- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Chemotherapy-Induced Neuropathic Pain and Underlying Mechanisms
Ziyi Liu, Sitong Liu, Yu Zhao and Qian Wang*
Tumor Center, The First Hospital of Jilin University, Changchun, Jilin 130021, China
Submission: July 05, 2023; Published: July 21, 2023
*Corresponding Address:Qian Wang, Tumor Center, The First Hospital of Jilin University, Changchun, 71 Xinmin Street, Changchun, Jilin 130021, China, Email: helloliudongxue@jlu.edu.cn
How to cite this article: Ziyi L, Sitong L, Yu Z, Qian W. Chemotherapy-Induced Neuropathic Pain and Underlying Mechanisms. Canc Therapy & Oncol Int J. 2023; 24(4): 556142. DOI:10.19080/CTOIJ.2023.24.556142
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Abstract
One of the most common issues caused by antineoplastic agents is chemotherapy-induced peripheral neuropathy (CIPN). In patients, CIPN is a sensory neuropathy accompanied by various motor and autonomic changes. With a high prevalence of cancer patients, CIPN is becoming a major problem for both cancer patients and for their health care providers. Nonetheless, there are lacking effective interventions preventing CIPN and treating the CIPN symptoms. Several current studies have demonstrated the cellular and molecular signaling pathways leading to CIPN using experimental models and the beneficial effects of some interventions on the CIPN symptoms related to those potential mechanisms. This review summarizes results obtained from recent human and animal studies, which include the abnormalities in mechanical and temperature sensory responses following chemotherapeutic agents such as representative bortezomib, oxaliplatin and paclitaxel. The underlying mechanisms of CIPN at cellular and molecular levels are also discussed for additional in-depth studies needed to be better explored. A better understanding of the risk factors and fundamental mechanisms of CIPN is needed to design effective preventive drugs and therapeutic strategies. Overall, this paper reviews the basic picture of CIPN and the signaling mechanisms of the most common antineoplastic agents and potential approaches to be considered to alleviate neuropathic pain observed during chemotherapy.
Keywords: Chemotherapeutic drugs; Inflammatory cytokines; Oxidative stress; Neuropathy
Abbreviations: CIPN: Chemotherapy-Induced Peripheral Neuropathy; BTZ: Bortezomib; OXL: Oxaliplatin; PLT: Paclitaxel; EA: Electroacupuncture; PIC: Proinflammatory Cytokine; IL-1β- Interleukin-1β; IL-6- Interleukin-6; TNF-α: Tumor Necrosis Factor-α; DRG: Dorsal Root Ganglion; PAR2: Proteinase-Activated Receptor 2; TRPA1: Transient Receptor Potential Ankyrin 1; PKC: Protein kinase C; PKA: Protein kinase A; TRPV1: Transient Receptor Potential Vanilloid 1; TRPM8: Transient Receptor Potential Channel Subfamily M8; CCR2: CC Chemokine Receptor 2; CCL2: C-C Motif Chemokine Ligand 2; TLR4: Toll Like Receptor 4; NF-κB: Nuclear Factor Kappa-Light-Chain-Enhancer of Activated B cells; IκBα: Endogenous Inhibitor of NF-κB Alpha; ATF3: Activating Transcription Factor 3; STAT3: Signal Transducer and Activator of Transcription-3; Nrf2: Nuclear Factor (Erythroid-derived 2)-like 2; ARE: Antioxidant Response Element; HO-1: Hemeoxygenase-1; ROS: Reactive Oxygen Species; PBN: N-Tert-Butyl-Α-Phenylnitrone; NOX: NADPH Oxidase; 8-iso PGF2α: 8-Iso-Prostaglandin F2α; 8-OHdG: 8-Hydroxy-2’-Deoxyguanosine; SOD: Superoxide Dismutase; PI3K: Phosphoinositide 3-Kinase; MAPK: Mitogen-Activated Protein Kinase; mTOR: Mammalian Target of Rapamycin; JNK- c: Jun N-Terminal Kinase; S6K1: Ribosomal Protein S6 Kinase Beta-1; 4E-BP-1: Eukaryotic Translation Initiation Factor 4e-Binding Protein 1
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Introduction
Cancer is a leading cause of mortality worldwide [1]. One of the most common and distressing symptoms suffered by patients with progression of cancer is pain [2]. Cancer pain mainly arises from a tumor compressing or infiltrating tissue; from nerve and other changes caused by a hormone imbalance or immune response; and/or from treatments and diagnostic procedures [2,3]. It is promising that effective treatments including chemotherapeutic agents can increase the number of cancer survivors [1] whereas the side effects are present. Among them, one of the most common problems caused by antineoplastic drugs is chemotherapy-induced peripheral neuropathy. It should be noted that chemotherapy produces painful conditions persisting long after treatment has ended [2,4,5]. This side effect caused by chemotherapeutic agents is still a serious clinical issue for cancer patients during treatment. As a result, how to effectively manage neuropathic pain related to these therapies becomes important for treatment and management of cancer patients in clinics. Nonetheless, treatment options for these abnormal sensations have been restricted, partly due to a poor understanding of the underlying mechanisms responsible for neuropathic pain induced by chemotherapeutic agents. Also, there are lacking of effective drugs applied to patients; and costs and side effects of drugs affect a treatment strategy of neuropathic pain in patients with chemotherapy. Thus, alternative treatment approaches of neuropathic pain are noteworthy during chemotherapy on the basis of study findings of basic signaling pathways.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Chemotherapy-induced peripheral neuropathy (CIPN)
CIPN is a nerve-damaging side effect of antineoplastic agents in the common cancer chemotherapy, which involves various symptoms such as tingling, pain, and numbness in the hands and feet in addition to motor, and/or autonomic functions [6]. Patients may experience numbness, tingling, altered touch sensation, gait and balance disturbances, burning pain, thermal allodynia or hyperalgesia, impaired vibration sense, extreme temperature sensitivity, paresthesia, and/ or dysesthesia as part of sensory damage [7]. These symptoms can impair activities of daily living, such as typing or dressing, reduce balance, and increase risk of falls and hospitalizations, which can cause to decrease and/or discontinue chemotherapy in cancer patients.
Nonetheless, there are lacking effective interventions preventing CIPN and treating the CIPN symptoms. Therefore, it is important to explore the underlying mechanisms leading to CIPN and develop effective preventive drugs and therapeutic strategies.
As following, we will discuss results obtained from recent human and animal studies, including the abnormalities in mechanical pain and cold sensitivity response following representative chemotherapeutic drugs. This provides a basic picture of CIPN and the signaling mechanisms of the most common antineoplastic agents such as representative bortezomib (BTZ), oxaliplatin (OXL) and paclitaxel (PLT). The underlying mechanisms of CIPN at cellular and molecular levels are also reviewed for additional in-depth studies needed to be better explored. A better understanding of the risk factors and underlying mechanisms of CIPN is needed to develop and design effective preventive drugs, therapeutic strategies and management of cancer patients.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Chemotherapeutic agents causing neuropathy
There are six main agent groups found in chemotherapy treatment that damage the sensory, motor, and autonomic neurons and therefore cause CIPN [1,6,7]. They include proteasome inhibitors, platinum-based compounds, taxanes, vinca alkaloids, epothilones, and immunomodulatory drugs (Table 1). In this review, we focus on the representative chemotherapeutic agents, which have been recently studied with respect to the signaling pathways responsible for neuropathic pain, potentially considered to be targeted in improving neuropathy as a new drug is developed and designed.
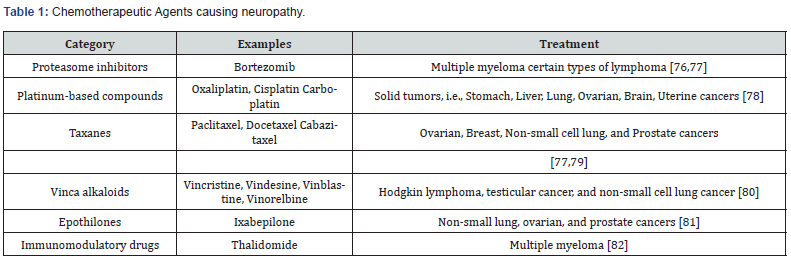
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
There are many signaling pathways in peripheral nerve system responsible for CIPN. The presented Figure 1 partly summarizes signaling pathways associated with neuropathic pain after antineoplastic treatment. Specifically, inflammation and oxidative stress are generally involved in the process of CIPN. The detailed discussion is followed as below. It is also interesting to pay attention to several interventions to improve neuropathic pain in experimental models discussed in each section.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Pro-inflammatory cytokines (PICs)
PICs are produced predominantly by activated macrophages and are involved in the up regulation of inflammatory reactions. There is abundant evidence that certain pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α are involved in the process of pathological pain. Furthermore, BTZ, OXL and PLT were reported to increase the level of IL-1β, IL-6 and TNF-α in the sensory nerve-dorsal root ganglion (DRG) of rats thereby inducing pain behaviors [8,9]. Consistent with these findings, blocking PIC signaling pathways inhibits mechanical and thermal hypersensitivity [10,11].
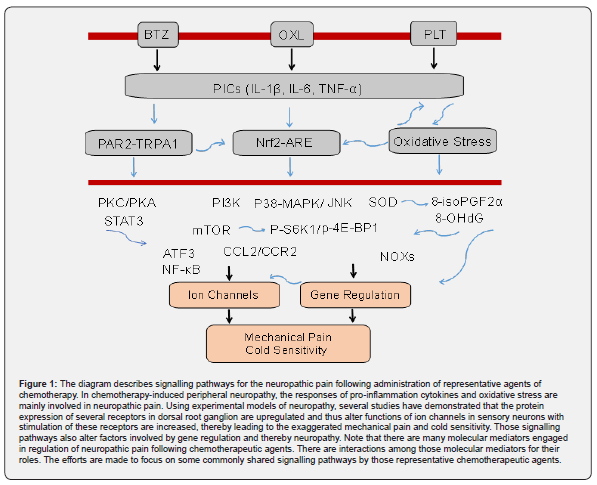
In particular, via PIC signaling proteinase-activated receptor 2 (PAR2) and transient receptor potential ankyrin 1 (TRPA1) are engaged in neuropathic pain induced by BTZ in rats [11]. Results of this prior study demonstrated that systemic injection of BTZ increased mechanical pain and cold sensitivity as compared with control animals. Data of this report further showed that blocking respective PAR2 and TRPA1 attenuated mechanical pain and cold sensitivity observed in control rats and BTZ rats. Notably, the attenuating effect of blocking PAR2 and TRPA1 on mechanical pain and cold sensitivity was significantly smaller in BTZ rats than that in control rats. In addition, protein expression of PAR2 and TRPA1 was upregulated in the lumbar DRG of BTZ rats, and inhibition of PAR2 decreased the levels of TRPA1 and attenuated its downstream pathways (namely, PKCɛ and PKA). This suggests that there is a specific regulatory pathway PAR2- TRPA1 in involvement of BTZ-induced neuropathic pain and that intracellular PKCɛ and PKA play a role [11]. In the similar way, blocking PAR2 also significantly decreased TRPA1 expression in the DRG tissue and attenuated mechanical pain and cold sensitivity observed in control rats and OXL rats [10]. Also, the attenuating effect of PAR2 on mechanical pain and cold sensitivity was significantly smaller in OXL-rats than that in controls.
On top of TRPA1, OXL induced up-regulation of the mRNA of the TRPV1 and TRPM8 in cultured DRG neurons [12] whereas OXL causes cold allodynia and enhance the sensitivity and expression of TRPM8 and TRPA1. Increased levels of CCL2 and CCR2 accompanied by mechanical hypersensitivity [13,14] were found in experimental model after application of OXL. IL-8 signaling pathway was also reported to be involved in neuroinflammation [15] and microbiota -TLR4 activation on macrophages.
In addition, several studies have implicated dysregulation of transcription factors in the DRG in the development of BTZinduced CIPN. Following BTZ administration, nuclear factor kappalight- chain-enhancer of activated B cells (NF-κB) is increased in intranuclear fractions, indicating activation of the NF-κB signaling pathway [16,17]. Consistent with this finding, transgenic mice expressing a dominant negative form of IκBα (an endogenous inhibitor of NF-κB), which inhibited NF-κB translocation to the nucleus by preventing endogenous IκBα degradation, did not develop CIPN [17]. Moreover, BTZ-induced axonal degeneration of the sciatic nerve was less severe in these transgenic mice, while decreased density of intraepidermal nerve fibers occurred at the same level observed in wild type mice [17]. Upregulation of activating transcription factor 3 (ATF3) is also reportedly involved in BTZ-induced CIPN. In DRG neurons of CIPN rodents, ATF3- immunoreactivity was increased in the nucleus, whereas vehicletreated animals rarely exhibited immunoreactivity [18,19]. The increase of ATF3 elicited by BTZ enhanced the recruitment of c-Jun, a transcription factor known to form a heterodimer with ATF3 that binds the promoter region of CCL2, ultimately resulting in upregulation of CCL2 and the development of neuropathic pain [19]. Signal transducer and activator of transcription-3 (STAT3) is also highly phosphorylated in DRG neurons following BTZ injection [20]. Indeed, both pharmacological or genetic inhibition of STAT3 and treatment prevent the development of BTZ-induced mechanical allodynia in rats and mice [20].
Likewise, PLT was observed to amplify IL-1β, IL-6 and TNF-α in the DRG whereas it increased mechanical and thermal hypersensitivity [21]. Interestingly, EA attenuated increases in IL-1β, IL-6 and TNF-α after PLT and attenuated mechanical and thermal hypersensitivity. Results of this previous report suggest that downregulation of IL-1β, IL-6 and TNF-α in the DRG is involved in the inhibitory effects of EA on mechanical pain and thermal hypersensitivity in PLT-induced neuropathy [21]. It should be noted that other general signaling pathways involved in BTZ and OXL were also found in experimental models of PLT including NF-κB, CCL2/CCR2, TLR4 etc [1,7,22]. This suggests that BTZ, OXL and PLT have a common regulatory mechanism in engagement of neuropathic pain and it is significant to target those signaling pathways for improve symptoms of neuropathy during administration of chemotherapeutic drugs in patients.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
The role of oxidative stress in CIPN has been studied. A large number of studies have demonstrated that BTZ, OXL and PLT increase products of oxidative stress and thus stimulate signaling pathways of oxidative stress in CINP [1,6,7,22]. The side effects due to antineoplastic agents occur due to activation of nuclear factor NF-κB and free radical injury is also involved in the antineoplastic agent-mediated neurotoxicity as dysregulation of nuclear factor (erythroid-derived 2)-like 2 (Nrf2) is observed. Note that Nrf2 is a transcription factor and as a basic leucine zipper protein it regulates the expression of antioxidant proteins protecting against oxidative damage triggered by injury and inflammation [23]. Numerous drugs that stimulate the Nrf2 pathway were used for treatment of diseases caused by oxidative stress [24]. This protects against the free radical injury by regulating glutathione S-transferases and hemeoxygenase-1 (HO-1). Thus, correcting the imbalance between NF-κB and Nrf pathways is suggested to affect CINP following chemotherapy [25]. Via those pathways, BTZ and OXL have been reported to increase activities of oxidative stress and alter Nrf2-ARE mechanisms thereby leading to neuropathic pain [26-29].
Specifically, as a non-specific reactive oxygen species (ROS) scavenger, N-tert-butyl-α-phenylnitrone (PBN) was reported to inhibit the development of PLT-induced mechanical hypersensitivity [30]. Also, tempol used for supplement of ROS scavenger inhibited the development and maintenance of PLT-induced mechanical hypersensitivity but was ineffective on cold allodynia [8,30]. In addition, the activity of different antioxidant enzymes in the DRG and peripheral sensory nerves were examined during the time course of PLT-induced painful neuropathy. Enhanced activity of mitochondrial and cellular endogenous antioxidant enzymes in the DRG and peripheral nerves was observed, however this was inadequate and delayed in its onset leading to excessive ROS in peripheral sensory axons [31]. Others have demonstrated an impaired mitochondrial antioxidant response after PLT [32].
In addition to antioxidants and/or free radical scavengers, it is also interesting to determine the effects of decreasing ROS generation on PLT-induced mechanical and thermal hypersensitivity. One source of ROS production is the enzyme family of NOX. The rodent genome encodes four genes that contain the catalytic NOX subunit, namely NOX1, NOX2, NOX3, and NOX4 [33]. This electron-transferring subunit is constitutively inactive in resting cells and generates ROS only upon activation, e.g. after noxious stimuli [34]. While NOX2 activation is predominantly associated with innate immunity mediated host defense and NOX1 with blood pressure control and related vascular mechanisms [35,36], NOX4 was shown highly expressed under ischemic conditions in the central nervous system, e.g. ischemic stroke [37]. Evidence has also identified activation of NOX4 as a causative factor that contributes to inflammatory or neuropathic pain role in the peripheral nervous system [38]. A recent study further determined the effects of EA intervention on expression levels of NOX4 in the DRG of PLT rats [39]. It was observed that NOX4 was upregulated after administration of PLT and EA attenuated upregulation of NOX4 as well as increases of oxidative 8-iso PGF2α/8-OHdG.
Moreover, results of a recent study demonstrated that the expression of Nrf2-ARE signal and SOD expression were downregulated in the DRG of PLT rats as compared with control rats [21,39]. The expression of NOX4, the levels of oxidative products 8-iso PGF2α/8-OHdG and PICs such as IL-1β, IL-6 and TNF-α were amplified in the DRG of PLT rats. In those prior studies, results further demonstrated that PICs were decreased, and mechanical and thermal hypersensitivity were attenuated in PLT rats after inhibition of NOX4 and systemic supplying of SOD and antioxidant vitamin C as well as EA [21,39]. Notably, results of those studies suggest that EA intervention 1) restored impairment of Nrf2-ARE and SOD in the DRG of PLT rats; 2) inhibited amplification of NOX4 and 8-iso PGF2α/8-OHdG and PICs, thereby alleviating neuropathic pain induced by PLT. This provides an alternate potential approach to alleviate neuropathic pain for cancer patients during chemotherapy, which should be considered for application in clinical practice before a better drug is developed and designed.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Interventions considered for beneficial effects on neuropathy
The beneficial role played some approaches in alleviating neuropathic pain induced by chemotherapeutic drugs have been recently investigated and they should be included in the future considerations.
Factors by gene regulation involved in neuropathy
MicroRNAs (miRNAs) are small noncoding endogenous RNA molecules, repressing their target mRNA through complementary binding in the message 3’-UTR [40]. They have important effects in processes of multiple physiological responses including cell death and survival, cellular response to stress, stem cell division, and pluripotency [41]. MiRNAs also contribute to disease processes such as cancer, cardiovascular disease and neurodegenerative diseases [42-44]. As a result of their small size, relative ease of delivery, and sequence specificity in recognizing their targets, miRNAs are reflected as therapeutic targets of drug development [45].
Notably, miRNA-155 (miR-155) plays a role in various physiological and pathological processes among various miRNAs [46-49]. For instance, MiR-155 is involved in chronic immune activity through T cells by the downregulation of lymphocyteassociated antigens [50]. In autoimmune diseases, miR-155 is found in patients’ tissues and synovial fibroblasts [48]³+. In multiple sclerosis, miR-155 is upregulated in resident myeloid cells of the nervous systems, blood monocytes and activated microglia [51].
The inflammatory process is involved in neuropathic pain [52,53]. PICs including IL-1β, IL-6 and TNF-α are elevated in the nervous system after nerve injury and/or inflammation, responsible for mechanical and thermal hypersensitization [54]. In particular, TNF-α has a role in regulating neuropathic pain [52,53]. In pain models, TNF-α in sensory nerves is upregulated following peripheral nerve injury [55]. TNF-α evokes hyperalgesia and allodynia in naive rats [56]. Chemotherapeutic drugs PLT or vincristine upregulate TNF-α [56]. A blockade of TNF-α with its inhibitor or genetic impairment attenuates mechanical hyperalgesia and allodynia [55]. BTZ treatment increases TNF-α in DRG and spinal dorsal horn [57,58] and TNF-α antibody inhibits allodynia by BTZ [59]. Moreover, a recent study showed that a blockade of TNF-α signal attenuates intracellular p38-MAPK and JNK in the DRG and this alleviates mechanical hyperalgesia and cold hypersensitivity by BTZ via decreasing TRPA1 expression [60]. This is based on that TRPA1 is presented in sensory nerves [61] and is involved in mechanical and cold hypersensitivity [61- 64,67]. Further studies indicate that TRPA1 mediates mechanical and cold hypersensitivity by chemotherapeutics [68,69].
Nonetheless, the role of miRNA-155 in control of TNF-α signal in neuropathy by BTZ has been revealed [70]. Results of a recent study showed that inhibition of miR-155 alleviates mechanical hyperalgesia and cold hypersensitivity by BTZ [70]. This previous study further showed that inhibition of miRNA-155 attenuates upregulation of TNF-α receptor (TNFR1), intracellular p38-MAPK and JNK signal and TRPA1 in the dorsal horn. Data suggest that blocking TNFR1-TRPA1 signal attenuates pain hypersensitivity induced by miRNA-155 mimics. Overall, this provides a base suggesting that targeting small noncoding endogenous RNA molecules is an effective approach to inhibit neuropathic pain caused by chemotherapeutic drugs. More studies are necessary to be explored in terms of some molecular mediators and mechanisms by gene regulation involved in neuropathic pain.
Alternative approaches applied to improve neuropathy
EA has been used for the clinical treatment of chronic pain caused by various diseases [71,72]. It has been well documented for the analgesic effects of EA stimulation in animal with peripheral nerve injuries and inflammatory and neuropathic pain [73-75]. Thus, the beneficial effects of EA on neuropathic pain as alternative treatment strategy were studied during PLT chemotherapy [39]. Data of this previous suggest the underlying molecular mechanisms by which EA improves PLT-induced neuropathic pain. Specifically, it was observed that NOX4 was upregulated after administration of PLT and EA attenuated upregulation of NOX4 as well as increases of oxidative 8-iso PGF2α/8-OHdG whereas EA enhanced Nrf2-ARE signaling pathways.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Future Perspective
CIPN is a main side effect of cancer in patients who experience chemotherapy, and this common symptom can alter treatment strategy on patients. Neuropathic pain associated with chemotherapy has been studied for providing basic mechanisms responsible for CIPN. It is noted that a suitable alternate intervention has benefits for patients with CIPN. Therefore, it is necessary to utilize interventions with an appropriate economic cost to examine the effect of alleviating CIPN in cancer patients with chemotherapy. Experimental strategies need to be considered to address in the future by 1) performing the similar studies using different experimental model of CIPN and/ or a combination of multiple models to address effectiveness of studied interventions, and 2) designing clinical studies and applying the similar experiments corresponding to humans to validate the effectiveness of interventions, Nevertheless, in the future experimental models and clinical studies determining CIPN pathophysiology are still desired to be designed and developed.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Author Contributions
The authors express their gratitude to people who were involved in this work. Ziyi Liu, Sitong Liu and Yu Zhao equally contributed to this review as first authorship. Corresponding author Qian Wang is responsible for overall intellectual perceptions.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Ethics Approval and Consent to Participate
None.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Acknowledgement
None.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Funding
None.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
Conflict of Interest
None.
- Review Article
- Abstract
- Introduction
- Chemotherapy-induced peripheral neuropathy (CIPN)
- Chemotherapeutic agents causing neuropathy
- Cellular and molecular mechanisms leading to neuropathy in peripheral nerve system
- Pro-inflammatory cytokines (PICs)
- Oxidative stress and Nrf2-antioxidant response element (Nrf2-ARE)
- Interventions considered for beneficial effects on neuropathy
- Future Perspective
- Author Contributions
- Ethics Approval and Consent to Participate
- Acknowledgement
- Funding
- Conflict of Interest
- References
References
- Finnerup NB, Kuner R, Jensen TS (2021) Neuropathic Pain: From Mechanisms to Treatment Physiological reviews 101(1): 259-301.
- Hanna Magdi, Zylicz Ben Z (2013) Introduction. Cancer Pain Springer.
- Pasetto LM, D’Andrea MR, Rossi E, Monfardini S (2006) Oxaliplatin-related neurotoxicity: How and why? Crit Rev Oncol Hematol 59(2): 159-168.
- Hoskin PJ (2008) Radiotherapy. In: Clinical pain management: Cancer Pain Hodder Arnold, London, UK, pp. 251-255.
- Portenoy RK (2011) Treatment of cancer pain. Lancet 377(9784): 2236-2247.
- Loprinzi CL, Lacchetti C, Bleeker J, Guido Cavaletti, Cynthia Chauhan, et al. (2020) Prevention and Management of Chemotherapy-Induced Peripheral Neuropathy in Survivors of Adult Cancers: ASCO Guideline Update J Clin Oncol 38(28): 3325-3348.
- Zajączkowska R, Kocot-Kępska M, Leppert W, Anna Wrzosek, Joanna Mika,et al. (2019) Mechanisms of Chemotherapy-Induced Peripheral Neuropathy Int J Mol Sci 20(6): 1451.
- Kim HK, Hwang SH, Abdi S (2017) Tempol Ameliorates and Prevents Mechanical Hyperalgesia in a Rat Model of Chemotherapy-Induced Neuropathic Pain Frontiers in pharmacology 7: 532.
- Kim HK, Hwang SH, Lee SO, Sung Hoon Kim, Salahadin Abdi,et al. (2016) Pentoxifylline Ameliorates Mechanical Hyperalgesia in a Rat Model of Chemotherapy-Induced Neuropathic Pain Pain physician 19: E589-600.
- Duan Z, Su Z, Wang H, Pang X (2018) Involvement of pro-inflammation signal pathway in inhibitory effects of rapamycin on oxaliplatin-induced neuropathic pain Molecular pain 14: 1744806918769426.
- Wang Q, Wang J, Gao D, Li J (2017) Inhibition of PAR2 and TRPA1 signals alleviates neuropathic pain evoked by chemotherapeutic bortezomib. J Biol Regul Homeost Agents 31(4): 977-983.
- Yang Y, Luo L, Cai X, Yuan Fang, Jiaqi Wang et al. (2018) Nrf2 inhibits oxaliplatin-induced peripheral neuropathy via protection of mitochondrial function Free radical biology & medicine 120: 13-24.
- Maruta T, Nemoto T, Hidaka K, Tomohiro Koshida, Tetsuro Shirasaka et al. (2019) Upregulation of ERK phosphorylation in rat dorsal root ganglion neurons contributes to oxaliplatin-induced chronic neuropathic pain. PloS one 14(11): e0225586.
- Tian L, Fan T, Zhou N, Hui Guo, Weijie Zhang, et al. (2015) Role of PAR2 in regulating oxaliplatin-induced neuropathic pain via TRPA1 Transl Neurosci 6(1): 111-116.
- Brandolini L, Castelli V, Aramini A Cristina Giorgio, Gianluca Bianchini, et al. (2019) DF2726A, a new IL-8 signalling inhibitor, is able to counteract chemotherapy-induced neuropathic pain. Scientific reports 9(1): 11729.
- Alé A, Bruna J, Morell M, Velde H V, Monbaliu J et al. (2014) Treatment with anti-TNF alpha protects against the neuropathy induced by the proteasome inhibitor bortezomib in a mouse model. Exp neurol 253: 165-173.
- Alé A, Bruna J, Calls A, Karamita M, Haralambous, et al. (2016) Inhibition of the neuronal NFκB pathway attenuates bortezomib-induced neuropathy in a mouse model. Neurotoxicology 55: 58-64.
- Carozzi VA, Renn CL, Bardini M, Chiorazzi A, Meregalli C, et al. (2013) Bortezomib-induced painful peripheral neuropathy: an electrophysiological, behavioral, morphological and mechanistic study in the mouse. PLoS one 8(9): e72995.
- Liu C, Luan S, OuYang H, Huang Z, Wu S, et al. (2016). Upregulation of CCL2 via ATF3/c-Jun interaction mediated the Bortezomib-induced peripheral neuropathy Brain Behav immunity 53: 96-104.
- Liu CC, Huang ZX, Li X, Shen K F, Liu M et al. (2018). Upregulation of NLRP3 via STAT3-dependent histone acetylation contributes to painful neuropathy induced by bortezomib Exp neurol302: 104-111.
- Miao H, Xu J, Xu D, X Ma, X Zhao, et al. (2019). Nociceptive behavior induced by chemotherapeutic paclitaxel and beneficial role of antioxidative pathways. Physiol Res 68(3): 491-500.
- Yamamoto S, Egashira N (2021). Pathological Mechanisms of Bortezomib-Induced Peripheral Neuropathy. Int J Mol Sci 22(2): 888.
- Moi P, Chan K, Asunis I, A Cao, Y W Kan (1994) Isolation of NF-E2-related factor 2 (Nrf2), a NF-E2-like basic leucine zipper transcriptional activator that binds to the tandem NF-E2/AP1 repeat of the beta-globin locus control region. Proc Natl Acad Sci USA 91(21): 9926-9930.
- Gold R, Kappos L, Arnold DL et al. (2012) Placebo-controlled phase 3 study of oral BG-12 for relapsing multiple sclerosis. N Engl J med 367(12): 1098-1107.
- Gupta P, Makkar TK, Goel L, Pahuja M (2022). Role of inflammation and oxidative stress in chemotherapy-induced neurotoxicity. Immunol Res 70(6): 725-741.
- Yardim A, Gur C, Comakli S, Selcuk Ozdemir, Sefa Kucukler, et al. (2022) Investigation of the effects of berberine on bortezomib-induced sciatic nerve and spinal cord damage in rats through pathways involved in oxidative stress and neuro-inflammation. Neurotoxicology 89: 127-139.
- Liu B, Wang H (2022) Oxaliplatin induces ferroptosis and oxidative stress in HT29 colorectal cancer cells by inhibiting the Nrf2 signaling pathway Exp Ther Med 23(6): 394.
- Lu Y, Wu S, Xiang B, Li L, Lin Y et al. (2020). Curcumin Attenuates Oxaliplatin-Induced Liver Injury and Oxidative Stress by Activating the Nrf2 Pathway Drug Des Devel Ther 14:73-85.
- Miao F, Wang R, Cui G, Li X, Wang T, et al. (2019) Engagement of MicroRNA-155 in Exaggerated Oxidative Stress Signal and TRPA1 in the Dorsal Horn of the Spinal Cord and Neuropathic Pain During Chemotherapeutic Oxaliplatin. Neurotox Res 36(4): 712-723.
- Fidanboylu M, Griffiths LA, Flatters SJ (2011). Global inhibition of reactive oxygen species (ROS) inhibits paclitaxel-induced painful peripheral neuropathy. PLoS one 6(9): e25212.
- Duggett NA, Griffiths LA, McKenna OE, Vittorio de Santis, Nutcha Yongsanguanchai et al. (2016) Oxidative stress in the development, maintenance and resolution of paclitaxel-induced painful neuropathy Neuroscience 333: 13-26.
- Janes K, Doyle T, Bryant L, Emanuela Esposito, Salvatore Cuzzocrea, et al. (2013) Bioenergetic deficits in peripheral nerve sensory axons during chemotherapy-induced neuropathic pain resulting from peroxynitrite-mediated post-translational nitration of mitochondrial superoxide dismutase Pain 154(11): 2432-2440.
- Altenhofer S, Kleikers PW, Radermacher KA, Peter Scheurer, J J Rob Hermans, et al. (2012) The NOX toolbox: validating the role of NADPH oxidases in physiology and disease. Cell Mol Life Sci 69(14): 2327-2343.
- Salvemini D, Little JW, Doyle T, Neumann WL (2011) Roles of reactive oxygen and nitrogen species in pain Free radical biology & medicine 51(5): 951-966.
- Lam GY, Huang J, Brumell JH (2010) The many roles of NOX2 NADPH oxidase-derived ROS in immunity Seminars in immunopathology 32(4): 415-430.
- Gavazzi G, Banfi B, Deffert C, Laurence Fiette, Michela Schappi, et al. (2006) Decreased blood pressure in NOX1-deficient mice FEBS letters 580: 497-504.
- Suzuki Y, Hattori K, Hamanaka J, Tetsuji Murase, Yusuke Egashira, et al. (2012) Pharmacological inhibition of TLR4-NOX4 signal protects against neuronal death in transient focal ischemia Scientific reports 2: 896.
- Kallenborn-Gerhardt W, Schroder K, Del Turco D, Ruirui Lu, Katharina Kynast, et al. (2012) NADPH oxidase-4 maintains neuropathic pain after peripheral nerve injury The Journal of neuroscience: the official journal of the Society for Neuroscience 32(30): 10136-10145.
- Zhao X, Liu L, Wang Y, Y Zhao, Y Zhang et al. (2019) Electroacupuncture enhances antioxidative signal pathway and attenuates neuropathic pain induced by chemotherapeutic paclitaxel Physiological research 68(3): 501-510.
- Bartel DP (2009) MicroRNAs: target recognition and regulatory functions. Cell 136(2): 215-233.
- Sayed D, Abdellatif M (2011) MicroRNAs in development and disease Physiological reviews 91(3): 827-887.
- Eacker SM, Dawson TM, Dawson VL (2009) Understanding microRNAs in neurodegeneration Nat Rev Neurosci 10(12): 837-841.
- Farazi TA, Spitzer JI, Morozov P, Tuschl T (2011) miRNAs in human cancer. The Journal of pathology 223: 102-115.
- Han M, Toli J, Abdellatif M (2011) MicroRNAs in the cardiovascular system Current opinion in cardiology 26(3): 181-189.
- Gambari R, Fabbri E, Borgatti M, Ilaria Lampronti, Alessia Finotti, et al. (2011). Targeting microRNAs involved in human diseases: a novel approach for modification of gene expression and drug development. Biochem Pharmacol 82(10): 1416-1429.
- Calame K (2007) MicroRNA-155 function in B Cells. Immunity 27(6): 825-827.
- Elton TS, Selemon H, Elton SM, Parinandi NL (2013) Regulation of the MIR155 host gene in physiological and pathological processes. Gene 532(1): 1-12.
- Faraoni I, Antonetti FR, Cardone J, Bonmassar E (2009) miR-155 gene: a typical multifunctional microRNA Biochimica et biophysica acta 1792(6): 497-505.
- O'Connell RM, Rao DS, Baltimore D (2012) microRNA regulation of inflammatory responses Annu Rev Immunol 30: 295-312.
- Sonkoly E, Janson P, Majuri ML, Terhi Savinko, Nanna Fyhrquist, et al. (2010) MiR-155 is overexpressed in patients with atopic dermatitis and modulates T-cell proliferative responses by targeting cytotoxic T lymphocyte-associated antigen 4 The Journal of allergy and clinical immunology 126(3): 581-589.e581-520.
- Moore CS, Rao VT, Durafourt BA, Barry J Bedell, Samuel K Ludwin, et al. (2013) miR-155 as a multiple sclerosis-relevant regulator of myeloid cell polarization Annals of neurology 74(5): 709-720.
- Clark AK, Old EA, Malcangio M (2013) Neuropathic pain and cytokines: current perspectives Journal of pain research 6: 803-814.
- Mika J, Zychowska M, Popiolek-Barczyk K, Ewelina Rojewska, Barbara Przewlocka (2013) Importance of glial activation in neuropathic pain European journal of pharmacology 716(1-3): 106-119.
- Moalem G, Tracey DJ (2006) Immune and inflammatory mechanisms in neuropathic pain Brain research reviews 51(12): 240-264.
- Miller RJ, Jung H, Bhangoo S, White FA (2009) Cytokine and chemokine regulation of sensory neuron function. In: Handbook of Experimental Pharmacology. Edited by: Canning BJ, Spina DV. Berlin Heidelberg, Springer, pp. 417-449.
- Carozzi VA, Canta A, Chiorazzi A (2015) Chemotherapy-induced peripheral neuropathy: What do we know about mechanisms? Neurosci Lett 596: 90-107.
- Zhang J, Su YM, Li D, Yu Cui, Zhen-Zhen Huang, et al. (2014) TNF-alpha-mediated JNK activation in the dorsal root ganglion neurons contributes to Bortezomib-induced peripheral neuropathy. Brain Behav Immun 38: 185-191.
- Li ZY, Zhang YP, Zhang J, Su-Bo Zhang, et al. (2016) The possible involvement of JNK activation in the spinal dorsal horn in bortezomib-induced allodynia: the role of TNF-alpha and IL-1beta J Anesth 30(1): 55-63.
- Chiorazzi A, Canta A, Meregalli C, Valentina Carozzi, Barbara Sala, et al. (2013) Antibody against tumor necrosis factor-alpha reduces bortezomib-induced allodynia in a rat model Anticancer research 33(12): 5453-5459.
- Li C, Deng T, Shang Z, Di Wang, Yi Xiao, et al. (2018) Blocking TRPA1 and TNF-alpha Signal Improves Bortezomib-Induced Neuropathic Pain. Cellular physiology and biochemistry: international journal of experimental cellular physiology, biochemistry, and pharmacology 51(5): 2098-2110.
- Jordt SE, Bautista DM, Chuang HH, David D McKemy, Peter M Zygmunt, et al. (2004) Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature 427(6971): 260-265.
- Kwan KY, Allchorne AJ, Vollrath MA, Adam P Christensen, Duan-Sun Zhang, et al. (2006) TRPA1 contributes to cold, mechanical, and chemical nociception but is not essential for hair-cell transduction. Neuron 50(2): 277-289.
- Story GM, Peier AM, Reeve AJ, Johannes Mosbacher, Todd R Hricik, et al. (2003) ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell 112(6): 819-829.
- Andersson DA, Gentry C, Moss S, Bevan S (2008) Transient receptor potential A1 is a sensory receptor for multiple products of oxidative stress. J Neurosci 28(10): 2485-2494.
- Bandell M, Story GM, Hwang SW, Veena Viswanath, Samer R Eid, et al. (2004) Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin Neuron 41(6): 849-857.
- Bautista DM, Movahed P, Hinman A, Helena E Axelsson, Olov Sterner, et al. (2005) Pungent products from garlic activate the sensory ion channel TRPA1. Proc Natl Acad Sci U S A 102(34): 12248-12252.
- Sawada Y, Hosokawa H, Matsumura K, Kobayashi S (2008) Activation of transient receptor potential ankyrin 1 by hydrogen peroxide. Eur J Neurosci 27(5): 1131-1142.
- Zhao M, Isami K, Nakamura S, Hisashi Shirakawa, Takayuki Nakagawa, et al. (2012) Acute cold hypersensitivity characteristically induced by oxaliplatin is caused by the enhanced responsiveness of TRPA1 in mice Molecular pain 8: 55.
- Nassini R, Fusi C, Materazzi S, Elisabetta Coppi, Tiziano Tuccinardi, et al. (2015) The TRPA1 channel mediates the analgesic action of dipyrone and pyrazolone derivatives. Br J Pharmacol 172(13): 3397-3411.
- Duan Z, Zhang J, Li J, Xiaochuan Pang, Hushan Wang,et al. (2020) Inhibition of microRNA-155 Reduces Neuropathic Pain During Chemotherapeutic Bortezomib via Engagement of Neuroinflammation. Front Oncol 10: 416.
- Kavoussi B, Ross BE (2007) The neuroimmune basis of anti-inflammatory acupuncture. Integr Cancer Ther 6: 251-257.
- McDonald JL, Cripps AW, Smith PK, Caroline A Smith, Charlie C Xue, et al. (2013) The anti-inflammatory effects of acupuncture and their relevance to allergic rhinitis: a narrative review and proposed model. Evid Based Complement Alternat Med 2013: 591796.
- Kim SK, Park JH, Bae SJ, Ji Hoon Kim, Byung Gil Hwang, et al. (2005) Effects of electroacupuncture on cold allodynia in a rat model of neuropathic pain: mediation by spinal adrenergic and serotonergic receptors. Exp Neurol 195(2): 430-436.
- Pittler MH, Ernst E (2008) Complementary therapies for neuropathic and neuralgic pain: systematic review Clin J Pain 24(8): 731-733.
- DeLeo JA, Yezierski RP (2001) The role of neuroinflammation and neuroimmune activation in persistent pain. Pain 90(1-2): 1-6.
- Saifee TA, Elliott KJ, Rabin N, Kwee L Yong, Shirley D'Sa, et al. (2010) Bortezomib-induced inflammatory neuropathy. J Peripher Nerv Syst 15(4): 366-368.
- Wadd N, Peedell C, Polwart C (2022) Real-World Assessment of Cancer Drugs Using Local Data Uploaded to the Systemic Anti-Cancer Therapy Dataset in England. Clin Oncol (R Coll Radiol) 34(8): 497-507.
- Ciarimboli G (2021) Anticancer Platinum Drugs Update. Biomolecules 11(11): 1637.
- Babajani A, Manzari-Tavakoli A, Jamshidi E, Roghayeh Tarasi, Hassan Niknejad, et al. (2022) Anti-cancer effects of human placenta-derived amniotic epithelial stem cells loaded with paclitaxel on cancer cells. Sci Rep 12(1): 18148.
- Zhang Y, Yang SH, Guo XL (2017) New insights into Vinca alkaloids resistance mechanism and circumvention in lung cancer Biomed Pharmacother 96: 659-666.
- Ibrahim NK (2021) Ixabepilone: Overview of Effectiveness, Safety, and Tolerability in Metastatic Breast Cancer Front Oncol 11: 617874.
- Richardson P, Hideshima T, Anderson K (2002) Thalidomide in multiple myeloma. Biomed Pharmacother 56(3): 115-128.






























