Comparison of p-XRF Spectrometers for Archaeological Research: A Case Study for Analysis of Tin-Rich Archaeological Bronzes
Boika Zlateva*
Faculty of Chemistry and Pharmacy, University of Sofia, James Bouchier, Sofia, Bulgaria
Submission: November 06, 2017; Published: December 11, 2017
*Corresponding author: Boika Zlateva, Faculty of Chemistry and Pharmacy, University of Sofia, James Bouchier blbd., 1, BG-1164, Sofia, Bulgaria, Email: ahbz@chem.uni-sofia.bg
How to cite this article: Boika Z. Comparison of p-XRF Spectrometers for Archaeological Research: A Case Study for Analysis of Tin-Rich Archaeological Bronzes. Glob J Arch & Anthropol. 2017; 2(4): 555591. DOI10.19080/GJAA.2017.02.555591
Abstract
The XRF (X-ray Fluorescence) spectroscopy is a non-destructive technique of analysis widely used in archaeometry for the identification of chemical elements in pigments, glass, metal alloys, and other materials. Specially attention in the last two decades is focused on portable XRF spectrometers, usually used for in the filed investigations in archaeological excavations or in museum collections.
In this paper using three different p-XRF instruments chemical composition of the tin-bronze artefacts form Bronze Age in Bulgaria and different standards are obtained. The results for the standard materials (MBH-33X GM7 J and MBH-33X RB1 A) are in very good coincidence with the certificated value for all type instruments, despite the results for archaeological objects, where exist significant differences in the chemical compositions obtained from the different models of p-XRF.
Introduction
Advancements in the scientific analysis of prehistoric objects, without the risk of damage, promise researchers access to previously hidden information. Furthermore, the increasing availability of portable XRF devices, as well as their moderate cost when compared to laboratory-based techniques, more frequently allows the incorporation of chemical examinations in material culture studies [1]. Use of portable X-ray fluorescence instrument for bulk alloy analysis on low corroded indoor bronzes. Spectrochimica Acta Part B, 89, 7-13. . However, such analyses have their limits and not all questions can be answered in this manner [2-4].
Materials and Method
The samples from copper alloy are collected from fibulae, from NE Bulgaria dated in Bronze Age. Part of them are analyzed by three different p-XRFs (Bruker Tracer III-V, Bruker S1 Titan and Thermo Niton XL3T-900 GOLDD models) and additional compared with the results obtained by ICP-AES (Perkin Elmer Optoma 7000 DV).
Results and Discussion
The results for SRMs using for the estimation of totally Uncertainty (U) of the p-XRFs shows very good agreement according two-way ANOVA test. All type of the p-XRFs are giving comparable results despite the number of the determining elements are quite different for the used instruments. The best results are obtained for Bruker S1 Titan model (all certificated elements in both SRMs), followed by Thermo Niton XL3T-900 GOLDD (without data for As, S, Bi and Mg) and the last one is the Bruker Tracer III-V, where the concentration of only major compounds are in good correlations with certificated data for the SRMs [5].
Due to the corrosion almost of archaeological copper-tin alloys objects have changes of the elemental composition from the bulk metal to the surface. The surface of corroded bronze ornaments consists mostly of copper carbonates, oxides, and chlorides.
For the archaeological bronze samples it is necessary to remove so called "patina|" to obtained chemical composition for the original alloy and bronzes, both modern, but especially archaeological bronzes, are characterized by different extend of homogeneity, depending on the composition and production techniques. Pb segregation is a well-known phenomenon in copper alloys and this problem was examined in combination with the metallographic investigations. At the fig.1 are shown the spectra obtained from Bruker S1 Titan for Sn-bronze sample before and after removing of green patina. It is easy to see that the photons generated in tin atoms (corresponding to the peak at 25.2 keV) penetrate easily through the patina while the copper and lead ones are strongly absorbed.
Additional the obtained results for archaeological bronze samples depending from the manufacturer and the used model of p-XRF. While the numbers of the determined elements are quite different from the all models p-XRFs it is resulting on the different composition of the original metal alloy. For example for the same sample the results for p-XRFs shows "Sn-bronze with As", "Sn-bronze with As and/or Pb", "Sn-bronze with Pb" despite the laboratory analysis carry out by ICP-AES shows only "Sn- bronze with Pb".
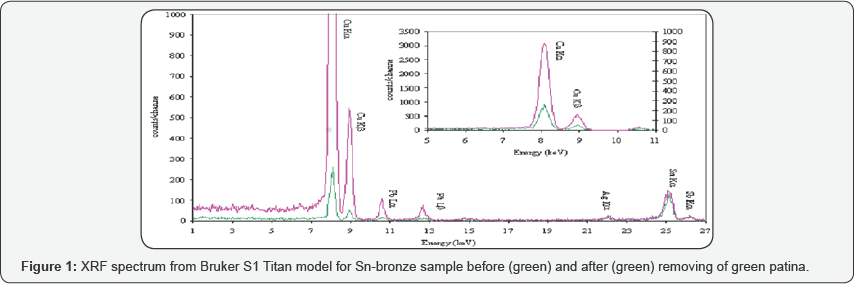
The level of the errors of the determination of tin content by p-XRF is increasing with increasing of the concentration in the samples and in some cases may achieved up to 56% of the real concentration (determined with ICP-AES). Using only internal calibration without correction and statistical treatment there are not possibilities for determination of bulk metal compositions. Even removing of the patina in dept 1-3 mm can provide unreal data-set of analyzed (and not only) Sn-bronzes samples. Chemical processes, such as decertification, change the element composition in such a manner that the original alloy cannot be traced with a non-destructive method (Figure 1).
Conclusion
Obviously the using of only p-XRF for determination of chemical content without external calibration by SRMs and removing of patina it is not suitable for archaeological (and not only) Sn-bronze objects. The best results for the investigated archaeological samples of Cu-Sn-rich-alloys (after removing of patina in dept 3 mm and using an external calibration with SRMs) are done by Bruker S1 Titan model. The new device-Silicon Drift Detector (SDD), cooled by a Pettier element, can reach a resolution of the order of 160eV FWHM at 6keV. This feature makes this device suitable for portable high-resolution XRF spectrometers in the analyses of Cu-alloy after removing of patina and using of external calibration.
References
- Šatović D, Desnica V, Fazinić S (2013) Use of portable X-ray fluorescence instrument for bulk alloy analysis on low corroded indoor bronzes. Spectrochimica Acta Part B: 89: 7-13.
- Killick D (2015) The awkward adolescence of archaeological science. J Arch Sci 56(1): 242-247.
- Lyubomirova V, Djingova, R, Kuleff I (2015) Comparison of analytical techniques for analysis of archaeological bronze. Archaeometry 57(4): 677-686.
- N0rgaard HW (2017) Portable XRF on Prehistoric Bronze Artefacts: Limitations and Use for the Detection of Bronze Age Metal Workshops. Open Archaeology 3(1): 101-122.
- Pernicka E (2014) Provenance Determination of Archaeological Metal Objects. In: Roberts BW & Thornton CP (Eds.), Archaeometallurgy in Global Perspective. Methods and Syntheses, London: Springer.






























