Morphology of Deciduous Canines in African Apes
Hiroyuki Yamada*
Ph.D. Former Dentist, Dental Anthropologist, Department of Oral Anatomy, School of Dentistry, Aichi-Gakuin University, Japan
Submission: January 13, 2024; Published: January 26, 2024
*Corresponding author: Hiroyuki Yamada, Ph.D. Former Dentist, Dental Anthropologist, Department of Oral Anatomy, School of Dentistry, AichiGakuin University, 15-16 Obatanaka 2 Chome, Moriyama-Ku, Nagoya 463-0013, Japan
How to cite this article: Hiroyuki Yamada*. Morphology of Deciduous Canines in African Apes. Glob J Arch & Anthropol. 2024; 13(4): 555867. DOI: 10.19080/GJAA.2024.13.555867
Abstract
The aim of this study was to determine the morphology and sexual dimorphism of deciduous canines in African great apes. The morphology of the deciduous canines of the chimpanzee and the gorilla was similar in both the male and the female. The occlusal view of the chimpanzee showed an ellipsoid shape in the maxilla and a more distorted quadrilateral shape in the mandible. The lingual view showed a pyramidal triangle in the maxilla and a distorted square in the mandible. Compared to chimpanzees, gorillas had a larger maxillary labiolingual diameter, a more elliptical shape, a higher crown height and more mesial grooves. The basal tubercles, spinous processes and distal accessory ridges observed in human primary canines were not observed in chimpanzees and gorillas. No significant sex differences were found for maxillary and mandibular primary canines in chimpanzees. However, in the maxilla, there were significant sex differences in gorillas for all variables except mesiodistal diameter. Compared to humans, chimpanzees were significantly larger for all variables except maxillary labiolingual diameter. On the other hand, the crown index was significantly larger in humans and the occlusal view was more rounded. The deciduous canines of chimpanzees and gorillas were similar in morphology to the permanent canines of females of both species. However, they differed significantly from the permanent canines of males.
Keywords: Deciduous Canines; Crown Morphology; Tooth Size; Sexual Dimorphism; African Apes
Introduction
The apes are the closest relatives of humans and are taxonomically classified in the superfamily Hominoidea. The great apes that live on the African continent are the chimpanzee/bonobo and the gorilla, while the small ape, the gibbon, and the great ape, the orangutan, live in Asia. Primate canines resemble the fanged morphology of carnivores which use their long, dagger-like canines to stab and bring down their prey [1]. This strongly suggests that primate canines have a weapon or intimidation function against predators [2]. The large canines of males also play a role in attracting females and in male intra-sexual selection [3]. As a result, male canines strongly reflect sexual dimorphism and are significantly larger [3-5]. In contrast, except for gibbons, female canines are small and of little evolutionary value as weapons [6]. The male maxillary canines of Cercopithecus are daggers with a high crown and a broad base [2,5], whereas the great apes have a broad, stout, massive apical cone [7] or an isosceles triangle lingually [8]. Apes are essentially omnivorous and eat a wide variety of foods. Chimpanzees, for example, eat figs and other fruits as a staple diet, as well as leaves, flowers, seeds, plants, ants and small monkeys. Numerous studies of primate canine teeth have been reported in relation to sexual dimorphism [3-10]. On the other hand, deciduous teeth are more conservative than permanent teeth, and primitive features not found in permanent teeth tend to be preserved in deciduous teeth [11-14]. Nevertheless, studies of primary teeth are limited in paleoanthropology because they are structurally weaker than permanent teeth and rarely survive as fossils [15-18]. Studies of deciduous teeth in living and extinct apes have been rare throughout their long evolutionary history. This is due to the difficulty of detecting sex differences due to their juvenile nature and small population sizes [12,19-21]. The aim of this study was to examine the morphology of the deciduous canines of the African great apes, chimpanzee and gorilla, and to compare interspecific differences and sexual dimorphism between the two species.
Materials and Methods
The materials consisted of specimens of deciduous canines of Pan troglodytes and Gorilla gorilla from the collections of the Kenya National Museum, the Laboratory of Physical Anthropology, Kyoto University, the Center for the Evolutionary Origins of Human Behaviour, Kyoto University (EHUB), the Japan Monkey Center, and the Higashiyama Zoo and Botanical Garden, Japan. The primary canines selected for the study were fully erupted from the alveolus. Impressions of the primary canines were taken with silicone impression material (Provil: Heraeus Kulzer GmbH) [8] and hard plaster casts were made. Photographs were taken from the occlusal view, with the crown axis aligned with the optical axis of the camera, and from the lingual view, with the crown axis perpendicular to the optical axis of the camera. Tooth sizes (mesiodistal and labiolingual diameters) were taken on the model using digital calipers (1/100 mm, Digimatic: Mitutoyo Co.). Deciduous canines were measured according to previous methods [5,7,8,12]. Johanson (1974) stated that the lower deciduous canines are only slightly rotated and that their long axis is approximately equal to the mesiodistal diameter of the crown [12]. Therefore, the maximum diameter of the deciduous mandibular canine is recorded as the mesiodistal dimension. The measurements were the maximum diameter of the tooth mesiodistally (MD), taken on a line parallel to the long axis of the tooth, and the maximum diameter of the tooth labiolingually (LL), taken at right angles to the mesiodistal diameter. Crown area (MD×LL) and crown index (LL/MD×100) were calculated from the measurements. The percentage of sex dimorphism in crown measurements was calculated using the formula [(male mean - female mean) / female mean x 100] [22-26]. Student’s t-test (P<0.05, P<0.01) was used to assess the significance of differences in means.
Results
Chimpanzee (Pan troglodytes )
Maxilla: Figure 1 shows the maxillary and mandibular deciduous canines of a male (no. 1534) and a female (no. 1367) chimpanzee. In the occlusal view, male canines are elliptical mesiodistally. The lingual view showed a pyramidal triangular shape, and the distal crest was concave, and asymmetrical mesiodistally. The apex was located mesially. The mesial shoulder was approximately 1/5 of the crown height from the cervical line. The mesial groove was only a trace. The lingual ridge was rounded and broad and ran from the apex to the cervical ridge in a widening pattern along the mesial crest. The distal lingual fossa was wide. The cervical ridge was well developed and more pronounced. Females have a similar crown morphology to males. In particular, they are phenotypically less robust than males, with ridges and grooves that are slender and poorly developed. Individual differences in their morphology have been observed. However, the outline of the crown, the position of the mesial shoulder, the lingual ridge and the cervical ridge were very similar to those in both sexes.
Mandible: Male mandibular deciduous canines were quadrangular when viewed from the occlusal side. The labial margin was strongly convex in the mesial half of the crown and slightly concave in the distal half. The lingual margin was straight in the mesial half and strongly convex in the distal half. When viewed lingually, the tooth had a distorted square shape. The mesial cristid was straight and the mesial margins of the crown were clearly angled at the shoulder. The distal cristid was concave, protruding outwards at the distal shoulder. The mesial shoulder was approximately 1/3 of the crown height from the cervical line, the distal shoulder was approximately 1/5 of the crown height. The mesial groove was not defined. The lingual ridge was thick. The mesial lingual fossa was wide and flat. A relatively deep groove ran between the lingual ridge and the distal cristid, forming a distal lingual fossa at its base. The cervical ridge was moderately swollen. The crown morphology of females was similar to that of males. In particular, the bending of the mesial margin of the crown was weaker than in males. However, this was within the range of individual differences (Figure 1).

Gorilla (Gorilla gorilla)
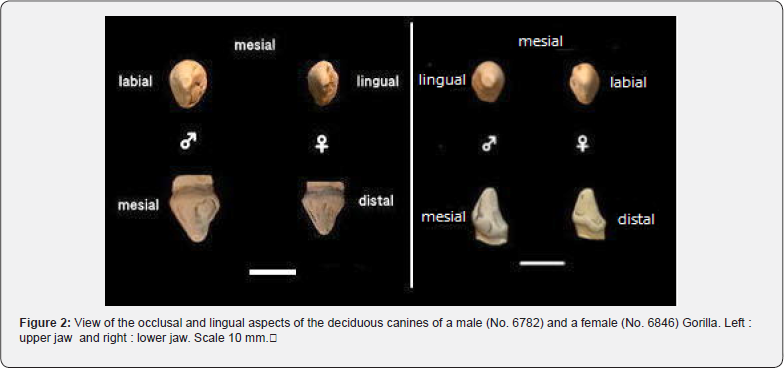
Maxilla: Figure 2 shows the maxillary and mandibular deciduous canines of a male (no. 6782) and a female (no. 6846) gorilla in occlusal and lingual views. The morphology of the gorilla canines was like that of chimpanzees. The differences with chimpanzees were that the occlusal plane was egg-shaped and more labiolingually symmetrical, the lingual view had a slightly higher crown height, a broad bulge in the middle of the distal lingual fossa, the mesial 2/3 of the cervical ridge was thicker and more swollen, while the distal 1/3 was narrower. The crown morphology of female primary canines was generally similar to that of males. In particular, the occlusal surface was flatter than in males, with less pronounced ridges and grooves. Individual differences in morphology were observed. However, the outline of the crown, the position of the mesial shoulder, the lingual ridge and the cervical ridge were very similar to those in both sexes.
Mandible: The morphology of the gorilla mandibular canine was like that of the chimpanzee. The differences were that the occlusal view was eliptical and labiolingually symmetrical, the crown height and mesial shoulder were higher than in chimpanzees, and the mesial groove was moderately deep. The female occlusal view showed almost the same morphology as the male, but the female labiolingual diameter was narrower and more flattened. The lingual view showed almost the same morphology as the male, but the phenology was weaker. There were some differences in the morphology of the mandibular primary canines, but these were within the range of individual differences (Figure 2).
Numerical analysis
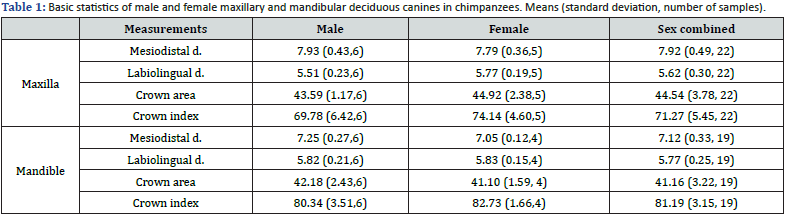
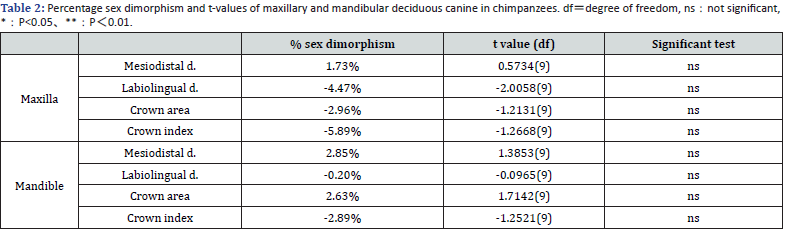
Chimpanzee: Table 1 shows the basic statistics for mesiodistal crown diameter, labiolingual diameter, crown area and crown index of chimpanzee upper and lower primary canines. Table 2 shows the significance tests (t-tests) of mean differences. In both the maxilla and mandible, mesiodistal diameters were larger in males, but labiolingual diameters were larger in females. No significant sex differences were found in either case. The percentages of sex differences for all variables ranged from -5.9% to 1.7% in the maxilla. Similarly, the percentage of sex differences ranged from -2.9% to 2.9% in the mandible (Table 1 & 2).
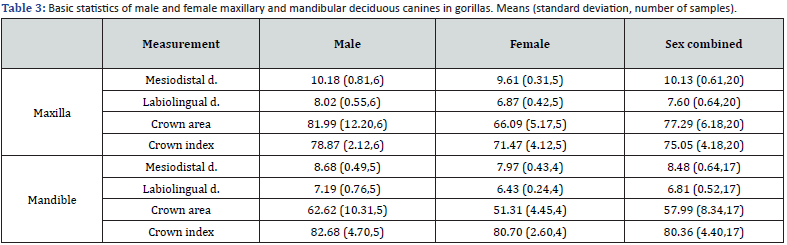
Gorilla: Table 3 shows the basic statistics for the gorilla measurements, and table 4 shows the significant tests and percentages of sex differences. In the maxilla, the mean deciduous canines of males exceeded those of females for all variables, and significant sex differences were found for all measurements except mesiodistal crown diameter. The proportion of sex differences ranged from 5.9% to 24.1%. Even in the mandible, males were on average larger than females for all variables. However, no significant sex differences were found. The percentage differences between the sexes ranged from 2.5% to 22.0%. The sex difference is greater for labiolingual diameter than for mesiodistal diameter (Table 3 & 4).
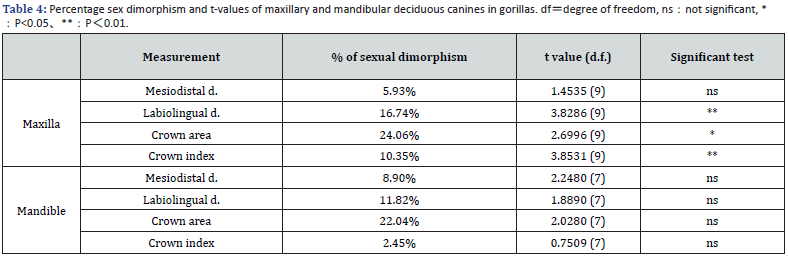
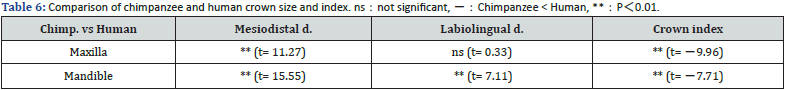
Comparison of chimpanzees with gorillas and humans: Table 5 compares chimpanzees and gorillas in terms of combined sex scores. On all variables except the mandibular crown index, gorillas scored significantly higher than chimpanzees (Table 5). The crown size and index of chimpanzee (combined sex) and human juveniles [27] are compared in Table 6. The mesiodistal and labiolingual diameters in chimpanzees were larger for both the maxilla and mandible and significantly different except for the maxillary labiolingual diameter. On the other hand, the crown index in humans was significantly larger for both the maxilla and mandible, with a more rounded shape (Table 6).

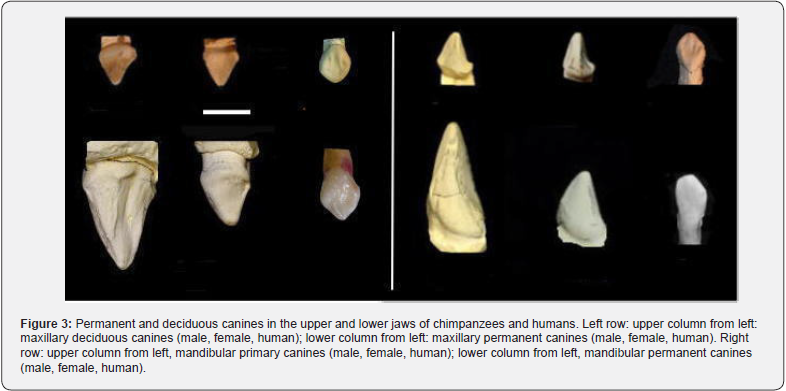
Comparison of canine morphology: Figure 3 compares male and female chimpanzee canines and deciduous canines, and human canines and deciduous canines. A comparison of chimpanzee permanent and deciduous canines for the maxilla showed that female permanent canines were more similar in shape to male and female deciduous canines than their male counterparts. However, the female permanent canines were approximately twice as large as deciduous canines. The male permanent canines were quite different in size and shape from the female and deciduous canines. Human permanent and deciduous canines are diamond-shaped and very different from those of chimpanzees. Human permanent canines are one to two times larger than chimpanzee deciduous canines. A similar situation applies to the mandibular canines (Figure 3).
Discussion
The primary canines of Platyrrhini and Catarrhini differ significantly in size and shape from the permanent canines of males but are very similar to those of females [2,6]. The occlusal view of the chimpanzee maxillary primary canines shows an elliptical shape, whereas that of the mandible is a distorted quadrilateral. The lingual view showed a pyramidal triangle in the maxilla and a distorted square in the mandible. In gorillas, the morphology is like that of chimpanzees, although the mesial grooves of the maxilla and mandible are slightly deeper in gorillas. However, this feature is also within the range of individual variation.
Sex differences in deciduous canines
It has been reported that sex hormones are responsible for sex differences in the size of permanent canines in humans [28,29]. However, it is less likely that there are sex differences in the morphology of primary teeth. Gil-Donoso et al (2023) investigated the sex difference in the enamel and dentin of human deciduous canines by three-dimensional analysis and argued that neither the volume nor the three-dimensional surface area of each structure can determine the sex of the tooth [26]. Sillman (1964) also found no significant sex differences in the mesiodistal diameter of deciduous teeth [30]. Lukas (2022) studied the degree of sexual dimorphism in human deciduous teeth in a number of populations and reported no significant sex differences in the mean % of sexual dimorphism in the mesiodistal and buccolingual diameters in any population [31]. Deciduous teeth begin to calcify in utero between 13 and 18 weeks after fertilisation [32,33] and are not affected by increased gonadotropins and sex steroids [26,34]. Calcification of the hard tissue of deciduous canines begins between 15 and 18 weeks after fertilisation and is completed by around 9 months of age, with eruption occurring between 16 and 18 months [35]. However, at 6 months of age, when deciduous teeth begin to erupt, crown formation of deciduous canines is already well advanced. The influence of sex hormones is therefore limited and no significant sex differences in crown size are observed [26,36]. Nevertheless, regarding the mesiodistal diameter of maxillary deciduous canines, the existence of significant sex differences in human primary dentition has been described [27,37-41]. Hanihara (1955) also found a significant sex difference [38] and Moorrees et al. (1957) stated that males were significantly larger [39].
In addition, the top-ranked tooth types in which sexual dimorphism is strongly expressed in the mesiodistal diameter are the first deciduous molars in the maxilla and the first deciduous incisors in the mandible [31]. Deciduous canines do not always show strong sex differences. This diversity is seen in deciduous teeth, but is uniform in permanent teeth, with canines being the most sexually dimorphic. However, it is not known what factors contribute to the phenomenon of significant sex differences in human deciduous dentition. On the other hand, Leigh et al (2005) examined the deciduous canines of five primate species (Saguinus fuscicollis, Macaca mulatta, Cercocebus atys, Papio hamadryas and Mandrillus sphinx) and reported no sexual dimorphism in size in any of them and suggested that sexual dimorphism in deciduous canine size may be related to the rate of eruption, age and duration of eruption [42]. In the great apes, such as gorillas and chimpanzees, the primary canines are the last to erupt, and quite advanced [43]. The gorillas in this study showed significant differences between the sexes, except for maxillary mesiodistal diameter. No significant sex differences were found in the mandible for all variables. The percentage of sex difference in gorilla permanent canines was 38.7% for mesiodistal diameter and 35.5% for labiolingual diameter, both significant sex differences and the strongest sex differences among great apes [8]. If sex differences are strong in the permanent canines, they may also appear in the preceding deciduous canines. This would require further investigation.
Comparison of permanent and deciduous canines
Viewed from the lingual aspect, the morphology of male chimpanzee and gorilla permanent maxillary canines is robustly an isosceles triangle and distally curved (8). In contrast, females are smaller and more triangular than males. The morphology of deciduous canines is triangular in both sexes, and resembles the permanent canines of females. Greenfield (1992) proposes a dual selection hypothesis for the similarity of female canine morphology to that of primary canines. He likens the size, morphology and occlusal relationship of canine teeth to two natural selection types, when used as weapons and when used as incisors [6]. In other words, in most anthropoids, male canines function as fangs because of their higher evolutionary value as weapons. On the other hand, female canines and deciduous canines are also selected for as weapons, but it is thought that they tend to become incisors because of their low evolutionary value.
Conclusion
The occlusal view of the chimpanzee maxillary deciduous canines was elliptical, while that of the mandible was a distorted quadrilateral. The lingual view showed a pyramidal triangle in the maxilla and a distorted square in the mandible. In gorillas, the morphology was like that of chimpanzees. There were no sex differences in the size of the deciduous canines in chimpanzees, but there were sex differences in gorillas. The deciduous canines of chimpanzees and gorillas were similar in morphology to the permanent canines of females of both species. However, they differed significantly from the permanent canines of males.
dropdown Start here
References
- Kelley J (1986) Species recognition and sexual dimorphism in Proconsul and Rangwapithecus. J Human Evol 15(6): 461-495.
- Lucas PW, Corlett RT, Luke DA (1986) Sexual dimorphism of tooth size in anthropoids. Human Evol 1: 23-39.
- Leutenegger W, Kelly JT (1977) Relationship of sexual dimorphism in canine size and body size to social, behavioral, and ecological correlates in Anthropoid primates. Primates 18: 117-136.
- Greenfield LO, Washburn A (1991) Polymorphic aspects of male Anthropoid canines. Am J Phys Anthropol 84(1):17-34.
- Plavcan JM, van Schaik CP (1993) Canine Dimorphism. Evol Anthropol 2(6): 208-214.
- Greenfield LO (1992) Origin of the human canine: A solution to an old Enigma. Yearbook of Physical anthropology 35(S15): 153-185.
- Plavcan JM (1993) Canine size shape in male anthropoid primates. Am J Phys Anthropol 92(2): 201-216.
- Yamada H, Hamada Y, Kunimatsu Y, Nakatsukasa M, Ishida H (2015) Canine crown morphology and sexual dimorphism in the Great apes. Anthropol Sci (Japanese Series) 123: 93-109.
- Plavcan JM, van Schaik CP (1992) Intrasexual competition and canine dimorphism in anthropoid primates. Am J Phys Anthropol 87(4): 461-477.
- Yamada H, Nakatsukasa M, Kunimatsu Y, Hamada Y, Ishida H (2022) Evolution of humans in view of maxillary canine morphology. Anthropological Science (Japanese Series) 64: 1-34.
- Remane A (1927) Studien Uber die Phylogenie desmenschlichen Eckzahns. Z Anat Entugesch 82: 391-481.
- Johanson DC (1974) An odontological study of the chimpanzee with some implications for hominoid evolution. The University of Chicago, PhD, pp. 1-444.
- Smith P (1978) Evolutionary changes in the deciduous dentition of the Near Eastern Populations. Journal of Human Evolution 7(5): 401-408.
- Hanihara K (1992) A story of teeth and anthropology. Ishiyaku Publishers, Inc. Tokyo.
- Weidenreich F (1937) The dentition of Sinanthropus pekinensis: A comparative odontography of the Hominids. New Series D 1: 121.
- Johanson DC, White TD, Coppens Y (1982) Dental remains from the Hadar formation, Ethiopia: 1974-1977 collections. Ame J Phys Anthropol 57(4): 545-603.
- Moggi-Cecchi J, Tobias PV, Beynon AD (2006) The mixed dentition and associated skull fragments of a juvenile fossil Homonid from Sterkfontein, South Africa. Am J Phys Anthropol 106(4): 425-465.
- Bailey SE, Hublin JJ (2006) Dental remains from the Grotte du Renne at Arcy-sur-Cure (Yonne). J Human Evol 50(5): 485-508.
- Ashton EH and Zuckerman FRS (1950) Some quantitative dental characteristics of the chimpanzee, gorilla and orang-outang. Philos Trans R Soc Lond B Biol Sci 234(616): 471-484.
- Ashton EH (1956) Sexual differences in the dimensions of the milk teeth of the Chimpanzee and Gorilla. Proc Zoologic Soc Lond 126(1): 121-125.
- Krogman WM (1969) Growth changes in skull, face, jaws, and teeth of the chimpanzee. In: The Chimpanzee, Karger, Basael. New York 1: 104-164.
- Schultz AH (1958) Cranial and dental variability in Colobus monkeys. Proc Zoologic Soc London 130(1): 79-105.
- Schultz AH (1960) Age changes and variability in the skulls and teeth of the central american monkeys Alouatta, Cebus and Ateles. Proc Zoologic Soc Lond 133(3): 337-390.
- Garn SM, Lewis AB, Swindler DR, Kerewsky RS (1967) Genetic control of sexual dimorphism in tooth size. J Dent Res 46(5): 963-972.
- Frisch JE (1963) Sex-differences in the canines of the Gibbon (Hylobates lar). Primates 4: 1-10.
- Gil-Donoso E, García-Campos C, Blasco-Moreno S, Modesto-Mata M, de Pinillos MM, et al. (2023) Sexual dimorphism of deciduous canine dental tissues dimensions of modern human populations. Anthropol Sci 131(2): 107-115.
- Funatsu T, Kondo S, Inoue M, Wakatsuki E, Sasa R (1999) Sexual differences in deciduous tooth crown diameters in a Japanese population. Japanese Soc Pediatric Dent 37: 700-707.
- Gingerich PD (1974) Size variability of the teeth in living mammals and the diagnosis of closely related sympatric fossil species. J Paleontol 48(5): 895-903.
- Zilberman U, Smith P (2001) Sex- and age-related differences in primary and secondary dentin formation. Adv Dent Res 15: 42-45.
- Sillman JH (1964) Dimensional changes of the dental arches: longitudinal study from bieth to 25 years. Am J Orthodontol 50(11): 824-842.
- Lucas JR (2022) Sexual dimorphism in deciduous tooth crown size: Variability within and between groups. Am J Human Biol 34(10): 1-17.
- Lunt RC, Law DB (1974) A review of the chronology of eruption of deciduous teeth. American Dental Association 89(4): 872-879.
- Sunderland EP, Smith CJ, Sunderland R (1987) A histological study of the chronology of initial mineralization in the human deciduous dentition. Arch Oral Biol 32(3): 167-174.
- Quigley CA (2002) The postnatal gonadotropin and sex steroid surge-insights from the androgen insensitivity syndrome. J Clin Endocrinol Metab 87(1): 24-28.
- Schour I, Massler M (1944) Chronology of the development of dentition. American Dental Association Chicago.
- Mendoza A, Solano E (2005) Desarrollo y erupción dentaria. In: Boj J, Catalá C, García-Ballesta C, and Mendoza A (eds.), Odontopediatrí Masson, Barcelona, pp. 55-65.
- Black III TK (1978) Sexual dimorphism in the tooth-crown diameters of the deciduous teeth. Am J Phys Anthropol 48(1): 77-82.
- Hanihara K (1955) Studies on the deciduous dentition of the Japanese and the Japanese-American hybrids. Ⅱ. Deciduous canines. J Anthropol Soc Nippon 64(3): 95-116.
- Moorrees CF, Thomsen S, Jensen E, Yen PKJ (1957) Mesiodistal crown diameters of the deciduous and permanent teeth in individuals. J Dent Res 36(1): 39-47.
- (1988) The chronology of deciduous and permanent dentition in Japanese children. The Japanese Society of Pedodontics. Shoni Shikagaku Zasshi 26(1): 1-18.
- The Japanese Society of Pediatric Dentistry (2019) Chronology of deciduous and permanent dentition in Japanese children. Ⅱ Part 2: Permanent dentition. 57(3): 363-373.
- Schultz AH (1944) Age changes and variability in Gibbons. A morphological study on a population sample of a man-like ape. American J Phys Anthropol 2(1): 1-129.
- Leigh SR, Setchell JM, Buchanan LS (2005) Ontogenetic Bases of Canine Dimorphism in Anthropoid Primates. American J Phys Anthropol 127(3): 296-311.






























