Rhizosphere Soil Bacterial Communities and Bacterial Root Endophytes in Cotton farming is Influence by Cultivation of Transgenic Bt and non-Bt Cotton Crop Varieties
Borkar SG1*, Bhosale AN2 and Ajayasree TS3
1Plant Disease Diagnostic and Research Centre, Endeavour Scientific Agriculture, India
2Department of Plant Pathology & Agricultural Microbiology, Mahatma Phule Agriculture University, India
3Department of Plant Pathology and Agricultural Microbiology, Mahatma Phule Agriculture University, India
Submission: May 18, 2023;Published: June 08, 2023
*Corresponding author: Borkar’s SG, Seed and Plant Disease Diagnostic Laboratory & Research Centre, India, Email id: borkarsg@yahoo.co.in
How to cite this article: Borkar SG, Bhosale AN, Ajayasree TS. Rhizosphere Soil Bacterial Communities and Bacterial Root Endophytes in Cotton farming is Influence by Cultivation of Transgenic Bt and non-Bt Cotton Crop Varieties. Ecol Conserv Sci. 2023; 2(5): 555597 DOI:10.19080/ECOA.2023.02.555597
Abstract
Presence of different bacterial communities in soil and their association with plant roots plays an important role in crop growth and cultivation. These bacterial communities varies with the soil type, soil ecology, soil environment, and plant species grown. However, there are no studies on the variation of bacterial communities present in transgenic and non-transgenic crop soils. In the present investigation, we studied the variation in rhizosphere soil bacterial communities and bacterial root endophytes in transgenic Bt and non-Bt cotton crop cultivation. Rhizosphere bacterial inhabitant under cultivation of Bt and non-Bt cotton hybrids varied and was related to the cotton hybrids grown. In the rhizosphere of RCH-2 Bt cotton, 5 different kinds of bacterial communities were observed with various population density whereas in rhizosphere of RCH-2 non-Bt cotton, bacterial community of only one type was observed indicating that the exudates of RCH-2 Bt were more favourable for the growth of rhizosphere bacterial flora/communities. Similarly, there was variation in the types of rhizosphere bacterial communities of Bunny Bt and Bunny non-Bt hybrids. Two types of bacterial communities were associated with Bunny Bt hybrid rhizosphere while 3 types of bacterial communities were associated with Bunny non-Bt hybrid rhizosphere and had one common bacterial community in rhizosphere of both the hybrids. The presence of bacterial root endophytes was detected in Bt and non-Bt cotton hybrids. In RCH-2 Bt, 2 types of bacterial root endophytes were found whereas 4 types of bacterial root endophytes were found in RCH-2 non-Bt. In this hybrid, only isolate no.2 was common. In Bunny Bt only one type of bacterial root endophyte was found while in Bunny non-Bt, 2 types of bacterial root endophytes were found, and all were different. Thus, both the Bt and non-Bt cotton hybrids contain root endophytes, and the types of endophytes differ from Bt-hybrid to non-Bt hybrid with variable population. The role of these rhizosphere soil bacterial communities and cotton root endophytes needs to be explore for their potential as cotton crop growth stimulant/ in root disease management of cotton crop in western Maharashtra cotton tract.
Keywords: Soil Bacterial Communities; Bacterial Root Endophytes; Cotton Crop; Transgenic and non-Transgenic Hybrids
Introduction
Cotton crop (Gossypium sp) also known as white gold in Indian sub-continent is grown in several countries of Asia region, parts of USSR, some European countries, American sub-continent, and in some African countries Khan [1]. The fabrics and textile industry of the world is dependent on the production of this crop. The area under cotton crop in the world is around 32,500 million hector with production of 25 million tons FAO [2] which comprises both transgenic Bt cotton and non-Bt cotton crop varieties. The acreage of cotton in India in 2019-20 was 12.58 million hector out of which 11.74 million hector (i.e. around 93.6 percent area) was under Bt cotton only MoA&FW [3]. Farm land soils are the basic medium for the growth of crop plants and their production are influenced by soil ecology, soil fertility/composition Long [4] and soil microbial richness. There are different soil microbial communities which functions differently in the crop growth and crop protection. These microbes not only confine to soil, but colonies its host plants as epiphytes and endophytes. Presence of bacterial endophytes is known in the plant system Chanway [5]and cotton is no exception Misaghi & Donndelinger [6]. Similarly, the presence of bacterial root endophytes are known in crops Haichun [7]. These bacterial endophytes are implicated with their role in plant growth and plant protection Lodwyckx [8]. The diversity of bacterial endophytes are affected by various factors including soil cultivation history Correa-Galeote [9]and the crop plant grown. However, is there any effect of transgenic Bt crop on rhizosphere soil bacterial communities and bacterial root endophytes has not yet studied. In the present investigation, therefore, we assess the bacterial communities in rhizosphere soil of Bt and non-Bt cotton hybrids grown in the same cotton cultivation plot and further the variation in bacterial root endophytes in transgenic Bt and non- Bt cotton varieties with their population densities. These results will help us to understand the influence of transgenic crop on the rhizosphere bacterial communities and communities of root endophytes.
Material and Methods
Studies on rhizosphere soil bacterial communities under cultivation of Bt and non-Bt cotton hybrids
To study rhizosphere soil bacterial communities, rhizosphere soil under two cotton hybrids, viz. RCH-2 and Bunny with their Bt and non-Bt counterparts were used. For the isolation of rhizosphere bacterial communities, 1 g soil from rhizosphere under each cotton variety was taken and serial dilution were made up to 10-5 dilution. 0.1 ml of each dilution was plated on Nutrient-Agar (NA) medium. The plates were incubated in BOD at 28 ± 10C temperature for 48 hrs and the appearance of different bacterial colony type and population density was recorded as per standard procedure Borkar [10].
Studies on root endophytes of Bt and non-Bt cotton hybrids
Roots of 2 cotton hybrids viz. RCH-2 and Bunny with their Bt and non-Bt counterparts were used to isolate the root endophytes. Tap root of 1 cm length of each cotton variety was taken for isolation of root endophytes. The root portion was washed thoroughly in water to remove soil and dust particles from the root surface. The root portion was then surface sterilized with 0.1 percent HgCl2 for 2 minutes, followed by 3 washing with distilled sterile water. This root portion was then macerated in 5 ml distilled sterile water in sterile mortar and pestle and 0.1 ml of macerated supernatant suspension was plated on NA medium by spread plate method. The plates were incubated in BOD at 28 ± 10C temperature for 48 hrs and the observations were recorded for isolated root endophytes Jacob [11].
Results and Discussion
Rhizosphere soil bacterial communities under cultivation of Bt and non-Bt cotton hybrids
The rhizosphere soil bacterial type and their population (cfu/g of soil) from the root zone of RCH-2 Bt and non-Bt cotton as well as Bunny Bt and non-Bt cotton was studied by dilution plate method. The results (Table 1) indicated that the rhizosphere soil bacterial community of RCH-2 Bt cotton was composed of different bacterial population which include white mucoid circular colonies with population density 4 x105 cfu/g of soil, yellow nonglistering circular colonies with population density 2 x 105 cfu/g , white rough irregular colonies with population density 2 x 105 cfu/g, yellow glistering circular colonies with population density 3 x 105 cfu/g, and transparent irregular mucoid colonies with population density 2 x 105 cfu/g of soil. However, the rhizosphere soil bacterial communities of RCH-2 non-Bt cotton had only one bacterial population i.e. rough white irregular colonies forming bacteria with population density of 5 x105 cfu/g of soil. Other bacteria population which were associated with RCH-2 Bt cotton rhizosphere were not found in RCH-2 non-Bt cotton rhizosphere soil of the same location. This indicated that the root exudates of the RCH-2 Bt cotton hybrid got ability to thrive various kind of bacterial population in its root rhizosphere as compared to RCH-2 non-Bt cotton hybrid. Similarly, the rhizosphere soil bacterial communities of Bunny Bt cotton composed of 2 bacterial population i.e. isolate no.3 with rough white irregular colonies with population density of 7 x 105 cfu/g; whereas another population of isolate no.6 was pink circular mucoid with population density of 1 x 105 cfu/g of soil. The bacterial population of isolate no.3 was universally present in rhizosphere of Bunny Bt and Bunny non-Bt, as well as RCH-2Bt and RCH-2 non-Bt cotton hybrids. However, isolate no.6 which was present in the rhizosphere of Bunny Bt was not present in the rhizosphere of Bunny non-Bt, RCH-2 Bt and RCH-2 non-Bt hybrids indicating that the type of Bt cotton hybrid had influence on the rhizosphere soil bacterial communities at the same cotton crop cultivation location.
Studies on root endophytes of Bt and non-Bt cotton hybrids
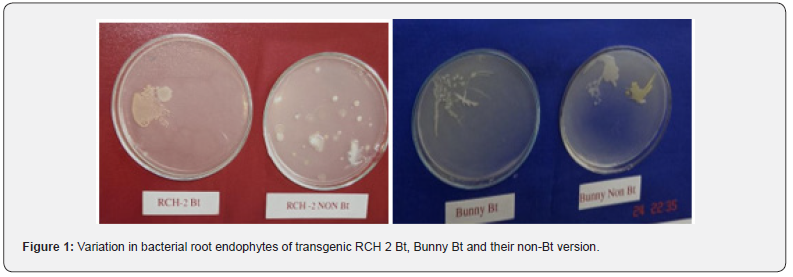
Presence of root endophytes in Bt and non-Bt cotton hybrids were estimated by spread plate technique. A root portion of 1 cm length was macerated in 5 ml of sterile water and suspension was plated by spreading on solidified NA medium to note the appearance of endophytes. The endophytic bacterium were differentiated on the basis of their colony characters. The results (Table 2) & (Figure 1) indicated that the RCH-2 Bt cotton root had 2 types of root endophytes. The one with pale yellow mucoid irregular colonies with a population density of 5 x 103 cfu/cm of root length and the other isolate no.2 forming white mucoid irregular colonies with population density of 50 cfu/cm of root length. In RCH-2 non-Bt cotton root four types of root endophytes were observed. Isolate no.2 had with population density 50 cfu/ cm of root length. Endophyte isolate no.3 was transparent pale yellow suppressed irregular colonies with population density of 250 cfu/cm of root length. Endophyte isolate no.4 was white raised mucoid circular colonies with population density of 250 cfu/cm of root length. Endophyte isolate no.5 was yellowish raised glistering circular colonies with population density 350 cfu/cm of root length. Bunny Bt root contained only 1 type of root endophyte i.e. isolate no.6 of rough white irregular colonies with a population density of <5 x 103 cfu/cm of root length . This root endophyte was not present in Bunny non-Bt root. Instead of this, Bunny non-Bt root contained 2 other endophytes. The one was isolate no.2 with a population density of <25 x 102 cfu/cm of root length and the other forming yellow raised irregular colony with population density of <25 x 102 cfu/cm of root length. These results clearly indicated that both Bt and non Bt hybrids contained root endophytes and the types of endophytes differed from hybrid to hybrid with variable population density. Studies on rhizosphere soil bacterial communities of Bt and non-Bt cotton hybrids indicated that the type of Bt cotton hybrids has influence on the rhizosphere bacterial communities at the same experimental site, which was noted for the first time in case of Bt cotton crop as no such reference is available.


Presence of endophytes in root was reported by many scientist. Tapia-Hernandez [12] reported the presence of endophytic diazotrophs in surface sterilized tissuies of roots of pine-apple plant. Chanway [5] reported nitrogen fixing bacterial endophytes of sugarcane colonized the internal root, stem and leaf tissues.
Barraquio [13] isolated diazothrophic and non-diazotrophic bacterial endophytes from roots and culms of rice. Our results showed the presence of root endophytes in cotton roots which is also reported by McInray [14] McInray [15], and Chen [16]. Further studies showed that the types of root endiphytes differed with cotton hybrids with variable proportion. Similar results was also reported by Adams [17]. Papik [18] reported the diversity in endophytic bacteria. The role of root endophytes are reported in plant growth promoting and plant protection activities Kobayashi and Palumbo [19]; Gaiero [20]; Selim [21]; Dubey [22]. The presence of diversified bacterial communities in rhizosphere soil can be used as a yardstick for soil richness for crop growth and the particular root endophytes as biocontrol agent to protect the crop for specific root pathogen.
References
- Khan MA, Wahid A, Ahmad M, Tahir MT, Ahmad M (2020) World cotton production and consumption: An Overview. In book: Cotton Production and Uses.
- FAO (2021) Recent trends and prospects in the world cotton market and policy development.
- MoA & FW (2020) Cultivation of GM crops. Directorate of Economics and Statistics.
- Long HH, Sonntag DG, Schmidt DD, Baldwin IT (2010) The structure of the culturable root bacterial endophyte community of Nicotiana attenuate is organised by soil composition and host plant ethylene production and perception. New Phytol 185(2): 554-567.
- Chanway CP (1998) Bacterial endophytes: ecology and practical implications. Sydowia 50: 149-170.
- Misaghi IJ, Donndelinger CR (1990) Endophytic bacteria in symptoms free cotton plants. Phytopathology 43: 140-146.
- Haichun C, Shahzad M, Pengfei He, Yixin Wu, Pengbo He (2022) Diversity of root endophytic bacteria from maize seedling involved in biocontrol and plant growth promotion. Egyptian Journal of Biological Pest Control 32: 129.
- Lodwyckx C, Vangronsveld J, Proteous F (2002) Endophytic bacteria and their potential application. Critical Review in Plant Sciences 21(6): 583-606.
- Correa-Galeote D, Bedmar EJ, Arone GJ (2018) Maize endophytic bacterial diversity as affected by soil cultivation history. Front Microbiol 9: 484.
- Borkar SG (2017) Laboratory Techniques in Plant Bacteriology. CRC Press pp: 342.
- Jacob MJ, Bugbee WM, Gabrielson DA (1985) Enumeration, location and characterization of endophytic bacteria within sugar-beet root. Canadian J Botany 63: 1262-1265.
- Tapia-Hernandez A, Bustillos-Cristales MR, Jimenez ST, Cabellero Mellado J, Fuentes Ramierez LE (2000) Natural endophytic occurrence of Acetobacter diazotrophicus in pineapple plants. Microb Ecol 39(1): 49-55.
- Barraquio WL, Revilla L, Ladha JK (1997) Isolation of endophytic diazotrophic bacteria from wetland rice. Plant & Soil: 15-24.
- Mc-Inroy JA, Kloepper JW (1995) Survey of indigenous bacterial endophytes from cotton and sweet corn. Plant & Soil 173: 337-342.
- Mc-Inroy JA, Kloepper JW (1991) Analysis of endophytic bacteria of maize and cotton in field 8: 328-331.
- Chen C, Bauske EM, Musson G, Rodriguez KR, Kloepper JW (1995) Biological control of fusarium wilt of cotton by use of endophytic bacteria. Biological control 5(1): 83-91.
- Adams PD, Kloepper JW (2002) Effect of host genotypes on indigenous bacterial endophytes in cotton. Plant & Soil 240: 181-189.
- Papik J, Folkmanova M, Polivkova M, Suman J, Uhlik O (2020) The invisible life inside plants: deciphering the riddles of endophytic bacterial diversity. Biotechnol Adv 44: 107614.
- Kobayashi DY, Palumbo JD (2002) Bacterial endophytes and their effects on plants and uses in agriculture. Microbial Endophytes pp: 199-233.
- Gaiero JR, Crystal AM, Karen AT, Nicola JD, Anna SB (2013) Inside the root microbiome: Bacterial root endophytes and plant growth promotion. Am J Bot 100 (9): 1738-1750.
- Selim HM, Gomaa NM, Essa AM (2016) Antagonistic effect of endophytic bacteria against some phytopathogens. Egypt J Bot 1: 74-81.
- Dubey A, Saiyam D, Kumar A, Abeer H, Allah EFA (2021) Bacterial root endophytes: Characterization of their competence and plant growth promotion in Soybean under drought stress. Int J Environ Res Public Health 18(3): 931.






























