dopaminergic neurons in the mouse olfactory bulb. biological psychiatry 61(11):1235-1243.
- Salo R T, Thomas E Nordahl, Michael H Buonocore, Yutaka Natsuaki, Christy Waters, (2009) Cognitive control and white matter callosal microstructure in methamphetamine-dependent subjects: a diffusion tensor imaging study. Biological psychiatry 65(2): 122-8.
- Barbalat G, Valerian Chambon, Nicolas Franck, Etienne Koechlin, Chlöé Farrer, et al. (2009) Organization of cognitive control within the lateral prefrontal cortex in schizophrenia. Archives of general psychiatry 66(4): 37- 86.
- Gonzalez R, Antoine Bechara, Eileen M Martin (2007) Executive functions among individuals with methamphetamine or alcohol as drugs of choice: preliminary observations. Journal of clinical and experimental neuropsychology 29(2):155-159.
- Hughes C, Andrew Graham (2002) Measuring executive functions in childhood: Problems and solutions?. Child and adolescent mental health 7(3): 131-142.
- Lundqvist T (2005) Cognitive consequences of cannabis use: comparison with abuse of stimulants and heroin with regard to attention, memory and executive functions. Pharmacology Biochemistry and Behavior 81(2): 319-30.
- Garner J K (2009) Conceptualizing the relations between executive functions and self-regulated learning. The Journal of psychology 143(4): 405-26.
- Mintzer M, Maxine LS (2002) Cognitive impairment in methadone maintenance patients. Drug and alcohol dependence 67(1):41-51.
- Kalivas P W, Nora DV (2005) The neural basis of addiction: a pathology of motivation and choice. American Journal of Psychiatry 162(8):1403-13.
- Noël X, Martial Van der Linden, Mathieu d'Acremont, Antoine Bechara, Bernard Dan, et al. (2007) Alcohol cues increase cognitive impulsivity in individuals with alcoholism. Psychopharmacology 192(2):291-8.
- Lubman D, Murat Yücel, Christos Pantelis (2004) Addiction, a condition of compulsive behaviour? Neuroimaging and neuropsychological evidence of inhibitory dysregulation. Addiction 99(12): 1491-502.
- Kalechstein AD, Thomas F Newton, Michael Green (2003) Methamphetamine dependence is associated with neurocognitive impairment in the initial phases of abstinence. The Journal of neuropsychiatry and clinical neurosciences 15(2):215-20.
- Crawford J R, Julie D Henry (2003) The Depression Anxiety Stress Scales (DASS): Normative data and latent structure in a large non‐clinical sample. British journal of clinical psychology 42(Pt 2): 111-131.
- Nyhus E, Francisco Barceló (2009) The Wisconsin Card Sorting Test and the cognitive assessment of prefrontal executive functions: a critical update. Brain and cognition 71(3): 437-451.
- Pau C W, Tatia MC L, Shui-fun F C (2002) The impact of heroin on frontal executive functions. Archives of clinical neuropsychology 17(7): 663-670.
- Scott J C, Steven P W, Georg E M, Rachel A M, Robert K H, et al. (2007) Neurocognitive effects of methamphetamine: a critical review and meta-analysis. Neuropsychology review 17(3): 275-297.
- Kim S J, In Kyoon Lyoo, Jaeuk Hwang, Ain Chung, Young Hoon Sung, (2006) Prefrontal grey-matter changes in short-term and long-term abstinent methamphetamine abusers. International journal of neuropsychopharmacology 9(2): 221-228.
- Cecilia S G, Esteve G F, Erwin R V G, Ferran P B, Araceli K M S, et al. (2024) Memory and Executive Function Deficits in Abstinent Patients with Methamphetamine Use Disorder. international Journal of Mental Health and Addiction 22: 1501-1514.
- Volkow N D, L Chang, G J Wang, J S Fowler, D Franceschi, (2009) Loss of dopamine transporters in methamphetamine abusers recovers with protracted abstinence. Journal of Neuroscience 21(23): 9414-9418.
- Sercan Karabulut (2023) A 6imonth Follow-up Study: cognitive impairment may predict more frequent use of methamphetamine. Substance abuse: research and treatment 16(17): 11782218231175811.
- Nordahl T E, Ruth Salo, Martin Leamon (2003) Neuropsychological effects of chronic methamphetamine use on neurotransmitters and cognition: a review. The Journal of neuropsychiatry and clinical neurosciences 15(3): 317-325.
- Nutthika C, Kraiwattanapirom N, Siripornpanich V, Chetsawang J, Chetsawang B, (2024) Manifestation of Psychosis and Impairments of Executive Functions Emphasize the Interaction of Psychological and Neurological Dysfunctions in People Who Use Methamphetamine. Psychiatry Research and Reviews 20(4): 388-400.
- Kim Y T, Do-Hoon Kwon, Yongmin Chang (2011) Impairments of facial emotion recognition and theory of mind in methamphetamine abusers. Psychiatry Research 186(1): 80-84.
- Gonzalez R J, Julie D Rippeth, Catherine L Carey, Robert K Heaton, David J Moore, et al. (2004) Neurocognitive performance of methamphetamine users discordant for history of marijuana exposure. Drug and alcohol dependence 76(2): 181-190.
- Ani Zerekidze, Meng Li, Nooshin Javaheripour, Laura Huff, Thomas Weiss, et al. (2023) Neural Correlates of Impaired Cognitive Control in Individuals with Methamphetamine Dependence: An fMRI Study. Brain Sci 13(2): 197.
- Glasner-Edwards S, Larissa J Mooney, Patricia Marinelli-Casey, Maureen Hillhouse, Alfonso Ang, et al. (2008) Risk factors for suicide attempts in methamphetamine-dependent patients. American journal on addictions 17(1): 24-27.
- London E D, Sara L Simon, Steven M Berman, Mark A Mandelkern, Aaron M Lichtman, et al. (2004) Mood disturbances and regional cerebral metabolic abnormalities inrecently abstinent methamphetamine abusers. Archives of general psychiatry 61(1): 73-84.






























