Targeting Overexpressed Aromatase in Estrogen Positive Human Cancers: Isorottlerin Emerged as Potential Inhibitor of Aromatase in Computational Docking and Dynamics Simulation Study
Phani Kumar Reddy1*, Aruna Talatam2, Anuradha Vanam3* and Rao Gollapudi4
1Internal Medicine, NRI Academy of Medical Sciences, Guntur-522503, India
2Pulmonary Medicine, NRI Academy of Medical Sciences, Guntur-522503, India
3Sri Venkateswara University, Tirupathi-517502, India
4The University of Kansas, Lawrence, Kansas-66045, USA
Submission: August 26, 2023; Published: September 08, 2023
*Corresponding Address:Phani Kumar Reddy, Internal Medicine, NRI Academy of Medical Sciences, Guntur-522503, India
How to cite this article: Phani Kumar R, Aruna T, Anuradha V, Rao G. Targeting Overexpressed Aromatase in Estrogen Positive Human Cancers: Isorottlerin Emerged as Potential Inhibitor of Aromatase in Computational Docking and Dynamics Simulation Study. Canc Therapy & Oncol Int J. 2023; 25(1): 556153. DOI:10.19080/CTOIJ.2023.25.556153
Abstract
Background: Aromatase enzyme activity is a key step in the biosynthesis of estrogens. Aromatase is a monooxygenase coded by gene CYP19A1 which catalysis myriad biological reactions associated with the biosynthesis of steroids. Over expression of aromatase is routinely observed in estrogen-responsive breast cancer cells of postmenopausal women. Aromatase inhibitors such as anastrozole, exemestane and letrozole are predominantly utilized to treat these hormone dependent breast cancers. However, drug resistance and side effects are commonly associated in such treatments.
Objective: The present study investigates the inhibitory activity of isorottlerin (a novel Mallotus philippinensis derived flavanone) compared against known aromatase kinase inhibitors, namely anastrozole, exemestane and letrozole.
Methods: Anastrozole, exemestane and letrozole together with isorottlerin were docked on human placenta aromatase. The 3D structure of aromatase (PDB id: 3s7s) employed for docking studies using Autodock vina, MGL tools as well as molecular dynamic studies utilizing the iMOD server.
Results: Computational studies revealed isorottlerin as a competitive aromatase kinase inhibitor compared against FDA approved drugs, specifically anastrozole, exemestane and letrozole. In addition, docking studies results suggested that similar interactions in the binding site of aromatase. The binding energies of docked anastrozole, exemestane, letrozole and isorottlerin complexes with aromatase kinase were -7.8, -10.7, -8.4 and -11.0 kcal/mol, respectively. The docking results were supported through molecular simulation studies using the iMOD server.
Conclusion: The in silico docking simulations of anastrozole, exemestane, letrozole and isorottlerin suggested that isorottlerin is a competitive inhibitor of aromatase kinase.
Keywords:Cancer; Aromatase kinase; Cell cycle; Anastrozole; Exemestane and letrozole; Isorottlerin; In silico docking; Molecular dynamics
Introduction
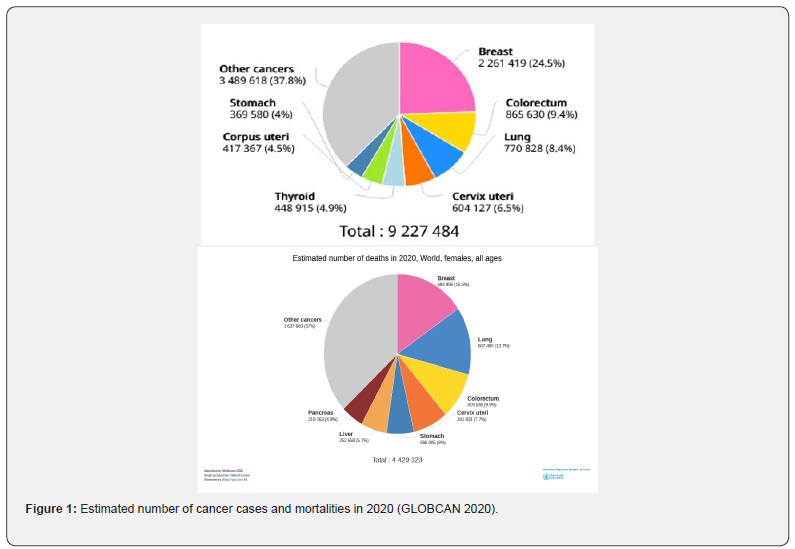
Cancer is responsible for the majority of deaths and health ailments in humans. In 2020, approximately 19.3 million new cancer cases were reported globally of which 10.0 million resulted in cancer related deaths. Despite the advancement in diagnostics and therapeutics, cancer incidences and associated mortalities continue to rise at an alarming rate [1]. Cancer occurs in various body parts resulting in a variety of cancer types. In stage 4, cancer metastasizes through angiogenesis to other body parts via circulatory and lymphatic systems. In the USA, lung and bronchial cancer mortalities are higher than any other cancer types [2]. In 2020, the number of cancer cases and mortalities were predicted as illustrated in the following pie chart (Figure 1) (GLOBCAN 2020).
The prognosis of breast cancer is relatively poor once it spreads to distant organs via metastasis. Metastatic breast cancer cells obtain aggressive characteristics from the tumor micro-environment through epithelial-mesenchymal transition and epigenetic regulatory mechanisms. Cytochrome p450 19A1 (CYP19A1; EXC 1.14.1) is an aromatase produced within endoplasmic reticulum by estrogen-secreting cells that converts androgens to estrogens. Human placental aromatase coded by gene CYP19A1 located in chromosome 15 is essential for aromatization of androstenedione to estrone. In female populations, breast cancer is the leading cause of death. In healthy women, estrogen is primarily secreted not only in ovaries but also in breast, bone, skin and adipose tissues. However, post-menopause, estrogen is produced primarily in breast tissue. In addition, around 60% of pre- menopausal and 75% of post-menopausal cancers are estrogen dependent. The transformation process of estrogen biosynthesis comprises hydroxylation of androgens 19-metyl group followed by concurrent elimination of the methyl group which resulted in the aromatization of the A-ring (Figure 2) [3].
The over expression of aromatase plays a vital role in the development and progression of estrogen receptor alpha (ERa) positive breast cancers ultimately acquiring “anoikis” resistance. To prevent metastasis, cells normally experience a form of apoptosis known as “anoikis”, a form of cell death as a result of loss of contact with the extracellular matrix or adjacent cells. Nevertheless, cancer cells develop “anoikis” resistance to survive after disengaging from the primary locations and migrating through the circulatory and lymphatic systems that distribute throughout the body leading to metastasis [4]. Estrogens overexpression promotes cancer cell growth, proliferation, metastasis, and recurrence of hormonal dependent breast cancers. Therefore, reduction of estrogen levels through catalytic aromatase inhibition is effective in breast cancer management. In the early 1970s, the most successful breast cancer treatment involved the discovery of drugs targeting estrogen signalling pathways [5]. Increased aromatase activity is generally influenced by age, alcohol consumption, obesity, as well as gonadotropin and insulin concentrations. In males, sudden bursts in testosterone levels contributed towards increased aromatase activity. The reduction of estrogen levels in males is a potential treatment for myriad of disorders including andropause, gynecomastia and pubertas praecox [6]. Furthermore, over expression of aromatase had also been noticed in hepatitis and hepatocellular carcinomas [7]. Multiple studies implicated that aromatase inhibitors are superior to anti-estrogens in the treatment of breast cancers [8]. A plethora of drugs in treatment of estrogen receptor-positive breast cancer commonly interfere with either estrogen production or estrogen action [9]. At present, third generation non-steroidal aromatase inhibitors like anastrozole and letrozole are utilized in the treatment of post-menopausal breast and ovarian cancers [10]. In addition, the steroid, exemenstane is used to irreversibly bind to the aromatase enzyme and thereby inhibiting the enzyme activity [11]. Nonetheless, the side effects of non-steroidal drugs, anastrozole and letrozole include joint and muscle pain, menopausal symptoms, depression, sleeplessness, osteoporosis, high cholesterol, carpal tunnel syndrome, blood clots and fatigue [12,13]. Therefore, there is an increased demand for discovery of potent and novel aromatase inhibitors with reduced side effects.

The plant species, Mallotus philippinensis (Family: Euporbiaceae) is a rich source of natural compounds which includes cardenolides, coumarins, diterpenoids, flavonoids, isocoumarins, phenols, steroids, and triterpenoids. The flavanones isolated from M. philippinensis such as isorottlerin, kamalachalcone A, kamalachalcone B, mallotophilippinen A-E and rottlerin exhibited antimicrobial, antioxidant, antiviral, anti-inflammatory, cytotoxicity, immuno-regulatory protein inhibition activity against variety of cancer cells [14-17]. A novel flavanone, isorottlerin with a distinctive ring system resulted by dimerization between a dimethylchromene ring and a phenoxyl group was isolated from kamala dye (M. philippinensis) [18]. In the present investigation, M. philippinensis flavonoids; isoallorottlerin, isorottlerin, kamalachalcone A, kamalachalcone B, mallotophilippen A-E and rottlerin (Figure 3) were investigated by molecular docking studies to establish their inhibitory potency against aromatase enzyme activity.
The resultant data was compared against approved aromatase inhibitor drugs, namely anastrozole, letrozole, and exemestane (co-crystallized with aromatase enzyme) (Figure 4). Subsequently, the most effective inhibitor, isorottlerin was subjected to molecular dynamic simulation studies along with exemestane to further support the results of the molecular docking observations.
Materials and Methods
Lipinski’s rule of five
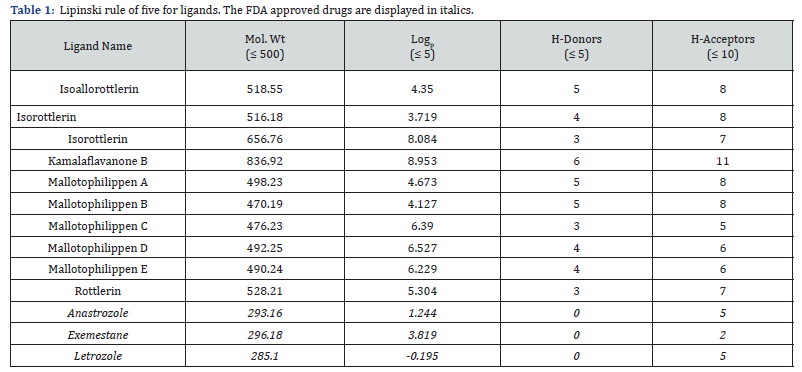
At the initial stages of drug discovery, the evaluation of druglikeness of lead molecules abets in decreasing the operational costs. The leading hits should not disobey more than one criterion as defined by the Lipinski’s “rule of five”. These rules include molecular weight (no greater than 500 Daltons or g/mol), octanol-water partition coefficient (Logp of 5 or less), hydrogen bond donors (5 or less) and acceptors (10 or less). The druglikeliness tool software (DruLiTo) was used to determine the physicochemical characteristics of isoallorottlerin, isorottlerin, kamalachalcone A, kamalachalcone B, mallotophilippen A-E and rottlerin including anastrozole, and letrozole along with co-crystallized ligand, exemenstane (RCSB PDB ID: 3s7s) [19]. Nonetheless, Lipinski’s rule of five does not apply to the substrates of biological transporters or natural products. Certain highprofile drugs such as lipitor and singulair fail more than one of the Lipinski’s rules. Besides, the mean molecular properties of new pharmacological compounds are still credited Lipinski compliant although their property distributions are distant from historical standards. Hence, widely used rules for new drug discovery do not have to comply with Lipinski’s rule of five [20].
Receptor protein preparation
The X-ray diffraction crystal structure of aromatase domain and co-crystalized ligand complex (PDB ID: 3s7s) with 2.75 Å resolution was retrieved from RCSB protein data bank. The protein structure of aromatase in complex with ligand and co-crystallized water contributed for elevated resolution and stability of protein. Therefore, water molecules and ligand were removed using Discovery Studio software, 2021 retaining chain A of aromatase for docking study.

Ligands and target preparations
The two-dimensional structures of isoallorottlerin, isorottlerin, kamalachalcone A, kamalachalcone B, mallotophilippen A-E, rottlerin, anastrozole, and letrozole were designed with the help of ChemDraw 21.0 and later converted to 3D structure using Chem3D 21.0 software. The 3D structure of exemestane was extracted from the aromatase co-crystalized ligand complex (PDB ID: 3s7s).
Docking between aromatase kinase and ligands
Molecular docking studies were achieved with the help of AutoDock 4.6.2 software to generate PDBQT files of ligands and aromatase kinase from PDB files. The Kollman partial charges were allocated to each specific atom of the protein for PDBQT format whereas Geister charges were assigned to ligands. Molecular docking of ligands with aromatase kinase was performed using Autodock Vina 1.1.2 (Windows) to explore the best sites for receptor-ligand bonding and to establish the most stable free energy of the receptor-ligand complex.
The 3D grid box with X=25, Y=25 and Z=25 dimensions containing a grid box center with x=86.012, y=54.602 and z=46.539 coordinates were used to cover the active binding site of the receptor molecule. These parameters were used to generate a configuration file for grid mapping and docking for Autodock Vina study. An independent run was conducted for each ligand and each apparent conformer was docked in a randomized order into the binding pocket of aromatase kinase. The data obtained for each docked receptor ligand complex was analyzed [21]. Subsequently, LigPlot+ v.4.5.3 software was employed to compute the number of hydrophobic and hydrogen bonds between ligands and aromatase kinase complexes at the binding site. The type and number of amino acids that are in the binding site were identified by LigPlot + 4.5.3 software [22].
Molecular dynamics simulation study
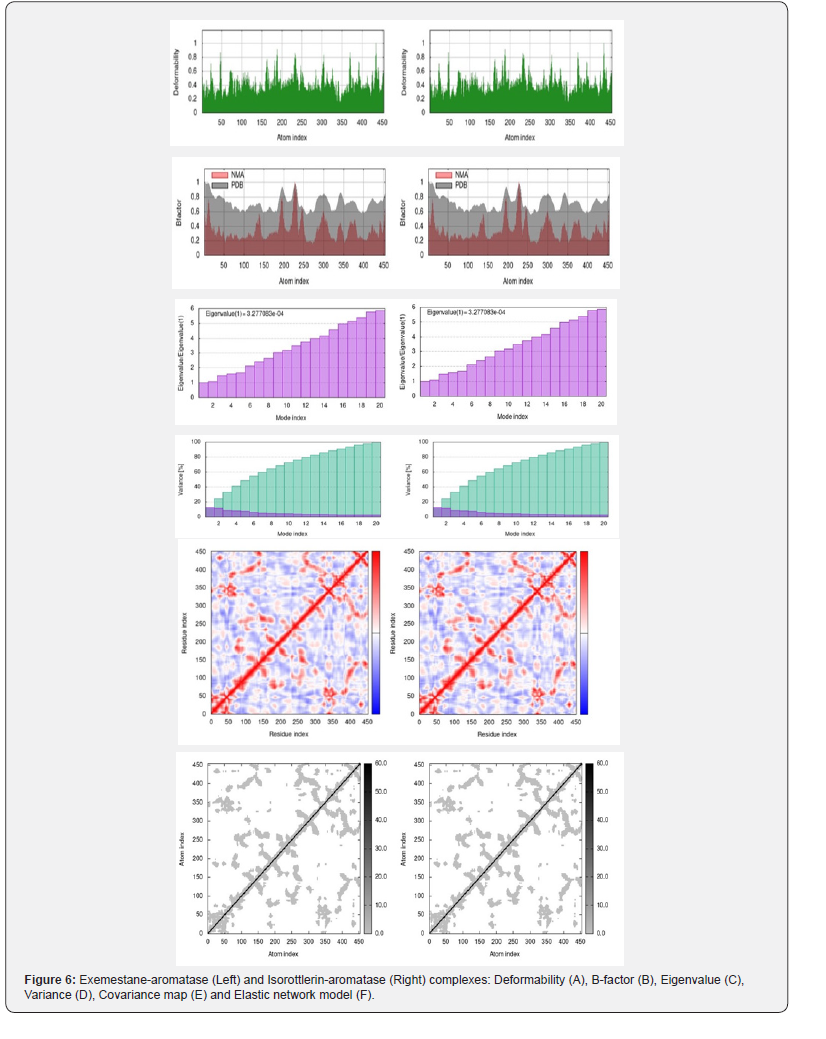
The iMOD software and server (iMODS) generates Molecular Dynamic (MD) simulations to conduct Normal Mode Analysis (NMA) in internal coordinates (IC) on protein atomic structures to reconstruct the collective functional motions of biological macromolecules. The iMOD server allows the generation of feasible transition pathways between two homologous molecular structures. The specific internal coordination increases the efficacy of normal mode analysis (NMA) while extending its application and simultaneously maintaining stereochemistry. Vibrational analysis, motion animations as well as morphing trajectories can be accomplished at different resolutions. In this framework, iMOD offers progressive visualization capabilities for depicting collective motions and an improved affine-model-based arrow depiction of domain dynamics. The resulting all-heavyatoms confirmations are applicable for more advanced modeling that utilizes structural flexibility. The binding effects of ligands on aromatase kinase were estimated basing on conformational stability of protein/ligand complex interactions with NMA using iMOD server. During this time, several properties such as deformability and eigenvalues of protein/ligand interactions, variance, covariance and elastic factors were considered [23,24].
Results
Lipinski’s rule of five for ligands
The drug likeliness tool software (DruLiTo) was utilized to evaluate the physicochemical features of the molecular weight, Logp, H-donors and H acceptors of isoallorottlerin, isorottlerin, isorottlerin, kamalaflavanone B, mallotophilippen A-E, rottlerin, anastrozole and letrozole along with the co-crystallized ligand exemestane with aromatase kinase (RCSB PDB ID: 3s7s) (Table 1).
Docking study
The anastrozole, exemestane and letrozole, along with eleven naturally occurring flavanonoids: isoallorottlerin, isorottlerin, isorottlerin, kamalachalcone A, kamalaflavanone B, mallotophilippen A-E, rottlerin docked with aromatase are listed in Table 2.
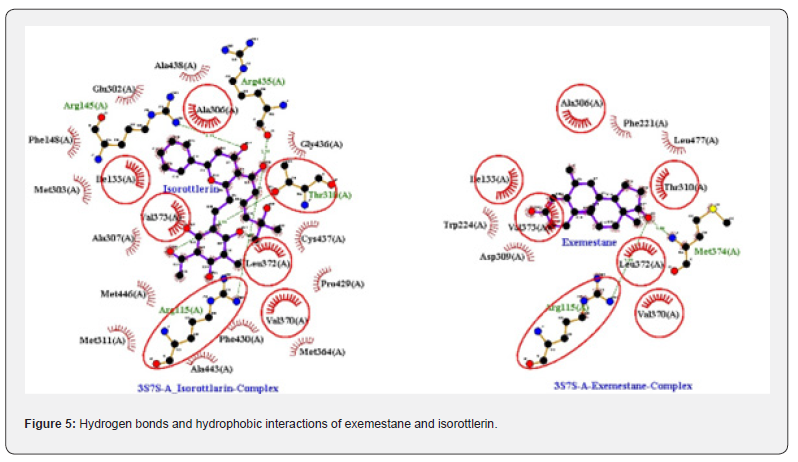
The results from molecular docking of isorottlerin with aromatase kinase were used to reveal the best sites for the interactions of ligand to receptor in addition to specify the most stable free energy of ligand/receptor along with exemestane. Isorottlerin formed four hydrogen bonds at Arg115 (2.82 Ao), Arg145 (3.19 Ao), Thr 310 (2.00 Ao) and Arg435 (2.74 Ao) as well as hydrophobic interactions at Ile133, Phe148, Glu302, Met303, Ala306, Ala307, Thr310, Met311, Met364, Val370, Leu372, Pro429, Ala438, Phe430, Gly436, Cys437, Ala443, Met446, amino acid residues of aromatase kinase. Nonetheless, exemestane formed two hydrogen bonds at Arg115 (2.80 Ao) and Met374 (2.80 Ao) as well as hydrophobic interactions Ile133, Phe221, Trp224, Ala306, Asp309, Thr310, Val370, Leu372, Val373 and Leu477 amino acid residues of aromatase kinase (Figure 5).
Molecular Dynamic Simulation study
The stability of each protein-ligand complex was assessed through Molecular Dynamic (MD) simulations utilizing iMOD software and server (iMODS) to conduct NMA in internal coordinates (IC) on protein atomic structure of aromatase. Comparative analysis of the aromatase-ligand complexes suggested that isorottlerin displayed equally good data analogous to those of exemestane. Specifically, the Deformability (A), B-factor (B), Eigenvalue (C), and Variance (D) plots for each complex were almost identical to one another. In addition, the binding interactions of isorottlerin and exemestane induced almost identical changes within the aromatase kinase receptor, as evidenced by the fact that the covariance maps (E) and elastic network models (F) for each complex were indistinguishable from one another. The molecular dynamics simulation data generated by the external iMOD server for Deformability (A), B-factor (B), Eigenvalue (C), Variance (D) plots; Covariance map (E) and Elastic network model (F) of aromatase -isorottlerin and aromataseexemestane complexes are presented in Figure 6. Exemestane (Left) vs Isorottlerin (Right) (No deviation observed in data)

mM = Micro-molar; nM = Nano-molar; pM= Pico-molar
Discussion
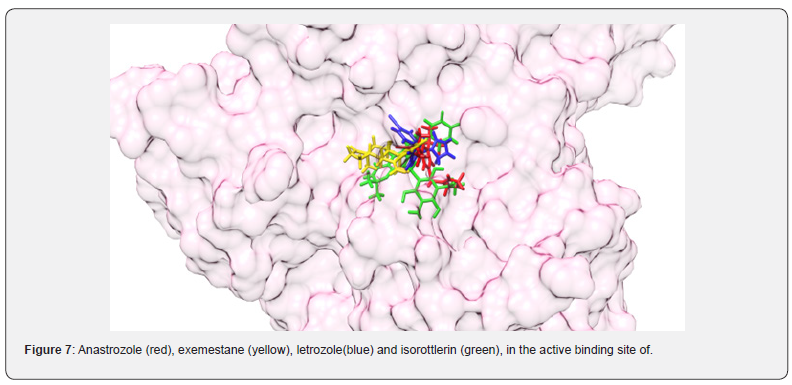
Aromatase inhibitors can reduce the over production of estrogen in the body. Estrogen receptor positive (ER+) breast cancers utilize estrogen for cell proliferation. Prior to the menopause (pre-menopause), estrogen is predominantly produced in the ovaries. The ovaries cease estrogen production after the menopause (post-menopause). However, small quantities of estrogen are still produced in adipose tissues with the aid of the aromatase enzyme. As such, aromatase inhibitors prevent the aromatase enzyme from generating estrogen and thus there is less estrogen present to induce further ER+ breast cancer cell proliferation [25]. The over expression of aromatase plays a vital role in the development and progression of “anoikis” resistant ERa breast cancers. Numerous FDA approved reversible nonsteroidal (anastrozole and letrozole) and irreversible steroidal (exemestane) aromatase kinase inhibitors induce promising in vitro as well as in vivo anticancer activities against a variety of cancer types [26]. The current study revealed that among these FDA approved therapeutics, exemestane (-10.7 kcal/mol) produced the greatest binding energy in complex with the aromatase kinase enzyme. Furthermore, in silico studies found that isorottlerin produced even greater binding energy (-11.0 kcal/mol) with the aromatase enzyme. In this context, isorottlerin being a nonsteroidal aromatase inhibitor with higher binding energy might be functioning as a reversible non-steroidal aromatase inhibitor. The iMODS data revealed that both complexes exhibited almost similar behaviour. Exemestane can produce a variety of negative side effects such as arthralgias and myalgias, decreased libido, and sleep disturbances [27]. Combined, these MD data generated by external iMOD server strongly suggests that isorottlerin could be a better alternative to exemestane. The interactions of anastrozole (red), exemestane (yellow), letrozole (blue) and isorottlerin (green), in the active binding site of aromatase kinase were generated using Chimera as displayed in Figure 7.
Conclusion
Cancer is the second most common cause of death in the United States, next to heart disease. Computational approaches have been utilized for the discovery and development of new drugs as multi-targeted inhibitors for diseases caused by a variety of overexpressed proteins. The present study focused on targeting the excessive aromatase kinase levels responsible for uncontrolled cell growth in a plethora of cancers. Molecular docking of aromatase kinase with a variety of inhibitors revealed that isorottlerin-aromatase complex (11.0 kcal/mol,) produced a higher binding energy compared to that of Exemestane-aromatase (10.7 kcal/mol). Thus, isorottlerin exhibited significantly higher binding affinity towards the active binding site of aromatase kinase comparable to FDA approved orphan drug, Exemestane. Therefore, isorottlerin could be a promising lead candidate helpful in the treatment of aromatase kinase overexpressed cancer for the prevention of cell cycle progression. Consequently, in vitro, and in vivo evaluation of isorottlerin as a novel aromatase kinase inhibitor is recommended.
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this manuscript.
Acknowledgment
R.G4. is grateful to Late Professor Lester A. Mitscher, Late Professor Delbert M. Shankel and Late Professor Andrew M. Torres in The University of Kansas (KU), Lawrence, Kansas, USA for introducing to computational docking studies at the earlier stages of drug discovery. The authors appreciate Professor Noboru Motohashi, Meiji University, Tokyo, Japan for his encouragement to pursue this study.
References
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, et al. (2021) Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 71(3): 209-249.
- Ridge CA, McErlean AM, Ginsberg MS (2013) Epidemiology of lung cancer. Semin Intervent Radiol 30(2): 93-98.
- Thomas MP, Potter BV (2013) The structural biology of oestrogen metabolism. J Steroid Biochem Mol Biol 137: 27-49.
- Wang TT, Phang JM (1995) Effects of estrogen on apoptotic pathways in human breast cancer cell line MCF-7. Cancer Res 55(12): 2487-2489.
- Jordan VC, Brodie AM (2007) Development and evolution of therapies targeted to the estrogen receptor for the treatment and prevention of breast cancer. Steroids 72(1): 7-25.
- de Ronde W, de Jong FH (2011) Aromatase inhibitors in men: effects and therapeutic options. Reprod Biol Endocrinol RB&E 9: 93.
- Hata S, Miki Y, Saito R, Ishida K, Watanabe M, et al. (2013) Aromatase in human liver and its diseases. Cancer Med 2(3): 305–315.
- Wang J, Gildea JJ, Yue W (2013) Aromatase overexpression induces malignant changes in estrogen receptor α negative MCF-10A cells. Oncogene 32(44): 5233–5240.
- Fontham ET, Thun MJ, Ward E, Portier KM, Balch AJ, et al. (2009) American Cancer Society perspectives on environmental factors and cancer. CA Cancer J Clin 59(6): 343–351.
- Varela C, Tavares da Silva EJ, Amaral C, Correia da Silva G, Baptista T, et al. (2012) New structure-activity relationships of A- and D-ring modified steroidal aromatase inhibitors: design, synthesis, and biochemical evaluation. J Med Chem 55(8): 3992–4002.
- Martin GD, Narvaez J, Bulmer R, Durrant MC (2016) Biotransformation and molecular docking studies of aromatase inhibitors. Steroids 113: 95–102.
- Dixon JM, Renshaw L, Langridge C, Young OE, McHugh M, et al. (2011) Anastrozole and letrozole: an investigation and comparison of quality of life and tolerability. Breast Cancer Res Treat 125(3): 741–749.
- Morimoto Y, Sarumaru S, Oshima Y, Tsuruta C, Watanabe,K (2017) Joint symptoms associated with anastrozole and letrozole in patients with breast cancer: a retrospective comparative study. J Pharm Health Care Sci 3: 25.
- Ma J, Hou Y, Xia J, Zhu X, Wang ZP (2018) Tumor suppressive role of rottlerin in cancer therapy. Am J Transl Res 10(11): 3345–3356.
- Gangwar M, Goel, RK, Nath G (2014) Mallotus philippinensis Muell. Arg (Euphorbiaceae): ethnopharmacology and phytochemistry review. BioMed Research International 2014: 213973.
- Lounasmaa M, Widen CJ, Tuuf CM, Huhtikangas A (1975) On the phloroglucinol derivatives of Mallotus philippinensis. Planta med 28(1): 16–31.
- Zaidi SF, Yoshida I, Butt F, Yusuf MA, Usmanghani K, et al. (2009) Potent bactericidal constituents from Mallotus philippinensis against clarithromycin and metronidazole resistant strains of Japanese and Pakistani Helicobacter pylori. Biol Pharm Bull 32(4): 631–636.
- Tanaka T, Ito T, Iinuma M, Takahashi Y, Naganawa H (1998) Dimeric chalcone derivatives from Mallotus philippensis. Phytochemistry 48(8): 1423–1427.
- Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (2001) Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Advanced Drug Delivery Reviews 46(1-3): 3–26.
- Bickerton GR, Paolini GV, Besnard J, Muresan S, Hopkins AL (2012) Quantifying the chemical beauty of drugs. Nature Chemistry 4(2): 90–98.
- Morris GM, Goodsell DS, Halliday RS, Huey R, Hart WE, et al. (1998) Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. Journal of Computational Chemistry 19(14): 1639-1662.
- Laskowski RA, Swindells, MB (2011) LigPlot+: multiple ligand-protein interaction diagrams for drug discovery. Journal of Chemical Information and Modeling 51(10): 2778–2786.
- Lopez-Blanco JR, Garzon JI, Chacon P (2011) iMod: multipurpose normal mode analysis in internal coordinates. Bioinformatics (Oxford, England) 27(20): 2843–2850.
- Kovacs JA, Chacon P, Abagyan R (2004) Predictions of protein flexibility: first-order measures. Proteins 56(4): 661–668.
- Chumsri S, Howes T, Bao T, Sabnis G, Brodie A (2011) Aromatase, aromatase inhibitors, and breast cancer. J Steroid Biochem Mol Biol 125(1-2): 13–22.
- Tenti S, Correale P, Cheleschi S, Fioravanti A, Pirtoli L (2020) Aromatase Inhibitors-Induced Musculoskeletal Disorders: Current Knowledge on Clinical and Molecular Aspects. Int J Mol Sci 21(16): 5625.
- Robinson A (2009) A review of the use of exemestane in early breast cancer. Ther Clin Risk Manag 5(1): 91–98.






























