Coupling Genetic Addiction Risk Score (GARS) and Pro Dopamine Regulation (KB220) to Combat Substance Use Disorder (SUD)
Kenneth Blum1-10*, Margaret A Madigan4, Lyle Fried7, Eric R Braverman6, John Giordano5 and Rajendra D Badgaiyan9
Department of Psychiatry & McKnight Brain Institute, University of Florida College of Medicine, USA
Department of Psychiatry and Behavioral Sciences, Keck Medicine University of Southern California, USA
Division of Applied Clinical Research & Education, Dominion Diagnostics, USA
Department of Neurogenetics, Igene, USA
National Institute for Holistic Addiction Studies, USA
Department of Clinical Neurology, Path Foundation NY, USA
Division of Neuroscience Based Addiction Therapy, The Shores Treatment & Recovery Center, USA
Eötvös Loránd University, Institute of Psychology, Europe
Department of Psychiatry, Wright State University Boonshoft School of Medicine and Dayton VA Medical Center, US
Division of Reward Deficiency Syndrome, Nupathways, Inc., Innsbrook, MO, USA
Submission: February 11, 2017; Published: February 23, 2017
*Corresponding author: Kenneth Blum, Department of Psychiatry, University of Florida College of Medicine, Gainesville, FL, USA; Tel:352-294-4911;Fax:352-392-9887; Email:drd2gene@gmail.com
How to cite this article: Kenneth B, Margaret A M, Lyle F, Eric R B, John G, Rajendra D B.Coupling Genetic Addiction Risk Score (GARS) and Pro 002 Dopamine Regulation (KB220) to Combat Substance Use Disorder (SUD). Glob J Add & Rehab Med. 2017; 1(2): 555556. DOI: 10.19080/GJARM.2017.01.555556
Abbreviations: RDS: Reward Deficiency Syndrome; SMART™: Systematic Medical Approach to Reward Transformation; GARS: Genetic Addiction Risk Score; DNA: Deoxyribonucleic Acid; DRD2: Dopamine Receptor D2 Gene; MOA: Monoamine Oxidase; SNPs: Single-Nucleotide Polymorphisms; SSRs: Simple Sequence Repeats
Introduction
We are proposing a generalized approach based on the Reward Deficiency Syndrome (RDS) conceptualization called the Systematic Medical Approach to Reward Transformation (SMART™). This system consists of: early pre-disposition diagnosis (even in children) using the Genetic Addiction Risk Score (GARS) [1]; a validated RDS questionnaire [2]; urine drug testing during actual treatment that uses comprehensive analysis of reported drugs to determine compliance with prescription medications and non-abstinence illicit drugs [3]; and adjunctive treatment with a glutaminergic-dopaminergic optimization nutraceutical (KB220) to prevent relapse by induction of dopamine homeostasis [4].
Understanding reward deficiency syndrome (RDS)
I. RDS conceptualizationThe biological processes of reward that underlie addiction to substances and all addictive, compulsive and impulsive behaviors are the basis of the RDS conceptualization [5,6]. RDS then is a deficiency, a hypodopaminergic condition that results from some combination of genetic variations, environmental stressors, and adverse molecular effects or blunting due to prolonged substance use or behavioral habituation [7-9]. The RDS concept was developed based on animal and human research that explored the molecular biology of neurotransmission, and behavioral genetics [8,10,11]. Understanding this concept, explained in the following paragraphs, is central to treating the abnormal psychology of personality and spectrum disorders, as well as, substance and non-substance (behavioral) addictions. To feel ordinary pleasure, complex interactions of neurotransmitters regulate the dopaminergic activity of the brain in the reward center -the mesolimbic system, particularly the nucleus accumbens. Individuals, who suffer from a lack of ordinary pleasure in their lives, are predisposed to use any means; substance or behavior, to activate dopamine release, relieve stress and feel healthy pleasure [12,13].
Genes are deoxyribonucleic acid (DNA), which directs the functional properties of proteins like neurotransmitters. Genetic alleles are unusual versions of a gene that can change genetic function they are called polymorphisms or variants. Early in the 1990’s a statistically significant association of severe alcoholism with a variant, the A1 allele of the Dopamine Receptor D2 Gene (DRD2) was discovered [14]. This variant was later associated with numerous other addictive, compulsive and impulsive behaviors. At the same time, a binding availability study found that functionally, the presence of the A1 allele resulted in lower dopamine receptor availability in the parts of the brain known to effect reward [15]. Other earlier studies had explored the role of neurotransmitters in pleasure. In the limbic neural circuitry serotonin, enkephalin, GABA, and dopamine work together in a complex cascade of activation and inhibition that result in the release of dopamine. Dopamine was identified as one of the most powerful neurotransmitters that control feelings of well-being and reward. Negative emotions and craving are the results of disruption of the intercellular brain reward cascade that leads to reduced dopamine availability [16].
II. Hypodopaminergic function Genes are deoxyribonucleic acid (DNA), which directs the functional properties of proteins like neurotransmitters. Genetic alleles are unusual versions of a gene that can change genetic function they are called polymorphisms or variants. Early in the 1990’s a statistically significant association of severe alcoholism with a variant, the A1 allele of the Dopamine Receptor D2 Gene (DRD2) was discovered [14]. This variant was later associated with numerous other addictive, compulsive and impulsive behaviors. At the same time, a binding availability study found that functionally, the presence of the A1 allele resulted in lower dopamine receptor availability in the parts of the brain known to effect reward [15]. Other earlier studies had explored the role of neurotransmitters in pleasure. In the limbic neural circuitry serotonin, enkephalin, GABA, and dopamine work together in a complex cascade of activation and inhibition that result in the release of dopamine. Dopamine was identified as one of the most powerful neurotransmitters that control feelings of well-being and reward. Negative emotions and craving are the results of disruption of the intercellular brain reward cascade that leads to reduced dopamine availability [16].
III. The epigenetics of stress and prolonged exposure In addition to genetic polymorphisms, which reduce the availability of dopamine in the synapse, prolonged stress and long -term substance abuse also result in reduced cascade function and decreased dopamine release and may have a cumulative effect on vulnerability to addiction and other RDS Behaviors [19,20]. Harmful molecular effects or blunting occur due to prolonged substance use [21,22]. The repeated release of high amounts of dopamine into the synaptic cleft induces prolonged, heightened postsynaptic receptor activity, resulting in receptor down-regulation and, for this reason, further decreases dopamine function. Also, hypodopaminergic function, caused by genetic variations impacted by epigenetics, can induceimpairments in the pre-frontal cortex-cingulated gyrus, which in turn leads to poor judgment and potential habit reinstatement or relapse [20,22].
Receptor down-regulation reported, in both obese rats and drug-addicted humans, is the reason habituated addicts require ever increasing substance or behavior to maintain the rewarding effect [20,23]. However after prolonged abstinence dopamine receptor super-sensitivity, an enhanced biochemical response develops, and reinstatement at the previous level of habituation in the case of substance abuse may lead to fatalities [24]. Environmentally induced epigenetic effects on the chromatin structure of the DNA due to stress or triggered by cues can increase craving. Stress-triggered craving involves the neurotransmitters corticotrophin-releasing factor and norepinephrine. These neurotransmitters necessitate the abundant release of dopamine (100X times resting state) and subsequently, temporary hypodopaminergic functioning, repeated, or prolonged stress can induce a chronic hypodopaminergic state. Cue-triggered craving involves the basolateral nucleus of the amygdala, the hippocampus, and through glutaminergic activation, causes the increased release of dopamine that if chronic ultimately leads to a hypodopaminergic state. Due to this hypodopaminergic trait (genetic) and state (environmental), it is known that drug intake or aberrant behaviors will escalate [25,26].
Genetic addiction risk score (GARS)
The Genetic Addiction Risk Score (GARS), is the first test to accurately predict vulnerability to pain, addiction, and other obsessive and compulsive behaviors, identified as RDS [27]. There is a need to classify patients at genetic risk to alcohol and drug-seeking behavior and relapse before or upon entry to pain and residential and or non-residential chemical dependency programs. Based on an extensive literature review, an addiction risk index consisting of 11 polymorphisms in 10 genes, involved in the neurological processing of reward, were identified and tested. The resulting genetic addiction risk score (GARS) included; six single-nucleotide polymorphisms (SNPs) in the DRD1, DRD2, DRD3, DRD4, COMT, and OPRM1 genes; four simple sequence repeats (SSRs) in the DAT1, DRD4, MAOA, and 5HTT transporter genes; and a dinucleotide polymorphism in the GABRA3 gene [9]. Blum’s laboratory sought to address genetic risk for alcohol and drug by evaluating whether the combined effect of reward gene polymorphisms that contribute to a hypodopaminergic trait, associate with RDS related substance abuse risk. Among those who consented to provide a saliva sample for DNA genotyping, 273 (derived from seven centers) also had ASI phenotypic information.
The patient population n=393, 17.6%, 80.7%, and 1.5% scored in the low, moderate and high severity range, respectively. The mean number of GARS alleles was 7.97 (S.D. = 2.34) and ranged between 3 and 17 alleles. The relationship between GARS genotype panel and the Alcohol Risk Severity Score using the Fishers Exact Test revealed a significant predicative relationship(Χ2 = 8.84, df = 1, p = 0.004, 2-tailed) that remained significant after controlling for age (p < 0.01). A similar, though less robust, relationship was obtained from chi-square (p = 0.05) and linear regression (b = -0.122, t = -1.91, p = 0.10, 2-tailed) analyses of the ASI Drug Severity Risk Score. Blum et al. [28,29] details the construction of a genetic addiction risk score (GARS™) and its predictive relationship with ASI -MV derived alcohol and drug severity risk scores. Innovative strategies to combat epidemic opioid/opiate abuse, and death, based on the role of dopaminergic tone in pain pathways, are proposed. Sensitivity to pain may reside in the mesolimbic projection system, where genetic polymorphisms associate with a predisposition to pain vulnerability or tolerance. Pharmacogenomic testing of candidate genes like CB1, mu receptors, and PENK might result in pharmacogenomic, personalized solutions, and improved clinical outcomes. Identifying genetic risk for all RDS behaviors, especially in compromised populations, may be a frontline tool to assist municipalities in providing better resource allocation and possibly precision medicine [30].
RDS questionnaire
In conjunction with Zsolt Demetrovics in the Eötvös Loránd University, Institute of Psychology, Budapest, Hungary, an unpublished, 29 item RDS questionnaire reduced from 51 items generated based on the RDS theory, has been validated in over 1726 individuals attending college. The general reward deficiency factor was associated with gender, sensation seeking and impulsivity. Females show ahigher degree of reward deficiency trait. Greater sensation seeking and impulsivity predict higher degrees of reward deficiency and risk seeking behaviors and are positively associated with sensation seeking and impulsivity [2].
Pro-Dopamine regulator (KB220)
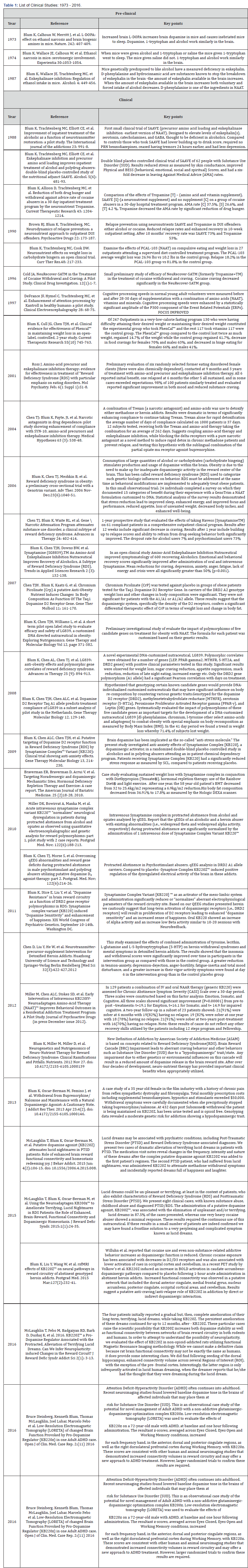
A glutaminergic-dopaminergic optimization nutraceutical called KB220 has been developed that supports the brain reward system and induces “dopamine homeostasis”. This agonistic nutraceutical has been shown to safely provide substantial clinical benefit to the victims of RDS and assist in recovery from addiction to opiates/opioids and other substance and non-substance addictions and behaviors [7,17,31-33]. DNAdirected compensatory over expression of the DA D2 receptors (a form of gene therapy) has been shown to result in a significant reduction in alcohol and cocaine craving behavior in drugpreferring rodents [34,35] and acute in vitro bromocriptine a strong agonist-induced D2 receptor proliferation in rats [36]. KB220 variants formulations have been studied extensively in both animals and humans. Pre-clinical and human trials using a variety of methodologies are reported on in a detailed review article [37] and Table 1 lists the studies of KB220 variants in a multiplicity of RDS populations. Interestingly, in abstinent heroin addicts, a pilot study of a single dose of KB220Z compared to placebo found improvement of the prefrontal-cerebellaroccipital neural network and activation of the NAc [38].
Conclusion
Recently the hypothesis [39-43] that KB220Z would enhance resting connectivity patterns between reward and cognitive brain regions was tested in placebo-controlled rsfMRI experiments in the rat. Additionally, qEEG studies in humans found that KB220Z modulates theta power in the anterior cingulate cortex [44,45]. Double-blind controlled studies and others [37,46-48] have demonstrated positive effects on both craving attenuation and relapse prevention [48-50] and enhanced compliance to KB220Z treatment was significant in obese carriers of the D2 receptors A1 allele vs. carriers of the usual complement of DA D2 receptors [51,52]. The rationale for evaluating KB220Z in the present article application is the evidence of clinical benefit KB220Z can give by epigenetically changing the neuro-mechanisms involved in producing dopamine homeostasis [53]. Based on this research and current literature new strategies to treat RDS are needed. The traditionally used therapeutic agents have failed to address the reduced connectivity patterns now seen in many addictions and have had limited success in relapse prevention and effective recovery from psychoactive substance abuse [4,54]. This concept would enable genetic testing and precise treatment of genetic and epigenetic deficits with formulations of KB220 [55,56]. Recovery and good health would be the goal of this SMART, holistic program design, based on this extensive research in a diverse but stable population [57]. In a diverse but stable population like African- Americans as well as other minority groups showing high genetic risk for all RDS behaviors.
References
- Blum K, Oscar-Berman M, Dinubile N, Giordano J, Braverman ER, et al. (2013) Coupling Genetic Addiction Risk Score (GARS) with Electrotherapy: Fighting Iatrogenic Opioid Dependence. J Addict Res Ther 4(163).
- Zsolt Demetrovics, Róbert Urbán, K Blum (2016) Reward Deficiency Syndrome and Addictive Disorders. Hungary 4th International Conference on Pathological Gambling and Other Behavioural Addictions, Europe..
- Blum K, Han D, Femino J, Smith DE, Saunders S, et al. (2014) Systematic evaluation of “compliance” to prescribed treatment medications and “abstinence” from psychoactive drug abuse in chemical dependence programs: data from the comprehensive analysis of reported drugs. PLoS One 9(9): e104275.
- Blum K, Febo M, Fahlke C, Archer T, Berggren U, et al. (2015) Hypothesizing Balancing Endorphinergic and Glutaminergic Systems to Treat and Prevent Relapse to Reward Deficiency Behaviors:Coupling D-Phenylalanine and N-Acetyl-L-Cysteine (NAC) as a Novel Therapeutic Modality. Clin Med Rev Case Rep 2(8).
- Blum K, Sheridan PJ, Wood RC, Braverman ER, Chen TJ, et al. (1996) The D2 dopamine receptor gene as a determinant of reward deficiency syndrome. J R Soc Med 89(7): 396-400.
- Blum K, Braverman ER, Holder JM, Lubar JF, Monastra VJ, et al. (2000) Reward deficiency syndrome: a biogenetic model for the diagnosis and treatment of impulsive, addictive, and compulsive behaviors. J Psychoactive Drugs 32(i-iv): 1-112.
- Blum K, Thanos PK, Badgaiyan RD, Febo M, Oscar-Berman M, et al. (2015) Neurogenetics and gene therapy for reward deficiency syndrome: are we going to the Promised Land? Expert Opin Biol Ther 15(7): 973-85.
- Comings DE, Blum K (2000) Reward deficiency syndrome: genetic aspects of behavioral disorders. Prog Brain Res 126: 325-341.
- Blum K, Oscar-Berman M, Giordano J, Downs B, Simpatico T, et al. (2012) Neurogenetic Impairments of Brain Reward Circuitry Links to Reward Deficiency Syndrome (RDS): Potential Nutrigenomic Induced Dopaminergic Activation. J Genet Syndr Gene Ther 3(4).
- Blum K, Febo M, Badgaiyan RD, Demetrovics Z, Simpatico T, et al. (2016) Common Neurogenetic Diagnosis and Meso-Limbic Manipulation of Hypodopaminergic Function in Reward Deficiency Syndrome (RDS): Changing the Recovery Landscape. Curr Neuropharmacol 15(1): 184- 194.
- Blum K, Futterman S, Wallace JE, Schwertner HA (1977) Naloxoneinduced inhibition of ethanol dependence in mice. Nature 265(5589): 49-51.
- Blum K, Gardner E, Oscar-Berman M, Gold M (2012) “Liking” and “Wanting” Linked to Reward Deficiency Syndrome (RDS): Hypothesizing Differential Responsivity in Brain Reward Circuitry. Curr Pharm Des 18(1): 113-118.
- K Blum, J Payne (1990) Alcohol & The Addictive Brain. The Free Press Simon & Schuster, New York, USA.
- Blum K, Noble EP, Sheridan PJ, Montgomery A, Ritchie T, et al. (1990) Allelic association of human dopamine D2 receptor gene in alcoholism. JAMA 263(15): 2055-2060.
- Noble EP, Blum K, Ritchie T, Montgomery A, Sheridan PJ (1991) Allelic association of the D2 dopamine receptor gene with receptor-binding characteristics in alcoholism. Arch Gen Psychiatry 48(7): 648-54.
- K. Blum and G. P. Kozlowski (1990) Ethanol and neuromodulators interaction: a cascade model of reward. In: Alcohol and Behavior. (Eds.) H Ollat, S. Parvez & H Parvez VSP Press Utrecht, The Netherlands, USA.
- Blum K, Febo M, Thanos PK, Baron D, Fratantonio J, et al. (2015) Clinically Combating Reward Deficiency Syndrome (RDS) with Dopamine Agonist Therapy as a Paradigm Shift: Dopamine for Dinner? Mol Neurobiol 52(3): 1862-1869.
- Blum K, Oscar-Berman M, Barh D, Giordano J, Gold M (2013) Dopamine Genetics and Function in Food and Substance Abuse. J Genet Syndr Gene Ther 4(121).
- Sotomayor-Zárate R, Abarca J, Araya KA, Renard GM, Andrés ME, et al. (2015) Exposure to repeated immobilization stress inhibits cocaineinduced increase in dopamine extracellular levels in the rat ventral tegmental area. Pharmacol Res 101: 116-123.
- Hill E, Han D, Dumouchel P, Dehak N, Quatieri T, et al. (2013) Long term Suboxone emotional reactivity as measured by automatic detection in speech. PLoS One 8(7).
- Stice E, Yokum S, Blum K, Bohon C (2010) Weight gain is associated with reduced striatal response to palatable food. J Neurosci 30(39): 13105-13109.
- Jenner P, Marsden CD (1987) Chronic pharmacological manipulation of dopamine receptors in brain. Neuropharmacology 26(7b): 931-940.
- Bogomolova EV, Rauschenbach IY, Adonyeva NV, Alekseev AA, Faddeeva NV, et al. (2010) Dopamine down-regulates activity of alkaline phosphatase in Drosophila: the role of D2-like receptors. J Insect Physiol 56(9): 1155-1159.
- Blum K, Chen TJ, Downs BW, Bowirrat A, Waite RL, et al. (2009) Neurogenetics of dopaminergic receptor supersensitivity in activation of brain reward circuitry and relapse: proposing “deprivationamplification relapse therapy” (DART). Postgrad Med 121(6): 176- 196.
- Martinez D, Saccone PA, Liu F, Slifstein M, Orlowska D, et al. (2012) Deficits in dopamine D(2) receptors and presynaptic dopamine in heroin dependence: commonalities and differences with other types of addiction. Biol Psychiatry 71(3): 192-198.
- Bonito-Oliva A, Södersten E, Spigolon G, Hu X, Hellysaz A (2016) Differential regulation of the phosphorylation of Trimethyl-lysine27 histone H3 at serine 28 in distinct populations of striatal projection neurons. Neuropharmacology 107: 89-99.
- Draine J, Wolff N, Jacoby JE, Hartwell S, Duclos C (2005) Understanding community re‐entry of former prisoners with mental illness: a conceptual model to guide new research. Behav Sci Law 23(5): 689- 707.
- K Blum, BC Haberstic, A Smolen, D Han, Marlene Oscar-Berman, et al. (2017) Genetic addiction risk score (GARS) predicts addiction severity index - MV alcohol and drug - Risk scores in a multi-center study. In, Submitted to PLOSONE.
- Kenneth Blum, David Han, Mary Hauser, Bernard William Downs, John Giordano, et al. (2013) Neurogenetic Impairments of Brain Reward Circuitry Links to Reward Deficiency Syndrome (RDS) as evidenced by Genetic Addiction Risk Score (GARS): A case study. The IIOAB Journal 4(1): 4-9.
- Kenneth Blum, B W Downs, Kristina Dushaj, Mona Li, Eric R Braverman, et al. (2016) The Benefits of Customized DNA Directed Nutrition To Balance The Brain Reward Circuitry And Reduce Addictive Behaviors. Precis Med (Bangalore) 1(1): 18-33.
- Blum K, Simpatico T, Badgaiyan RD, Demetrovics Z, Fratantonio J, et al. (2015) Coupling Neurogenetics (GARS) and a Nutrigenomic Based Dopaminergic Agonist to Treat Reward Deficiency Syndrome (RDS): Targeting Polymorphic Reward Genes for Carbohydrate Addiction Algorithms. J Reward Defic Syndr 1(2): 75-80.
- McLaughlin T, Febo M, Badgaiyan RD, Barh D, Dushaj K, et al. (2016) KB220Z™ a Pro-Dopamine Regulator Associated with the Protracted, Alleviation of Terrifying Lucid Dreams. Can We Infer Neuroplasticityinduced Changes in the Reward Circuit? J Reward Defic Syndr Addict Sci 2(1): 3-13.
- Steinberg B, Blum K, McLaughlin T, Lubar J, Febo M, et al. (2016) Lowresolution electromagnetic tomography (LORETA) of changed brain function provoked by pro-dopamine regulator (KB220z) in one adult ADHD cases. Open J Clin Med Case Rep 2(11): 11-21.
- Thanos PK, Katana JM, Ashby CR Jr, Michaelides M, Gardner EL, et al. (2005) The selective dopamine D3 receptor antagonist SB-277011-A attenuates ethanol consumption in ethanol preferring (P) and nonpreferring (NP) rats. Pharmacol Biochem Behav 81(1): 190-197.
- Thanos PK, Michaelides M, Umegaki H, Volkow ND (2008) D2R DNA transfer into the nucleus accumbens attenuates cocaine selfadministration in rats. Synapse 62(7): 481-486.
- Boundy VA, Pacheco MA, Guan W, Molinoff PB (1995) Agonists and antagonists differentially regulate the high affinity state of the D2L receptor in human embryonic kidney 293 cells. Mol Pharmacol 48(5): 956-964.
- Blum K, Oscar-Berman M, Stuller E, Miller D, Giordano J, et al. (2012) Neurogenetics and Nutrigenomics of Neuro-Nutrient Therapy for Reward Deficiency Syndrome (RDS): Clinical Ramifications as a Function of Molecular Neurobiological Mechanisms. J Addict Res Ther 3(5): 139.
- Blum K, Liu Y, Wang W, Wang Y, Zhang Y, et al. (2015) rsfMRI effects of KB220Z on neural pathways in reward circuitry of abstinent genotyped heroin addicts. Postgrad Med 127(2): 232-241.
- Duong TQ, Yacoub E, Adriany G, Hu X, Ugurbil K, et al. (2002) Highresolution, spin-echo BOLD, and CBF fMRI at 4 and 7 T. Magn Reson Med 48(4): 589-593.
- Febo M, Ferris CF (2014) Oxytocin and vasopressin modulation of the neural correlates of motivation and emotion: results from functional MRI studies in awake rats. Brain Res 1580: 8-21.
- Madularu D, Yee JR, Kenkel WM, Moore KA, Kulkarni P, et al. (2015) Integration of neural networks activated by amphetamine in females with different estrogen levels: a functional imaging study in awake rats. Psychoneuroendocrinology 56: 200-212.
- Goense JB, Logothetis NK (2006) Laminar specificity in monkey V1 using high-resolution SE-fMRI. Magn Reson Imaging 24(4): 381-392.
- Liang Z, King J, Zhang N (2011) Uncovering intrinsic connectional architecture of functional networks in awake rat brain. J Neurosci 31(10): 3776-3783.
- Miller DK, Bowirrat A, Manka M, Miller M, Stokes S, et al. (2010) Acute intravenous synaptamine complex variant KB220 “normalizes” neurological dysregulation in patients during protracted abstinence from alcohol and opiates as observed using quantitative electroencephalographic and genetic analysis for reward polymorphisms: part 1, pilot study with 2 case reports. Postgrad Med 122(6): 188-213.
- Blum K, Chen TJ, Morse S, Giordano J, Chen AL, et al. (2010) Overcoming qEEG abnormalities and reward gene deficits during protracted abstinence in male psychostimulant and polydrug abusers utilizing putative dopamine D(2) agonist therapy: part 2. Postgrad Med 122(6): 214-226.
- Blum K, Chen AL, Giordano J, Borsten J, Chen TJ, et al. (2012) The addictive brain: all roads lead to dopamine. J Psychoactive Drugs 44(2): 134-143.
- Chen TJ, Blum K, Payte JT, Schoolfield J, Hopper D, et al. (2004) Narcotic antagonists in drug dependence: pilot study showing enhancement of compliance with SYN-10, amino-acid precursors and enkephalinase inhibition therapy. Med Hypotheses 63(3): 538-548.
- Brown RJ, Blum K, Trachtenberg MC (1990) Neurodynamics of relapse prevention: a neuronutrient approach to outpatient DUI offenders. J Psychoactive Drugs 22(2): 173-187.
- Chen TJ, Blum K, Waite RL, Meshkin B, Schoolfield J, et al. (2007) Gene \Narcotic Attenuation Program attenuates substance use disorder, a clinical subtype of reward deficiency syndrome. Adv Ther 24(2): 402- 414.
- K Blum, D Allison, MC Trachtenberg, RW Williams (1988) Reduction of both drug hunger and withdrawal against advice rate of cocaine abusers in a 30 day inpatient treatment program by the neuronutrient Tropamine. Curr Ther Res 43(6): 1204-1214.
- Blum K, Chen AL, Chen TJ, Braverman ER, Reinking J, et al. (2008) Activation instead of blocking mesolimbic dopaminergic reward circuitry is a preferred modality in the long term treatment of reward deficiency syndrome (RDS): a commentary. Theor Biol Med Model 5: 24.
- Dahlgren A, Wargelius HL, Berglund KJ, Fahlke C, Blennow K, et al. (2011) Do alcohol-dependent individuals with DRD2 A1 allele have an increased risk of relapse? A pilot study. Alcohol Alcohol 46(5): 509- 513.
- Szutorisz H, DiNieri JA, Sweet E, Egervari G, Michaelides M, et al. (2014) Parental THC exposure leads to compulsive heroin-seeking and altered striatal synaptic plasticity in the subsequent generation. Neuropsychopharmacology 39(6): 1315-1323.
- K Blum, RD Badgaiyan, Z Demotrovics, J Fratantonio, G Agan, et al. (2015) Can Genetic Testing Provide Information to Develop Customized Nutrigenomic Solutions for Reward Deficiency Syndrome? Clin Med Rev Case Rep 2(1).
- Blum K, Meshkin B, Downs BW (2006) DNA based customized nutraceutical “gene therapy” utilizing a genoscore: a hypothesized paradigm shift of a novel approach to the diagnosis, stratification, prognosis and treatment of inflammatory processes in the human. Med Hypotheses 66(5): 1008-1018.
- Thanos PK, Hamilton J, O’Rourke JR, Napoli A, Febo M, et al. (2016) Dopamine D2 gene expression interacts with environmental enrichment to impact lifespan and behavior. Oncotarget 7(15): 19111- 19123.
- K Blum, J Femino, ST Teitelbaum, J Giordano, M Oscar-Berman, GMS (2013) Molecular Neurobiology of Addiction Recovery. The 12 Steps Program and Fellowship. Springer, New York, USA..






























