Preparation of Patterned Polymeric Scaffolds by Soft Lithographic Method and their Characterization for Tissue Engineering
Ayse Zehra Aroguz*
Istanbul-Cerrahpasa University, Turkey
Submission: December 10, 2018;Published: January 16, 2019
*Corresponding author: Ayse Zehra Aroguz, Faculty of Engineering, Department of Chemistry, Istanbul Cerrahpasa University, Istanbul, 34850, Turkey
How to cite this article: Ayse Zehra Aroguz. Preparation of Patterned Polymeric Scaffolds by Soft Lithographic Method and their Characterization for Tissue Engineering. Glob J Add & Rehab Med. 2019; 6(3): 555688. DOI: 10.19080/GJARM.2019.06.555688.
Abstract
Nowadays, cell growth studies on patterned surfaces have received increasing interest due to their cell control and substrate adhesion properties. In this study, patterned scaffolds were prepared by using Soft Lithographic Method. The structural and morphological properties of the prepared materials were analyzed for their use within cell growth systems. Microscope glasses were used as the bottom holder and coated with different polymers; polyethylene glycol dimethyl acrylate (PEG-DMA), polyvinyl chloride (PVC), and polymethyl methacrylate (PMMA) in order to compare the efficiency of the samples on the cell growth. The coated surfaces were patterned using poly (dimethyl siloxane) (PDMS) molds which were fabricated originally as soft materials for pattern transfer and used as stamps and as scaffolds.
Keywords: Soft Lithography; Pattern; Scaffold; Cell Growth; PDMS Mould
Introduction
Many studies have been reported on the preparation of functional scaffolds for cell growth systems [1-3]. Cell morphology and growth observation studies were performed on the novel, patterned polymeric surfaces. Several techniques were developed for the fabrication of the patterned surfaces, including laser cutting, soft-lithographic patterning, electrochemical deposition, etc [4]. Lee and co-workers reported three dimensional micropatterning hydrogels including bioactive properties to guide three dimensional cell migration [5]. Chen produced micro patterned surfaces to control the cell shape, and its position [6]. Whiteside’s and co-workers have widely studied the development of the soft-lithography for cell growth applications [7,8]. Some polymers and polymeric blends such as, SU-8, Poly (dimethyl siloxane) (PDMS), and polyethylene glycol dimethyl acrylate (PEG-DMA) are widely used for production of patterned surfaces [9-11]. Among these materials, PDMS is preferably used as pattern transfer stamp to produce the nanopatterned polymeric scaffold because of its superior properties like transparency and durability [12,13]. Vozzi and co-workers fabricated polymeric scaffolds by using different techniques to create different structures. They compared the advantages and limitations of these techniques with respect to resolution, cost, and preparation variables [14]. In this work, Soft Lithographic Method was used to fabricate patterned surfaces on microscope glass. The microscope glasses were covered by the polymers before pattering. PEG-DMA, PMMA, PVC were used as coating polymers. PDMS molds were originally produced in this study as soft material stamps to transfer the desired pattern. PDMS was also used as a scaffold to compare their efficiency for the cell growth systems. Cell growth was monitored using neutral red absorption assay [15]. Cell viability was observed on these patterned surfaces and the results were compared.
Experimental
Materials
Poly (ethylene glycol dimetacrylate) (PEG-DMA) was supplied from Sigma Aldrich company. Poly (dimethyl siloxane) (PDMS) and hardener were purchased from Dow Corning Corporation. Poly (vinyl chloride (PVC) was obtained from Fluke.
Preparation of the Scaffolds
In this work, the preparation of patterned surfaces involves successive steps following each other in a certain order. These steps are:
a) Drawing and the printing of the pattern (photomask).
b) Production of the elastomeric PDMS stamp which is used for the pattern transfer.
c) Coating of the microscopic glass with a photoresist polymer.
d) Using the photomask, the surface of the photoresist is patterned by passing UV light along the pattern.
e) Production of the PDMS stamp by pouring liquid PDMS onto the photoresist material.
f) The pattern was transferred to the PDMS after drying process. The PDMS stamp replicates the opposite of the master.
g) The PDMS mould was contacted with the polymer (PEGDMA, PVC, PMMA) covered microscope surfaces using a force approximately 10 kN after being immersed into an alkanethiol solution.
Cell Growth Experiments
The polymer coated and patterned microscope glasses were used as scaffolds for the cell growth studies. L929 mouse fibroblasts were used in vitro cell culture experiments. Cell growth after adhesion of the cells was observed on these materials. The number of the cells was measured by neutral red uptake assay.
Results and Discussions
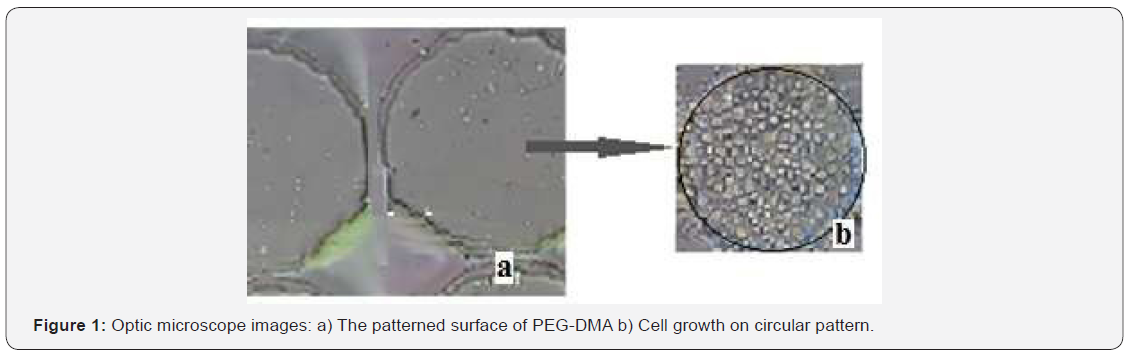
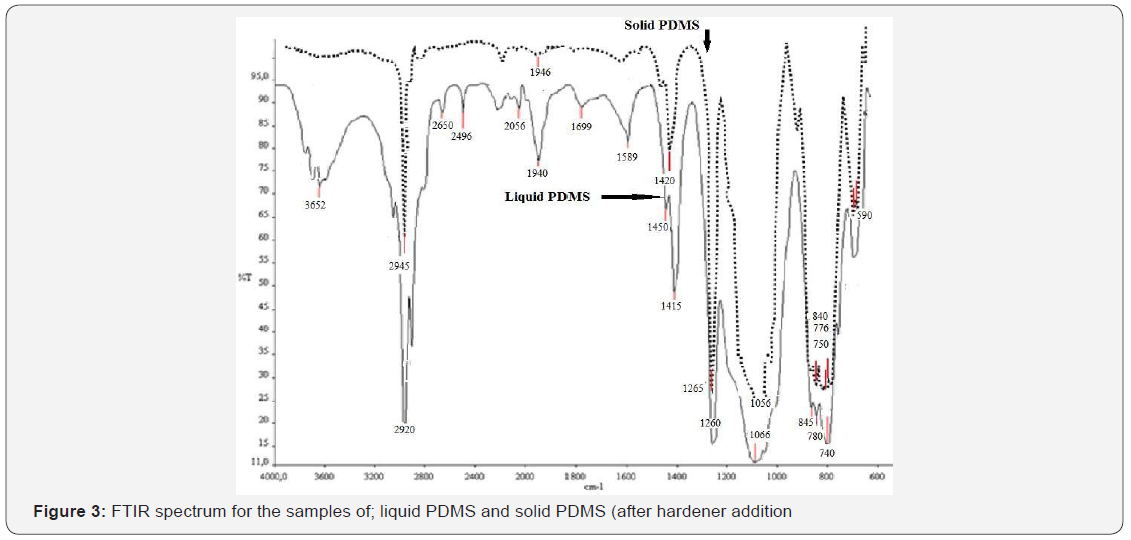
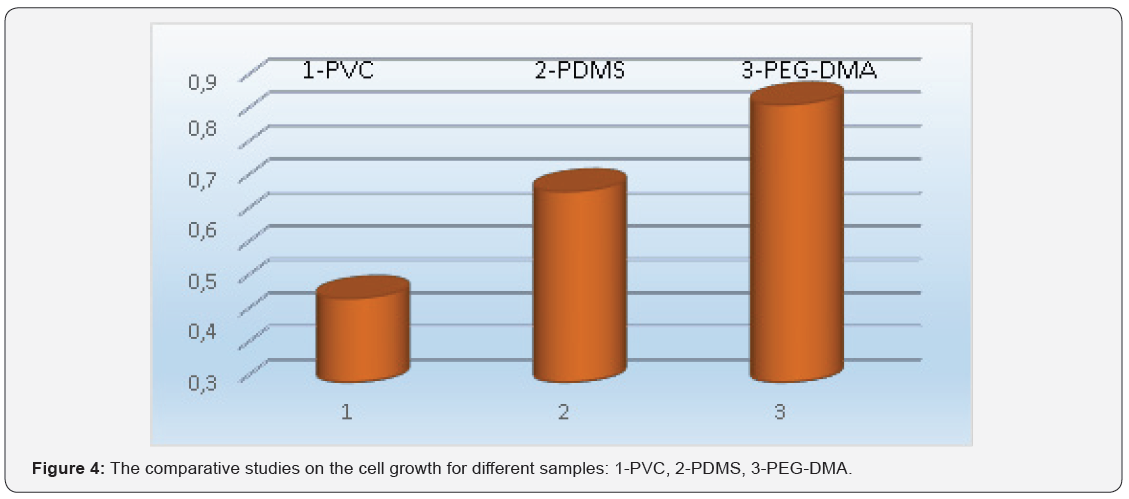
In this work, PVC, PEG-DMA and PMMA covered microscope glasses were patterned by using PDMS mould and cell growth studies were performed on these samples. The pattern and the cell growth properties were followed by an optic microscope and a scanning electron microscope (SEM). The optic microscope images of the samples were analyzed before and after cell growth experiments. The proliferation of the cells was observed after 72 hours incubation at 37°C. The results are shown in Figure 1. Figure 2 shows scanning electron microscope results and cell grown on the patterned surface. The microscope results confirmed the adhesion of cells on these substances. FTIR spectra of solid and liquid PDMS were compared and the results are shown in Figure 3. According to Figure 3 it can be seen that the strong band on the FTIR spectra for solid and liquid PDMS at 1260–1265 cm−1 are related to the Si-CH3 bending and stretching vibrations. The peaks of liquid and solid spectra correspond to the hardening effect, before and after the addition to the sample. The siloxane bonds (Si-O-Si) observed at 1066 cm−1 shifted to a broad band around 1056 cm−1 following the addition of the hardener. On FTIR spectra in (Figure 3) no significant change is observed. These results show that the structure of PDMS is not significantly changed in the presence of the hardener. The cell grown experiments were performed on different samples to compare the result. These comparative studies are shown in Figure 4. Cell growth was monitored in each group of experiments and successful results were obtained (except PMMA-covered samples). Cell growth studies were also attempted on the PMMA covered samples but cell growth did not occur on the surface of these samples.
SEM and optic microscope results as seen in Figures 1 & 2 support the successful adhesion of cells on the scaffolds. Both microscope results show the patterns were transferred onto the polymer coated surface from PDMS stamps. UV-exposure time chosen as 10 mins was good enough for these experiments. The high absorbance value in Figure 4 was obtained for the samples of PEG-DMA scaffold. PDMS was also used as scaffold to examine the cell growth results and it was found that PDMS covered and patterned surface is more effective than PVC covered surface for the cell growth studies.

Conclusion
The patterned scaffold materials are used effectively in tissue engineering and biotechnological applications among cell multiplication studies. The results of the cell growth experiments indicate that the materials prepared and patterned in this study by using PEG-DMA, PVC and PDMS are suitable for cell viability studies.
References
- H Yu, S Xiong, CY Tay, WS Leong, LP Tan (2012) A novel and simple microcontact printing technique for tacky, soft substrates and/or complex surfaces in soft tissue engineering. Acta Biomaterialia 8(3): 1267-1272.
- X Wu, Y Liu, X Li, P Wen, Y Zhang, et al. (2010) Preparation of aligned porous gelatin scaffolds by unidirectional freeze-drying method, Acta Biomaterialia 6(3): 1167-1177.
- L Chen, C Yan, Z Zheng (2018) Functional polymer surfaces for controlling cell behaviors. Materials Today 21(1): 38-59.
- D Qin, Y Xia, GM White sides (2010) Soft lithography for micro- and nanoscale patterning Nature protocols 5(3): 491-502.
- SH Lee, JJ Moon, JL West (2008) Three-dimensional micropatterning of bioactive hydrogels via two-photon laser scanning photolithography for guided 3D cell migration. Biomaterials 29(20): 2962-2968.
- CS Chen, M Mrksich, S Huang, GM White sides (1998) Micropatterned surfaces for control of cell shape, position, and function, Biotechnol Prog 14(3): 356-363.
- DJ Lipomi, RV Martinez, L Cademartiri, GM Whitesides (2012) Soft Lithographic Approaches to Nanofabrication Polymer Science: A Comprehensive Reference 7
- X Jiang, S Takayama, RG Chapman, RS Kane, GM White sides, et al. (2014) Chapter-19 Micro-scale patterning of cells and their environment; Principles of Tissue Engineering (Fourth Edition) Academic press Pages 359- 384.
- MC Lensen, P Mela, A Mourran, J Groll, J Heuts, et al. (2007) Micro- and Nanopatterned Star Poly (ethylene glycol) (PEG) Materials Prepared by UV-Based Imprint Lithography, Langmuir 23(14): 7841-
- P Kim, DH Kim, B Kim, SK Choi, SH Lee, et al. (2005) Fabrication of nanostructures of polyethylene glycol for applications to protein adsorption and cell adhesion. Nanotechnology 16(10): 2420-
- Aroguz AZ, Baysal BM (2000) Thermal, mechanical, and morphological characterization studies of poly (2,6- dimethyl-1,4-phenylene oxide) blends with polystyrene and brominated polystyrene. J Appl Polymer sci. 75(2): 225-231.
- CA Farcau, S Astilean (2005) Optical and structural characterization of periodic silver polystyrene nanocomposites, J Optoelectron Adv M 7: 2721-
- Z Adiguzel, SA Sagnic, AZ Aroguz (2017) Preparation and characterization of polymers based on PDMS and PEG-DMA as potential scaffold for cell growth. Materials Science and Engineering 78: 942-948.
- G Vozzia, CR Flaimb, A Ahluwaliaa, S Bhatiab (2003) Fabrication of PLGA scaffolds using soft lithography and micro syringe deposition. Biomaterials 24(14): 2533-
- K Baysal, AZ Aroguz, Z Adiguzel, BM Baysal (2013) Chitosan/alginate crosslinked hydrogels: preparation, characterization and application for cell growth purposes. Int J Biol Macromol 59: 342-






























