Introduction
Melanoma is an extremely aggressive and deadly form of skin cancer. With the emergence of immune checkpoint inhibitors and targeted therapies, significant progress has been made in the systemic treatment of melanoma. Neoadjuvant therapy has emerged as a promising approach in the management of locally advanced and metastatic melanoma. This strategy involves administering systemic therapy before surgery, aiming to improve recurrence-free survival (RFS) and overall survival (OS) while potentially de-escalating surgical intervention. Immune checkpoint inhibitors (ICIs) and targeted therapies have demonstrated significant efficacy in the neoadjuvant setting, leading to high rates of pathological complete response (pCR) and major pathological response (MPR).
Particularly, the use of PD-1 blockade alone or in combination with CTLA-4 blockade has shown promising results in terms of reducing the risk of relapse and mortality. Initial data also show that neoadjuvant immunotherapy allows de-escalation of lymph node surgery. Similarly, neoadjuvant BRAF-MEK inhibition for melanoma with BRAF mutations has produced comparable outcomes, although the response durability is lower compared to immunotherapy. Moreover, emerging data from trials such as SWOG S1801 [1] and OpACIN-neo [2] suggest that neoadjuvant approaches may outperform conventional adjuvant therapy. Recent findings of the NADINA trial, demonstrated the superiority of neoadjuvant ipilimumab plus nivolumab compared to adjuvant nivolumab in macroscopic stage III melanoma Neoadjuvant therapy offers several advantages over traditional adjuvant therapy, including early evaluation of treatment response, potential reduction in surgical burden, and the opportunity to study tumor tissue for research purposes. However, uncertainties remain regarding optimal treatment protocols, including dosing, duration, timing relative to surgery, and integration with other modalities. This article reviews the current landscape of neoadjuvant therapy for melanoma, highlighting key clinical trials, predictive biomarkers, and ongoing research efforts. We also discuss the challenges and unanswered questions that need to be addressed to optimize the use of neoadjuvant therapy in clinical practice.
Discussion
Improved Prognosis through Modern Therapies
The prognosis of unresectable or metastatic melanoma has significantly improved with the development and approval of immune checkpoint inhibitors (ICI) and targeted substances (TT). In stage IV, the median overall survival is now 6.5 years [2]. Highly effective PD-1 blockers Nivolumab and Pembrolizumab, as well as BRAF-MEK inhibition with Dabrafenib/Trametinib (in the presence of a BRAF-V600E/K mutation), are also approved for adjuvant melanoma therapy after complete resection in stage III (Nivolumab also in stage IV) and lead to a significant reduction in recurrence risk [3-5]. Whether, and if so, to what extent such adjuvant therapy extends the overall survival of patients remains unclear. The latest approach to further improve the course of melanoma in stages IIIB-D or IV is neoadjuvant therapy. Here, systemic therapeutics are primarily used before a planned operation of clinically detectable metastases. The goal is less about improving operability and more about increasing recurrence-free survival (RFS) and overall survival (OS) and potentially even de-escalating surgical care for patients. Additionally, unlike adjuvant therapy, neoadjuvant therapy allows early evaluation of the response to a therapeutic agent. Furthermore, tissue material can be obtained for research purposes. Although the data on neoadjuvant melanoma therapy are very promising, there is currently no explicit approval for any substance for this therapeutic intention in melanoma. As shown in Table 1, several studies have investigated the efficacy of neoadjuvant immunotherapy for melanoma.

Data on neoadjuvant therapy of melanoma
Dabrafenib plus Trametinib
In a neoadjuvant intent, targeted therapy with the BRAF inhibitor Dabrafenib in combination with the MEK inhibitor Trametinib was tested in patients with histologically confirmed, surgically resectable melanoma in stage III or oligometastatic melanoma in stage IV with a BRAF-V600E or -V600K mutation [6]. The two treatment arms included either immediate surgery with the possibility of subsequent standard adjuvant therapy or neoadjuvant plus adjuvant therapy with Dabrafenib and Trametinib over a total of 8 weeks before surgery with a total treatment duration of 52 weeks. The neoadjuvant plus adjuvant therapy approach was clearly superior to the standard arm; however, only a few patients in the comparison arm received adjuvant therapy with Dabrafenib plus Trametinib. This was because superiority in the neoadjuvant arm was seen early, leading to the premature closure of the study and thus fewer patients in the adjuvant arm. An interpretation of these data regarding the actual difference between the two approaches is therefore not possible. In the recently conducted REDUCTOR study in inoperable, locally advanced melanoma, short-term neoadjuvant therapy with Dabrafenib plus Trametinib over 8 weeks enabled radical resection of metastases in 81% of patients [7]. Due to the single-arm study design, interpreting these data regarding clinical benefit is difficult.
Ipilimumab plus Nivolumab
The phase III NADINA trial investigated the efficacy of neoadjuvant ipilimumab plus nivolumab compared to adjuvant nivolumab in patients with macroscopic, resectable stage III melanoma. The study randomized 423 patients to receive either two cycles of neoadjuvant ipilimumab (1 mg/kg) plus nivolumab (3mg/kg) followed by therapeutic lymph node dissection (TLND) and, in case of not achieving a major pathologic response (MPR), adjuvant dabrafenib plus trametinib or adjuvant nivolumab versus TLND followed by adjuvant nivolumab.
The primary endpoint of event-free survival (EFS) was significantly improved in the neoadjuvant arm compared to the adjuvant arm (HR 0.32, 99.9% CI 0.15-0.66, p<0.0001), with estimated 12-month EFS rates of 83.7% versus 57.2%, respectively. In the subgroup of BRAF-mutated melanoma, estimated EFS rates were 83.5% and 52.1% for neoadjuvant versus adjuvant, respectively, and in BRAF wild-type melanoma, the rates were 83.9% and 62.4%, respectively. The neoadjuvant arm also showed a high rate of MPR (58.0%), with 12-month RFS rates of 95.1% for MPR, 76.1% for partial pathological response (PPR), and 57.0% for pathological non-response (PNR). Grade ≥3 systemic treatment-related adverse events were seen in 29.7% and 14.7% of patients in the neoadjuvant and adjuvant arms, respectively, with one death due to toxicity (pneumonitis) in the adjuvant arm. The NADINA trial is the first phase III study to evaluate neoadjuvant immunotherapy against standard of care in melanoma and the first phase III trial in oncology to evaluate a neoadjuvant regimen consisting of immunotherapy alone. Based on these results, neoadjuvant ipilimumab plus nivolumab followed by response-driven adjuvant treatment should be considered a new standard of care treatment in macroscopic stage III melanoma
High pathological response rates have been shown in three large studies testing the combined regimen of the CTLA-4 blocker Ipilimumab and the PD-1 blocker Nivolumab in neoadjuvant intent [8-10]. In the OpACIN study, a clinical, randomized phase Ib/II study, neoadjuvant therapy with Ipilimumab (3 mg/kg BW) and Nivolumab (1 mg/kg BW) was investigated. Part of the patients received two doses before and two doses after surgery. This treatment scheme was compared with the adjuvant arm, where patients were operated on immediately and subsequently received four doses of the same combination as adjuvant therapy. The efficacy of the neoadjuvant treatment scheme was already demonstrated here but at the cost of significant side effects [9]. In clinical phase II study OpACIN-neo, three different combination schemes were investigated. The scheme with two cycles of Ipilimumab 1 mg/kg BW and Nivolumab 3mg/kg BW q3w (Arm 2) showed a pCR rate of 47% and the occurrence of immune-mediated adverse events (irAE) of grades 3-4 according to Common Terminology Criteria for Adverse Events (CTCAE) in 20% of treated patients, showing the best ratio of efficacy to toxicity. Recent updated survival analyses of the OpACINneo study resulted in a 3-year RFS rate of 82% for all patients, 95% for patients achieving a pathological response, and 37% for patients showing no response (pNR; p < 0.001) [2,11]. The PRADO extension cohort of the OpACIN-neo study also examined the correlation between the pathological response in an index lymph node (ILN) and RFS in patients undergoing neoadjuvant treatment with Ipilimumab 1 mg/kg BW plus Nivolumab 3 mg/ kg BW [12]. In patients who achieved MPR (“major pathological response”; less than 10% vital tumor cells) or pCR in their ILN, no therapeutic lymph node dissection (TLND) or adjuvant therapy was performed, and therapy was de-escalated based on the pathological response. This reduction was associated with fewer postoperative complications [12].
Neoadjuvant plus Adjuvant versus Adjuvant Therapy with Pembrolizumab
In the SWOG-1801 study, a randomized phase II study, it was investigated whether neoadjuvant plus adjuvant therapy with the PD-1 blocker Pembrolizumab is superior to the sole adjuvant administration of Pembrolizumab [1]. One group received three doses of Pembrolizumab (200mg q3w), followed by surgery and subsequent administration of 15 more doses of Pembrolizumab (200mg q3w). The other group underwent immediate surgery and received 18 doses of Pembrolizumab (200mg q3w) exclusively adjuvantly after surgery. A total of 313 patients participated in this large study encompassing 90 US centers. After a median follow-up period of 14.7 months, the 2-year event-free survival (EFS) rate in the neoadjuvant and adjuvant group was significantly higher (72%; 95% confidence interval, 95%-CI: 64-80) than in the adjuvant group (49%; 95%-CI: 41-59; p = 0.004 by the log-rank test). Events for the endpoint EFS included recurrences after surgery (local or distant), progression of melanoma, or toxicity leading to inoperability, initiation of adjuvant therapy > 84 days after surgery, and death of patients. Radiological assessment according to Response Evaluation Criteria in Solid Tumors (RECIST) criteria showed a response rate of 47% in the neoadjuvant plus adjuvant group, while the rate of pathological complete remissions (pCR) after evaluation of the surgical specimens was 21%. The rate of grade 3-4 adverse events according to CTCAE was 12% in the neoadjuvant and adjuvant group and 14% in the adjuvant group.
Other Substances
T-VEC
Talimogen Laherparepvec (T-VEC), an oncolytic virus specifically adapted for selective replication in tumor cells and induction of host immunity, was approved for local treatment of advanced, inoperable melanoma in stages III and IV (M1a) [13]. In a randomized phase II study, neoadjuvant intratumoral T-VEC administration (6 doses; Arm 1) was compared with surgical resection and subsequent standard adjuvant therapy (Arm 2) [14]. The pCR rate was 17.1% for the neoadjuvant Arm 1. An ongoing single-arm study is currently investigating neoadjuvant therapy with T-VEC in combination with the PD-1 blocker Nivolumab every 2 weeks over 9 weeks [15]. The study is currently in the recruitment phase and has not yet published results.
Relatlimab plus Nivolumab
A fixed-dose combination of Nivolumab and Relatlimab (anti- LAG3 antibody) was recently approved after the RELATIVITY-047 study validated the regimen as an effective treatment strategy for patients with inoperable or metastatic melanoma [16]. This study showed a significant advantage for progressionfree survival (PFS) over Nivolumab alone. In locally advanced, operable melanoma, a phase II study investigated two doses of neoadjuvant therapy with Nivolumab 480 mg plus Relatlimab 160 mg i.v. 4 weeks apart, followed by surgery and subsequently 10 doses of adjuvant combination therapy (q4w) [17]. The pCR rate was 57%, and the radiological overall response rate (ORR) was also 57%. The 1- and 2-year RFS rates were 100% and 92% for patients showing a pathological response, compared to 88% and 55% for patients showing no pathological response (p = 0.005). No treatment-related grade 3 or 4 adverse events occurred during the neoadjuvant treatment, whereas these occurred in 26% of treated patients in the adjuvant phase [18-23].
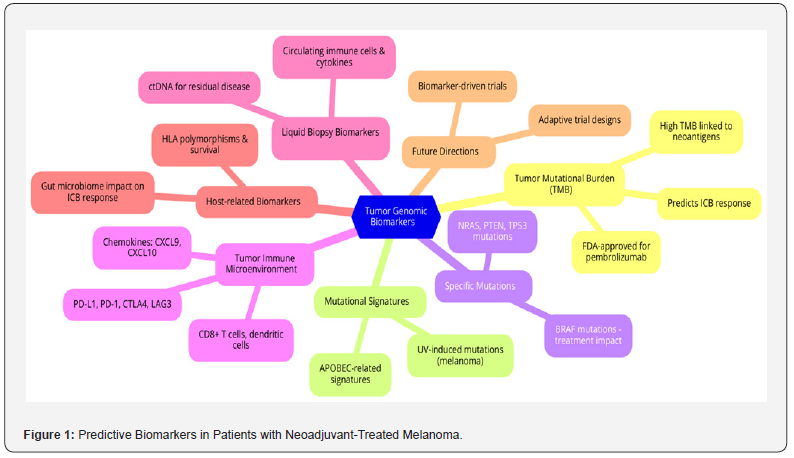
Predictive biomarkers in patients with neoadjuvanttreated melanoma
Tumor Genomic Biomarkers
The concept that TMB predicts immunotherapy response is rooted in the neoantigen theory, which posits that a greater number of mutations leads to a higher likelihood of generating neoantigens that can trigger an immune response [24]. While TMB has shown promise as a biomarker in other settings, its role in neoadjuvant immunotherapy for melanoma is still under investigation. Some studies suggest that TMB may be associated with response to neoadjuvant ICIs in melanoma patients [25,26]. For example, a study by Blank et al. found that patients with both elevated interferon-gamma (IFN-γ) and TMB had a 100% partial pathologic response rate and no recurrence at 2 years [27,28]. While the role of TMB in neoadjuvant immunotherapy for melanoma is still under investigation, some studies suggest it may be associated with treatment response. Clinical trials have demonstrated the potential of neoadjuvant immunotherapy for melanoma, but further research is needed to optimize treatment strategies and identify predictive biomarkers like TMB. Driver mutations, such as those in the BRAF gene, can influence tumor growth and immune evasion. In advanced melanoma, BRAFmutated tumors have shown improved survival with combined ipilimumab and nivolumab compared to nivolumab monotherapy [29]. However, in the neoadjuvant setting, BRAF mutation status has not significantly impacted pathologic response [12]. Other mutations, such as those in NRAS, PTEN, BCLAF1, and TP53, have been associated with ICB response or resistance in advanced melanoma but have not been investigated in the neoadjuvant setting [30].
Tumor Immune Microenvironment Phenotype Biomarkers
The presence and diversity of tumor-infiltrating lymphocytes (TILs), particularly CD8+ T cells, are associated with response to neoadjuvant ICB [8]. The phenotype of these T cells, including expression of PD-1, CD39, and TCF7, also correlates with improved response [31]. T-cell clonality and diversity in the tumor microenvironment (TME) are thought to be predictive of ICB response, with a diverse T-cell repertoire at baseline and a more clonal repertoire during therapy associated with better outcomes [32]. Dendritic cells (DCs), particularly BATF3+ DCs, play a crucial role in antigen presentation and T-cell recruitment. A low BATF3+ DC gene signature in pretreatment tumor biopsies is associated with relapse after neoadjuvant ICB. Chemokines such as CXCL9 and CXCL10, produced by BATF3+ DCs, are also associated with improved ICB response [26]. Inflammatory gene expression signatures, such as the IFNγ signature, offer a broader representation of the antitumor immune response. The IFNγ signature has been predictive of pathologic response and relapse in several neoadjuvant melanoma trials. Combining TMB and IFNγ signatures has shown high predictive value for pathologic response, with response rates of 90-100% in patients with high TMB and high IFNγ signatures [26].
Liquid Biopsy Biomarkers
Circulating tumor DNA (ctDNA) is a powerful tool for detecting residual disease after neoadjuvant therapy. The presence of ctDNA before or after surgery is associated with poor response to ICB and decreased relapse-free survival (RFS) [33]. ctDNA can also be used to estimate blood TMB, which has been predictive of ICB response in metastatic NSCLC. In melanoma, ctDNA may assist in selecting patients for adjuvant or neoadjuvant treatment [34]. Posttreatment circulating PD1+ CD8+ T cells and EOMES+ CD8+ T cells have been associated with favorable outcomes in melanoma. Circulating cytokines, such as IFNγ, IL6, and IL8, have also been proposed as biomarkers for ICB response and toxicity [35]. High levels of IL10 and IL17 are associated with disease progression and toxicity, respectively, in patients treated with neoadjuvant ipilimumab [36].
Host-Related Biomarkers
HLA genes encode proteins responsible for antigen presentation to T cells. HLA heterozygosity, particularly at HLA-I loci, is associated with improved survival after ICB, likely due to broader antigen presentation [37]. Loss of heterozygosity (LOH) of HLA-I genes and downregulation of HLA-I expression are associated with ICB resistance [26]. HLA polymorphisms have not been extensively studied in the neoadjuvant setting but may become relevant as neoadjuvant immunotherapy becomes more common. The gut microbiome influences the immune system and response to ICB. A diverse microbiome, particularly dominated by Ruminococcaceae, is associated with higher response rates and lower toxicity in melanoma patients treated with neoadjuvant ipilimumab and nivolumab [38]. Antibiotic use, which reduces microbiome diversity, is associated with decreased ICB response [39]. Certain bacterial species, such as Akkermansia muciniphila and Faecalibacterium prausnitzii, have been linked to improved responses to anti-PD-1 therapy in melanoma patients [40]. While still in its infancy, research into the interaction between the microbiome and neoadjuvant therapy holds promise for developing novel strategies to enhance treatment outcomes. For a visual representation of the predictive biomarkers in patients with neoadjuvant-treated melanoma, see the Figure 1.
Data gaps and outlook
Although some of the studies presented on neoadjuvant therapy also included patients with stage IV melanoma, their total number is rather low, and the relevance of neoadjuvant therapy in this stage remains to be seen. Overall, it is now clear that oligometastatic melanoma can only be controlled in the long term by surgery in a few patients, so drug therapy should be regularly performed in such patients. It is therefore advisable to generally postpone surgical intervention in these patients and primarily use systemic therapy. Another group underrepresented in the mentioned studies are patients with stage III primary tumors and/or in-transit metastases. In principle, neoadjuvant therapy also seems to lead to remissions here, but discordant responses of the different tumor manifestations can occur [20-22]. The SWOG- 1801 study clearly shows that neoadjuvant plus adjuvant therapy is superior to adjuvant pembrolizumab alone, but it remains unclear whether continuation of therapy after surgery is actually necessary. Data on the neoadjuvant use of ipilimumab plus nivolumab suggest that neoadjuvant therapy alone can already lead to very high rates of patients without recurrence. To what extent this can be increased or consolidated by adjuvant therapy (depending on the category of pathological response) remains unclear. Here, the double-blind, randomized, and placebocontrolled IMMUNED study provides promising data in patients with stage IV melanoma in remission. It shows a significant prolongation of relapse-free survival through the adjuvant use of ipilimumab plus nivolumab compared to nivolumab monotherapy or placebo [23]. For combination therapy, unlike for pembrolizumab, there are also data on de-escalation of TLND. The activity of pembrolizumab as monotherapy demonstrated in the SWOG-1801 study is lower, with a pCR rate of about 20%, than under the combination of ipilimumab and nivolumab. Therefore, it is necessary to build on the promising results of this study and explore more effective therapies. Currently, the phase III study NADINA is comparing the efficacy of neoadjuvant treatment with ipilimumab (1mg/kg BW) and nivolumab (3 mg/ kg BW) with adjuvant treatment with nivolumab in resectable stage III melanoma, with patient survival as the primary endpoint (NCT04949113).
Despite promising early-phase trial results, uncertainties remain regarding optimal treatment protocols, including duration, dosing, timing relative to surgery, and integration with other modalities. Shorter courses may reduce toxicity and avoid surgical delays, while longer durations could enhance response rates and survival. The PRADO trial showed a 57% MPR rate with two cycles of neoadjuvant anti-PD-1, but the impact of extending therapy is unknown. Prolonged immunotherapy increases the risk of immune-related adverse events, potentially delaying surgery or requiring immunosuppressive interventions. Further research is needed to determine the optimal duration of therapy and whether early biomarkers can guide adaptive dosing. Standard adjuvant dosing is often used, but neoadjuvant-specific regimens are unexplored. The PRADO trial used a fixed 200 mg pembrolizumab dose, but weight-based or response-adapted dosing could improve outcomes. The NADINA trial reported a 67% pCR rate with nivolumab plus ipilimumab but 35% grade 3-4 irAEs, leading to the exploration of reduced ipilimumab dosing to improve tolerability. Further research should investigate the optimal dosing strategies for neoadjuvant immunotherapy, including the potential benefits of combination therapy and staggered dosing. BRAF/MEK inhibitors induce rapid tumor shrinkage but lack durable immune memory, and combining them with immunotherapy risks overlapping toxicities. Preclinical studies suggest radiotherapy enhances antigen release and abscopal effects, but clinical data are limited. Future research should explore the optimal integration of neoadjuvant immunotherapy with other modalities, including targeted therapies and intratumoral therapies. The unanswered questions surrounding neoadjuvant immunotherapy for melanoma highlight the need for robust clinical trials, biomarker-driven approaches, and multidisciplinary collaboration. Addressing these gaps will pave the way for personalized, effective, and safe treatment protocols that optimize outcomes for patients with locally advanced melanoma [40].
Conclusion
Neoadjuvant therapy represents a paradigm shift in the treatment of melanoma, offering the potential to improve patient outcomes and personalize treatment strategies. The success of neoadjuvant immunotherapy, particularly with PD-1-based regimens, has been demonstrated in clinical trials, leading to high pCR rates and reduced surgical burden. Targeted therapies like BRAF-MEK inhibition also show efficacy, especially in BRAFmutated melanomas, but their long-term benefits may be limited compared to immunotherapy. Key biomarkers, such as tumor mutational burden (TMB), PD-L1 expression, and circulating tumor DNA (ctDNA), are being investigated to predict response and guide treatment decisions. However, critical questions remain regarding the optimal timing, duration, and sequencing of neoadjuvant interventions, as well as their integration with surgery and other modalities. Future research must focus on refining adaptive dosing strategies, exploring novel combinations, and validating emerging biomarkers to ensure safe and effective implementation of neoadjuvant therapy in clinical practice. As the field continues to evolve, multidisciplinary collaboration and robust clinical trials will be essential to fully realize the potential of this innovative approach for patients with melanoma.
References
- Sapna P Patel, Megan Othus, Yuanbin Chen, G Paul Wright, Kathleen J Yost, et al. (2023) Neoadjuvant–Adjuvant or Adjuvant-Only Pembrolizumab in Advanced Melanoma. N Engl J Med 388(9): 813-823.
- JM Versluis, AM Menzies, K Sikorska, EA Rozeman, RPM Saw, WJ van Houdt, et al. (2023) Survival update of neoadjuvant ipilimumab plus nivolumab in macroscopic stage III melanoma in the OpACIN and OpACIN-neo trials. Ann Oncol Off J Eur Soc Med Oncol 34(4): 420-430.
- Paolo A Ascierto, Michele Del Vecchio, Mario Mandalá, Helen Gogas, Ana M Arance, et al. (2020) Adjuvant nivolumab versus ipilimumab in resected stage IIIB-C and stage IV melanoma (CheckMate 238): 4-year results from a multicentre, double-blind, randomised, controlled, phase 3 trial. Lancet Oncol 21(11): 1465-1477.
- Alexander MM Eggermont, Christian U Blank, Mario Mandalà, Georgina V Long, Victoria G Atkinson, et al. (2021) Adjuvant pembrolizumab versus placebo in resected stage III melanoma (EORTC 1325-MG/KEYNOTE-054): distant metastasis-free survival results from a double-blind, randomised, controlled, phase 3 trial. Lancet Oncol 22(5): 643-654.
- Reinhard Dummer, Jan C Brase, James Garrett, Catarina D Campbell, Eduard Gasal, et al. (2020) Adjuvant dabrafenib plus trametinib versus placebo in patients with resected, BRAFV600-mutant, stage III melanoma (COMBI-AD): exploratory biomarker analyses from a randomised, phase 3 trial. Lancet Oncol 21(3): 358–372.
- Rodabe N Amaria, Peter A Prieto, Michael T Tetzlaff, Alexandre Reuben, Miles C Andrews, et al. (2018) Neoadjuvant plus adjuvant dabrafenib and trametinib versus standard of care in patients with high-risk, surgically resectable melanoma: a single-centre, open-label, randomised, phase 2 trial. Lancet Oncol 19(2): 181-193.
- Stéphanie A Blankenstein, Maartje W Rohaan, Willem Martin C Klop, Bernies van der Hiel, Bart A van de Wiel, et al. (2021) Neoadjuvant Cytoreductive Treatment With BRAF/MEK Inhibition of Prior Unresectable Regionally Advanced Melanoma to Allow Complete Surgical Resection, REDUCTOR: A Prospective, Single-arm, Open-label Phase II Trial. Ann Surg 274(2): 383-389.
- Rodabe N Amaria, Sangeetha M Reddy, Hussein A Tawbi, Michael A Davies, Merrick I Ross, et al. (2018) Neoadjuvant immune checkpoint blockade in high-risk resectable melanoma. Nat Med 24(11): 1649-1654.
- Christian U Blank, Elisa A Rozeman, Lorenzo F Fanchi, Karolina Sikorska, Bart van de Wiel, et al (2018) Neoadjuvant versus adjuvant ipilimumab plus nivolumab in macroscopic stage III melanoma. Nat Med 24(11): 1655-1661.
- EA Rozeman, EP Hoefsmit, ILM Reijers, RPM Saw, JM Versluis, O Krijgsman, et al. (2021) Survival and biomarker analyses from the OpACIN-neo and OpACIN neoadjuvant immunotherapy trials in stage III melanoma. Nat Med 27(2): 256–263.
- Elisa A Rozeman, Alexander M Menzies, Alexander CJ van Akkooi, Chandra Adhikari, Carolien Bierman, et al. (2019) Identification of the optimal combination dosing schedule of neoadjuvant ipilimumab plus nivolumab in macroscopic stage III melanoma (OpACIN-neo): a multicentre, phase 2, randomised, controlled trial. Lancet Oncol 20(7): 948–960.
- Irene LM Reijers, Alexander M Menzies, Alexander CJ van Akkooi, Judith M Versluis, Noëlle M J van den Heuvel, et al. (2022) Personalized response-directed surgery and adjuvant therapy after neoadjuvant ipilimumab and nivolumab in high-risk stage III melanoma: the PRADO trial. Nat Med 28(6): 1178-1188.
- Robert HI Andtbacka, Howard L Kaufman, Frances Collichio, Thomas Amatruda, Neil Senzer, et al. (2015) Talimogene Laherparepvec Improves Durable Response Rate in Patients With Advanced Melanoma. J Clin Oncol Off J Am Soc Clin Oncol 33(25): 2780-2788.
- R Dummer, DE Gyorki, J Hyngstrom, AC Berger, R Conry, L Demidov, et al (2021) Neoadjuvant talimogene laherparepvec plus surgery versus surgery alone for resectable stage IIIB-IVM1a melanoma: a randomized, open-label, phase 2 trial. Nat Med 27(10): 1789-1796.
- Rohaan MW, Stahlie EHA, Franke V, Zijlker LP, Wilgenhof S, et al. (2022) Neoadjuvant nivolumab + T-VEC combination therapy for resectable early stage or metastatic (IIIB-IVM1a) melanoma with injectable disease: study protocol of the NIVEC trial. BMC Cancer 22(1): 851.
- Hussein A Tawbi, Dirk Schadendorf, Evan J Lipson, Paolo A Ascierto, Luis Matamala, et al. (2022) Relatlimab and Nivolumab versus Nivolumab in Untreated Advanced Melanoma. N Engl J Med 386(1): 24-34.
- Rodabe N Amaria, Michael Postow, Elizabeth M Burton, Michael T Tetzlaff, Merrick I Ross, et al (2022) Neoadjuvant relatlimab and nivolumab in resectable melanoma. Nature 611(7934): 155-160.
- MT Tetzlaff, JL Messina, JE Stein, X Xu , RN Amaria, et al. (2018) Pathological assessment of resection specimens after neoadjuvant therapy for metastatic melanoma. Ann Oncol Off J Eur Soc Med Oncol 29: 1861–1868.
- Alexander M Menzies, Rodabe N Amaria, Elisa A Rozeman, Alexander C Huang, Michael T Tetzlaff, et al. (2021) Pathological response and survival with neoadjuvant therapy in melanoma: a pooled analysis from the International Neoadjuvant Melanoma Consortium (INMC). Nat Med 27(2): 301-309.
- Weber J, Glutsch V, Geissinger E, Haug L, Lock JF, et al. (2020) Neoadjuvant immunotherapy with combined ipilimumab and nivolumab in patients with melanoma with primary or in transit disease. Br J Dermatol 183(3): 559-563.
- Judith M Versluis, Irene L M Reijers, Elisa A Rozeman, Alexander M Menzies, Alexander CJ van Akkooi, et al (2021) Neoadjuvant ipilimumab plus nivolumab in synchronous clinical stage III melanoma. Eur J Cancer Oxf Engl 1990 148: 51–57.
- Patrick Schummer, Valerie Glutsch, Lukas Haug, Mathias Rosenfeldt, Johann Lock, et al. (2023) Neoadjuvant ipilimumab plus nivolumab in locally advanced melanoma: A real-world single-centre retrospective study. EJC Skin Cancer 1: 100003.
- Elisabeth Livingstone, Lisa Zimmer, Jessica C Hassel, Michael Fluck, Thomas K Eigentler, et al. (2022) Adjuvant nivolumab plus ipilimumab or nivolumab alone versus placebo in patients with resected stage IV melanoma with no evidence of disease (IMMUNED): final results of a randomised, double-blind, phase 2 trial. Lancet Lond Engl 400(10358): 1117-1129.
- Qin Y, Huo M, Liu X, Li SC (2024) Biomarkers and computational models for predicting efficacy to tumor ICI immunotherapy. Front Immunol 15: 1368749.
- Ning B, Liu Y, Wang M, Li Y, Xu T, Wei Y (2022) The Predictive Value of Tumor Mutation Burden on Clinical Efficacy of Immune Checkpoint Inhibitors in Melanoma: A Systematic Review and Meta-Analysis. Front Pharmacol 13: 748674.
- Hoeijmakers LL, Reijers ILM, Blank CU (2023) Biomarker-Driven Personalization of Neoadjuvant Immunotherapy in Melanoma. Cancer Discov 13(11): 2319-2338.
- Hieken TJ, Kreidieh F, Aedo-Lopez V, Block MS, McArthur GA, et al. (2023) Neoadjuvant Immunotherapy in Melanoma: The Paradigm Shift. Am Soc Clin Oncol Educ Book 43: e390614.
- Harriet M Kluger, Christopher R Zito, Gabriela Turcu, Marina K Baine, Hongyi Zhang, et al (2017) PD-L1 Studies Across Tumor Types, its Differential Expression and Predictive Value in Patients Treated with Immune Checkpoint Inhibitors. Clin Cancer Res Off J Am Assoc Cancer Res 23(15): 4270-4279.
- Jedd D Wolchok, Vanna Chiarion-Sileni, Piotr Rutkowski, C Lance Cowey 1, Dirk Schadendorf, et al. (2025) Final, 10-Year Outcomes with Nivolumab plus Ipilimumab in Advanced Melanoma. N Engl J Med 392(1): 11-22.
- Gajic ZZ, Deshpande A, Legut M, Imieliński M, Sanjana NE (2022) Recurrent somatic mutations as predictors of immunotherapy response. Nat Commun 13(1): 3938.
- Grace Heloise Attrill, Carina N Owen, Tasnia Ahmed, Ismael A Vergara, Andrew J Colebatch, et al. (2022) Higher proportions of CD39+ tumor-resident cytotoxic T cells predict recurrence-free survival in patients with stage III melanoma treated with adjuvant immunotherapy. J Immunother Cancer 10: e004771.
- Georgia M Beasley, Michael C Brown, Norma E Farrow, Karenia Landa, Rami N Al-Rohil, et al. (2022) Multimodality analysis confers a prognostic benefit of a T-cell infiltrated tumor microenvironment and peripheral immune status in patients with melanoma. J Immunother Cancer 10: e005052.
- Zeynep Eroglu, Shifra Krinshpun, Ekaterina Kalashnikova, Sumedha Sudhaman, Turkan Ozturk Topcu, et al. (2023) Circulating tumor DNA-based molecular residual disease detection for treatment monitoring in advanced melanoma patients. Cancer 129(11): 1723-1734.
- GV Long, K Desai, T Tang, JS Weber, S Dolfi, et al. (2022) 788O Association of pre-treatment ctDNA with disease recurrence and clinical and translational factors in patients with stage IIIB-D/IV melanoma treated with adjuvant immunotherapy (CheckMate 915). Ann Oncol 33(7): S904.
- Yared Hailemichael, Daniel H Johnson, Noha Abdel-Wahab, Wai Chin Foo, Salah-Eddine Bentebibel, et al. (2022) Interleukin-6 blockade abrogates immunotherapy toxicity and promotes tumor immunity. Cancer Cell 40(5): 509-523.e6.
- Johannet P, Liu W, Fenyo D, Wind-Rotolo M, Krogsgaard M, et al. (2022) Baseline Serum Autoantibody Signatures Predict Recurrence and Toxicity in Melanoma Patients Receiving Adjuvant Immune Checkpoint Blockade. Clin Cancer Res Off J Am Assoc Cancer Res 28(18): 4121-4130.
- Cho U, Im S, Park HS (2024) Exploring histological predictive biomarkers for immune checkpoint inhibitor therapy response in non-small cell lung cancer. J Pathol Transl Med 58(2): 49-58.
- Rebecca C Simpson, Erin R Shanahan, Marcel Batten, Irene LM Reijers, Mark Read, et al. (2022) Diet-driven microbial ecology underpins associations between cancer immunotherapy outcomes and the gut microbiome. Nat Med 28(11): 2344–2352.
- Pinato DJ, Howlett S, Ottaviani D, et al (2019) Association of Prior Antibiotic Treatment with Survival and Response to Immune Checkpoint Inhibitor Therapy in Patients with Cancer. JAMA Oncol 5(12): 1774-1778.
- Yousefi Y, Baines KJ, Maleki Vareki S (2024) Microbiome bacterial influencers of host immunity and response to immunotherapy. Cell Rep Med 5(4): 101487.






























