Abstract
Introduction: The biological characteristics of the tumor are used to estimate prognosis and select appropriate systemic therapy for patients with breast cancer. The advent of molecular technology has incorporated new biomarkers along with immunohistochemical and serum biomarkers. Immunohistochemical markers are often used to guide treatment decisions, to classify breast cancer into subtypes that are biologically distinct and behave differently, and both as prognostic and predictive factors. Steroid hormone receptors, markers of tumour proliferation, and factors involved in angiogenesis and apoptosis are of scientific interest. We consider the utility of established immunohistochemical markers, and discuss the challenges involved in integrating novel molecular markers into clinical practice. Immunohistochemistry has an important role in the pathology of breast disease. There is a growing list of Available antibodies or antigen retrieval techniques, which all contribute to the broader utility of immunohistochemistry for solving diagnostic problems or for determining prognosis and response to therapy in breast pathology. Myoepithelial markers are useful in helping to distinguish benign lesions from malignant lesions. The most common immunohistochemical breast cancer prognostic and therapeutic markers used include estrogen receptor, human epidermal growth factor receptor-2, Ki-67, progesterone receptor, and p53. In addition, markers of angiogenesis and apoptosis are also important.
Discussion: It has been categorized into different subsets to make this very reviewed article simple to understand by every oncologist as given below.
Keywords:Biomarkers; Immunohistochemistry; Predictive factors
Abbreviations: IHC: Immunohistochemistry; CK: Cytokeratins; EMA: Epithelial membrane antigen; ER: Estrogen receptor; PR: Progesterone receptor; DCIS: Ductal carcinoma in situ; SMA: Smooth muscle actin; GCDFP-15: Gross cystic disease fluid protein 15; HER2: Human epidermal growth factor receptor 2
Introduction
Immunohistochemistry (IHC) is used to characterize intracellular proteins or various cell surfaces in all tissues. Individual markers or more often panels of various marker proteins can be used to characterize various tumour subtypes, confirm tissue of origin, distinguish metastatic from primary tumour and provide additional information which may be important for prognosis, predicting response to therapy or evaluating residual tumor post-treatment. There is a growing list of available products (antibodies) or antigen retrieval techniques, which all contribute to the broader utility of immunohistochemistry for solving diagnostic problems or for determining prognosis and response to therapy in breast pathology. Diagnostic and prognostic markers are described although some of them can be included in both.
Diagnostic Markers
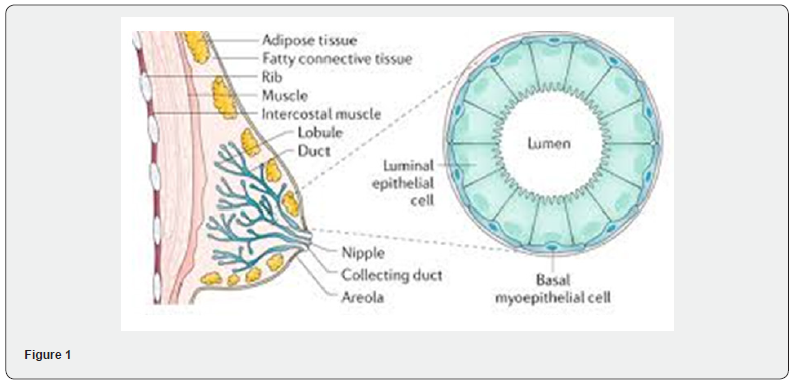
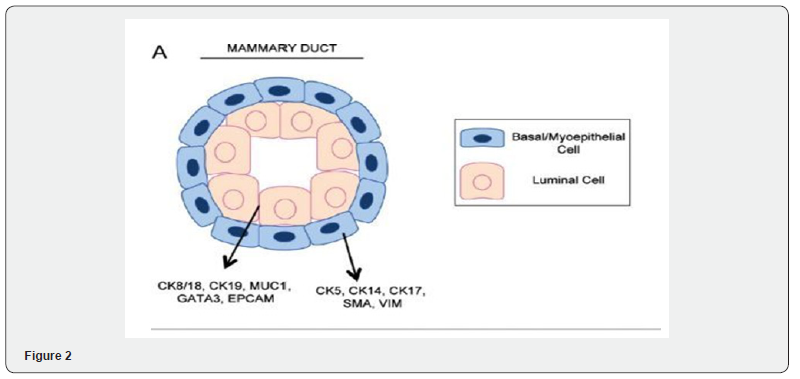
The most important diagnostic problems that occur in mam mary gland tumor pathology are the differential diagnosis of various types of benign lesions and carcinoma, differentiating between carcinoma in situ and invasive carcinoma, diagnosis and differentiation of microinvasion and its imitating lesions and confirming the breast as the primary site in metastatic carcinoma. In the absence of advanced molecular biological techniques, IHC can be used to identify histological subtypes or molecular phenotypes. Some of these problems can be solved using IHC markers (Table 1). It is well known that normal glandular breast tissue is composed of three cell types. Which express different subsets of proteins: luminal, basal and myoepithelial. The luminal cells express cytokeratins (CK7, 8, 18, 19), epithelial membrane antigen (EMA), milk fat globule membrane antigen (MFGM), α-lactalbumin, estrogen receptor (ER), and progesterone receptor (PR). Myoepithelial cells express basal cell type CKs and specific markers: smooth muscle action, calponin, S100 and p63 (Figures 1-3).

ER=Estrogen Receptor; PR= Progesterone Receptor; HER2= Human Epidermal Growth Factor Receptor 2; HER1 = Human Epidermal Growth Factor 1; CK = Cytokeratin
Myoepithelial markers SMA, Calponin, p63, SMMHC
Myoepithelial markers are useful in helping to distinguish invasive carcinoma from benign proliferations with a similar morphological appearance, benign proliferative lesions and most preinvasive lesions with an intact myoepithelium. Invasive carcinomas lack the myoepithelial cell layer that normally surrounds benign breast glands. There is an exception, microglandular adenosis, a benign proliferative lesion which lacks the myoepithelial cell layer in the same context, to assess intraductal proliferative lessions, high-molecular-weight cytokeratins (cytokeratin 14 and cytokeratin 5/6) can be helpful in distinguishing ductal hyperplasia from low-grade ductal carcinoma in situ (DCIS). Atypical ductal hyperplasia or in situ carcinoma can arise in otherwise benign papillary lesions and is defined as a type of ductal hyperplasia that morphologically simulates DCIS. Characteristically, atypical ductal hyperplasia has a uniform population of cells and most lesions. are small and focal, involving only a portion of a duct or only a few small ducts measuring less than 2 mm. Using IHC, positive myoepithelial staining is seen in the benign area with attenuated or absent staining in areas of atypia or in situ carcinoma. It is possible that the area of atypia or in situ carcinoma may not even be represented in the limited sample from a core needle biopsy. Smooth muscle actin (SMA) has long been used as a myoepithelial marker in breast pathology diagnosis as a Sensitive marker of myoepithelial differentiation, even if it is not specific, because any cell with substantial expression of action is positive for SMA (myofibroblasts and blood vessels are positive for SMA). This becomes problematic in lesions where there are either myofibroblasts or blood vessels near the epithelial lesion. One pitfall is the presence myofibroblasts within desmoplastic stroma adjacent to nests/ glands of invasive carcinoma being misinterpreted as myoepithelial cells, resulting in a false-negative diagnosis. This is why the use of a panel of markers (p63, calponin, smooth muscle myosin, CD10, S100) or a more specific marker such as p63 are recommended.
One option is calponin, a protein belonging to the contractile apparatus in smooth muscle cells, which is considered to have the same sensitivity as SMA, however, like SMA, staining of myofibroblasts and smooth muscle in blood vessels can be obtained. As with SMA, cytoplasmic staining of myoepithelial cells tends to encircle the nucleus as opposed to the staining pattern of myofibroblasts. Compared to other markers (p63 or smooth muscle myosin heavy chain (SMMHC)), it tends to show more complete staining of the myoepithelial layer. p63 is a homolog of p53 and has been shown to be expressed exclusively in myoepithelial cells in normal breast and can be very useful in differential diagnosis involving benign lesions such as sclerosing adenosis, radial scars and papillary lesions. The advantage of using p63 is its nuclear localization and absence of staining in smooth muscle cells, such as myofibroblasts and blood vessels. Thus, it provides almost 100% specificity, however, its sensitivity has been reported to be approximately 90%. This is demonstrated by the so-called “focal gaps” in staining in the myoepithelial layer, partly due to the plane of section. In addition, it has now been shown that about 10% to 15% of invasive tumors, particularly high-grade and metaplastic carcinomas, express p63, although the staining is usually weaker than that seen in myoepithelial cells. Similarly, the force of squamous differentiation stains positively.
Like other smooth muscle markers, SMMHC is associated with contractile elements and is present in all cells with such properties. It is expressed primarily in myoepithelial cells but is also expressed in blood vessels. An advantage of SMMHC is that it demonstrates less crossreactivity in my fibroblasts than calponin and SMA. Overall, the studies so far suggest that among smooth muscle markers, SMMHC provides the best results, in terms of both sensitivity and specificity. When inflammation or reactive fibrosis obscure the interface between involved ducts and adjacent stroma in some cases of DCIS, IHC can help to clarify the integrity of the duct wall. Usually, ductal carcinoma cells are negative for myoepithelial cells markers: S100, SMA, SMMHC, calponin, CK5, CK14, CK17, CD10, and p63. The specific markers among these are SMMHC, calponin, and p63, these as well as some basal CKs have an advantage in that they do not stain myofibroblasts. Is this correct? In most laboratories, however, the choice between these markers depends on individual experience, preference or financial resources.
Lobular or ductal carcinoma:
E-cadherin, CK8: Determining whether an in-situ lesion is lobular carcinoma, or ductal carcinoma has clinical management implications and is another situation in which IHC proves its worth. IHC E-cadherin is currently used to differentiate between the two. Most ductal carcinomas express cytoplasmic E-cadherin, whereas most lobular carcinomas lack expression of E-cadherin [. In addition, the differences in CKs expression may be used: high molecular- weight CK (clone 34βE12) is usually expressed by lobular carcinomas but is absent or expressed at low levels in most cases of DCIS.
Identification of subtypes of breast cancer
Analysis of both adjuvant and neoadjuvant trials has shown that not all Chemotherapeutics have equal effects on breast cancer patients, therefore, further individualization of chemotherapy may be required. Data on differences in chemotherapy sensitivity to taxanes and anthracyclines suggest that there are significant differences across disease subtypes, which if further validated, could be used to guide the best decision-making in patient treatment. The St Gallen expert panel which met at the 12th International Breast Cancer Conference held at St Gallen (Switzerland) in March 2011, identified four subtypes of breast cancer according to estrogen and progesterone receptors, and over expression and/ or amplification of the hu man epidermal growth factor receptor 2 (HER2) oncogene. The four subtypes were luminal A, luminal B, Erb-B2 over expression and basal-like. The expert panel provided systemic treatment recommendations for the subtypes including endocrine therapy alone for luminal A, endocrine ± cytotoxic therapy for luminal B (HER2 negative); cytotoxics + anti-HER2 + endocrine therapy for luminal B (HER2 positive); cytotoxics + anti- HER2 for HER2 positive (non-luminal); and cytotoxics for triple negative [1-6].
Markers for mammary origin in metastatic carcinoma
GCDFP15, mamaglobin, CEA-In the case of small metastasis of infiltrating lobular carcinomas, false negative results are far more frequent than those in infiltrating ductal carcinoma Medullary carcinoma metastasis or other subtypes of mammary carcinoma (lobular, sarcomatoid) can often be mistaken for malignant lymphoma (with “signet ring” cells, clear cells, with carcinoma pattern, sarcomatoids). In these situations, a positive reaction for CK and lack of reactivity for lymph markers suggest a diagnosis of metastasis. In the case of large metastasis in the axillary lymph nodes, IHC can demonstrate by a positive reaction for epithelial markers the carcinomatous nature of cells, difficult to appreciate as epithelial in the case of axillary metastasis of infiltrating lobular carcinoma (relatively uniform appearance of tumor cells and low mitotic activity). For small metastasis of infiltrating lobular carcinoma, false negative results are much more common than in infiltrating ductal carcinoma [7-9]. In addition, medullary carcinoma metastasis or other subtypes of breast carcinoma can sometimes be confused with malignant lymphoma (cells in the socalled “signet ring”, clear cell); in these situations, a positive reaction for CK and lack of reactivity for lymphoma markers suggest a diagnosis of metastasis. The identification of metastatic carcinoma of the breast may be difficult in the absence of a previous history of breast cancer. Markers for mammary origin include receptors for hormones, such as androgen receptors (ARs) and gross cystic disease fluid protein 15 (GCDFP-15). GCDFP-15 is present in the liquid of breast cysts and any apocrine cells: mammary glands, salivary glands, sweat, Paget’s disease, etc. which is why it is important to add other markers to the diagnostic panel such as ER, PR, AR, and HER-2/neu, mammaglobin, and CKs (7 and 20). In this context, ARs and/or HER-2/neu are given additional value in a great number of ER-negative high grade ductal carcinomas. Lately, mammaglobin has been described as a breast cancer-specific gene, and its utility as a novel breast cancer marker has been confirmed. Mammaglobin A and B identified in breast cells are overexpressed in breast cancer. Mammaglobin A is more specific for breast and gynecologic organs, while mammaglobin B may be found in several other tumors, especially gastrointestinal malignancies. Many studies have suggested that elevated mammaglobin levels in breast cancer are associated with clinical and biological features defining a less aggressive tumor phenotype. Mammaglobin expression is not changed at the metastatic or lymph node site. It can help, in combination with other markers, to establish the correct diagnosis of metastatic breast carcinoma. Although many carcinomas would not be included in the differential diagnosis of breast carcinoma, the specificity of this marker was 92% [10-15].
In the same study, when the immunohistochemical staining pattern of mammaglobin was compared with GCDFP-15 in the breast carcinomas, mammaglobin had higher sensitivity than GCDFP-15. During the diagnosis of breast carcinoma, it should be taken into consideration that the sensitivity of mammaglobin is better than that of GCDFP-15. Carcinoembryonic antigen (CEA) is a well-known tumour marker glycoprotein of 180 kDa. The polyclonal antibody reacts strongly and diffusely with ductal mammary carcinomas, lung and large intestine carcinomas; CEAD-14 clone reacts with a small subset of mammary carcinomas, usually high grade, which is useful in the evaluation of metastatic mammary carcinoma in the lung, liver, brain and lymph nodes; 13% of breast carcinomas are positive for CEAD-14, with a focal reaction, but diffuse in high-grade carcinomas. A negative CEAD-14 pulmo nary tumour is more likely to be metastasis and not a primitive lung tumor, which is positive for other specific markers (such as thyroid transcription factor-1, TTF1) [16-18].
Markers Of Prognosis and Response to Therapy
The most common immunohistochemical breast cancer prognostic and therapeutic markers used include: ER, HER2, Ki-67, PR, and p53. In addition, markers of angiogenesis and apoptosis are used.
Hormone receptors
Nowadays, immunohistochemical detection of ER and PR is part of the routine work-up of breast cancer, and in some cases of DCIS the presence of ERs is an indication for tamoxifen therapy. There are many scoring systems, and many studies have compared their ability to predict treatment response and correlations with outcome. The first scoring system counted the percentage of positive cells and ignored staining intensity. When we determine the proportion of positive stained cells, at least 1% is considered a hormonally treatable state. According to the International Breast Cancer Study Group scheme which is the basis of the most recent St Gallen treatment guidelines, breast cancer is divided into three groups based on the percentage of positive cells: responsive (10%), response uncertain (1%-9%), and nonresponsive (0%). In other words, a threshold of 1% positive cells indicates the option for hormonal therapy. These guidelines are widely followed in many countries from Europe and the United States, but they seem to be insufficient. Many users report results as an Allred score, which comprises both the percentage of positive cells and staining intensity [. A total score of 3 or more, corresponding to 1% to 10% positive cells, characterizes the lowest positive result and corresponds to the St Gallen endocrine response uncertain category in which case adjuvant hormone treatment can be recommended, but has an uncertain benefit. Immunohistochemical staining for ER in DCIS, without associated invasive lesions, has a role in estimating the potential positive effect of tamoxifen. The National Surgical Adjuvant Breast and Bowel Project Protocol B-24, in patients with DCIS treated with partial mastectomy and then irradiation, who received placebo or tamoxifen for five years showed a conclusive reduction in both ipsilateral and contralateral breast cancer in the adjuvant tamoxifen group.
HER-2/Neu expression
HER-2/neu was one of the first oncogenes studied in samples of invasive breast cancer and it is identified in 10%-20% of breast cancer patients. It is a marker for sensitivity to Herceptin (trastuzumab), and resistance to tamoxifen. Although Her-2/neu can be detected using many methods, only two are currently approved and recommended for its detection: IHC and fluorescence in situ hybridization (FISH). IHC evaluates overexpression of the receptor protein at the surface of the cells, while FISH evaluates the status of the HER2 gene in the nucleus. In most HER2-positive cancers, HER2 protein overexpression is the result of gene amplification, thus both methods should be highly correlated. Immunohistochemistry reactions for HER-2 aree scored by Hercept Test where 0 and 1+ scores are negative, 2+ is weakly positive and 3+ is positive. A positive HER-2 result consists of a uniform and intense membrane staining of more than 30% of tumour cells. Weakly positive or equivocal or 2+ cases should be tested for gene amplification by FISH. Selection of the best treatment, especially if the patient is a candidate for HER2-targeted therapy, depends on accurate laboratory results of the assessment of HER2 status.
Markers of apoptosis and cell proliferation
Ki-67 proliferation index, BCl-2, p53: Ki-67, a non-histone protein, involved in the early stepsof polymerase I-dependent ribosomal RNA synthesis is a predictive and prognostic marker in cancers and has been extensively studied. When Ki-67 level is above 10%-14%, breast cancer patients are defined as high-risk. According to the St. Gallen Consensus (2009), the Ki-67 index is useful for selecting patients with hormone receptor-positive breast cancers for the addition of chemotherapy to endocrine therapy. Thus, breast tumours are classified as low, intermediate, and highly proliferating according to a Ki-67 labelling index of under 15%, 16%-30%, and over 30%, respectively. Data from the Clinical Cancer Registry Regensburg showed that Ki-67 expression was associated with common histopathological parameters, especially grading and survival, but is an additional independent prognostic parameter for disease free survival and overall survival in breast cancer patients. The neoadjuvant setting is useful for analyzing the value of Ki67 as a predictive and prognostic tool.
Most studies investigating complete pathological response have identified a high Ki67 proliferation rate as a predictive factor for a higher rate of complete pathological response. However, it was found that patients in whom progression occurred had a higher proliferation rate than those who responded to neoadjuvant chemotherapy. This suggests a nonlinear effect of Ki67 on treatment response, Ki-67 expression has been used to determine the effects of different doses of tamoxifen on breast cancer proliferation. The change in Ki-67 expressions induced by lower doses of tamoxifen was comparable to that achieved with the standard dose, indicating that tamoxifen retains antiproliferative activity at low doses. Dowsett et al, in a small study, showed that a higher Ki-67 labelling index after two weeks of neoadjuvant therapy with tamoxifen was associated with shorter recurrence-free survival, whereas higher Ki67 expression at baseline was not. Another proliferation marker in tumour tissue is the Ki-S2 antibody. This antibody recognises a proliferation specific nuclear protein expressed exclusively in the cell cycle phase S, G2, and M. Therefore, actively proliferating cells that constitute a subset of the population recognized by Ki-67 were specifically labelled. Alterations in cell cycle regulation at the G1-S transition strongly influence breast cancer progression. Prognosis is probably indicated by the percentage of cells in S phase. Regarding the molecular breast cancers, high Ki-67 proliferation index can be used to classify triple negative breast cancer into subtypes with different prognoses or responses to treatment. For this purpose, the number of Ki-67 positive cells among the total is more than 30% of tumour cells. Weakly positive or equivocal or 2+ cases should be tested for gene amplification by FISH. Selection of the best treatment, especially if the patient is a candidate for HER2-targeted therapy, depends on accurate laboratory results of the assessment of HER2 status scientists have reported that a p53 mutation has no influence on the outcome and therefore, the value of p53 status is too weak to be recommended as a routine marker in clinical practice [19].
Angiogenesis markers
Tumor growth and metastasis are dependent on tumour angiogenesis and this complex process involves a delicate balance between angiogenic and antiangiogenic factors. Numerous studies have investigated the relationship between tumour angiogenesis, prognosis and response to antiangiogenic drugs. Analysis of these factors in tumour or serum of breast cancer patients by IHC or multiplex protein assay (FASTQuant® Microspot Assays) can improve diagnosis and prognosis of the disease. There is a large list regarding angiogenesis markers: Angiogenin, Ang2, keratinocyte growth factor (KGF), fibroblast growth factor basic, intercellular adhesion molecule (ICAM)-1, platelet-derived growth factor- BB and the vascular endothelial growth factor family. About these markers, it has been observed that patients with breast cancer exhibited high levels, as well as high serum levels when compared to patients with benign breast diseases. When some of these markers were evaluated either in tumour or serum in breast cancer patients, they showed an association with standard clinical parameters, ER status and intratumoural microvessel density of tumours.The commonly used method to determine angiogenesis is counting intratumoral blood vessels (MVD) stained with factor Ⅷ related antigen or anti CD31 or CD34 using light microscopy. The main difficulty is the significant variability in density between different areas of tumor and among observers. Counting newly formed stained microvessels is a useful tool in the early detection of metastatic potential and in the selection of patients for whom anti- angiogenesis drugs might be beneficial.
The reactivity level of CD34 antigen was assessed by IHC in all types of invasive ductal breast cancer and its level seems to be a useful predictor for the development of local lymph node metastasis and can indicate the benefit of antiangiogenic treatment Anti-angiogenic drugs have been approved recently for the therapy of advanced cancers, including breast cancers. These drugs, alone or in combination with chemotherapy, can improve overall or progression free survival in cancer patients. Unfortunately, the lack of validated biomarkers to allow the selection of patients who are most likely to benefit from targeted drugs such as bevacizumab, sunitinib, sorafenib and pazopanib, limits the rational use of these drugs and the ability to determine optimal dose and scheduling of these drugs. Most of the biological and clinical activity of the antiangiogenic drugs currently approved for cancer therapy is against the VEGF-related pathways. The VEGF system is part of the platelet-derived growth factor gene family and interacts with its specific receptors; VEGFR-1 (flt-1) and VEGFR-2 (flt-2) for VEGF-A, a very potent angiogenic growth factor. VEGF-B, interacting with VEGFR-1, seems to have an important role in the maintenance of existing vessels, but this protein is not well studied. VEGF A and B, their receptors VEGFR-1 and 2 are expressed in a variety of normal cells, and over expression has been described in malignant tumors. There are different techniques used to assess VEGF-A, IHC being the most convenient in routine diagnosis as well as research, as it allows single cell analysis combined with morphology. The results are currently based on visual examination of IHC-stained tissue slides and several different scoring systems have been used. The prognostic importance of VEGF in invasive breast cancer is associated with tumour stage and ER status and inversely correlated with tumour grade and measurement of tumour VEGF, as an indicator of angiogenesis, which is more reliable prognostically than measurement of micro vessel density or serum VEGF. In addition, tamoxifen treatment was associated with higher circulating and platelet-derived VEGF levels [20,21].
Conclusion
IHC has become an integral part of the pathology laboratory It is a more mature technology and accessible to most pathology laboratories. IHC can be used for diagnostic issues, estimating prognosis or predicting response to therapy. The best approach in the use of immunohistochemical markers is to combine them with the examination of standard hematoxylin-eosin slides and use of markers. O Reactivity for steroid receptors was observed to be decreasing with increasing grade. Grade III tumors were more negative as compared to grade 1 and grade II tumor. This showed the same inverse relation between receptor status and increasing tumor grade.
Conflict of Interest
Author claims there is no conflict of interest.
Acknowledgement
We thank Cosmin Borcea, Romania, for harvesting the mistletoe plants.
References
- Yadav R, Sen R, Preeti (2012) Role of receptors in breast cancer. IJABR 2(4): 561-571.
- Siddiqui MS, Kayani N, Sulaiman S, et al. (2000) Breast carcinoma in Pakistani females; a morphological study of 572 breast specimens. J Pak Med Assoc 50(6): 174-177.
- Ahmedin Jemal, Rebecca Siegel, Elizabeth Ward, Taylor Murray, Jiaquan Xu, et al. (2007) Cancer statistics. CA cancer J Clin 57(1): 43-66.
- P I Borgen, GY Wong, V Vlamis, C Potter, B Hoffmann, et al. (1992) Current management of male breast cancer. A review of 104 cases. Ann Surg 215(5): 451-457.
- Fentiman IS, Fourquet A, Hortobagyi GN (2006) Male breast cancer. Lancet 367(9510): 595-604.
- Burstein HJ, Harris JR, Morrow M (2011) Malignant tumors of the breast. In: De Vita VT, Lawrence TS, editors. Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia: Lippincott Williams & Wilkins 2: 1401-1446.
- Jatoi I, Miller AB (2003) Why is breast cancer mortality declining? Lancet Oncol 4(4):251-254.
- Muhammad Ashraf Sharif, Nadira Mamoon, Sajid Mushtaq, Muhammad Tahir Khadim (2009) Morphological profile and association of Her–2 Neu with prognostic markers in breast carcinoma in Northern Pakistan. J Coll Phy Surg Pak 19(2):99-103.
- Rastelli F, Crispino S (2008) Factors predictive response to hormone therapy in breast cancer. Tumouri 94(3): 370-383.
- Obeagu EI, Obeagu GU (2024) Breast Cancer: A Review of Risk Factors and Diagnosis. Medicine 103(3): e36905.
- Bodewes FTH, van Asselt AA, Dorrius MD, Greuter MJW, de Bock GH (2022) Mammographic Breast Density and the Risk of
- Breast Cancer: A Systematic Review and Meta-Analysis. Breast 66: 62-68.
- Mokhtary A, Karakatsanis A, Valachis A (2021) Mammographic Density Changes over Time and Breast Cancer Risk: A Systematic Review and Meta-Analysis. Cancers 13(19): 4805.
- Mann RM, Athanasiou A, Baltzer PAT, Camps-Herrero J, Clauser P, et al. (2022) Breast Cancer Screening in Women with Extremely Dense Breasts Recommendations of the European Society of Breast Imaging (EUSOBI). Eur Radiol 32(6): 4036-4045.
- Lubinski J, Kotsopoulos J, Moller P, Pal T, Eisen A, et al. (2024) MRI Surveillance and Breast Cancer Mortality inWomenWith BRCA1 and BRCA2 Sequence Variations. JAMA Oncol 10(4): 493-499.
- Trentham-Dietz A, Newcomb PA, Nichols HB, Hampton JM (2007) Breast Cancer Risk Factors and Second Primary Malignancies among Women with Breast Cancer. Breast Cancer Res. Treat 105(2): 195-207.
- Brewer HR, Jones, ME, Schoemaker MJ, Ashworth A, Swerdlow AJ (2017) Family History and Risk of Breast Cancer: An Analysis Accounting for Family Structure. Breast Cancer Res. Treat 165: 193-200.
- Liu L, Hao X, Song Z, Zhi X, Zhang S, Zhang J (2021) Correlation between Family History and Characteristics of Breast Cancer. Sci. Rep 11: 6360.
- Collins A, Politopoulos I (2011) The Genetics of Breast Cancer: Risk Factors for Disease. Appl Clin Genet 4: 11-19.
- Baretta Z, Mocellin S, Goldin E, Olopade OI, Huo D (2016) Effect of BRCA Germline Mutations on Breast Cancer Prognosis: A Systematic Review and Meta-Analysis. Medicine 95: e4975.
- Gorodetska I, Kozeretska I, Dubrovska A (2019) BRCA Genes: The Role in Genome Stability, Cancer Stemness and Therapy Resistance. J Cancer 10: 2109-2127.






























