Abstract
Synthesis and cycloaddition reactions of some N-Substituted-4-(Trifluoromethyl)-C-phenyl nitrones using mechanochemical procedure has been reported. This solvent free procedure with the addition of sodium bicarbonate showed a remarkable faster reaction rate and enhanced workup process as well as yields of the reported fluoronitrones and fluoro cycloadducts compared to microwave and coventional cycloaddition procedures. Solid phase mechanochemical procedure is thus found to be more suitable than other greener techniques. The present study reports synthesis of few N-Substituted-4- (Trifluoromethyl)-C-phenyl nitrones and their cycloaddition reactions with activated alkenes and electron deficient alkynes. Few synthesized new fluoro cycloadducts found to exhibit significant anticancer activities too.
Keywords:Mechanochemistry; Fluoro nitrones; Intermolecular cycloaddition reaction; Fluoro Isoxazolidine; Isoxazolines; Anticancer activity
Introduction
Perhaps nitrones are one of the most important reaction intermediates in synthetic organic chemistry as far as the constructions of 5-membered oxygen-nitrogen heterocyclic molecules are concerned [1]. The synthesis, isolation and cycloaddition reactions of nitrones with activated olefins/ alkynes are always a point of great interest not only because of the synthetic potentials of cycloadducts (isoxazolidine & isoxazoline derivatives) but also for their significant biological activities [2-4]. Many new approaches in this chemistry are widely known but greener synthesis and cycloaddition reactions of fluoro nitrones and fluoro cycloadducts have not reported too many [5-7]. Due to versatile applications of fluoro nitrones, they are of special interest in organic synthesis. The introduction of fluorine atoms or atoms in a particular position of an organic molecule can significantly change the stability and the bioactivity of the molecule [8]. Three important factors or properties of fluorine atoms are expected to play the crucial role for this significant change and bioactivity. These are the high electronegativity of the fluorine, the strong C-F bonding and the same size of the halogen and hydrogen atoms respectively. Hence fluorinated isoxazolidinyl and isoxazoline derivatives are point of attraction not only to synthetic organic chemists but also in the field of pharmacology as well [9]. The reported works in literature reveals that fluorinated compounds exhibit excellent bioactive properties and may be suitably used as main ingredients in medicines (letermovir, pexidartinib, celecoxib and osilodrostat) [10], drug developments [10,11] including new cancer drugs [12] as well as in the agrochemical industries [5]. In the pharmaceutical industry, fluorinated heterocycles are getting more importance day by day compared to common heterocycles due to its low polarizability and high lipophilicity which increases relative metabolic stability and improves the bioavailability of the new heterocyclic compounds compared to its hydrocarbon analogues [7].
Our group has reported already many environments friendly (green chemistry) procedures in nitrone cycloaddition reactions and further applications of cycloadducts including peptide synthesis [13-20]. CF3 as substituent plays vital role in synthesis and also, we have found its many applications of CF3 substituted isoxazolidine and isoxazoline derivatives [10,11,12], we report in this communication synthesis of some important new stable fluoro cycloadducts (isoxazolidine & isoxazoline derivatives) using mechanochemical procedure (Scheme 1; Tables 1 & 2) [21,22] and also significant anticancer activities of few new fluoro cycloadducts. We have compared the synthetic of fluoro cycloadducts studied in microwave and conventional methodologies and found significant acceleration in reaction rate and yield of the cycloadducts in mechanochemical procedure. Mechanochemical procedures involving ball-milling technique are very popular in today’s scenario for its many environmentally friendly aspects and hence attracted the attention of mainly synthetic organic chemists [21]. Conducting organic synthesis under environment friendly conditions is always a challenge therefore we have conducted ball-milling technique as a lucrative methodology in our synthesis. After utilizing almost all the greener procedures and reported the results, we would recommend “mechanochemical procedure” as one of the best procedures for future organic chemists [22]. Like majority of usual nitrones, fluoro nitrones synthesized in our laboratory are stable (melting points ranges between 680C to 850C, therefore utilization of these nitrones in cycloadditions reaction becomes very easy. Moreover, a few newly synthesized fluoro-cycloadducts found to exhibit significant anticancer activities. Hence, these fluoro- nitrones may be regarded as an important precursor for the synthesis of new anticancer drugs which could attract organic and medicinal chemists in research.

aReaction conditions: nitrone (1 mmol), dipolarophiles (1 equivalent), ball-milling, (40-70Hz)
bAll products were characterized by IR,1H NMR, 13C NMR and MS sp ectral data.
cIsolated yield after purification. Figures in parentheses indicate yields obtained under MWI.

aReaction conditions: nitrone (1 mmol), dipolarophiles (1 equivalent), ball-milling, (40-70Hz)
bAll products were characterized by IR,1H NMR, 13C NMR and MS sp ectral data.
cIsolated yield after purification. Figures in parentheses indicate yields obtained under MWI.
Results & discussion
In our new endeavor of N-substituted-4-(Trifluoromethyl)- C-phenyl nitrone (1) synthesis, we have conducted the reactions taking one equivalent each of nitrone 1 (a, b & c) and N- substituted hydroxylamine hydrochloride along with one equivalent of sodium bicarbonate for the synthesis of fluoro nitrones 1 (Scheme 1; R = Me, Ph, Bz). The synthesized fluoro nitrones have been used for cycloaddition reactions without further purification. 4-Trifluoromethyl benzaldehyde derived fluoro nitrones were stable (m.p: 680C to 850C) and cycloaddition reactions were performed with dipolarophiles in 1:1 ratio. In ball-milling procedure, usually heat and slight pressure are developed in the reaction vessel in our study, best results have been obtained when 1:1 ratio of starting materials were used but we have also observed incomplete conversion to molecules when tried with different (1:2 and 2:1) ratios of starting materials. We added sodium bicarbonate in the reaction mixture in the synthesis of fluoro nitrones because we had observed that it could activate the N- substituted hydroxylamines. The probable reason could be due to the addition of sodium bicarbonate the reaction mixture becomes faintly alkaline and the liberated HCl is neutralized. We have also observed that if we conduct the ball-milling procedure without adding sodium bicarbonate, the reaction leads to develop a paste, or gum and ingredients do not mix well. Solid material also helps in free flow proceedings in ball-milling techniques. This was our purpose to use solid sodium bicarbonate for more efficiency in the process and to obtain the best results.
1H NMR spectrum of crude products has been also studied at a lower frequency (10-20Hz) but we didn’t see any indications of developments of our target molecules. May be at a lower frequency the reaction rates were very slow and needed almost 45 minutes for the completion of the reaction and in few reactions, the ingredients 4-Trifluoromethylbenzalaldehyde and hydroxylamines were also present along with the crude products. It could be due to lesser amount of energy per impact is involved and ball-milling procedure with an interval develops lower conversion rate from starting materials to products. In mechanochemistry, the development of nitrones were fast and therefore it becomes easy for conducting in situ cycloaddition reactions if researchers so desire. But in our study, we wanted to study the stability and characterize the fluoro nitrones, therefore we h conducted intermolecular cycloaddition reactions of the fluoro nitrones with various activated double bonded dipolarophiles (maleimides) as well as electron deficient dipolarophiles (alkynes) respectively. After successful study using various reaction conditions and trials, finally we decided to run the cycloaddition reactions in ball-milling process at a frequency of 40-70Hz and found excellent development of fluoro cycloadducts in 5-7 minutes of ball-milling. We have also studied these reactions using acetonitrile (1 ml) as solvent (polar solvent) in ball-milling procedure and found slight less developments and lower yields of fluoro cycloadducts. The yields of fluoro cycloadducts using mechanochemical procedure were also compared (Table 1 & 2) with microwave technology (MWI) under solvent-free conditions [23,24]. We found lower yields of fluoro nitrones and fluoro cycloadducts in MWI. Microwave methodology, for the synthesis of fluoro nitrones and fluoro cycloadducts required high temperature (120-1300C) and average time required for the synthesis of fluoro nitrones and fluoro cycloadducts were found to be 40 – 60 minutes respectively. In addition, the complete conversion of the starting materials to our target molecules was not satisfactory. Probably, it could be due to the degradation of N-substitutedhydroxylaminehydrochlorides. Continuing our efforts, we have also studied DMF as solvent in MWI methodology for cycloaddition reactions, but yields (fluoro isoxazolidine & isoxazolines) were found to be 75-80% which was regarded as poor in comparison with ball-milling process.
1H NMR spectroscopy technique was mainly used for the confirmation of the structures of newly synthesized molecules (fluoro isoxazolidine & isoxazoline derivatives) and fluoro nitrones [25- 27]. In addition, also 13C NMR, MS & IR spectroscopic techniques have been used successfully. 1H NMR spectrum of the fluoro isoxazolidine and isoxazoline derivatives (2-7 & 8-13) reveals that the structures are expected to be symmetrical in nature. Nitrone 1(a, b &c) exists exclusively in Z configuration and syn cycloadducts are believed to develop from Z nitrone via an exo transition state geometry. The configurations of C3, C4, C5 protons of the fluoro cycloadducts are expected to be syn in nature because the coupling constant (J ~ 2.00-3.00Hz, for C4-C5 & J ~ 2.00-3.00 Hz, for C3-C4) values are in good agreement with reported values in our previously published research articles in literature [25-28] as well. Therefore, we may assume from these J values that the dipolarophiles with syn configuration produced syn fluoro cycloadducts and the addition of fluoro nitrones to electron rich alkenes (maleimides) are stereo specifically syn in nature. In maleimide cycloadducts, 3-H, 4-H protons are syn orientated and are enantioselective in nature. The coupling constant (J) values of 3-H, 4-H protons (J3,4~ 2.00 Hz) [25,26,29] are also in good agreement in favor of cis oriented structure. Due to the absence of 4-H protons, in case of fluoro isoxazoline derivatives, exact geometry could not be determined (Figure 1).
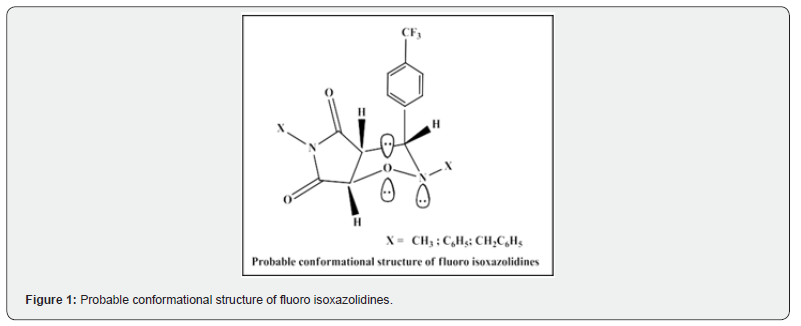
The probable conformational structure indicates the substituents at the C-3, C-4 & C-5 positions tries to have equatorial positions while all the H-atoms at these positions are axial respectively. We have observed and obtained expected fragmentation peaks in the mass spectrum including molecular ion (M+) and base peaks (BP) in all the fluoro isoxazolidine derivatives. In case of fluoro isoxazoline derivatives in addition to M+ other prominent peaks are also obtained. This is due to the fragmentations of COOCH3, Ph and COOH groups which subsequently undergoes development of aziridine derivatives. Though we have not studied theoretical concepts and studies on fluoro nitrones and fluorocycloadducts but recent studies by B. Rachid and coworkers [30] reveals that using “Conceptual Density Function Theory” (CDFT) this kind of one step mechanism is under kinetic control and the potential energy surface of the reaction pathways can be mapped. The corresponding Gibbs free energy may reveal that the exo fluoro cycloadducts are kinetically and thermodynamically more favored than endo cycloadducts. We have reported in favour of exo fluoro cycloadducts in this communication following 1H NMR spectroscopy. At present we are conducting mechanochemical synthesis of N-Methyl-4-(trimethylsilyl)-C-phenyl nitrone derived from 4-trimethylsilyl benzaldehyde and cycloaddition reactions. Also, we shall compare the effect of trimethylsilyl group at the 4-position of benzaldehyde molecule with trifluoro methyl benzaldehyde molecule.
Anticancer study
Majority of the newly synthesized isoxazolidine & isoxazoline derivatives in recent years are screened for cancer studies [12,31] as their probability to act as anticancer drugs are very high. Cytotoxicity of the compounds were determined based on measurement of in vitro growth inhibition of tumor cell lines [31,32], A549 derived from human alveolar adenocarcinoma epithelial cells (ATCC No.CCL-180), HeLa derived from human cervical cancer cells (ATCC No. CCL-14), MDA-MB-167 derived from human breast adenocarcinoma cells (ATCC No. HTB-35) and MCF7 derived from human breast adenocarcinoma cells (ATCC No. HTB-40) using the MTT assay. The IC50 values (50% inhibitory concentration in μM) are expressed as the average of two independent experiments The effect of cycloadducts [2-13] on the growth of cancer cell lines were determined following the general procedure used by the National Cancer Institute for vitro anticancer drug study. The procedure uses the protein-binding dye Sulphorhodamine B for the estimation of cell growth [33,34]. In due course of time, the growth of the cells was counted (95 cells per well in 100 mL medium) in 90 microtitre plates. The study has been conducted keeping the cells for incubation for 40 hrs at 200C. The experimental set-up of three different wells was conducted where the cells were kept for 36 hrs. This was followed by reacting the cells with 30% cold (5-100C) TCA. It was left for 2 hrs at 200C and then washed and dried in air. All the cells were stained with Sulphorhodamine B dye. The dye was dissolved in tris-buffer solution. The plates under study were taken in shaker and kept for 20-30 minutes. The cell growth was calculated using optical density (OD) study and the results were reported in terms of IC50 values. Doxorubicin was considered as standard reference. From the study of IC50 values, it has been found that six (6) newly synthesized cycloadducts showed significant cytotoxicity against human alveolar adenocarcinoma epithelial cells, human cervical cancer cells, human breast adenocarcinoma cells and human breast adenocarcinoma cells respectively. Among all the tested cycloadducts [2-13], 2, 4 & 7 showed comparatively more potent IC50 value against (ATCC No.CCL-180), HeLa derived from human cervical cancer cells (ATCC No. CCL-14), MDA-MB-167 derived from human breast adenocarcinoma cells (ATCC No. HTB-35) and MCF7 derived from human breast adenocarcinoma cells (ATCC No. HTB-40) as compared to other fluoro cycloadducts. Based upon the study, two most potent fluoro isoxazolidine derivatives 2 and 4 were taken for cell cycle analysis. Cell cycle analysis is going on at present (Table 3).

Experimental
Unless otherwise stated, all reagents were obtained from commercial sources and used without further purification. Progress of all the reactions were monitored by TLC using 0.25 mm silica gel plates (Merck 60F254, UV indicator). Column chromatography was performed with silica gel (E.Merck, Germany) with 60–200 mesh. All other reagents and solvents were purified before starting reactions or column chromatography. 1H NMR spectra were recorded on a Bruker DRX 300 (300 Hz) spectrometer at an ambient temperature.13C NMR spectra were recorded on a Bruker DRX 300 (75 Hz) spectrometer at ambient temperature. The coupling constants (J) were expressed in Hz. Infra-red spectra were recorded on a Perkin-Elmer RX 1-881 machine as a film or KBr pellets. Mass-spectrometry data was recorded using a Joel SX- 102 (FAB). The ball mill used was a Retsch MM500 mixer mill digital GmbH,42781Haan, Germany. Reactions were carried out using stainless steel jars from Retsch. Milling balls were purchased from Germany. General procedure 1– General procedure for the synthesis of N-substituted-4-(Trifluoromethyl)- C- phenyl nitrone (1) in ball-milling procedure N-methyl hydroxylamine hydrochloride (1 mmol), 4-(Trifluoromethyl) benzaldehyde (1 equivalent), and NaHCO3 (1.0 equivalent) were mixed together and ball-milled at 40 Hz for 5 min in a 25 mL steel vessel and 15 mm diameter balls. After the completion of reaction, the reaction mixture was taken in CH2Cl2. It was filtered on cotton for the removal of NaCl. The filtrate was evaporated under vacuum to afford N-methyl-4-(Trifluoromethyl)- C- phenyl nitrone 1a as yellowish white crystalline solid with high purity (90%, m.p; 740 C). Same methodology was used for the synthesis of other fluoro nitrones (1b & 1c)
1a: N-methyl-4-(Trifluoromethyl)-C- phenyl nitrone
Spectroscopic data for nitrone 1a: White crystals. M.P: 740C. UV λmax 235 nm; IR (KBr): υmax 3030 (m), 2230 (m), 1680 (m), 1620 (s), 1440 (m), 1150 (m), 786 (s) cm-1. 1H NMR (CDCl3): δ 7.58-7.20 (m, 4H, phenyl protons), 2.34 (s, 3H, CH3), 1.55 (s, 1H,- CH=N+); 13CNMR (CDCl3): δ 142.12 (CH=N+), 134.82, 134.33, 134.15, 133.97 (phenyl carbons), 30.55 (CH3), 24.50 (CF3); Calculated for C9H8F3ON: C 52.42, H 3.88, N 6.79%; Found : C 52.37, H 3.80, N 6.64%.
1b: N-benzyl-4-(Trifluoromethyl)-C- phenyl nitrone
Spectroscopic data for nitrone 1b: White crystals. M.P: 820C. UV λmax 234nm; IR (KBr): υmax 3010 (m), 2215 (m), 1670 (m), 1625 (s), 1430 (m), 782 (s) cm-1. 1H NMR (CDCl3): δ 7.90-7.40 (m, 5H), 7.39-6.96 (m, 4H), 3.36 (s, 2H, CH2Ph), 1.48 (s, 1H,- CH=N+); 13CNMR (CDCl3): δ 143.00 (CH=N+), 134.70, 133.88, 133.50, 133.20 (C-phenyl carbons), 130.66, 130.45, 130.30, 129.85, 128.73 (phenyl carbons of benzyl group), 26.00 (CF3), 24.15 (CH2Ph) ; Calculated for C15H12F3ON: C 64.28, H 4.28, N 5.00%; Found : C 64.17, H 4.15, N 4.86%.
1c: N-phenyl-4-(Trifluoromethyl)-C- phenyl nitrone
Spectroscopic data for nitrone 1c: White crystals. M.P: 880C. UV λmax 238nm; IR (KBr): υmax 3036 (m), 2220 (m), 1660 (m), 1635 (s), 1436 (m), 1200 (m), 780 (s) cm-1. 1H NMR (CDCl3): δ 7.82-7.20 (m, 5H & m, 4H merged signals of phenyl ring protons), 1.78 (s, 1H, -CH=N+); 13CNMR (CDCl3): δ141.80 (CH=N+), 137.60, 137.45, 137.20, 136.90, 134.55, 134.40, 134. 14, 133.90, 133.75 (two phenyl carbons),26.50 (CF3); Calculated for C14H10F3ON: C 62.04, H 3.73, N 5.22%; Found: C 61.90, H 3.66, N 5.16%. 2 General procedure II–Mechanochemical synthesis of fluoro isoxazolidine & isoxazolidine derivatives from N-substituted-4-(Trifluoromethyl)- C- phenyl nitrone (1) (Table 1; entry 1). N-methyl-4-(Trifluoromethyl)- C-phenyl nitrone (1a; 1 equivalent), N-methyl maleimide (1 equivalent) and NaHCO3 (1 equivalent) were mixed and ball-milled at 40 Hz for 5 min in a 25 mL steel vessel and 15 mm diameter balls. After the completion of reaction, the reaction mixture was taken in CH2Cl2. It was filtered on cotton for the removal of NaCl. The filtrate was evaporated under vacuum to afford crude fluoro isoxazolidine derivative as white crystals (95%). The crude product was directly charged on silica gel column and eluted with a mixture of ethyl acetate: n-hexane (1:8) resulting pure isoxazolidine 2 (entry 1, Table 1, Scheme 1). Same methodology was adopted for the synthesis of other fluoro isoxazolidine & isoxazoline derivatives [2-13].
(3S)-2-methyl-3-(4-trifluoromethyl)-dihydro-5-methyl- 2H-pyrrolo[3,4-d]isoxazole-4,6(5H,6a-H)-dione, 2
White crystals. M.P 1260C; Yield 95%; Rf = 0.62; IR (KBr): υmax 3310 (m), 2940 (m), 2830 (m), 1760 (s), 1670 (s), 1485 (m), 1320 (m), 780 (s) cm-1; 1H NMR (CDCl3): δ 7.76 – 7.28 (m, 4H, aromatic protons), 6.38 (d, 1H, J = 2Hz, C5H), 6.34 (d, 1H, J = 2 Hz, C3H), 3.40 ( s, 6H, 2XCH3), 2.24 (dd, 1H, J = 2.00, 2.00 Hz, C4H); 13C NMR (CDCl3): δ 169.00 (carbonyl carbons), 138.46, 138.10, 137.20, 131.42, 130.80. 128.15 (aromatic carbons carbons), 85.40 (C5), 76.82 (C3), 62.20 (C4), 30.20, 28.00 (CH3 carbons), 24.50 (CF3); FAB-MS: m/z 314 (M+), 245, 169 (BP, 100%), 145, 69; Calculated for C14H13F3O3N2: C 53.48, H 4.16, N 8.91%; Found: C 53.34, H 4.06, N 8.70%.
(3S)-2-phenyl-3-(4-trifluoromethyl)-dihydro-5-methyl- 2H-pyrrolo[3,4-d]isoxazole-4,6(5H,6a-H)-dione, 3
White solid. M.P 1400C; Yield 92%; Rf = 0.60; IR (KBr): υmax 3236 (m), 2980 (m), 2775 (m), 1760 (s), 1685 (s), 1440 (s), 1262 (m), 784 (s) cm-1; 1H NMR (CDCl3): δ 7.68 – 7.20 (m, 5H; m, 4H merged signals of phenyl ring protons), 6.42 (d, 1H, J = 2.10 Hz, C5H), 6.36 (d, 1H, J = 2.10 Hz, C3H), 3.36 (s, 3H, N-CH3), 2.34 (dd, 1H, J = 2.00, 2.00 Hz, C4H); 13C NMR (CDCl3): δ 170.50, 170.00 (carbonyl carbons), 134.60, 134.30, 134.20, 134.00, 130. 40, 130.20, 129.00, 128.50, 128.30, 127.40 (phenyl carbons), 85.50 (C5), 77.50 (C3), 61.00 (C4), 28.30 (N-CH3), 23.50 (CF3); FAB-MS: m/z 376 (M+),361, 307, 299, 231 (BP, 100%), 231, 77, 69; Calculated for C19H15F3O3N2: C 60.14, H 3.98, N 7.38%; Found: C 60.02, H 3.85, N 7.27%.
(3S)-2-methyl-3-(4-trifluoromethyl)-dihydro-5-phenyl- 2H-pyrrolo[3,4-d]isoxazole-4,6(5H,6a-H)-dione, 4
White crystals. M.P 1360C; Yield 91%; Rf = 0.58; IR (KBr): υmax 3230 (m), 2975 (m), 2770 (m), 1760 (s), 1680 (s), 1440 (s), 1260 (m), 780 (s) cm-1; 1H NMR (CDCl3): δ 7.68 – 7.20 (m, 5H; m, 4H merged signals of phenyl ring protons), 6.42 (d, 1H, J = 2.10 Hz, C5H), 6.36 (d, 1H, J = 2.10 Hz, C3H), 3.36 (s, 3H, N-CH3), 2.34 (dd, 1H, J = 2.00, 2.00 Hz, C4H); 13C NMR (CDCl3): δ 170.50, 170.00 (carbonyl carbons), 134.30, 134.20, 134.00, 130. 40, 130.20, 129.00, 128.50, 128.30, 127.40 (phenyl carbons), 85.00 (C5), 76.50 (C3), 60.00 (C4), 28.50 (N-CH3), 23.00 (CF3); FAB-MS: m/z 376 (M+),361, 307, 299, 231 (BP, 100%), 231, 77, 69; Calculated for C19H15F3O3N2: C 60.14, H 3.98, N 7.38%; Found: C 60.05, H 3.70, N 7.20%.
(3S)-2-methyl-3-(4-trifluoromethyl)-dihydro-5-benzyl- 2H-pyrrolo[3,4-d]isoxazole-4,6(5H,6a-H)-dione, 5
White solid. Yield 92%; Rf= 0.60; IR (KBr): υmax 3310(m), 2910 (m), 2840 (m), 1765 (s), 1678 (s), 1460 (s), 1330 (m), 785 (s) cm-1;1H NMR (CDCl3): δ 7.67 – 7.23 (m, 5H & m, 4H,merged phenyl ring protons), 6.42 (d, 1H, J = 2.00 Hz, C5H), 6.24 (d, 1H, J = 2.00 Hz, C3H), 3.38 (s, 3H, N-CH3), 2.38 (dd, 1H, J = 2.00, 2.00 Hz, C4H), 1.20 (s,2H, CH2); 13C NMR (CDCl3): δ 170.00, 168.20 (carbonyl carbons), 138.00, 137.80, 137.20, 136.80, 130.50, 130.10, 129.80, 127.60, 126.30 (phenyl carbons), 80.10 (C5), 77.20 (C3), 60.20 (C4), 28.50 (N-CH3), 25.00 (CF3), 22.50 (CH2); FAB-MS: m/z 390 (M+),284, 245 (BP, 100%), 145, 106, 91, 77; Calculated for C20H17O3F3N2 : C 61.51%, H 4.39%, N 7.18%. Found C 61.32%, H 4.26%, N 7.04%.
(3S)-2-phenyl-3-(4-trifluoromethyl)-dihydro-5-phenyl- 2H-pyrrolo[3,4-d]isoxazole-4,6(5H,6a- H)-dione, 6
Yellow solid. M.P 1170C; Yield 91%; Rf = 0.66; IR (KBr): υmax 3250 (m), 2960 (m), 2835 (m), 1760 (s), 1685 (s), 1460 (s), 1360 (m), 782 (s) cm-1; 1H NMR (CDCl3): δ 7.68 – 7.20 (m, 2x5H; m, 4H, merged phenyl ring protons), 6.42 (d, 1H, J = 2.00 Hz, C5H), 6.24 (d, 1H, J = 2.00 Hz, C3H), 2.34 (dd, 1H, J = 2.00, 2.00 Hz, C4H); 13C NMR (CDCl3): δ 170.20, 169.50 (carbonyl carbons), 135.90, 135.70, 135.30, 135.00, 134.80, 134.00, 133.70, 133.45, 133.20, 132.70, 131.60, 131.30, 130.20, 129.50, 128.40, 128.10 (phenyl ring carbons), 82.60 (C5), 75.60 (C3), 55.50 (C4), 24.00 (CF3); FAB- MS: m/z 441 (M+), 364, 287, 256 (BP,100%), 145, 77; Calculated for C24H17F3O3N2: C 65.02, H 3.88, N 6.35%; Found : C 64.90, H 3.66, N 6.13%.
(3S)-2-benzyl-3-(4-trifluoromethyl)-dihydro-5-phenyl- 2H-pyrrolo[3,4-d]isoxazole-4,6(5H,6a-H)-dione, 7
Brown crystals. M.P 1080C; Yield 91%; Rf = 0.60; IR (KBr): υmax 3240 (m), 2940 (m), 2830 (m), 1760 (s), 1680 (s), 1462 (s), 1366 (m), 786 (s) cm-1; 1H NMR (CDCl3): δ 7.76 – 7.20 (m, 2x5H; m, 4H, merged phenyl ring protons), 6.40 (d, 1H, J = 2.00 Hz, C5H), 6.32 (d, 1H, J = 2.00 Hz, C3H), 2.34 (dd, 1H, J = 2.00, 2.00 Hz, C4H), 1.24 (s, 2H, CH2); 13C NMR (CDCl3): δ 171.35, 168.50 (carbonyl carbons), 136.88, 135.80, 135.30, 135.10, 134.80, 134.20, 133.70, 133.20, 133.00, 132.40, 131.90, 131.30, 130.20, 128.560, 128.40, 128.00 (phenyl ring carbons), 80.50 (C5), 74.60 (C3), 57.50 (C4), 26.00 (CH2), 24.00 (CF3); FAB-MS: m/z 455 (M+), 364, 287, 219 (BP,100%), 145, 91, 77; Calculated for C25H19F3O3N2: C 65.93, H 4.18, N 6.15%; Found: C 65.75, H 4.06, N 6.05%.
(S)-methyl-2-methyl-3-(4-trifluoromethyl)-2,3-dihydro- 5-phenylisoxazole-4-carboxylate, 8
Red thick liquid. Yield 91%; Rf = 0.70; IR (KBr): υmax 3380 (m), 2250 (m), 1760 (s), 1700 (s), 1680 (s),14820(s),1210 (s), 780 (s) cm-1; 1H NMR (CDCl3): δ 7.80 – 7.30 (m, 5H), 7.30-7.20 (m, 4H), 3.88 (s, 3H, -COOCH3), 2.64 (s, 3H, N-CH3), 1.26 (s, 1H, C3H); 13C NMR (CDCl3): δ 170.00 (-COOCH3), 138.40, 138.00, 137.40, 137.10, 136.40, 136.30, 135.70, 135.20 (phenyl carbons), 90.00 (C5), 79.40 (C3), 63.00 (C4), 30.00 (-COOCH3), 26.20 (NCH3), 24.00 (CF3); FAB - MS (m/z): 363 (M+), 304, 286, 218 (BP), 145, 77, 59; Calculated for C19H16F3O3N: C 62.27, H 4.40, N 3.82%; Found: C 62.13, H 4.10, N 3.66%.
(S)-dimethyl-2-methyl-3-(4-trifluoromethyl)-2,3-dihydroisoxazole- 4,5-dicarboxylate, 9
Red liquid. Yield 92%; Rf = 0.60; IR (KBr): υmax 3290 (m), 2180 (m), 1740 (s), 1670 (s), 1620 (s), 1260 (s), 870 (m), 776 (s) cm-1; 1H NMR (CDCl3): δ 7.76 – 7.24 (m, 4H, phenyl protons), 3.84 (s, 6H, 2X -COOCH3), 1.84 (s, 3H, N-CH3), 1.28 (s, 1H, C3H); 13C NMR (CDCl3): δ 172.00, 170.00 (-COOCH3, carbonyl carbons of the ester group), 134.00, 133.40, 130.00, 129.50, 79.00 (C5H), 66.50 (C3H), 54.50 (C4H), 32.00 (N-CH3), 26.50 (CF3); FAB - MS (m/z): 345 (M+), 330, 296, 200 (BP), 145, 59; Calculated for C15H14F3O5N: C 51.72, H 4.02, N 4.12%; Found: C 51.60, H 3.90, N 3.97%.
(S)-2-methyl-3-(3-trifluoromethyl)-2,3-dihydroisoxazole- 4,5-dicarboxylic acid, 10
Colourless thick liquid. Yield 91%; Rf = 0.64; IR (KBr): υmax 3180 (m), 2990 (br), 1760 (s), 1485 (s), 1360 (s), 1210 (s), 790 (s) cm-1; 1H NMR (CDCl3): δ 10.00 (s, 2H, 2XCOOH), 7.68 – 7.40 (m, 4H, phenyl protons), 2.36 (s, 1H, C3H), 1.24 (s,3H, N-CH3); 13C NMR (CDCl3): δ 173.20, 172.50 (carboxyl carbons), 138.50, 137.00, 135.00, 134.50, 130. 00 (phenyl carbons), 79.50 (C5), 56.00 (C3), 46.80 (C4), 28.00 (N-CH3 carbon), 25.00 (CF3); FAB - MS (m/z): 317 (M+), 302, 272, 172 (BP), 145, 45; Calculated for C13H10F3O5N: C 48.74, H 3.14, N 4.37%; Found: C 48.58, H 3.07, N 4.18%.
(S)-2-benzyl-3-(3-trifluoromethyl)-2,3-dihydroisoxazole- 4,5-dicarboxylic acid, 11
Colorless thick liquid. Yield 90%; Rf = 0.68; IR (KBr): υmax 3280 (m), 2980 (br), 1760 (s), 1485 (s), 1350 (s), 1220 (s), 780 (s) cm-1; 1H NMR (CDCl3): δ 10.02 (s, 2H, 2XCOOH), 7.70 – 7.20 (m, 5H & m, 4H, merged phenyl protons), 2.36 (s,1H, C3H), 1.26 (s,2H, CH2); 13C NMR (CDCl3): δ 170.00, 160.00 (carboxyl carbons), 137.50, 137.00, 136.50, 135.50, 135.00, 134.00, 133.50, 130.00, 128.50 (phenyl carbons), 77.00 (C5), 64.00 (C3), 54.00 (C4), 29.00 (CH2 carbon), 27.00 (CF3); FAB – MS (m/z): 396 (M+), 305, 160 (BP), 145, 91, 77; Calculated for C19H14F3O5N: C 57.57, H 3.53, N 3.53%; Found : C 57.450, H 3.40, N 3.34%.
(S)-2-phenyl-3-(4-trifluoromethyl)-2,3-dihydro- 5-phenylisoxazole-4-carboxylate, 12
Red thick liquid. Yield 92%; Rf = 0.66; IR (KBr): υmax 3230 (m), 2240 (m), 1740 (s), 1715 (s), 1680 (s), 1320 (s), 780 (s) cm- 1; 1H NMR (CDCl3): δ 7.87 – 7.40 (m, 2x5H, phenyl protons), 7.39- 7.20 (m, 4H, phenyl protons), 3.86 (s, 3H, -COOCH3), 1.25 (s,1H, C3H); 13C NMR (CDCl3): δ 168.50 (- COOCH3), 137.00, 136.00, 135.00, 134.30, 134.00, 132.00, 131.00, 130.50, 127.00, 126.50, 125.00, 123.70 (phenyl carbons), 82.50 (C5), 66.00 (C3), 55.00 (C4), 31.00 (-COOCH3), 28.00 (CF3); FAB -MS (m/z): 425 (M+), 348, 280 (BP), 271, 154, 77; Calculated for C24H18F3O3N: C 67.28, H 4.20, N 3.27%; Found: C 67.07, H 4.05, N 3.13%.
(S)-dimethyl-2-benzyl-3-(4-trifluoromethyl)-) 2,3-dihydroisoxazole- 4,5-dicarboxylate, 13
Yellow liquid. Yield 91%; Rf = 0.70; IR (KBr): υmax 3310 (m), 2190(m), 1740 (s), 1680 (s), 1660 (s), 1440 (s), 1265(s), 1220 (s), 788 (s) cm-1; 1H NMR (CDCl3): δ 7.68 – 7.30 (m, 5H & m, 4H, merged signals of phenyl protons), 3.88 (s, br, 2X3H, -COOCH3), 1.70 (s, 3H, C3H), 1.25 (s,2H, CH2); 13C NMR (CDCl3): δ 172.00, 162.00 (-COOCH3, carbonyl carbons of the ester group), 136.00, 134.00, 133.50, 133.00, 132.50, 131.50, 130.00, 129.00 (phenyl carbons), 82.50 (C5), 74.00 (C3), 58.00 (C4), 29.00 (CH2), 26.50 (CF3); FAB - MS (m/z): 421 (M+), 344, 330, 276 (BP), 91, 77; Calculated for C21H18F3O5N: C 59.43, H 4.24, N 3.30%; Found: C 59.26, H 4.10, N 3.15%.
Conclusion
We have adopted one of the most favorable green chemistry methodologies (mechanochemistry: ball-milling) in a solid phase for the synthesis of few N-substituted-4-(Trifluoromethyl)-C-phenyl nitrones and fluoro cycloadducts with excellent yields in a minimum time frame. We believe this simple, cost efficient and time-saving methodology will be adopted by many more researchers in synthetic organic chemistry. Though there are other 1.3-doples aside nitrones, like azides, nitrile oxides etc., but we have not yet studied mechanochemical procedure for conducting synthesis and cycloaddition reactions with these dipoles. Finally, we have found promising anticancer activities in few newly synthesized fluoro cycloadducts and we are hopeful in near future we shall be able to establish these molecules as “anti-cancer drugs after proper applications of cancer screening.
Conflicts of Interest
There are no conflicts to declare.
Acknowledgement
The authors are grateful to Department of Biotechnology, New Delhi, Government of India for providing Overseas Associateship- Fellowship (Grant no: BT/20/NE/2011) and administrative support and help for conducting the research work at the School of Chemistry, Cardiff University, Wales, UK.
References
- a) S Thakur, A Das, T Das (2021) 1,3-Dipolar cycloaddition of nitrones: synthesis of multisubstituted, diverse range of heterocyclic compounds New J Chem 45(26): 11420-11456.
- b) X Tian, T Xuan, J Gao, J Zhang, T Liu, Y Wang, et al. (2024) Catalytic enantioselective nitrone cycloadditions enabling collective syntheses of indole alkaloids. Nature Commun 15: 6429.
- W Patterson, PS Cheung, MJ Ernest (1992) J Med Chem 35: 507.
- E Wagner, L Becan, E Nowakowska (2004) Synthesis and pharmacological assessment of derivatives of isoxazolo [4,5-d] pyrimidineBio Org Med Chem 12(2): 265-272.
- a) PN Confalone, EM Huie (1988) The [3 + 2] Nitrone–Olefin Cycloaddition Reaction Org React 36: 1-173. Chapter 1
- b) IA Grigor’ev, H Feuer (2008) Nitrile Oxides, Nitrones and Nitronates in Organic Synthesis. Nitrones: Novel Strategies in Synthesis (Wiley-Interscience, Hoboken, NJ) 2: 129-434.
- c) RCF Jones, A Padwa, WH Pearson (2002) Synthetic Applications of 1,3-Dipolar Cycloaddition Chemistry Toward Heterocycles and 5. Natural Products (John Wiley & Sons, Inc, NY), The Chemistry of Heterocyclic Compounds 59: Ch1, 1-82.
- S Cao, XH Qian, GH Song, QC Huang (2002) J Fluorine Chem 63: 117.
- A Arnone, R Bernardi, F Blasco, R Cardillo, II Gerus, VP Kukhar (1998) Tetrahedron 54: 2809.
- a) RD Chambers (2005) Fluorine in Organic Chemistry, (Blackwell Publishing: Oxford, UK).
- b) P. Kirsch (2004) Modern Fluoroorganic Chemistry, (Wiley-VCH: Weinheim, Germany).
- D See Bach (1990) Organic Synthesis-Where now? Angew Chem Int Engl 29: 1320.
- JT Welch (1987) Tetrahedron 43:3123.
- W Wu, Y You, Z weng (2022) Chinese Chem Lett 33(10): 4517.
- M Rahaman, AK bagdi, DS Kopchuk, IS Kovaley, GV Zyvanoy, et al. (2020) Recent advances in the synthesis of fluorinated compounds via an aryne intermediate. Org Biomolecular Chem 47: 9562.
- DN Binjawhar, FA Al-salmi, MAA Alqahtani, E Fayad, RM saleem, I Zaki (2024) ACS Omega 9(16): 18505.
- B Chakraborty, MS Chettri, GP Leutel (2017) J Heterocyclic Chem 54(2): 1611.
- B Chakraborty, GP Leutel (2015) J Heterocyclic Chem 52(3): 726.
- B Chakraborty (2020) J Heterocyclic Chem 57: 477.
- B Chakraborty, E Chhetri (2018) J Heterocyclic Chem 55: 1157.
- B Chakraborty, PK Sharma, S Kafley (2013) Green Chem Lett Rev 6(2): 141.
- B Chakraborty, GP Luitel (2013) Tetrahedron Lett 54: 765
- B Chakraborty, PK Sharma (2012) Synth Commun 42: 1804.
- B Chakraborty, N Rai (2018) J Heterocyclic Chem 55: 1053.
- B Ranu, A Stolle (2015) Ball Milling Towards Green Synthesis: Applications, Projects and Challenges (Royal Society of Chemistry, UK).
- I Huskic, I Halasz, T Friscic, H Vancik (2012) Mechanosynthesis of nitrosobenzenes: a proof-of-principle study in combining solvent-free synthesis with solvent-free separations. Green Chem 14: 1597-1600.
- For selected recent examples of organic synthesis in ball mills, see:
- JG Hernandez, E Juaristi (2011) Asymmetric Aldol Reaction Organocatalyzed by (S)-Proline-Containing Dipeptides: Improved Stereoinduction under Solvent-Free Conditions. J Org Chem 76(5): 1464-1467.
- JG Hernandez, V Garcıa-Lopez, E Juaristi (2012) “Solvent-Free Asymmetric Aldol Reaction Organocatalyzed by (S)-Proline-Containing Thiodipeptides under Ball- Milling Conditions,” Tetrahedron 68(1): 92-97.
- YF Wang, RX Chen, K Wang, BB Zhang, BZ Lib, DQ Xu (2012) Green Chem 14: 893.
- M Jorres, S Mersmann, G Raabe (2013) Organocatalytic solvent-free hydrogen bonding-mediated asymmetric Michael additions under ball milling conditions. Green Chem 15: 612-616.
- R Trotzki, O Hoffmann, B Ondruschka (2008) The Knoevenagel condensation at room temperature. Green Chem 10: 873-878.
- J Mack, M Shumba (2007) Rate enhancement of the Morita–Baylis–Hillman reaction through mechanochemistry. Green Chem 9: 328-330.
- DC Waddell, J Mack (2009) An environmentally benign solvent-free Tishchenko reaction. Green Chem 11: 79-82.
- F Schneider, T Szuppa, A Stolle, B Ondruschka, H Hopf (2009) Energetic assessment of the Suzuki-Miyaura reaction: a curtate life cycle assessment as an easily understandable and applicable tool for reaction optimization. Green Chem 11: 1894-1899.
- G Cravotto, D Garella, D Tagliapietra, A Stolle, S Schusler, et al. (2012) New J Chem 36: 1304.
- DA Fulmer, WC Shearouse, ST Medonza, J Mack (2009) Solvent-free Sonogashira coupling reaction via high-speed ball milling. Green Chem 11: 1821-1825.
- R Thorwirth, A Stolle, B Ondruschka (2010) Fast copper-, ligand- and solvent-free Sonogashira coupling in a ball mill. Green Chem 12: 985-991.
- R Thorwirth, A Stolle, B Ondruschka, A Wild, US Schubert (2011) Fast, ligand- and solvent-free copper-catalyzed click reactions in a ball mill. Chem Commun 47: 4370-4372.
- TL Cook, TL, JA Walker, J Mack (2013) Scratching the catalytic surface of mechanochemistry: a multi-component CuAAC reaction using a copper reaction vial. Green Chem 15: 617-619.
- n) V Estevez, M Villacampa, CJ Menendez (2013) Three-component access to pyrroles promoted by the CAN–silver nitrate system under high-speed vibration milling conditions: a generalization of the Hantzsch pyrrole synthesis. Chem Commun 49: 591-593.
- o) W Su, J Yu, Z Li, Z. Jiang (2011) Solvent-free cross-dehydrogenative coupling reactions under high-speed ball-milling conditions applied to the synthesis of functionalized tetrahydroisoquinolines. J Org Chem 76(21): 9144-9150.
- p) V Strukil, B Bartolec, T Portada, I Đilovic, I Halasz, et al. (2012) One-pot mechanosynthesis of aromatic amides and dipeptides from carboxylic acids and amines. Chem Commun 48: 12100-12102.
- GJ Hernandez, GE Juaristi (2010) J Org Chem 75: 7107.
- D Tan, V Strukil, C Mottillo, CT Friscic (2014) Mechanosynthesis of pharmaceutically relevant sulfonyl-(thio)ureas. Chem Commun 50: 5248-5250.
- Y Fang, N Salame, Simon Woo, T Friscic, C Cuccia, et al. (2014) Rapid and facile solvent-free mechanosynthesis in a cell lysis mill: preparation and mechanochemical complexation of aminobenzoquinones. Cryst Eng Comm 16: 7180.
- K Crossey, KRN Cunningham, P Redpath, ME Migaud (2015) Atom efficient synthesis of pyrimidine and purine nucleosides by ball milling Check for updates RSC Adv 5: 58116-58119.
- I IDokli, M Gredicak (2015) Eur J Org Chem 2727.
- PFM, Oliveira, M Baron, A Chamayou, C Andre-Barres, B Guidetti, M Baltas (2014) Solvent-free mechanochemical route for green synthesis of pharmaceutically attractive phenol-hydrazones RSC Adv 4: 56736-54742.
- TX Metro, J Bonnamour, T Reidon, A Duprez, J Sarpoulet, et al. (2015) Comprehensive study of the organic-solvent-free CDI-mediated acylation of various nucleophiles by mechanochemistry. Chem Eur-J 21(36): 12787-12796.
- TK Achar, S Maiti, P Mal (2014) IBX works efficiently under solvent free conditions in ball milling RSC Adv 4: 12834.
- A Loupy (2006) Ed: Microwaves in Organic Synthesis (Wiley-VCH: Weinheim).
- A Banerji, PK Biswas, P Sengupta, S Dasgupta, M Gupta (2004) Indian J Chem 43B: 880.
- P Deshong, P W Li, JW Kennington, HL Ammon (1991) J Org Chem 56: 1364.
- R Gandolfi, P Grunanger (1999) “The chemistry of heterocyclic compounds (Wiley Interscience)49(2): 774
- Y Yu, M Ohno, S Eguchi (1993) Tetrahedron 49: 824.
- R Huisge (1976) J Org Chem 41: 403.
- KN Houk, K Yamaguchi (1984) 6 1,3-Dipolar Cycloaddition Chemistry (Wiley, New York) 2: 407.
- B Rachid, K Abbiche, MD Mellaoui, A Imjjad (2023) Insights into the mechanism of [3+2] cycloaddition reactions between N-benzyl fluoro nitrone and maleimides, its selectivity and solvent effects. J Computational Chem 45(5): 284-299.
- Chetana Sanjai, Sushruta S Hakkimane, Bharath Raja Guru, Santosh L Gaonkar (2024) A Comprehensive Review on Anticancer Evaluation Bioorg Chem 142: 106973.
- D Campoccia, S Ravaioli, S Santi, V Mariani, CR Arciola, et al. (2021) Exploring the anticancer effects of standardized extracts of poplar-type propolis: In vitro cytotoxicity toward cancer and normal cell lines Biomedicine & Pharmacotherapy 141: 111895.
- L Cai, X Qin, Y Song, H Jiang, Y Wu, H Ruan (2019) Comparison of Cytotoxicity Evaluation of Anticancer Drugs between Real-Time Cell Analysis and CCK-8 Method ACS-Omega 4(7): 12036-12042.
- B Chakraborty, MN Roy (2022) Recent Advances in Noble Metal (Pt, Ru, and Ir)-Based Electrocatalysts for Efficient Hydrogen Evolution Reaction ACS-Omega 7: 4457.






























