Use of Dexmedetomidine and Ketamine for Rapid Opioids Detoxification in General Anesthesia
Ali Jabbari1*, Ebrahim Alijanpour2*, Vahid Khuri3 and Alireza Jahangirifard4
1Assistant professor, Department of Anesthesiology and Intensive Care Medicine, Golestan University of Medical Sciences, Iran
2Associated professor, Department of Anesthesiology and Critical Care Medicine, Babol University of Medical Sciences, Iran
3Professor of Ischemic Disorders Research Center of Golestan University of Medical Sciences, Iran
4Associate professor, Shahid Beheshti University of Medical Sciences, Iran
Submission: February 12, 2019;Published: March 01, 2019
*Corresponding author: Ebrahim Alijanpour, Ali Jabbari, Department of Anesthesiology and Critical Care Medicine, Ischemic Disorders Research Center of Golestan University of Medical Sciences, Iran
How to cite this article: Ali Jabbari, Ebrahim Alijanpour, Vahid Khuri, Alireza Jahangirifard. Use of Dexmedetomidine and Ketamine for Rapid Opioids Detoxification in General Anesthesia. Glob J Add & Rehab Med. 2019; 6(3): 555690. DOI: 10.19080/GJARM.2019.06.555690.
Abstract
Deliverance from opioids dependency is challenging subjects and there are varieties of methods. There are new approaches base on anesthesia techniques for rapid opioids’ detoxification and recently, these methods are increasingly popular. These methods aimed at reducing the time of opioid detoxification and decreasing intensity of opioids withdrawal. Our protocol for opiate detoxification under general anesthesia by using Propofol, Dexmedetomidine plus ketamine while patients receive Naloxone represents a potentially effective treatment, and it could constitute as a safe method. We found less hemodynamic changes, withdrawal signs like involuntary movement, muscle twitch, lacrimation and sweating comparison with a combination of Propofol, Midazolam, and Atracurium. We believe that a well- designed protocol will accelerate detoxification and attenuate withdrawal symptoms.
Keywords: Rapid opioid detoxification; Propofol; Dexmedetomidine; ketamine; Naloxone
Abbreviations: GA: General Anesthesia, ICU: Intensive Care Unit, CINA: Clinical Institute Narcotic Assessment, DPKA: Dexmedetomidine, Propofol, Ketamine, Atracorium, PMA: Propofol, Midazolam, Atracorium
Opinion
Opioids addiction is one of the most known social damages. Addiction to the opioids can begin after an initial short time of substances abuse. This period is variable base on the kind of substance and the root of abuse. Opioids addiction is associated with extremely unpleasant collateral social, economic and psychological damages. Deliverance from opioids dependency and substance abuse management are challenging subjects and there are varieties of methods for this purpose [1-3]. A list of serious complications such as respiratory distress, hemodynamic instability, cardiovascular collapse and shock, Anxiety, mood disorder, seizure, and psychotic episodes have been described when opioids are stopped. Choosing an appropriate method associated with minimal side effects is a wise selection [3-6].
Conventional detoxification protocol includes tapering doses of a long-acting substitute agonist drug like Methadone, the use of Clonidine which, has been used in combination with μ receptor antagonists to reduce opioids tendency. This traditional technique requiring at least 3-4 weeks times and associated with the chronic withdrawal syndrome which; may necessitate admission in hospital. Consequently, significant initial dropout rates have been seen [7-8]. There are new approaches base on anesthesia technique for rapid opioids detoxification and recently, these methods are increasingly popular for detoxifying patients who addicted to opiates. This different approach to detoxification from opioids is based on the administration of a high-dose μ-receptor antagonist during General Anesthesia (GA). This concept is supported by the several studies; which demonstrated that biochemical indexes of opioid withdrawal primarily involving the nucleus locus coeruleus that could return to the near baseline within 8-10 h after administration of high doses of the opioid antagonist [8-9].
Recent publications showed that the use of alpha 2-adrenoreceptor agonists and N-Methyl Disparate (NMDA) antagonists such as Dexmedetomidine and ketamine could minimize tolerance development to opioids agents. Ketamine is a drug that has traditionally been used as an anesthetic medicine. It could display interesting qualities for dealing with some symptoms relating to opioids withdrawal [9-12]. We considered these concepts and set a practical protocol for rapid opioids detoxification under generalized anesthesia. Our sample size was about 5 patients and we compare our observational data with past experiences (10 patients) in rapid opioids detoxification that we used the combination of propofol, Midazolam, Atracurium (PMA method) with or without inhalational agents.
Protocol and Results
After 8-10 hours withdrawing opioids and receiving 4mg Ondansetron and 150 mg Ranitidine intravenously, patients were transported to the Intensive care unit (ICU). Fluid loading for dehydration compensation was calculated by assuming that each patient had an 8 hours deficit. Ringer’s solution was infused through an 18-gauge peripheral intravenous catheter. Monitors included Electrocardiography, noninvasive blood pressure, pulse oximetry, and skin temperature probe. After induction of GA, a urinary catheter, a nasogastric tube and a 20-G arterial line catheter for repetitive arterial blood gas analyzing and lab data sampling was prepared.
The hemodynamic parameter was recorded, and Withdrawal scores were determined using an adapted scoring system referred to as the Clinical Institute Narcotic Assessment (CINA) withdrawal scale.
Initially, Patients received 0.5 mcg/kg Dexmedetomidine during 30 minutes in the ICU. We proposed prescribed Dexmedetomidine was decreasing systemic effects of the withdrawal that would soon occur as a result of the administration of the antagonist [12-13]. GA was induced with 2 mg/kg propofol and 0.5 mg/kg Atracurium. The trachea was intubated, and the patients were mechanically ventilated with fractional inspired oxygen concentration about 40 percent. GA was maintained by Propofol (150-200 mcg/kg/min) infusion combined with ketamine (20 mg/h), Dexmedetomidine (0.3 mcg/h) and Atracurium (0.01- 0.15 mg/kg/min); DPKA method.
The patients’ vital signs were recorded every 15 minutes. Low dose Atropine (0.1- 0.2 mg) was used to control gastrointestinal secretions and repeated if need. In our patients, we prescribed 0.2 mg Naloxone for the partial reversal of opioid effects 15 minutes after the induction of GA. We intended to test the intensity of opioid withdrawal on hemodynamic achieved by prescribing the titrated dose of opioids antagonist. The patients were observed for signs of withdrawal (piloerection, lacrimation, hemodynamic changes) for 15 min. We diluted Naloxone to 4 mcg/mL in Normal Saline and if; we could not have any unexpected and uncontrolled reaction then, continued Naloxone infusion 0.2- 0.8 mg/hours for 8 hours.
After the Naltrexone infusion time was over, administration of Atracurium and Ketamine was discontinued, allowing involuntary recovery under Propofol 50mcg/kg/min and Dexmedetomidine 0.3 mcg/h infusion over the next 8 hours to avoid having unexpected reaction and patients discomfort. Patients were emerged from GA and monitored for withdrawal signs for 8 hours after extubation. In other words, we design a 24 hours rapid opioid detoxification in 3 phases. Then, patients transfer to a general ward for 24 hours and discharge from hospital if they could tolerate oral intake, absent vomiting, controlled diarrhea and muscle ache by medication, normal urine output, unassisted ambulation and normal vital sign with acceptable mental status.
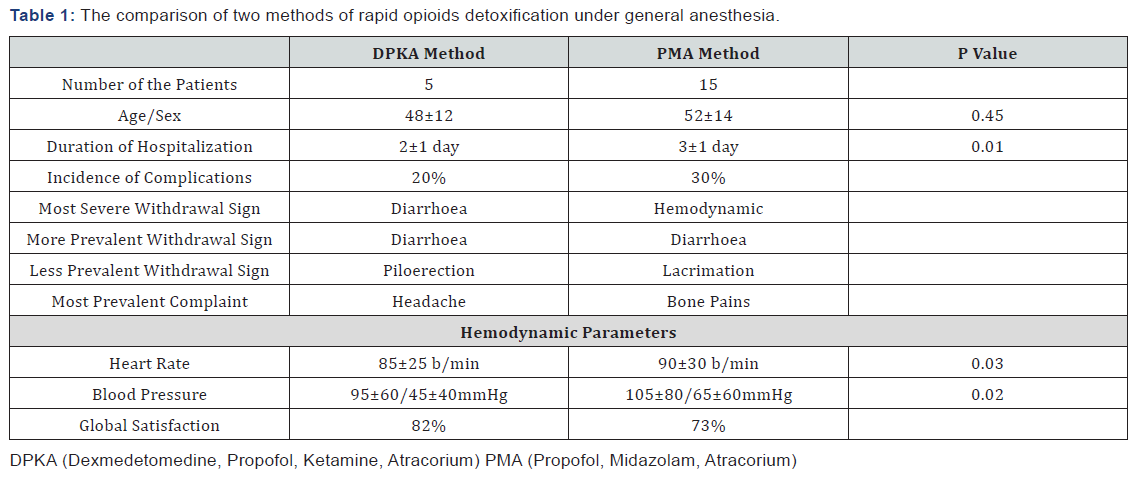
Based on our observation and registered data in patients’ profiles, we found less hemodynamic changes, withdrawal signs like involuntary movement, muscle twitch, lacrimation and sweating comparison with the combination of Propofol, Midazolam, and Atracurium (PMA- routine technique) (Table 1). None of our patients underwent a life-threatening complication and only one patient was discharged from hospital by his own determination and legally informed consent, out of our plan. Patients were more comfort and satisfy by DPKA methods due to short time hospitalization.
Conclusion
Rapid opiate withdrawal precipitated by Naloxone during GA, usually with the addition of α-2 adrenergic agonists, is an increasingly popular technique. These techniques aimed at reducing the time for opioid detoxification and decreasing intensity of opioids withdrawal by using GA base on Propofol coupled with Ketamine and Dexmedetomidine. The prospect of avoiding or shortening the worst discomforts of withdrawal may encourage more opiate addicts to attempt detoxification [8,10,12,14-17]. We believe that a well-designed protocol will accelerate detoxification and attenuate withdrawal symptoms.
Although, our protocol in opiate detoxification under GA by using Propofol, Dexmedetomidine plus ketamine represents a potentially effective treatment and we evaluate these methods as a safe technique but, there are criticisms to be made. We could not present a powerful statistical analysis due to our small sample size and contentment to our observations and clinical judgments. We could not evaluate our patients for a long term for possible side effects assessment and psychotic disorder of our treatment besides we have not a control group simultaneously.
References
- Raith K, Hochhaus G (2004) Drugs used in the treatment of opioid tolerance and physical dependence: a review. Int J Clin Pharmacol Ther 42(4): 191-203.
- Nasr DA, Omran HA, Hakim SM, Mansour WA (2011) Ultra-rapid opiate detoxification using dexmedetomidine under general anesthesia. J Opioid Manag 7(5): 337-344.
- Simon DL (1997) Rapid opioid detoxification using opioid antagonists: history, theory and the state of the art. J Addict Dis 16(1): 103-122.
- Gold ML, Sorensen JL, Mc Canlies N, Trier M, Dlugosch G (1988) Tapering from methadone maintenance: Attitudes of clients and staff. J Subst Abuse Treat 5(1): 37-44.
- Alison M Diaper, Fergus D Law, Jan K Melichar (2013) Pharmacological strategies for detoxification. Br J Clin Pharmacol 77(2): 302-314.
- Maccioli GA (2003) Dexmedetomidine to facilitate drug withdrawal. Anesthesiology 98(2): 575-577.
- Collins ED, Kleber HD, Whittington RA, Heitler NE (2005) Anesthesia-assisted vs buprenorphine- or clonidine-assisted heroin detoxification and naltrexone induction: a randomized trial. JAMA 294(8): 903-913.
- Dijkstra BA, De Jong CA, Bluschke SM, Krabbe PF, vander Staak CP, et al. (2007) Does naltrexone affect craving in abstinent opioid-dependent patients? Addict Biol 12(2): 176-182.
- Rasmussen K, Beitner Johnson DB, Krystal JH, Aghajanian GK, Nestler EJ, et al. (1990) Opiate withdrawal and the rat locus coeruleus: Behavioral, electrophysiological, and biochemical correlates. J Neurosci 10(7): 2308-2317.
- Griffiths R (2010) Opioid-induced hyperalgesia: low-dose ketamine does work for some Orthopaedic problems already. Br J Anaesth 104(5): 660-661.
- Morgan CJ, Curran HV (2012) Independent Scientific Committee on Drugs. Ketamine use: a review. Addiction 107(1): 27-38.
- Finkel JC, Elrefai A (2004) The use of dexmedetomidine to facilitate opioid and benzodiazepine detoxification in an infant. Anesth Analg 98(6): 1658-1659.
- Tobias JD (2006) Dexmedetomidine to treat opioid withdrawal in infants following prolonged sedation in the pediatric ICU. J Opioid Manag 2(4): 201-205.
- Seoane A, Carrasco G, Cabre L, Puiggros A, Hernandez E, et al. (1997) Efficacy and safety of two new methods of rapid intravenous detoxification in heroin addicts previously treated without success. Br J Psychiatry 171: 340-345.
- Collins ED, Kleber HD, Whittington RA, Heitler NE (2005) Anesthesia-assisted vs buprenorphine- or clonidine-assisted heroin detoxification and naltrexone induction: a randomized trial. JAMA 294(8): 903-913.
- Ito H, Sobue K, Hirate H, Sugiura T, So M, et al. (2006) Use of ketamine to facilitate opioid withdrawal in a child. Anesthesiology 104(5): 1113.
- Nasseri K, Ahsan B, Farhadifar F, Shami S (2010) Shortening Anesthesia Duration does not Affect Severity of Withdrawal Syndrome in Patients Undergoing Ultra Rapid Opioid Detoxification. Acta Med Iran 48(1): 27-32.






























