Abstract
Ewing sarcoma (ES) is a high-grade bone or soft tissue cancer in children and young people. While the EWS/FLI1 fusion mutation produces a unique oncogenic driver and therapeutic target in ES, detailed analysis of ES molecular biology has revealed numerous new vulnerabilities which provide alternative strategies for drug development. In this review article, we will discuss the biological rationale of using small-molecule inhibitors, as well as their preclinical and clinical applications, in ES treatment.
Keywords:Ewing Sarcoma; Small-Molecule Inhibitors; Targeted Therapy; Epithelial; Vascular Endothelial
Abbreviations:EMT: Epithelial-to-Mesenchymal Transition; ES: Ewing Sarcoma; EWS: Ewing Sarcoma Breakpoint Region 1; FLI1: Friend Leukemia Integration 1; HDACi: Histone Deacetylase Inhibitors; IGF-1R: Insulin-like Growth Factor-1 Receptor; PARPi: Poly-ADP-Ribose Polymerase Inhibitors; PDGFRs: Platelet-derived Growth Factor Receptors; Pol1: RNA Polymerase I; RHA: RNA Helicase A; rRNA: Ribosomal RNA; RTKs: Receptor Tyrosine Kinases; SL1: Selective Factor 1; TKI: Tyrosine Kinase Inhibitors; VEGFRs: Vascular Endothelial Growth Factor Receptors
Main Text
Ewing sarcoma (ES) is a malignant tumor arising in bone or soft tissue that predominantly affects children and young adults. The current standard treatment for ES is a chemotherapy regimen that includes alternating treatments of vincristine, doxorubicin, and cyclophosphamide with a combination of ifosfamide and etoposide (VDC/IE), alongside radiation therapy and surgery. This regimen has increased the 5-year survival rate of patients with localized ES to around 70-73% [1]. ES is particularly aggressive, with metastasis occurring in around 25% of cases.
Recurrence rates within 5 years are approximately 40% in patients with localized disease and 60-80% in patients with metastatic disease. The prognosis for patients with metastatic or recurrent ES is much lower despite similar treatment to localized cases, with a 6-year survival rate of <30% [2]. A phase III trial (NCT01231906) introducing additional chemotherapies (topotecan and cyclophosphamide) to the standard regimen did not result in increased survival, indicating that a more intense treatment did not cause better outcomes [3], further highlighting the need for targeted therapies as either monotherapy or combination therapy options to further bolster current chemotherapeutic regimens.
ES is characterized by a chromosomal translocation most commonly between members of the FET and ETS protein family, with 85% of cases resulting from a fusion between Ewing sarcoma breakpoint region 1 (EWS) and friend leukemia integration 1 (FLI1), forming EWS/FLI1 [t(11;22)(q24;q12)]. The resulting oncoprotein contains the transactivation domain of EWS and the DNA-binding domain of FLI1, creating an aberrant transcription factor that drives tumorigenesis [4]. The intrinsically disordered structure of EWS/FLI1 makes direct targeting of the oncoprotein difficult, and previous attempts have been lackluster due to poor solubility and resistance [5].
Instead, attempts to disrupt the EWS/FLI1 complex has become a focus of targeted therapy for ES treatment. YK-4-279 is a small molecule that has the ability to interrupt EWS-FLI1 interaction with RNA helicase A (RHA) and partially inhibit its transcriptional activity, leading to increased apoptosis and decreased tumor growth in ES xenograft mice [6-8]. However, clinical study of the YK-4-279 analog TK216 in a phase I/II trial (NCT02657005) showed only limited benefit for recurrent ES patients, with a progression-free survival rate of 11.9% [9].
Beyond EWS/FLI1-targeted therapy, multiple small-molecule inhibitors have been explored in ES treatment. Tyrosine kinase inhibitors (TKI) appear to be the most tested small molecule in recurrent ES clinical trials, given the fact that several receptor tyrosine kinases (RTKs), such as Insulin-like Growth Factor-1 Receptor (IGF-1R), Platelet-derived Growth Factor Receptors (PDGFRs), c-KIT and Vascular Endothelial Growth Factor Receptors (VEGFRs), are overexpressed in ES and contribute directly to cell proliferation, metastasis and immune evasion through activation of PI3K/AKT and MAPK/ERK pathways.
Preclinical testing of TKI has revealed promising anti-cancer response in ES models [10]. In support of these findings, the FDA-approved multi-kinase inhibitor regorafenib is currently being explored in patients with primary and metastatic ES in seven clinical trials across the world. Similar reagents such as cabozantinib and trametinib have also entered clinical trials for ES treatment. WEE1 is a non-receptor tyrosine kinase found to be upregulated in a subset of ES specimens [11].
Building on the role of WEE1 in regulation of the cell-cycle checkpoint, it was reported that inhibition of WEE1 activity by small molecules (such as AZD1775 and MK1775) triggers impaired DNA repair [12] and, when used in combination with DNA damaging agents, exhibits robust anti-cancer activity in ES [13,14]. Poly-ADP-ribose polymerase (PARP) is a family of proteins involved in DNA damage response [15], and ES has been characterized as having defects in DNA repair, making it particularly sensitive to PARP inhibition [16,17]. While clinical trials revealed modest outcomes when testing PARPi as a monotherapy [18], preclinical studies have shown positive results in ES when combining PARPi (such as talazoparib and olaparib) with the chemotherapeutic agents temozolomide and irinotecan [19].
Histone deacetylase inhibitors (HDACi) have been applied to ES treatment, initially because of their inhibitory activity against ES metastasis through acetylation of the RNA-binding protein YB-1 and downregulation of oxidative stress-adaptive factors [20]. Interestingly, recent studies in ES have also linked the mechanism of HDACi action with DNA replication stress and demonstrated a strong synergy between HDACi and the chemotherapeutic agent doxorubicin [21,22]. Thus, further exploring the biological interaction of WEE1, PARP and HDAC inhibitors with chemotherapy may lay out optimum combination strategies beneficial for ES patients.
Ribosomes are molecular machines that are synthesized into proteins which are vital for cell survival and growth, especially in many types of cancer that rely on hyperactivated ribosome production to promote tumorigenesis and proliferation [23]. ES cell lines were identified to be particularly sensitive to perturbations in genes associated with ribosome biogenesis via CRISPR loss-of-function screenings, indicating inhibition of this pathway could be a promising novel therapeutic option [24]. The rate-limiting step of ribosome production is the transcription of ribosomal RNA (rRNA) via RNA polymerase I (Pol1), and smallmolecule inhibitors of Pol1 have shown promising results in many cancer types.
In our prior study, two Pol1 inhibitors (CX-5461 and BMH- 21) were used on patient-derived ES cell lines with significant decrease in cell survival and migratory potential observed. BMH-21 exhibited a more robust response in ES cells, with cell viability in BMH-21-treated cells <10% and in CX-5461-treated cells around 50%. This difference in response is likely due to the distinct mechanisms of drug action. BMH-21 is considered as a direct Pol1 inhibitor, which blocks Pol1 initiation and elongation by causing proteasomal degradation of RPA194, a core catalytic subunit of the Pol1 complex [25]. In contrast, as the first-inhuman Pol1 inhibitor, CX-5461 exhibits an indirect mode of action through disconnecting Pol1 from its ancillary partner, Selective Factor 1 (SL1), to prevent rRNA gene promoter release [26].
Two phase I studies (NCT02719977 and NCT04890613) have released encouraging clinical data on the tolerability of CX-5461 in hematological cancers and solid tumors [27,28], although the therapeutic efficacy was modest. These findings could reflect an incomplete understanding of CX-5461 action or potential offtarget effects. Indeed, accumulating evidence suggests that Pol1 may not represent a primary target of CX-5461 in cancer cells [29- 31]. Since neither BMH-21 nor its derivatives have been assessed in clinical trials, the true significance of interfering with Pol1 function in cancer therapy remains an open question.
Despite the above-noted discrepancy in the mechanism of drug action, CX-5461 has been shown to work synergistically with numerous small molecule compounds having therapeutic potential in ES treatment. For instance, CX-5461 demonstrates promising efficacy when combined with the WEE1 inhibitor adavosertib in glioblastoma [32], the HDAC inhibitor panobinostat in multiple myeloma [33], and the PARP inhibitor talazoparib in both castrate-proficient prostate cancer [34] and high grade serous ovarian cancer [35]. Further research into effective combinations synergizing with CX-5461 may create new treatment strategies for ES patients.
At present, there is a very limited understanding of BMH- 21 interaction with existing drugs in cancer therapy. One study found that inhibition of Pol1 activity by BMH-21 can impair the epithelial-to-mesenchymal transition (EMT) property of breast cancer cells to enhance their sensitivity to chemotherapy drugs (including cyclophosphamide, 5FU, cisplatin, doxorubicin, gemcitabine, and paclitaxol) in mouse models [36]. In a second study also involving breast cancer, BMH-21 displayed strong cytotoxic synergy with CX-5461 through activation of p53 tumor suppression pathways [37].
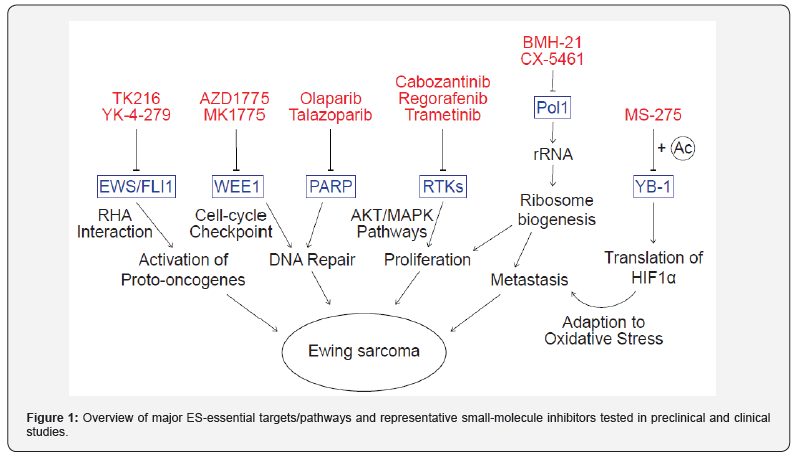
Further studies are required to apply synergistic analysis to high-throughput chemical screening for the most effective combinations with Pol1 inhibition in ES. In summary, advances in the knowledge of ES molecular biology have led to discovery of various druggable targets and cellular pathways/processes (schematically shown in Figure 1). Once the crosstalk between ES-essential signaling events is clearly understood in mechanistic terms, it may be possible to optimize the use of existing therapeutic compounds to develop effective anti-ES strategies.
Acknowledgement
This work was supported by grants from St. Baldrick’s Foundation (Career Development Award) and Children’s Cancer Research Fund (Accelerating Impact for Hard-to-Treat Cancers Award).
References
- Holcombe E Grier, Mark D Krailo, Nancy J Tarbell, Michael P Link, Christopher J H Fryer, et al. (2003) Addition of ifosfamide and etoposide to standard chemotherapy for Ewing's sarcoma and primitive neuroectodermal tumor of bone. N Engl J Med 348(8): 694-701.
- Van Mater D, L Wagner (2019) Management of recurrent Ewing sarcoma: challenges and approaches. Onco Targets Ther 12: 2279-2288.
- Leavey PJ, NN Laack MD Krailo, et al. (2021) Phase III Trial Adding Vincristine-Topotecan-Cyclophosphamide to the Initial Treatment of Patients with Nonmetastatic Ewing Sarcoma: A Children's Oncology Group Report. J Clin Oncol 39(36): 4029-4038.
- Uren A, JA Toretsky (2005) Ewing's sarcoma oncoprotein EWS-FLI1: the perfect target without a therapeutic agent. Future Oncol 1(4): 521-528.
- Moinuddin Jiauddin, Kirtana Reddy, Hashiya Preeya Ravi, Balaji Ramachandran (2025) Druggable upregulated proteins in EWS-FLI-driven Ewing sarcoma as emerging new therapeutic targets. Am J Transl Res 17(3): 1580-1603.
- Julie S Barber-Rotenberg, Saravana P Selvanathan, Yali Kong, Hayriye V Erkizan, Tara M Snyder, et al. (2012) Single enantiomer of YK-4-279 demonstrates specificity in targeting the oncogene EWS-FLI1. Oncotarget 3(2): 172-182.
- Hayriye Verda Erkizan, Jeffrey A Schneider, Kamal Sajwan, Garrett T Graham, Brittany Griffin, et al. (2015) RNA helicase A activity is inhibited by oncogenic transcription factor EWS-FLI1. Nucleic Acids Res 43(2): 1069-1080.
- Tsion Zewdu Minas, Jenny Han, Tahereh Javaheri, Sung-Hyeok Hong, Michaela Schlederer, et al. (2015) YK-4-279 effectively antagonizes EWS-FLI1 induced leukemia in a transgenic mouse model. Oncotarget 6(35): 37678-37694.
- Paul A Meyers, Noah Federman, Najat Daw, Peter M Anderson, Lara E Davis, et al. (2024) Open-Label, Multicenter, Phase I/II, First-in-Human Trial of TK216: A First-Generation EWS::FLI1 Fusion Protein Antagonist in Ewing Sarcoma. J Clin Oncol 42(31): 3725-3734.
- Ahmad Assi, Mohamad Farhat, Rami Mohanna, Maria Catherine Rita Hachem, Ziad Zalaquett, et al. (2025) Tyrosine kinase inhibitors in Ewing's sarcoma: a systematic review. BMC Cancer 25(1): 735.
- Upendra Kumar Soni, Yuhua Wang, Ram Naresh Pandey, Ryan Roberts, Joseph G Pressey, et al. (2023) Molecularly Defined Subsets of Ewing Sarcoma Tumors Differ in Their Responses to IGF1R and WEE1 Inhibition. Clin Cancer Res 29(2): 458-471.
- Stacia L Koppenhafer, Kelli L Goss, William W Terry, David J Gordon (2020) Inhibition of the ATR-CHK1 Pathway in Ewing Sarcoma Cells Causes DNA Damage and Apoptosis via the CDK2-Mediated Degradation of RRM2. Mol Cancer Res 18(1): 91-104.
- Jenny M Kreahling, Jennifer Y Gemmer, Damon Reed, Douglas Letson, Marilyn Bui, et al. (2012) MK1775, a selective Wee1 inhibitor, shows single-agent antitumor activity against sarcoma cells. Mol Cancer Ther 11(1): 174-182.
- Jeffrey C Martin, Jennie R Sims, Ajay Gupta, Andrei V Bakin, Joyce E Ohm (2022) WEE1 inhibition augments CDC7 (DDK) inhibitor-induced cell death in Ewing sarcoma by forcing premature mitotic entry and mitotic catastrophe. Cancer Res Commun 2(6): 471-482.
- Wang Y, W Luo, Y Wang (2019) PARP-1 and its associated nucleases in DNA damage response. DNA Repair (Amst) 81: 102651.
- Elizabeth Stewart, Ross Goshorn, Cori Bradley, Lyra M Griffiths, Claudia Benavente, et al. (2014) Targeting the DNA repair pathway in Ewing sarcoma. Cell Rep 9(3): 829-841.
- Amanda Balboni Iniguez, Björn Stolte, Emily Jue Wang, Amy Saur Conway, Gabriela Alexe, et al. (2018) EWS/FLI Confers Tumor Cell Synthetic Lethality to CDK12 Inhibition in Ewing Sarcoma. Cancer Cell 33(2): 202-216 e6.
- Edwin Choy, James E Butrynski, David C Harmon, Jeffrey A Morgan, Suzanne George, et al. (2014) Phase II study of olaparib in patients with refractory Ewing sarcoma following failure of standard chemotherapy. BMC Cancer 14: 813.
- Florian Engert, Cornelius Schneider, Lilly Magdalena Weiβ, Marie Probst, Simone Fulda (2015) PARP Inhibitors Sensitize Ewing Sarcoma Cells to Temozolomide-Induced Apoptosis via the Mitochondrial Pathway. Mol Cancer Ther 14(12): 2818-2830.
- Amal M El-Naggar, Syam Prakash Somasekharan, Yemin Wang, Hongwei Cheng, Gian Luca Negri, et al. (2019) Class I HDAC inhibitors enhance YB-1 acetylation and oxidative stress to block sarcoma metastasis. EMBO Rep 20(12): e48375.
- Stacia L Koppenhafer, Elizabeth L Geary, Mary V Thomas, Emma E Croushore, Jessica AO Zimmerman, et al. (2025) Histone Deacetylase Inhibitors Target DNA Replication Regulators and Replication Stress in Ewing Sarcoma Cells. Cancer Res Commun 5(6): 1034-1048.
- Kaitlyn H Smith, Erin M Trovillion, Chloe Sholler, Divya Gandra, Kimberly Q McKinney, et al. (2024) Panobinostat Synergizes with Chemotherapeutic Agents and Improves Efficacy of Standard-of-Care Chemotherapy Combinations in Ewing Sarcoma Cells. Cancers (Basel)16(21): 3565.
- Amr R Elhamamsy, Brandon J Metge, Heba A Alsheikh, Lalita A Shevde, Rajeev S Samant (2022) Ribosome Biogenesis: A Central Player in Cancer Metastasis and Therapeutic Resistance. Cancer Res 82(13): 2344-2353.
- Alexis Bruce, Gavin Anchondo, Rebekah Carter, Sergio Guerrero Pacheco, Hema Sri Kambala, et al. (2025) Targeting RNA Polymerase I in Ewing Sarcoma Treatment. Cancer Sci 116(8): 2317-2319.
- Ruth Q Jacobs, Abigail K Huffines, Marikki Laiho, David A Schneider (2022) The small-molecule BMH-21 directly inhibits transcription elongation and DNA occupancy of RNA polymerase I in vivo and in vitro. J Biol Chem 298(1): 101450.
- Denis Drygin, Amy Lin, Josh Bliesath, Caroline B Ho, Sean E O'Brien, et al. (2011) Targeting RNA polymerase I with an oral small molecule CX-5461 inhibits ribosomal RNA synthesis and solid tumor growth. Cancer Res 71(4): 1418-1430.
- Amit Khot, Natalie Brajanovski, Donald P Cameron, Nadine Hein, Kylee H Maclachlan, et al. (2019) First-in-Human RNA Polymerase I Transcription Inhibitor CX-5461 in Patients with Advanced Hematologic Cancers: Results of a Phase I Dose-Escalation Study. Cancer Discov 9(8): 1036-1049.
- John Hilton, Karen Gelmon, Philippe L Bedard, Dongsheng Tu, Hong Xu, et al. (2022) Results of the phase I CCTG IND.231 trial of CX-5461 in patients with advanced solid tumors enriched for DNA-repair deficiencies. Nat Commun 13(1): 3607.
- Peter M Bruno, Mengrou Lu, Kady A Dennis, Haider Inam, Connor J Moore, et al. (2020) The primary mechanism of cytotoxicity of the chemotherapeutic agent CX-5461 is topoisomerase II poisoning. Proc Natl Acad Sci USA 117(8): 4053-4060.
- Min Pan, William C Wright, Richard H Chapple, Asif Zubair, Manbir Sandhu, et al. (2021) The chemotherapeutic CX-5461 primarily targets TOP2B and exhibits selective activity in high-risk neuroblastoma. Nat Commun 12(1): 6468.
- Donald P Cameron, Jirawas Sornkom, Sameerh Alsahafi, Denis Drygin, Gretchen Poortinga, et al. (2024) CX-5461 Preferentially Induces Top2alpha-Dependent DNA Breaks at Ribosomal DNA Loci. Biomedicines 12(7): 1514.
- Meng Cheng, Rui Wang, Ying Pang, Xu Chen, Jing Zhang, et al. (2025) Combined inhibition of WEE1 by AZD1775 synergistically enhances CX-5461 mediated DNA damage and induces cytotoxicity in glioblastoma. Tissue Cell 97: 103093.
- Kylee H Maclachlan, Kezia Gitareja, Jian Kang, Andrew Cuddihy, Yuxi Cao, et al. (2024) Targeting the ribosome to treat multiple myeloma. Mol Ther Oncol 32(1): 200771.
- Elaine Sanij, Katherine M Hannan, Jiachen Xuan, Shunfei Yan, Jessica E Ahern, et al. (2020) CX-5461 activates the DNA damage response and demonstrates therapeutic efficacy in high-grade serous ovarian cancer. Nat Commun 11(1): 2641.
- Mitchell G Lawrence, Laura H Porter, Nicholas Choo, David Pook, Jeremy P Grummet, et al. (2021) CX-5461 Sensitizes DNA Damage Repair-proficient Castrate-resistant Prostate Cancer to PARP Inhibition. Mol Cancer Ther 20(11): 2140-2150.
- Yi Ban, Yue Zou, Yingzhuo Liu, Sharrell B Lee, Robert B Bednarczyk, et al. (2024) Targeting ribosome biogenesis as a novel therapeutic approach to overcome EMT-related chemoresistance in breast cancer. bioRxiv.
- Chung JY, KN Nguyen, BA Knutson (2025) Dual RNA Polymerase I Inhibition with CX-5461 and BMH-21 Synergizes in Breast Cancer by Activating p53-Dependent Stress. bioRxiv.






























